Lesson 10 Thermal Decomposition Chemistry Key Stage 3






- Slides: 14

Lesson 10 - Thermal Decomposition Chemistry- Key Stage 3 Energetics 1

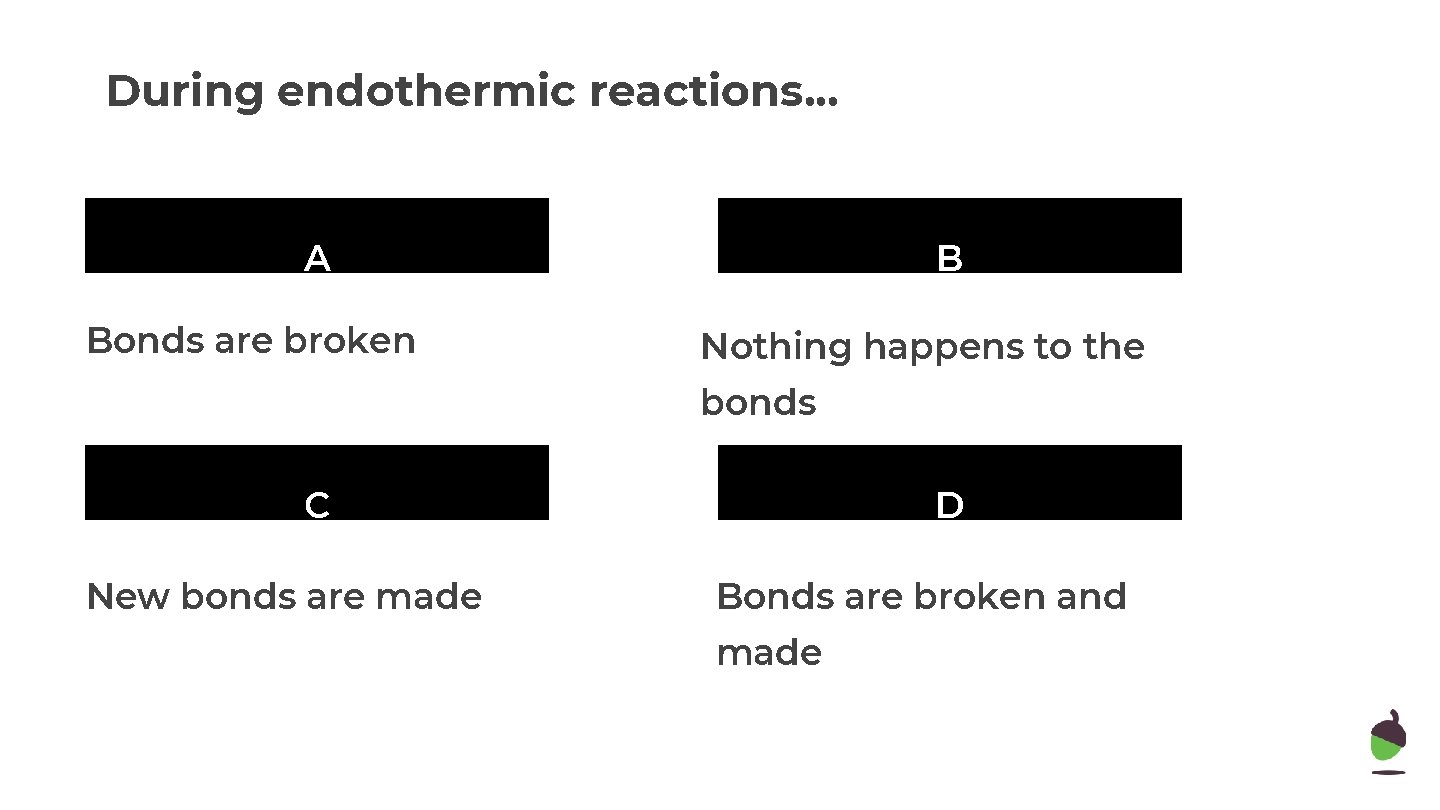
