Lesson 1 Pharmaceutical Products and Drug Action D













































- Slides: 59


Lesson 1: Pharmaceutical Products and Drug Action D. 1 Monday, March 7 th

Understandings • In animal studies, therapeutic index is the lethal dose of a drug for 50% of the population (LD 50) divided by the minimum effective dose for 50% of the population (ED 50). • In humans, therapeutic index is the toxic dose of a drug for 50% of the population (TD 50) divided by the minimum effective dose for 50% of the population (ED 50). • For ethical and economic reasons, animal and human tests of drugs (for LD 50/ED 50 and TD 50/ED 50 respectively) should be kept to a minimum. • The therapeutic window is the range of dosages between the minimum amount of the drug that produce the desired effect and a medically unacceptable adverse effect. • Dosage, tolerance, addiction, and side-effects are considerations of drug administration. • Bioavailability is the fraction of the administered dosage that reaches the target part of the human body. • The main steps in the development of synthetic drugs include identifying the need and structure, synthesis, yield, and extraction. • Drug–receptor interactions are based on the structure of the drug and the site of activity.

Applications and Skills • Discussion of experimental foundations for therapeutic index and therapeutic window through both animal and human studies. • Discussion of drug administration methods. • Comparison of how functional groups, polarity, and medicinal administration can affect bioavailability.

Your Body – Natural Defenses • Metabolism is the sum of all your body’s natural processes • Your body has a number of ways to maintain homeostasis and ward off illness and infection, recover from injury, and staying in working order. However, sometimes these systems are not enough. • Medicinal chemistry is a study into how we supplement our bodies natural defenses when things go wrong

Terms • We consider microorganisms that can cause us illness as invaders and our body’s immune defenses as lines of defense • Often, what we consider sickness is really a byproduct of our body’s line of defense (i. e. a fever is a way to kill off invaders whose proteins change and denature at higher temperatures)

Drugs A drug is any substance that, when applied to or introduced into a living organism, brings about a change in biological function through its chemical action. The change in biological function may be for the better – in the treatment of diseases – or for the worse – poisons that cause toxicity.

Drug Action • A drug produces an effect on the body by interacting with a particular target molecule. This target molecule is usually a protein such as an enzyme or receptor, but may be another molecule such as DNA or a lipid in a cell membrane.

Drugs can be: • relatively crude preparations, obtained by extracting plant or animal materials • pure compounds isolated from natural sources • semi-synthetic compounds, produced by chemical modification of pure natural compounds • synthetic compounds.

Medicine • A medicine is something that treats, prevents or alleviates the symptoms of disease – they have a therapeutic action. • Medicine are usually compound preparations, which means that they contain a number of ingredients – the active drug itself plus nonactive substances that improve the preparation in some way such as taste, consistency or administration of the drug.

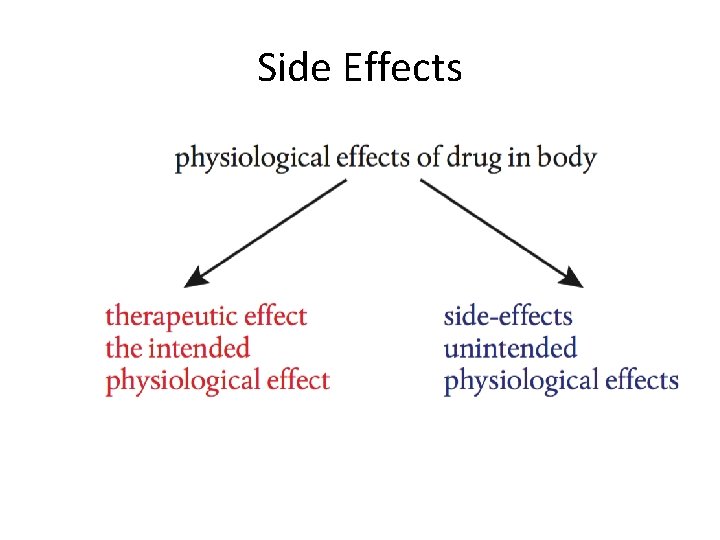
Therapeutic Effect • The therapeutic effect is the beneficial effect of a medical treatment. • The placebo effect is a phenomenon where a patient believing they are being given a medicine experience the benefits even when not taking the medicine; this is the reason when developing new drugs they must be tested against a placedo

