Lesson 3 2 Naming Compounds J Venables Northwestern

Lesson 3. 2 – Naming Compounds J. Venables Northwestern High School Chemistry 1 Honors

• Naming of compounds, nomenclature, is divided into organic compounds (those containing C) and inorganic compounds (the rest of the periodic table). • Cations formed from a metal have the same name as the metal. Example: Na+ = sodium ion. • If the metal can form more than one cation, then the charge is indicated in parentheses in the name. Examples: Cu+ = copper(I); Cu 2+ = copper(II). • Cations formed from non-metals end in -ium. Example: NH 4+ = ammonium ion.

Negative Ions • Monatomic anions (with only one atom) are called -ide. Example: Cl- is ______. Exceptions: hydroxide (OH-), cyanide (CN-), peroxide (O 22 -). Polyatomic anions (with many atoms) containing oxygen end in -ate or -ite. (The one with more oxygen is called -ate. ) – see sheet. Examples: NO 3 - is nitrate, NO 2 - is _______.

• Chemical nomenclature is a systematic way of naming compounds. – Name the cation followed by the anion. – For monatomic cations use the element name. – For monatomic anions, use the root element name and the suffix –ide. – To distinguish between different oxidation states of the same element, the oxidation state is written in parentheses after the name of the cation. (no roman numerals for group 1, group 2, Al, Zn, Cd, Ag) – When the compound contains a polyatomic ion, name the cation followed by the name of the polyatomic ion.

Naming Ionic Compounds • Name the cation then anion for the ionic compound. Example: Ba. Br 2 = barium bromide. Examples – Name each: 1. Ca(HSO 3)2 2. Ba(Cl. O 3)2 3. Na 3 N 4. Al(CN)3 5. (NH 4)3 PO 4 6. Li. NO 3 7. Cu(NO 2)2

Naming Ionic Compounds • Name the cation then anion for the ionic compound. Example: Ba. Br 2 = barium bromide. Examples – Name each: 1. Ca(HSO 3)2 calcium hydrogen sulfite 2. Ba(Cl. O 3)2 barium chlorate 3. Na 3 N sodium nitride 4. Al(CN)3 aluminum cyanide 5. (NH 4)3 PO 4 ammonium phosphate 6. Li. NO 3 lithium nitrate 7. Cu(NO 2)2 copper (II) nitrite

Examples of Writing Formulas Example: barium chlorate = Ba(Cl. O 3)2. Examples – Write the formula for each: 1. Ammonium chloride 2. Potassium carbonate 3. Aluminum sulfate 4. Magnesium acetate 5. Cobalt (III) nitrate 6. Lead (IV) bromide 7. Mercury (II) chlorite

Examples of Writing Formulas Example: barium chlorate = Ba(Cl. O 3)2. Examples – Write the formula for each: 1. Ammonium chloride NH 4 Cl 2. Potassium carbonate K 2 CO 3 3. Aluminum sulfate Al 2(SO 4)3 4. Magnesium acetate Mg(C 2 H 3 O 2)2 5. Cobalt (III) nitrate Co(NO 3)3 6. Lead (IV) bromide Pb. Br 4 7. Mercury (II) chlorite Hg(Cl. O 2)2
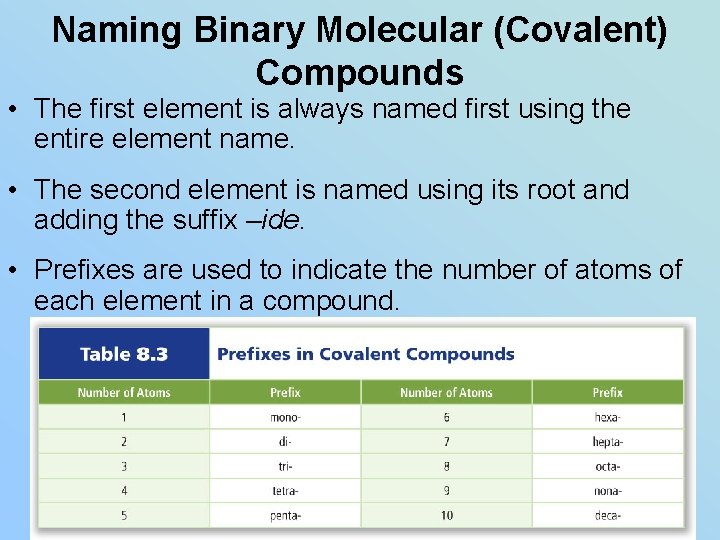
Naming Binary Molecular (Covalent) Compounds • The first element is always named first using the entire element name. • The second element is named using its root and adding the suffix –ide. • Prefixes are used to indicate the number of atoms of each element in a compound.

Examples 1. 2. 3. 4. 5. 6. Dinitrogen tetroxide Sulfur hexafluoride Dihydrogen monoxide P 2 O 5 Xe. F 4 PF 3

Examples 1. 2. 3. 4. 5. 6. Dinitrogen tetroxide N 2 O 4 Sulfur hexafluoride SF 6 Dihydrogen monoxide H 2 O P 2 O 5 diphosphorus pentoxide Xe. F 4 xenon tetrafluoride PI 3 phosphorus triiodide

Names and Formulas for Acids • Acids are compounds that start with hydrogen (H). H is bonded to a negative ion • The names of acids are related to the names of anions: -ide becomes hydro-…. -ic acid; -ate becomes -ic acid; -ite becomes -ous acid.

Examples 1. HCl 2. HNO 3 3. H 2 SO 3 4. Chlorous acid 5. Perbromic acid 6. Hydrosulfuric acid
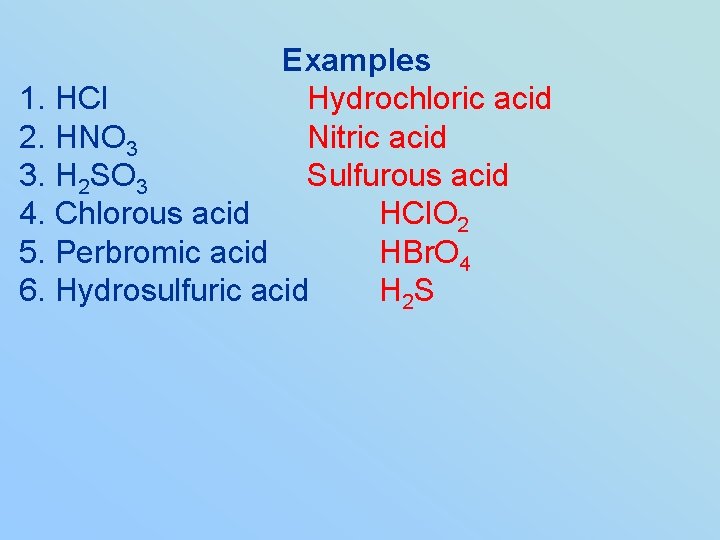
Examples 1. HCl Hydrochloric acid 2. HNO 3 Nitric acid 3. H 2 SO 3 Sulfurous acid 4. Chlorous acid HCl. O 2 5. Perbromic acid HBr. O 4 6. Hydrosulfuric acid H 2 S
- Slides: 14