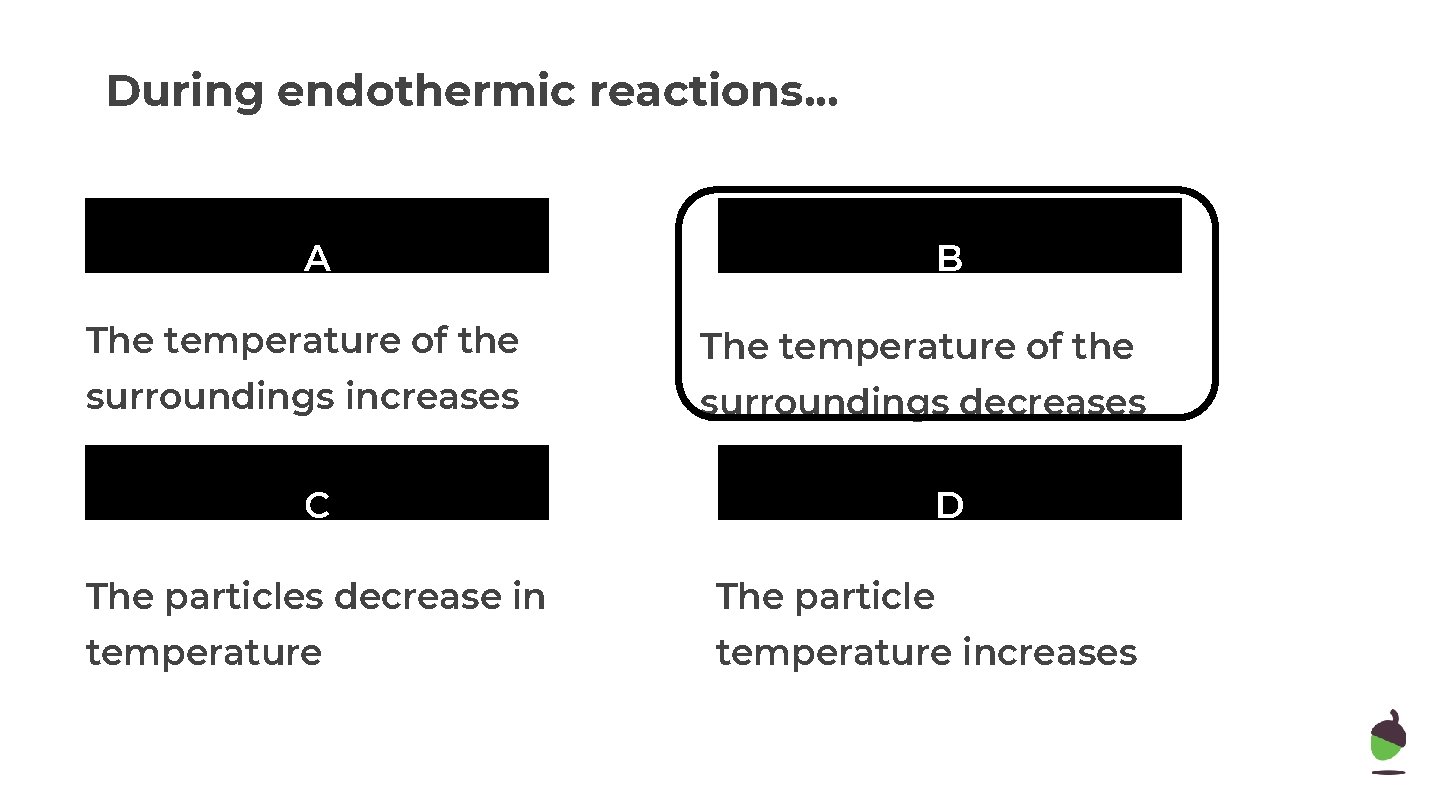
During endothermic reactions. . . A B The temperature of the surroundings increases surroundings decreases C D The particles decrease in The particle temperature increases