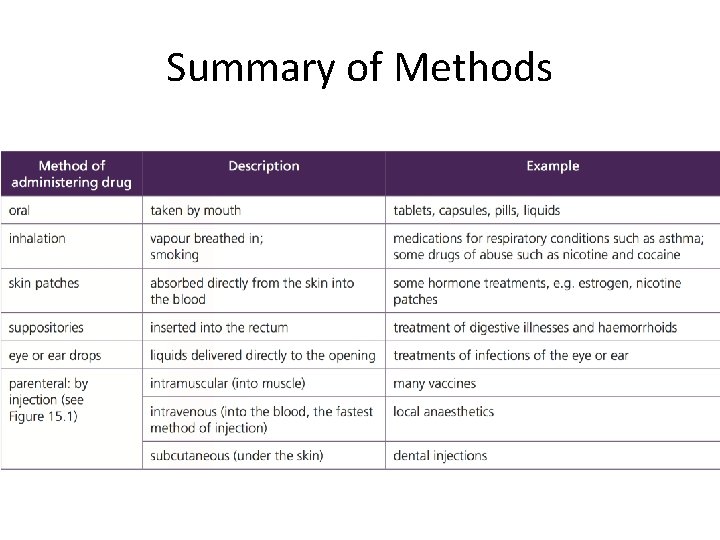
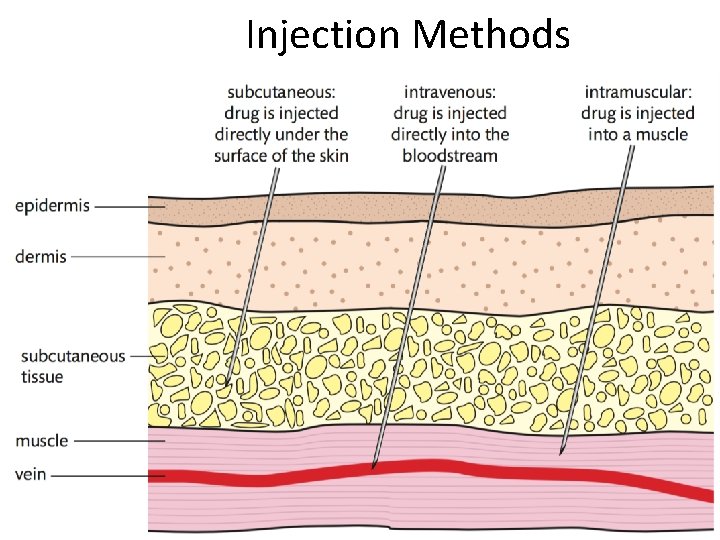
Drug Administration • Drugs can be administered in a number of way 1. Oral 2. Rectal 3. Injected a. Intravenous b. Intramuscular c. Subcutaneous 4. Pulmonary 5. Topical

Summary of Methods

Injection Methods

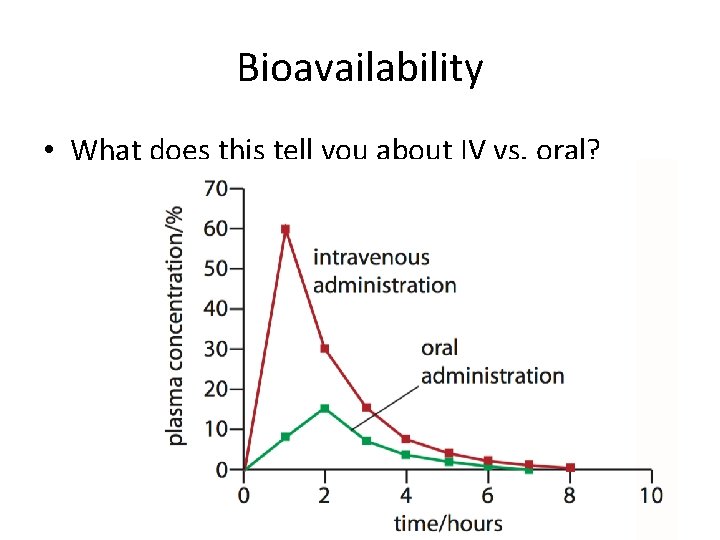
Bioavailability of Drugs • Regardless of how the drug is administered, not all of the drug will make its way to the intended target • Bioavailability is the fraction of the administered dosage that reaches the bloodstream. • Bioavailability is an important consideration when calculating how much of a drug to administer, known as the dosage.

