Lesson 5 1 States of Matter Chemistry 1









- Slides: 12

Lesson 5. 1 – States of Matter Chemistry 1 Honors Dr. J. Venables Northwestern High School

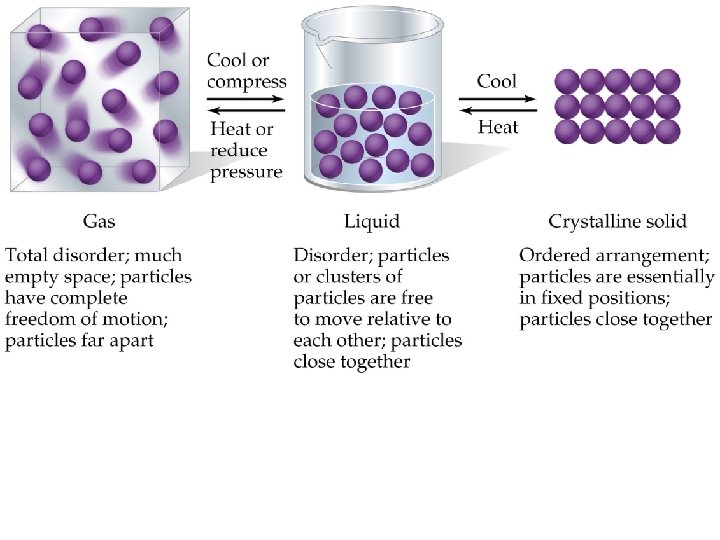
A Comparison of Solids, Liquids, and Gases • Physical properties of substances are understood in terms of kinetic molecular theory: – Gases are highly compressible, assume shape and volume of container: • Gas molecules are far apart and do not interact much with each other. – Liquids are almost incompressible, assume the shape but not the volume of container: • Liquids molecules are held closer together than gas molecules, but not so rigidly that the molecules cannot slide past each other.

– Solids are incompressible and have a definite shape and volume: • Solid molecules are packed closely together. The molecules are so rigidly packed that they cannot easily slide past each other.


• Converting a gas into a liquid or solid requires the molecules to get closer to each other: – cool or compress. • Converting a solid into a liquid or gas requires the molecules to move further apart: – heat or reduce pressure. • The forces holding solids and liquids together are called intermolecular forces. • Hydrogen Bonds • Dipole-dipole forces • London dispersion forces

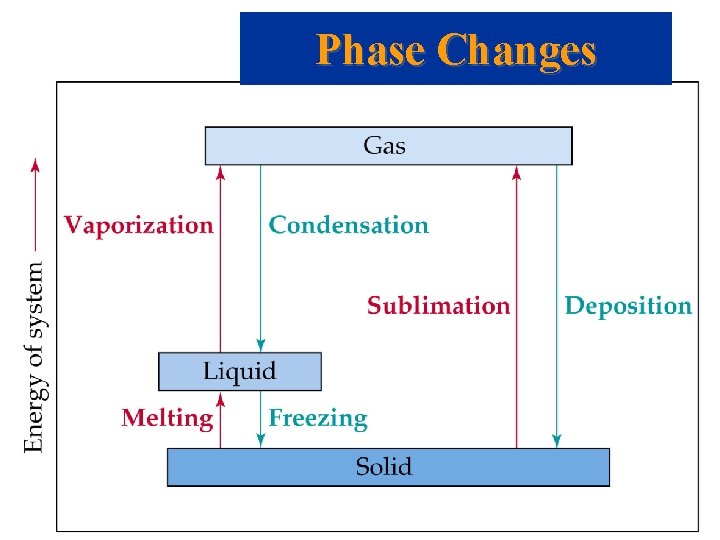
Phase Changes

Energy Changes Accompanying Phase Changes • All phase changes are possible under the right conditions. • The sequence heat solid melt heat liquid boil heat gas is endothermic. • The sequence cool gas condense cool liquid freeze cool solid is exothermic.

• • • Heating and Cooling Curves Plot of temperature change versus heat added is a heating curve. Plot of temperature change versus heat removed is a cooling curve. During a phase change, adding heat causes no temperature change. – These points are used to calculate Hfus and Hvap. Supercooling: When a liquid is cooled below its melting point and it still remains a liquid. Achieved by keeping the temperature low and increasing kinetic energy to break intermolecular forces.

From origin to “a” = From a to b = From b to c = From c to d = From d up =

From origin to a = solid From a to b = melting/freezing point From b to c = liquid From c to d = boiling/condensation point From d up = gas

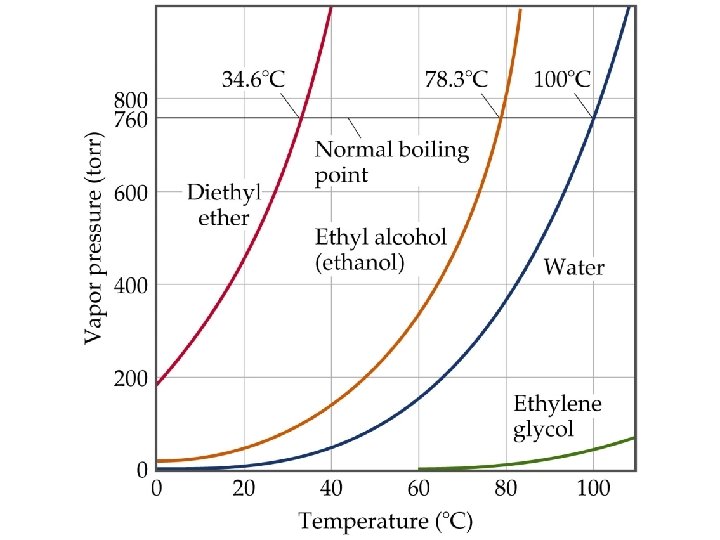
Vapor Pressure and Boiling Point • Liquids boil when the external pressure equals the vapor pressure. • Temperature of boiling point increases as pressure increases. • Two ways to get a liquid to boil: increase temperature or decrease pressure. • Pressure cookers operate at high pressure. At high pressure the boiling point of water is higher than at 1 atm. Therefore, there is a higher temperature at which the food is cooked, reducing the cooking time required. • Normal boiling point is the boiling point at 760 mm. Hg (1 atm).