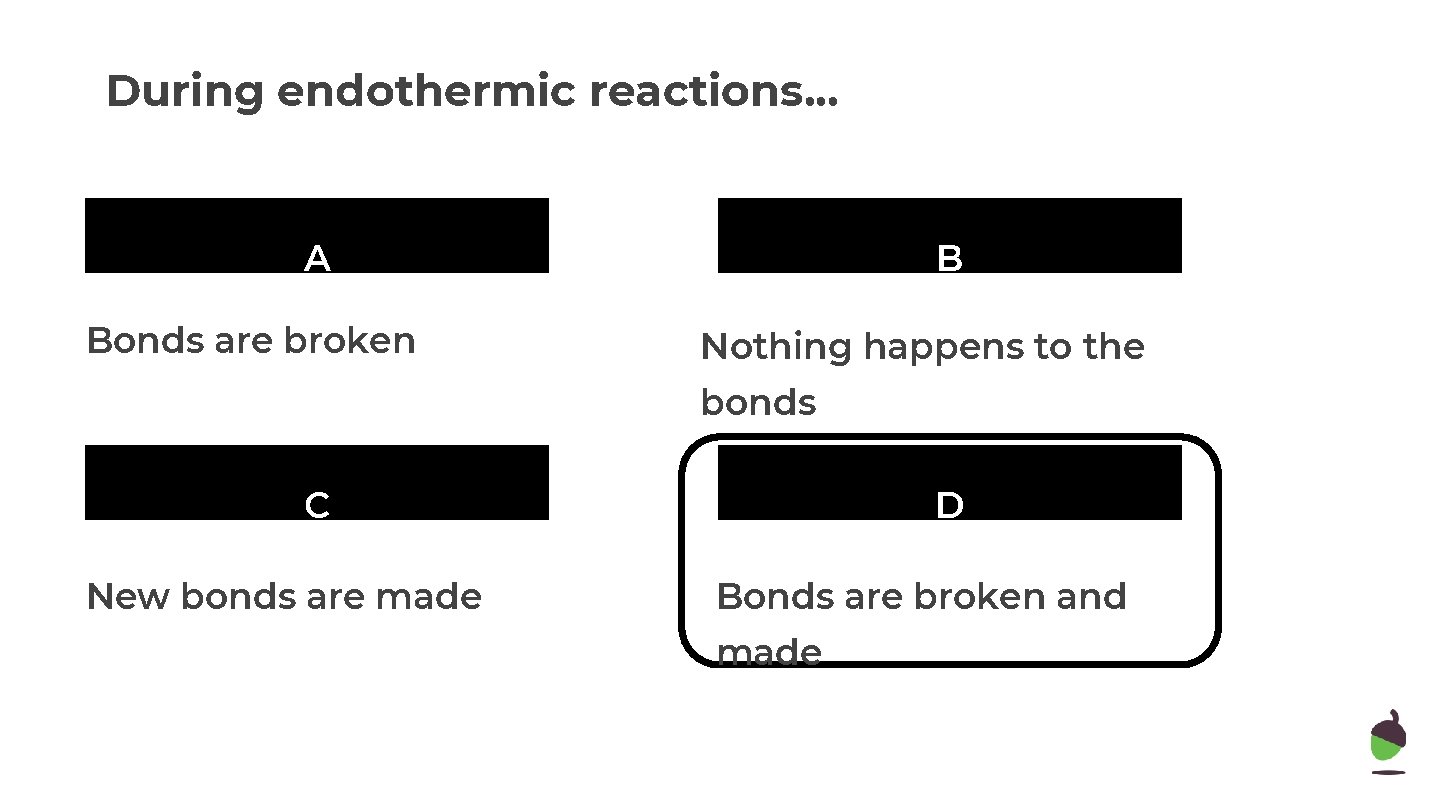
During endothermic reactions. . . A Bonds are broken B Nothing happens to the bonds C New bonds are made D Bonds are broken and made