Bioavailability • What does this tell you about IV vs. oral?

Oral Bioavailability • Various factors affect the fraction of a drug dose that survives to reach the general circulation – for instance, the formulation of the tablets, their solubility, how easily it is absorbed through the intestinal wall, and the susceptibility to being broken down by enzymes in the gut and liver all affect bioavailability.

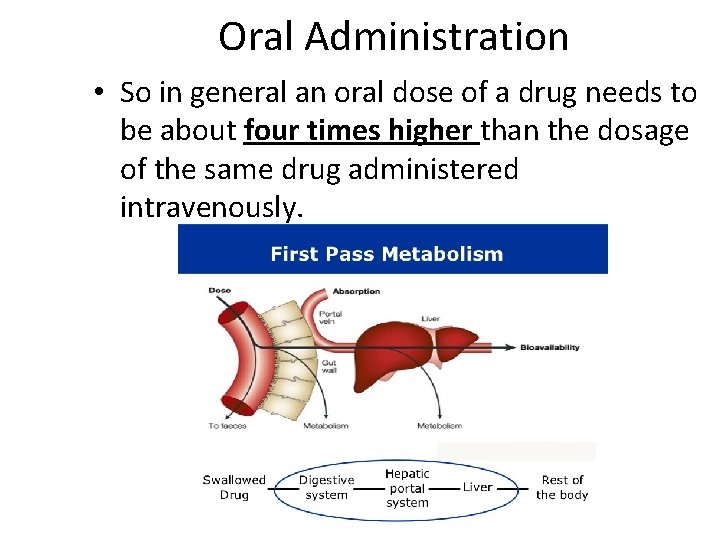
First Pass Effect • The relatively low bioavailability of a drug taken orally is known as the first-pass effect, and means that as little as 20– 40% of an orally ingested drug may reach the bloodstream. • This is because after swallowing, these drugs pass into the digestive system where biological catalysts known as enzymes may alter them chemically. Once absorbed from the digestive system, they are passed in the blood to the liver where further metabolic breakdown reactions occur.

Oral Administration • So in general an oral dose of a drug needs to be about four times higher than the dosage of the same drug administered intravenously.

Exam Tip • When asked to define bioavailability in the exam you should define it according to the syllabus definition: the fraction of the administered dosage that reaches the target part of the human body.

Solubility • Water solubility is important for circulation in the aqueous solution in the blood, but lipid solubility helps in the passage of the drug through membranes during absorption. • Only individual molecules of a drug can pass through the wall of the intestine, therefore it is essential that a drug is soluble in water – the medium of the gastrointestinal tract. • Several factors relating to the structure of drug molecules affect solubility – the presence of polar groups (e. g lots of OH groups) and/or functional groups that can undergo ionization (e. g. COOH and NH 2).

Functional Groups • Functional groups in the drug can also in influence bioavailability, particularly acid–base groups. • The p. Ka and p. Kb values of these groups in the molecule will determine the charges carried on the drug at different p. H values, and therefore its reactivity and solubility in different parts of the body.

