Lesson 2 Determining Density Lesson 2 Determining Density




















- Slides: 23

Lesson 2 Determining Density

Lesson 2 – Determining Density • You will use mass and volume measurements to calculate the densities of water, regular shaped objects, and irregular shaped objects.

Lesson 2 –COS 1. 0 1. 1 1. 2 1. 3 1. 4 1. 5

Objectives • Discuss the terms “mass” and “volume”. • Find the mass of a known volume of water. • Calculate the mass of 1. 0 cubic centimeter of water. • Measure the mass and volume of some regular and irregular objects. • Calculate the density of these objects.

Getting Started • In your notebook, write what you think the difference is between mass and volume. • What units of measurement are used for mass and volume? • Read “Useful Calculations” on page 16.

Lesson 2 - (1 -2) gram - a metric unit used to measure mass milliliter – unit used to measure volume cm 3 – unit used to measure volume g/cm 3 – unit of measure of density water displacement – method used to find the volume of irregular-shaped objects

Vocabulary • Density: The mass of a known volume of a substance, usually measured in g/cm 3. • Mass: A measure of the amount of matter in an object, usually measured in grams or kilograms • Volume: The amount of space occupied by a sample of matter, usually measured in liters (L), milliliters (m. L) for liquids and cubic centimeters (cm 3) and cubic meters (m 3) for solids.

Lesson 2 - (2 -2) Weight: A measure of the force of gravity, measured in Newtons. L x W x H: Method used to find the volume of a square or rectangular object 1 g/cm 3: The density of water Characteristic property: An attribute that can be used to help identify a substance. It is not affected by the amount or shape of a substance

Lesson 2. 1 – • Does changing the volume of water change the density of water?

Lesson 2. 1 If…. then… because…. .

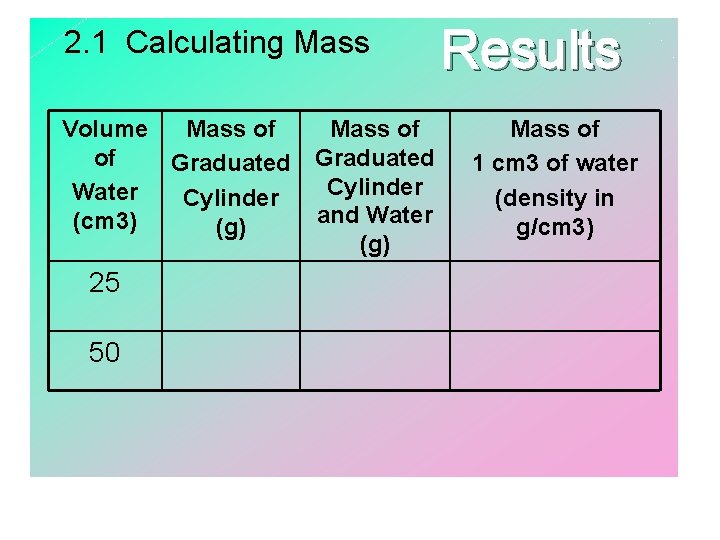
Lesson 2. 1 1. Use the balance to find the mass of the graduated cylinder. 2. Add 50 m. L of water to the graduated cylinder. Mass the graduated cylinder with water. 3. Subtract the initial mass of the graduated cylinder from the mass of the graduated cylinder with water.

2. 1 Calculating Mass Volume Mass of of Graduated Water Cylinder (cm 3) (g) 25 50 Mass of Graduated Cylinder and Water (g) Results Mass of 1 cm 3 of water (density in g/cm 3)

Lesson 2. 1 • Density is calculated by dividing the mass by the volume; measured in grams per cubic centimeter (g/cm 3). • Volume is the amount of space taken up by an object; measured in m. L or cm 3. (space) • Changing the amount of a substance does not change the density of the substance. • Density is a characteristic property of matter. • Characteristic property - property that is independent of mass, volume, and shape….

Lesson 2. 2 – • Do the substances making up different regular-shaped objects have the same density?

Lesson 2. 2 If…. then… because…. .