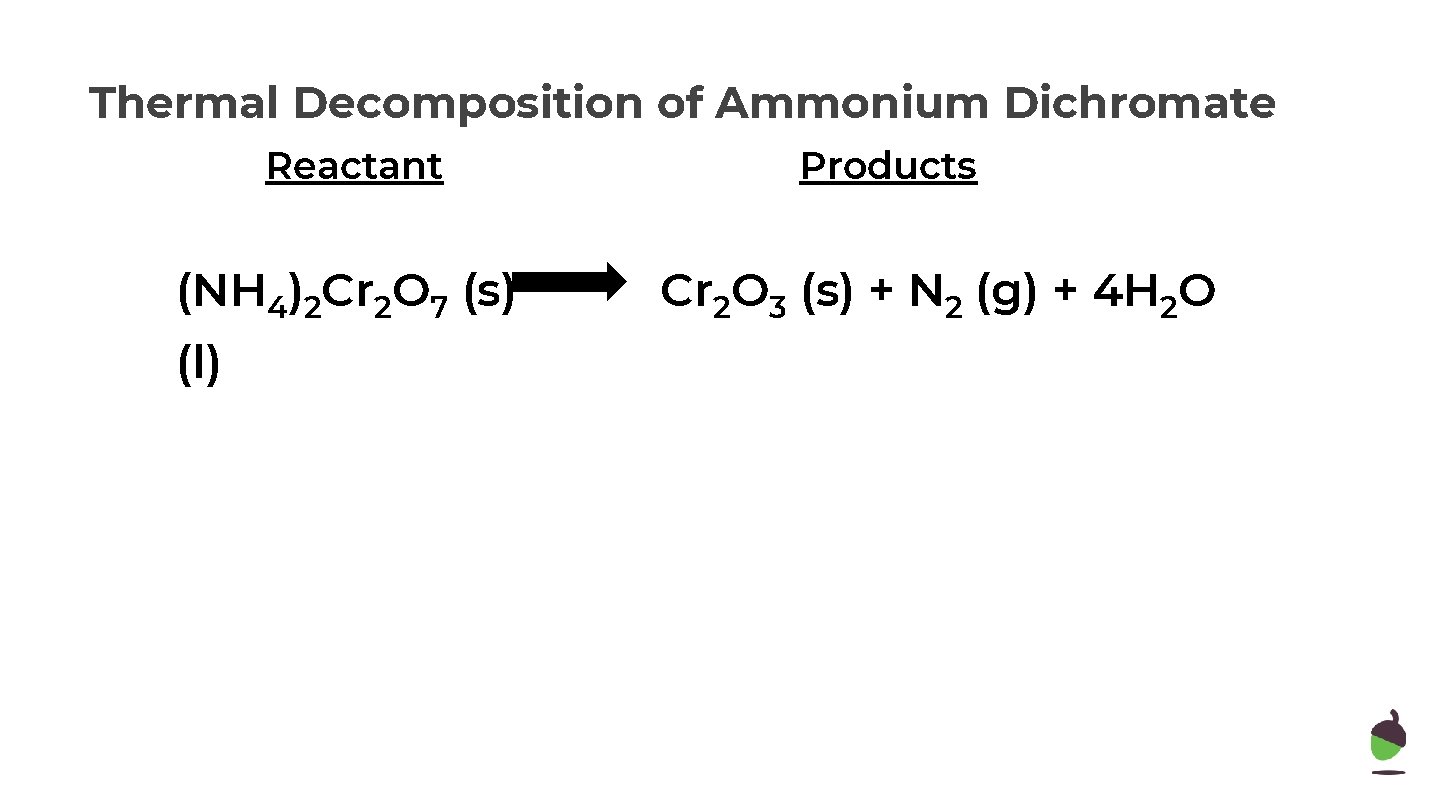
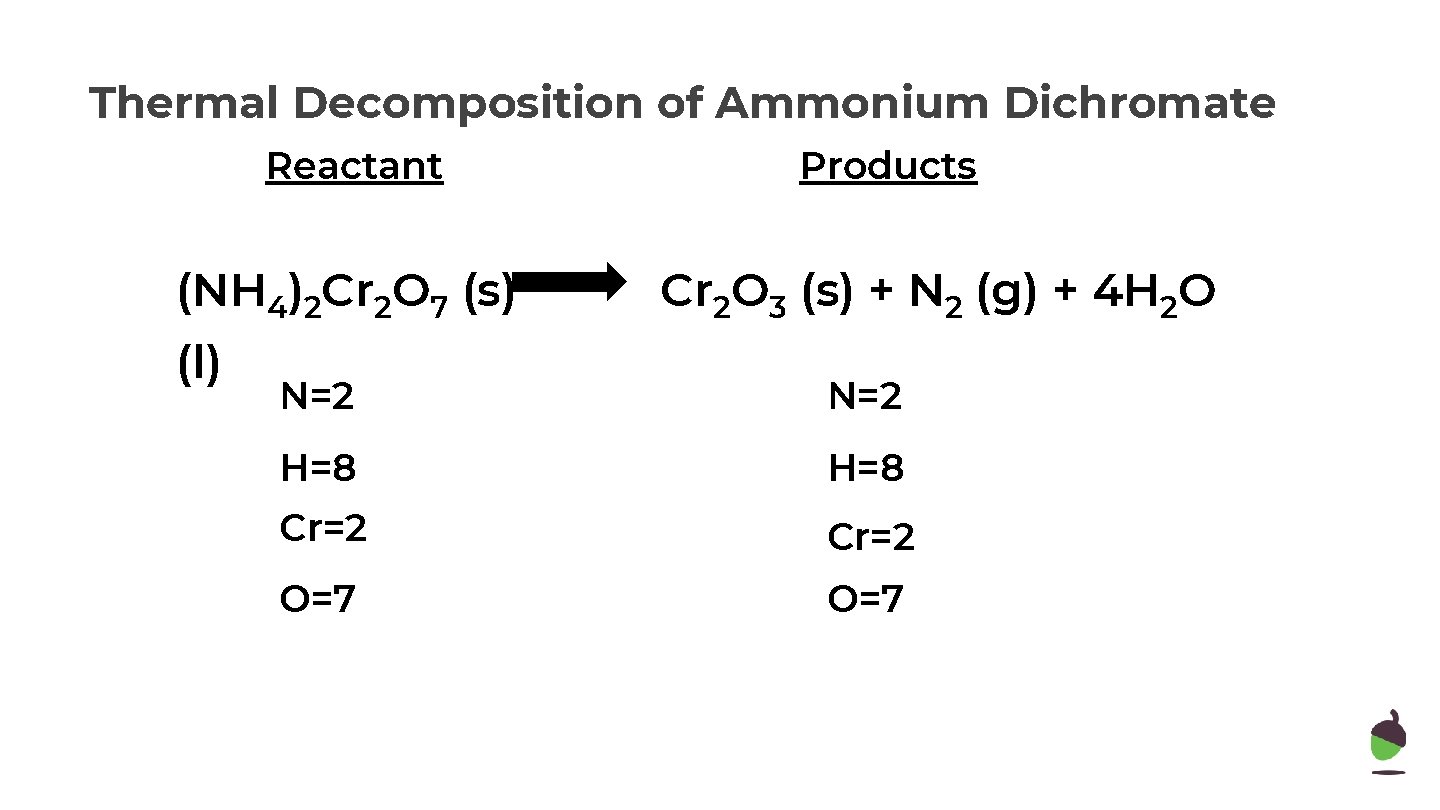
Thermal Decomposition of Ammonium Dichromate Reactant (NH 4)2 Cr 2 O 7 (s) (l) Products Cr 2 O 3 (s) + N 2 (g) + 4 H 2 O