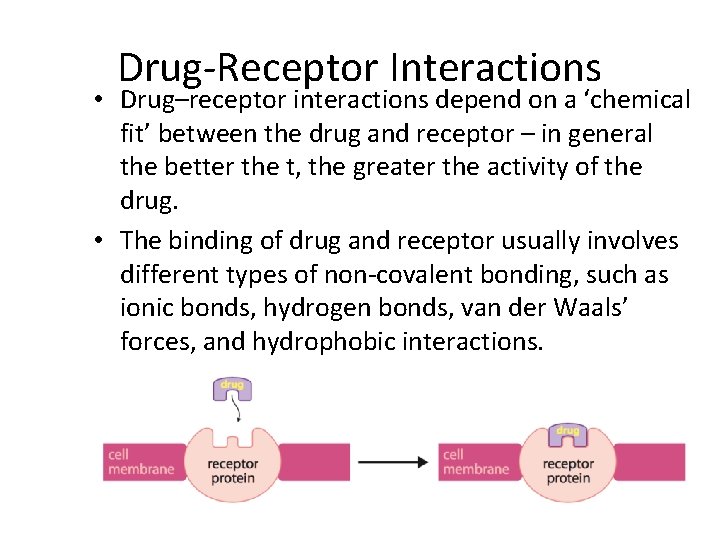
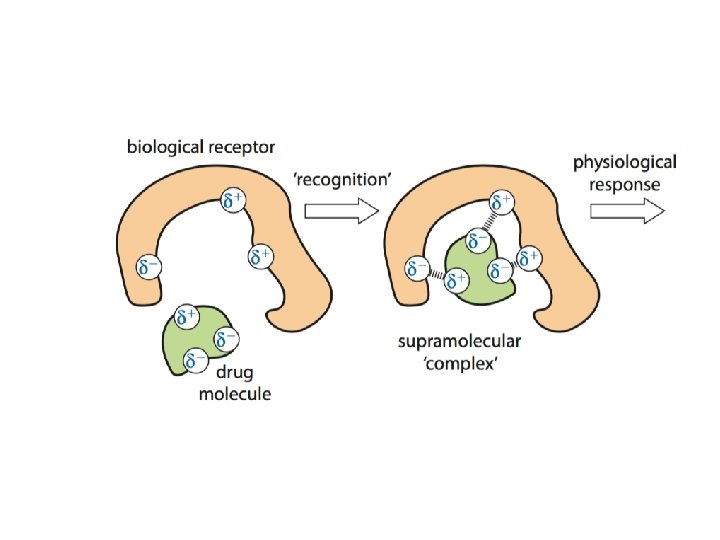
Drug-Receptor Interactions • A drug can act in various ways on receptors, for example: – it can bind to a cell-membrane protein receptor, mimicking the effect of the normal molecule that binds and cause a series of reactions in a cell – i. e. it turns a particular process in the cell on/off; in this case the drug is called a receptor agonist – it can bind to a cell-membrane protein receptor so that the normal messenger molecule can’t – it prevents a particular response from a cell; in this case the drug is called a receptor antagonist. • A drug, wherever possible, should be specific and bind to only one particular type of receptor

Drug-Receptor Interactions • Drug–receptor interactions depend on a ‘chemical fit’ between the drug and receptor – in general the better the t, the greater the activity of the drug. • The binding of drug and receptor usually involves different types of non-covalent bonding, such as ionic bonds, hydrogen bonds, van der Waals’ forces, and hydrophobic interactions.


Side Effects • Side-effects are defined as physiological effects which are not intended, and they vary greatly from one drug to another, and with the same drug in different people. • Sometimes side-effects may be beneficial, such as the fact that aspirin, taken for pain relief, helps protect against heart disease. Other times the side-effects may be relatively benign, such as causing drowsiness, nausea, or constipation. But of greater concern are side-effects which are much more adverse, such as causing damage to organs.

Side Effects

Tolerance and Addiction • When a person is given repeated doses of a drug, tolerance can develop, which means a reduced response to the drug for the same dose. So higher doses are needed to produce the same effect, and this increases the chances of toxic side-effects.

Addiction • A related but different condition is dependence or addiction. This occurs when a patient becomes dependent on the drug in order to feel normal, and suffers from withdrawal symptoms if the drug is not taken. • Symptoms can be mild, such as headaches suffered on withdrawal from dependence on caffeine, or serious if the drug is toxic or shows tolerance, such as opiates, alcohol, and barbiturates.

Drug Development Tuesday, March 8

Drug Dosing Regimes • The dosing regime for a drug refers to the specific quantity of drug to be taken at one time, and the frequency of administration. • Calculations of dosage must take bioavailability into account, as well as possible side-effects and potential problems of tolerance and addiction. • Determining appropriate dosage is usually quite dif cult as there are so many variables involved – for example the age, sex, and weight of the patient, as well as factors such as diet and environment. Interactions with other drugs must also be considered. • Ideally the dosage should result in constant levels of the drug in the blood, but this is almost impossible to achieve other than by a continuous, intravenous drip.

