Lesson 6 Hess law and enthalpy cycles using

















- Slides: 22

Lesson 6 – Hess’ law and enthalpy cycles using enthalpy of combustion Saturday, June 12, 2021

Hess’ Law using enthalpy of combustion Objective: to recall and use Hess’s Law for enthalpy of combustion data Success criteria: • Recall Hess’ Law • Interpret an enthalpy cycle using Hess’ Law • Construct an enthalpy cycle using Hess’ Law, for combustion data • To calculate an enthalpy change from combustion data using Hess’s Law • Know reasons why the enthalpy change cannot always be measured directly

Starter - Test 1. 2. 3. 4. What are standard conditions? Define ΔHc Define ΔHf Define ΔHr Describe the following equations as ΔHc, ΔHr or ΔHf 5. 3 C(s) + 4 H 2(g) C 3 H 8(g) 6. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) 7. C 2 H 4(g) + H 2(g) C 2 H 6(g) 8. 2 C 2 H 6(l) + 7 O 2(g) 4 CO 2(g) + 6 H 2 O(l) Write the equation for ΔHc for 9. H 2 10. CH 3 OH 11. C

Starter - Answers 1. 2. 3. 4. Standard conditions are 298 K, 100 k. Pa and 1 M for solutions. All substances should be in their standard states The is the enthalpy change that takes place when one mole of a substance reacts completely with oxygen under standard conditions, all reactants and products being in their standard states. The is the enthalpy change that takes place when one mole of a compound is formed from its constituent elements in their standard states under standard conditions. The is the enthalpy change that accompanies a reaction in the molar quantities expressed in a chemical equation under standard conditions, all reactants and products being in their standard states.

Starter - Answers Describe the following equations as ΔHc, ΔHr or ΔHf 5. 3 C(s) + 4 H 2(g) C 3 H 8(g) 6. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(l) 7. C 2 H 4(g) + H 2(g) C 2 H 6(g) 8. 2 C 2 H 6(l) + 7 O 2(g) 4 CO 2(g) + 6 H 2 O(l) Write the equation for ΔHc for 9. H 2(g) + ½ O 2(g) H 2 O(l) 10. CH 3 OH(l) + O 2(g) CO 2(g) + H 2 O(l) 11. C(s) + O 2(g) CO 2(g)

Measuring enthalpy changes • Using a calorimeter for a combustion reaction or or other reaction can often give the ΔHr directly (think about the spirit burners and copper sulphate experiments you have done). • Bond enthalpies can also be used to estimate ΔHr

Problems There may be: 1. a high activation energy 2. a slow reaction rate 3. more than one reaction taking place Take for example This is virtually impossible to measure directly - think of the number of compounds of hydrogen and carbon that could form!

Solution Measuring enthalpy of reaction indirectly • We can use Hess’ law to measure the energy change of reactions indirectly. • We use enthalpy changes that we can measure to calculate ones that we can’t. • This means we need to work out a “cycle” of reactions.

Using ΔHC Ө Many things burn and chemists love burning things! So we have huge tables of ΔHC Ө including very many of the most common chemicals. In a reaction which we cannot measure directly, ΔHC Ө gives us a link between reactants and products.

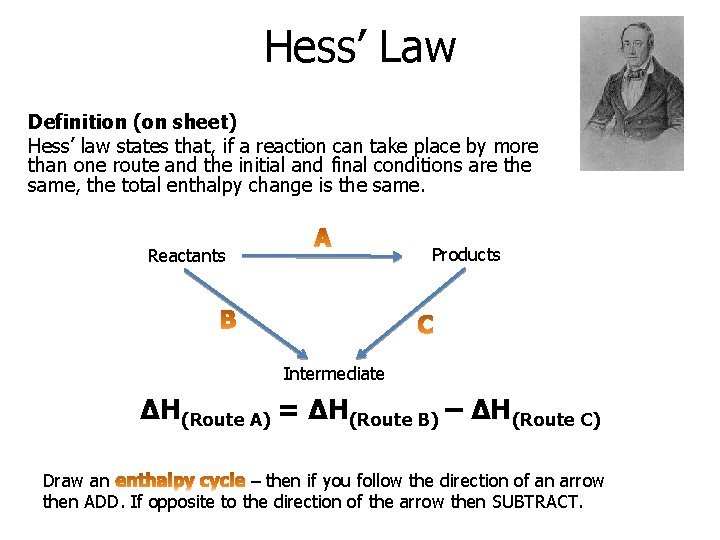
Hess’ Law Definition (on sheet) Hess’ law states that, if a reaction can take place by more than one route and the initial and final conditions are the same, the total enthalpy change is the same. Products Reactants Intermediate ΔH(Route A) = ΔH(Route B) – ΔH(Route C) Draw an – then if you follow the direction of an arrow then ADD. If opposite to the direction of the arrow then SUBTRACT.