Naming products of thermal decomposition Copper → Copper + Carbon dioxide carbonate oxide Cu. CO 3 1. __________ → Cu. O + CO 2 → Magnesium + Carbon dioxide 1. Zinc → Zinc carbonate ____ + ___________ Symbols Mg= Magnesium Zn= Zinc O= Oxygen C= Carbon

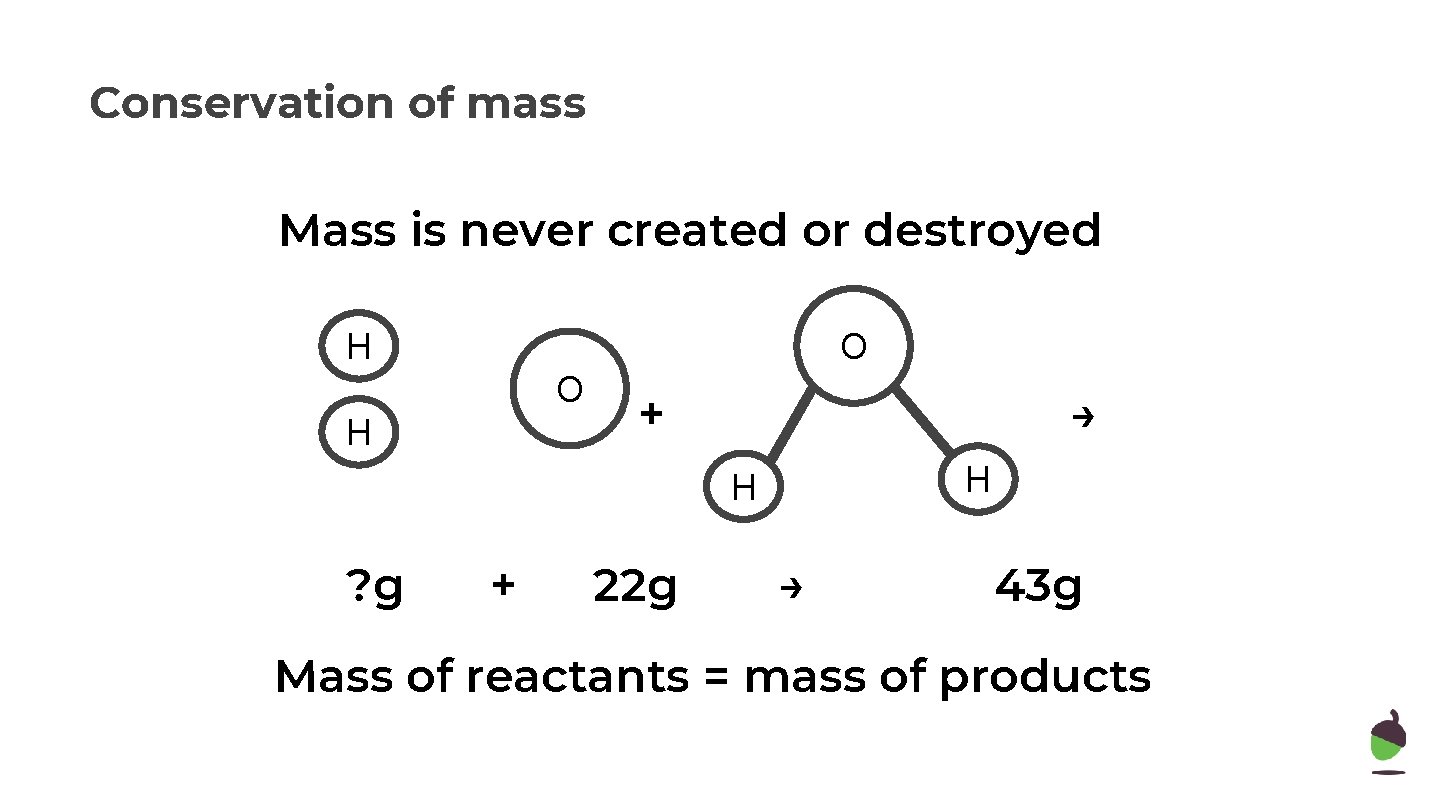
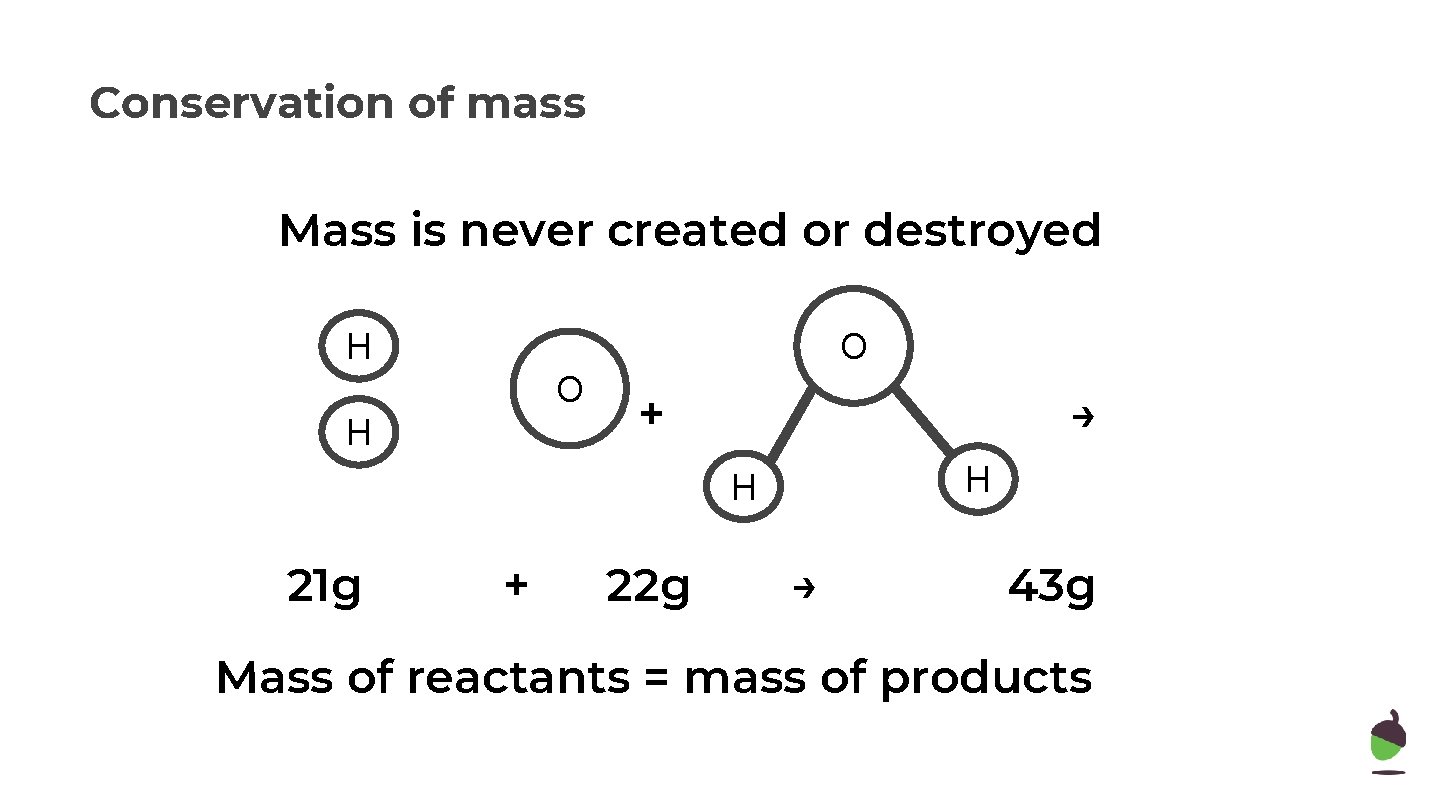
Conservation of mass Mass is never created or destroyed H O O H + → H H ? g + 22 g → 43 g Mass of reactants = mass of products