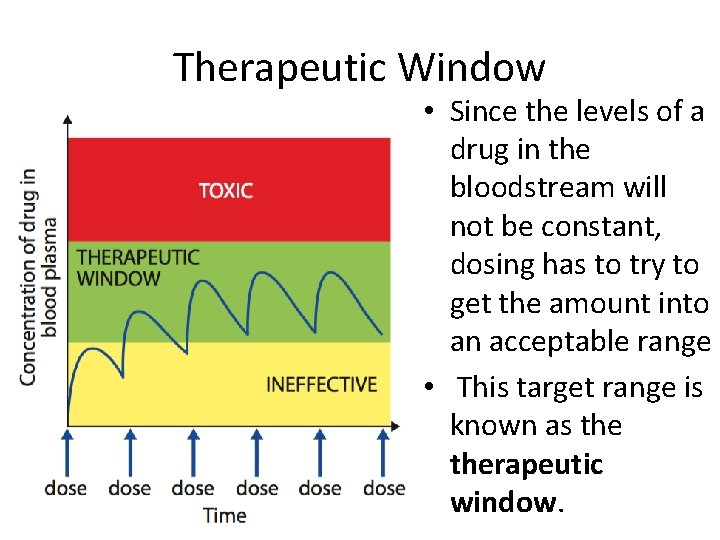
Therapeutic Window • Since the levels of a drug in the bloodstream will not be constant, dosing has to try to get the amount into an acceptable range • This target range is known as therapeutic window.

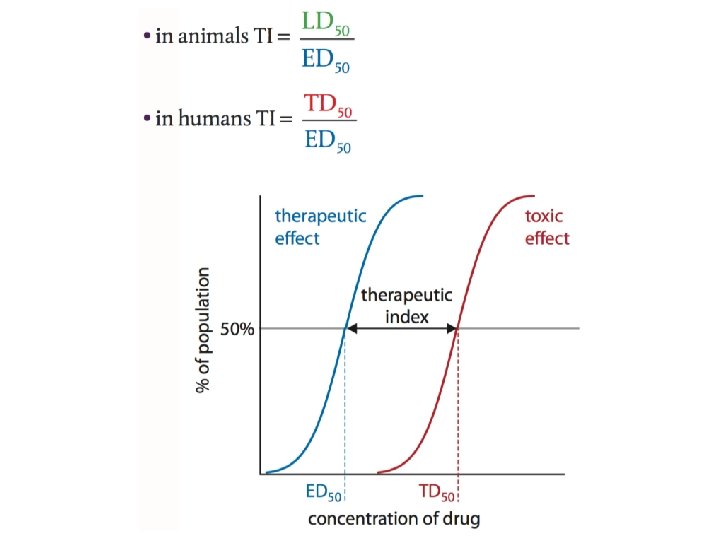
Therapeutic Index • The therapeutic window can be quanti ed as therapeutic index (TI). This is the ratio of the dose that produces toxicity to the dose that produces a clinically effective response in a population. The relevant terms in the equation are: 1. The minimum effective dose, ED 50, is the dose that produces therapeutic effect in 50% of the population. 2. The lethal dose, LD 50, is the dose that is lethal to 50% of the population. This is used in animal trials. 3. The toxic dose, TD 50, is the dose that is toxic to 50% of the population. This is used in human studies.


Animal vs. Human Studies • In animal studies lethal doses are determined; however, in human trials the upper limit is the toxic dose. • Measuring an LD 50 can result in the deaths of a large number of animals – many countries have phased out this test in favor of others in which few or no animal deaths result. • Both have benefits and drawbacks. The situation is way more complicated than this simple formula!

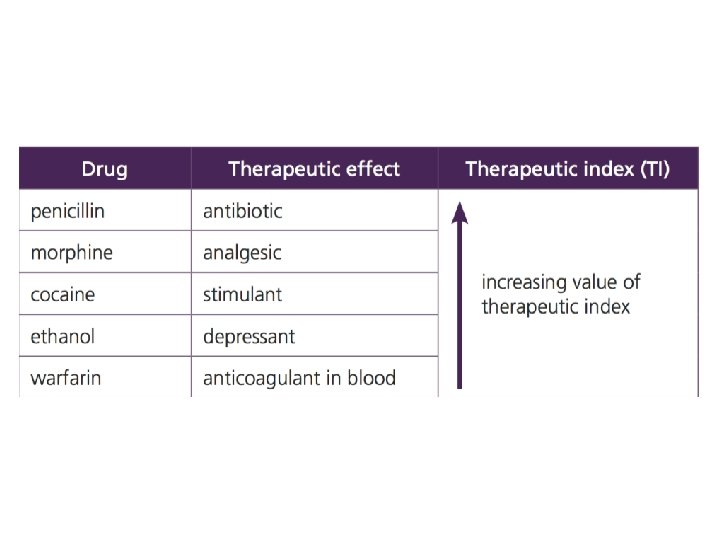
What does TI mean? • If a drug has a high (or wide) therapeutic index, this means that there is a large difference between the dose of the drug that causes a therapeutic effect compared with the dose that causes a toxic effect. • For example, if a TI is 100 then TD 50 is 100 times larger than ED 50, so it would require a 100 -fold increase in therapeutic dose to cause a toxic effect in 50% of the population; a high therapeutic index is therefore a desirable property of a drug.


