Lesson 5 Aim and Objectives Aim How are

Lesson 5: Aim and Objectives Aim: How are ionic bonds differently depicted than covalent bonds? Objectives: 1. Learn to draw Lewis Dot Structures for ionic compounds. 2. In terms of electrical conductivity, how do ionic bonds differ from covalent bonds.

Lewis Dot Structure Covalent Bonds
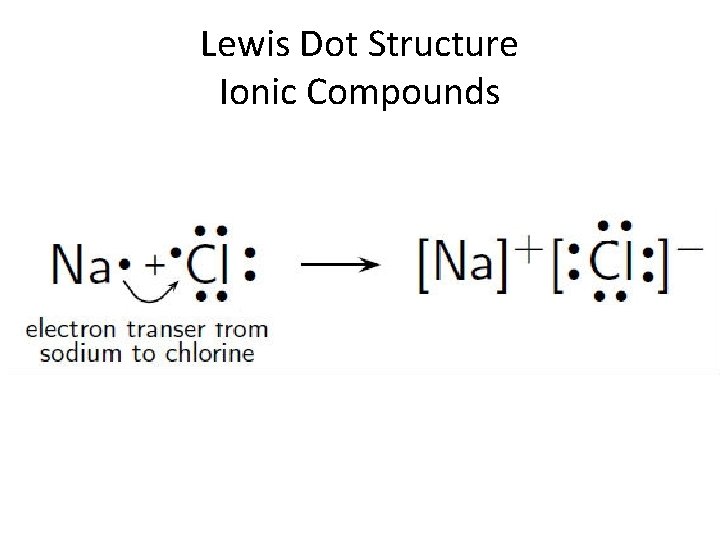
Lewis Dot Structure Ionic Compounds

Write the Lewis Dot Structure • • Na and Cl K and F Ca and O Mg and Cl Ca and I Al and Br Al and O

Writing the Lewis Dot Structure 1. 2. 3. 4. 5. Is it covalent or ionic? If it is covalent, electrons have to be drawn… _____ electrons per bond Add up all the electrons to equal… If it is ionic, you have to figure out how many positive ions and how many negative ions you need. 6. You must draw _______ and the positive ion has _____ electrons and the negative ion has _____ electrons.
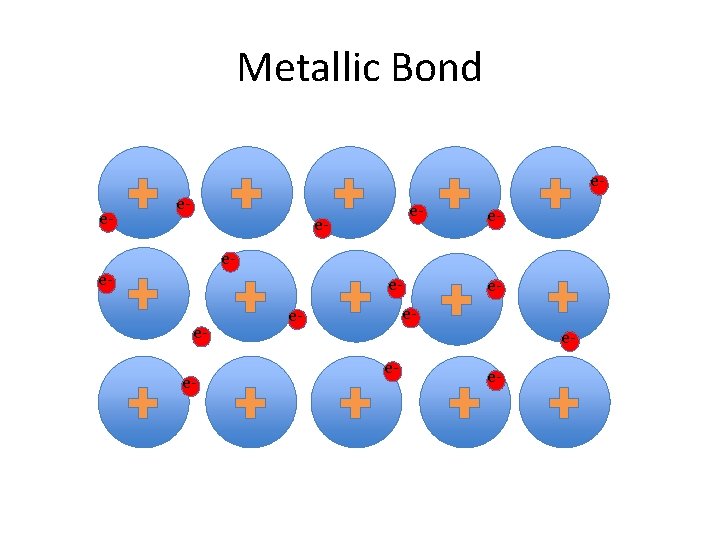
Metallic Bond ee- e- eee- e-

Metals and Electrical Conductivity • http: //cd 1. edb. hkedcity. net/cd/science/chemi stry/resource/animations/metallic_bond/met allic. html • http: //projectsharetexas. org/resource/chemic al-bonding-metallic-bonds-ontrack-chemistrymodule-3 -lesson-5 • http: //www. mhhe. com/physsci/chemistry/ess entialchemistry/flash/molvie 1. swf • http: //www. northland. cc. mn. us/biology/Biolo gy 1111/animations/dissolve. html

Ionic Compounds and Electrical Conductivity http: //bcs. whfreeman. com/thelifewire/content/ch p 02/02020. html http: //highered. mheducation. com/olcweb/cgi/plug inpop. cgi? it=swf: : 100%: : /sites/dl/free/00725 12644/117354/rev 01_Ionic_vs_Covalent. swf: : Ionic %20 vs. %20 Covalent%20 Bonding http: //www. mhhe. com/physsci/chemistry/essentia lchemistry/flash/molvie 1. swf

Homework 5 Define the following: 1. Hydrogen bonds 2. Dipole-dipole interactions 3. Van der Waals forces (Dispersion forces)
- Slides: 9