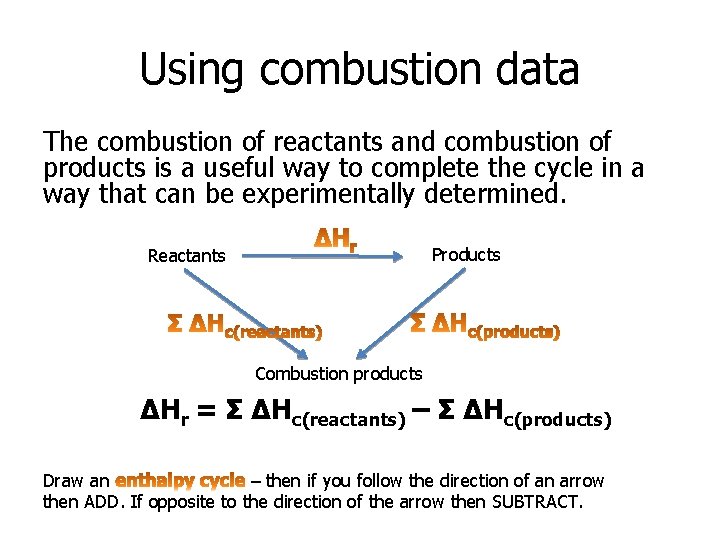
Using combustion data The combustion of reactants and combustion of products is a useful way to complete the cycle in a way that can be experimentally determined. Products Reactants Combustion products ΔHr = Σ ΔHc(reactants) – Σ ΔHc(products) Draw an – then if you follow the direction of an arrow then ADD. If opposite to the direction of the arrow then SUBTRACT.

• Example on worksheet

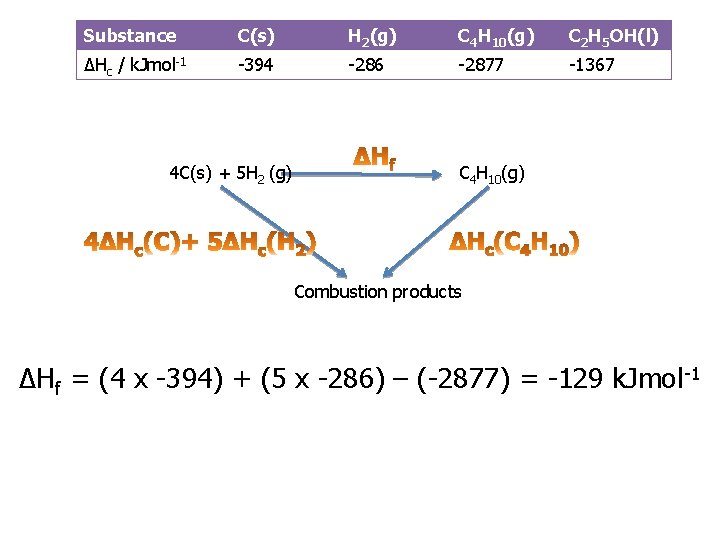
Practice question 1 a You are provided with the following enthalpy changes of combustion Substan ce C(s) H 2(g) C 4 H 10(g) C 2 H 5 OH( l) ΔHc / k. Jmol-1 -394 -286 -2877 -1367 Determine the enthalpy change for the following reaction: 4 C(s) + 5 H 2(g) C 4 H 10(g)

Substance C(s) H 2(g) C 4 H 10(g) C 2 H 5 OH(l) ΔHc / k. Jmol-1 -394 -286 -2877 -1367 4 C(s) + 5 H 2 (g) C 4 H 10(g) Combustion products ΔHf = (4 x -394) + (5 x -286) – (-2877) = -129 k. Jmol -1

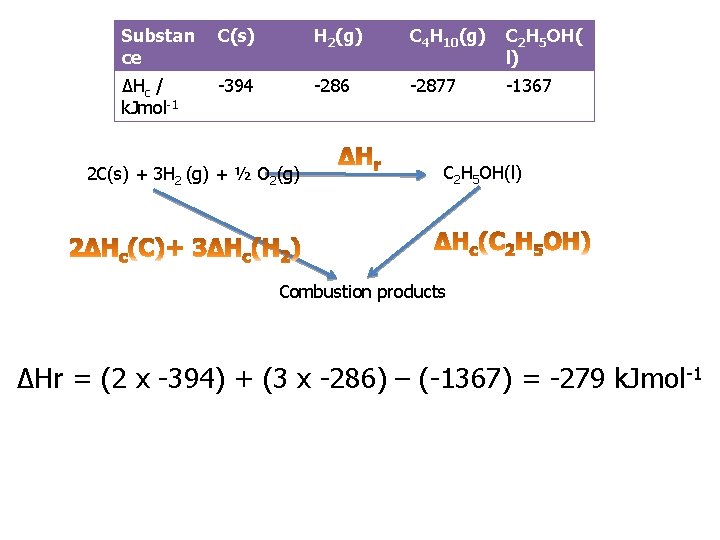
Practice question 1 b You are provided with the following enthalpy changes of combustion Substance C(s) ΔHc / k. Jmol- -394 1 H 2(g) C 4 H 10(g) C 2 H 5 OH(l) -286 -2877 -1367 Determine the enthalpy change for the following reaction: 2 C(s) + 3 H 2(g) + ½O 2(g) C 2 H 5 OH(l)

Substan ce C(s) H 2(g) C 4 H 10(g) C 2 H 5 OH( l) ΔHc / k. Jmol-1 -394 -286 -2877 -1367 2 C(s) + 3 H 2 (g) + ½ O 2(g) C 2 H 5 OH(l) Combustion products ΔHr = (2 x -394) + (3 x -286) – (-1367) = -279 k. Jmol -1

Exam questions (Edexcel) printed as booklet Hess Cycles both and enthalpy of combustion to finish for homework

Examination question


Mark scheme

Mark scheme

Hess’ Law using enthalpy of combustion Objective: to recall and use Hess’s Law for enthalpy of combustion data Success criteria: • Recall Hess’ Law • Interpret an enthalpy cycle using Hess’ Law • Construct an enthalpy cycle using Hess’ Law, for combustion data • To calculate an enthalpy change from combustion data using Hess’s Law • Know reasons why the enthalpy change cannot always be measured directly