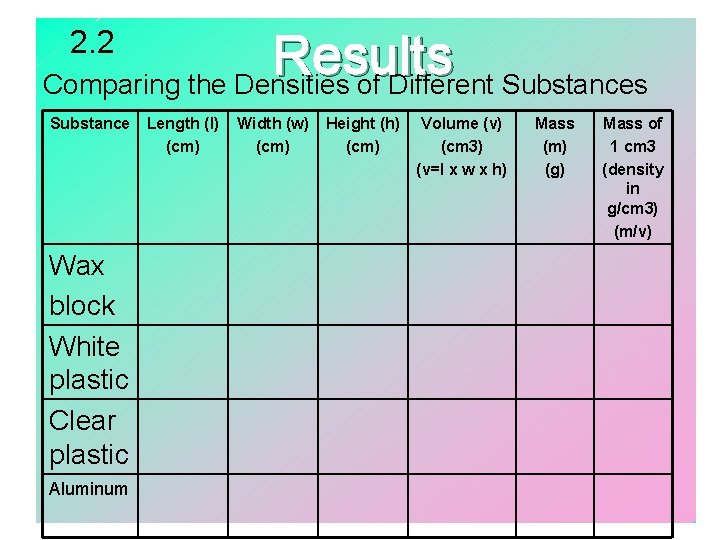
Lesson 2. 2 – Materials: Metric ruler Electronic balance A variety of objects 1. Gather materials. 2. Construct a data table. 3. Use the metric ruler to measure the length, width, and height of each object. 4. Use the electronic balance to mass each object 5. Calculate the density of each object.

2. 2 Results Comparing the Densities of Different Substances Substance Wax block White plastic Clear plastic Aluminum Length (l) (cm) Width (w) (cm) Height (h) (cm) Volume (v) (cm 3) (v=l x w x h) Mass (m) (g) Mass of 1 cm 3 (density in g/cm 3) (m/v)

Lesson 2. 2 Conclusion • The shape of an object does not change the density of the substance of which it is composed. • The volume of a regular shaped object is found by measuring l x w x h; measured in cm 3(one m. L = one cm 3) • The mass of a regular-shaped object is found by using an electronic balance; measured in grams. • Density is a characteristic property of matter; measured in g/cm 3. • Characteristic property - property that is independent of mass, volume, and shape….

Lesson 2. 3 – • Do the substances making up different irregular-shaped objects have the same density?

Lesson 2. 3 If…. then… because…. .

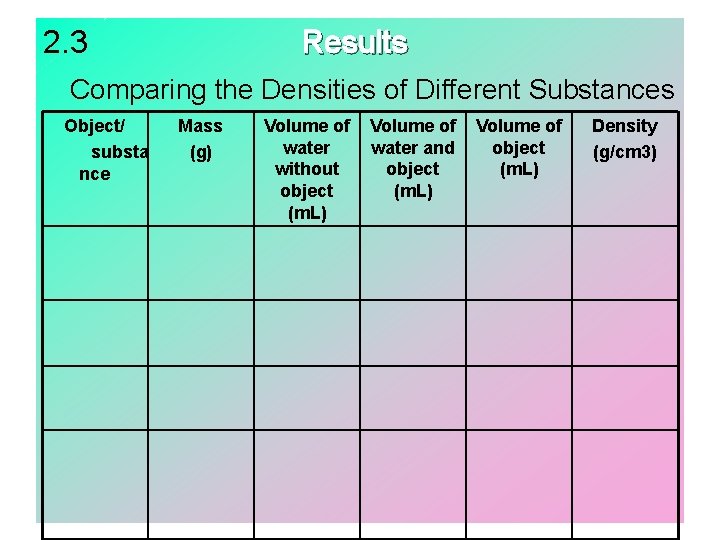
Lesson 2. 3 – Materials: Graduated cylinder Water Electronic balance Irregular-shaped objects 1. 2. 3. 4. 5. Gather materials. Construct a data table. Use the electronic balance to mass each object. Use the graduated cylinder and water to find the volume of each object by water displacement. Calculate the density of each object.

2. 3 Results Comparing the Densities of Different Substances Object/ substa nce Mass (g) Volume of water without object (m. L) Volume of water and object (m. L) Volume of object (m. L) Density (g/cm 3)

Lesson 2. 3 • Different irregular-shaped objects made of the same material will have the same density (characteristic property). Different materials have different densities.