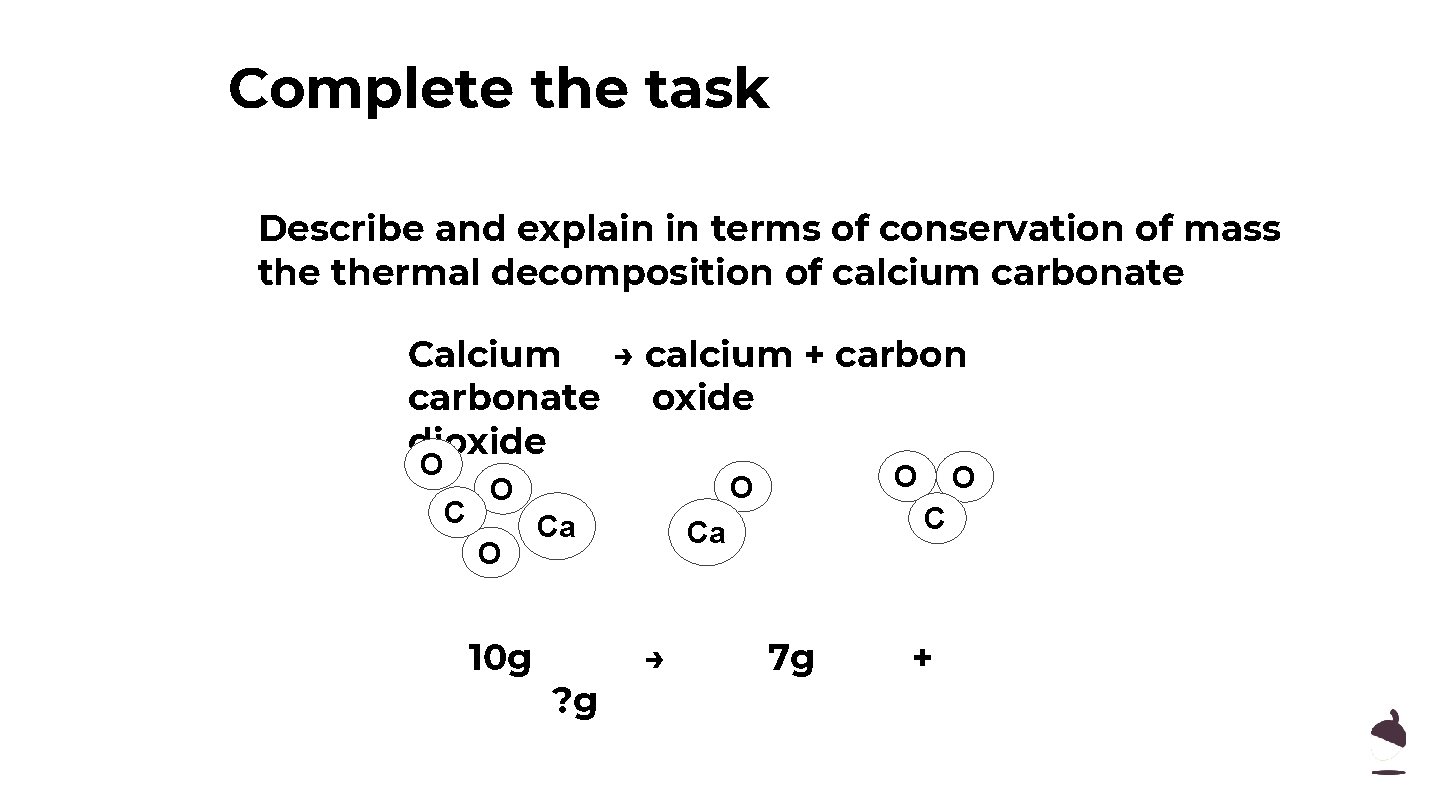
Complete the task Describe and explain in terms of conservation of mass thermal decomposition of calcium carbonate Calcium → calcium + carbonate oxide dioxide O C O 10 g O O O Ca ? g C Ca → O 7 g +

Answers 8

During endothermic reactions. . . A B The temperature of the surroundings increases surroundings decreases C D The particles decrease in The particle temperature increases

During endothermic reactions. . . A Bonds are broken B Nothing happens to the bonds C New bonds are made D Bonds are broken and made

Thermal Decomposition of Ammonium Dichromate Reactant (NH 4)2 Cr 2 O 7 (s) (l) Products Cr 2 O 3 (s) + N 2 (g) + 4 H 2 O N=2 H=8 Cr=2 O=7

Naming products of thermal decomposition 1. Magnesium → Magnesium + carbon dioxide carbonate oxide Mg. CO 3 1. Zinc → → carbonate Zn. CO 3 Mg. O Zinc + + carbon oxide → Zn. O CO 2 dioxide + CO 2

Conservation of mass Mass is never created or destroyed H O O H + → H H 21 g + 22 g → 43 g Mass of reactants = mass of products

Explain in terms of conservation of mass thermal decomposition of calcium carbonate ● When calcium carbonate decomposes, it breaks down and produces calcium oxide and carbon dioxide. ● The mass of product = the mass of reactants. ● mass of Ca. CO 3 = 10 g, mass of Ca. O+CO 2= 10 g. ● 10 g-7 g (Ca. O)= 3 g (CO 2)