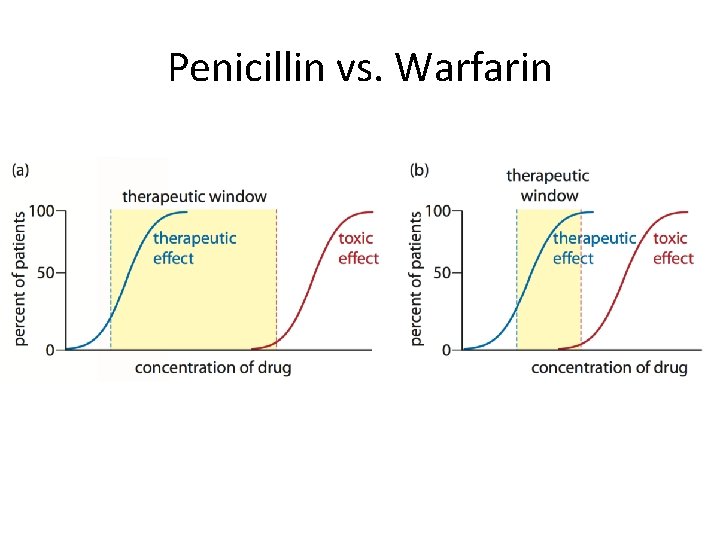
Penicillin vs. Warfarin

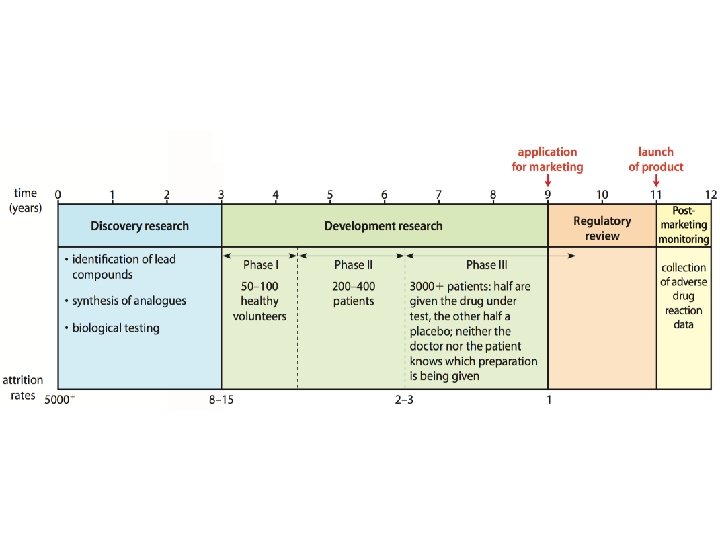
Drug Development • Extremely expensive • Industry is very selective about which diseases/conditions to go after; want to make money off of investment • Downside to universal healthcare? • The average time for development of a drug from its first identification to the market is about 10– 12 years. – Patent issues

Rational Drug Design • Knowledge of drug–receptor interactions has revolutionized the process by which new drugs are developed. • Most research now focuses on identifying a suitable molecular target in the body and designing a drug to interact with it. • This approach, known as rational drug design, is very different from the time when pharmaceutical companies worked mostly on a ‘trial and error’ basis, starting with a natural remedy and trying to improve on nature with no real insight into the mechanism of the action of the drug at the molecular level.

Lead Compounds • Once a target molecule has been identified, the next step is to find a lead compound – one that shows the desired pharmaceutical activity which will be used as a start for the drug design and development process. • The effectiveness of the lead compound is optimized by synthesizing and testing many chemically related compounds known as analogues. A process called combinatorial chemistry enables the production and testing of vast numbers of candidate medicines in a very short time.

Compound Libraries • Electronic databases that contain molecules which have been isolated or synthesized and tested by pharmaceutical companies for possible pharmaceutical properties • Pharmaceutical companies use such libraries to identify ‘lead’ compound for a particular ‘target’ molecule such as an enzyme, DNA or a receptor.

Combinatorial Chemistry • https: //www. youtube. com/watch? v=MVgs. X 7 PM 4 F 4

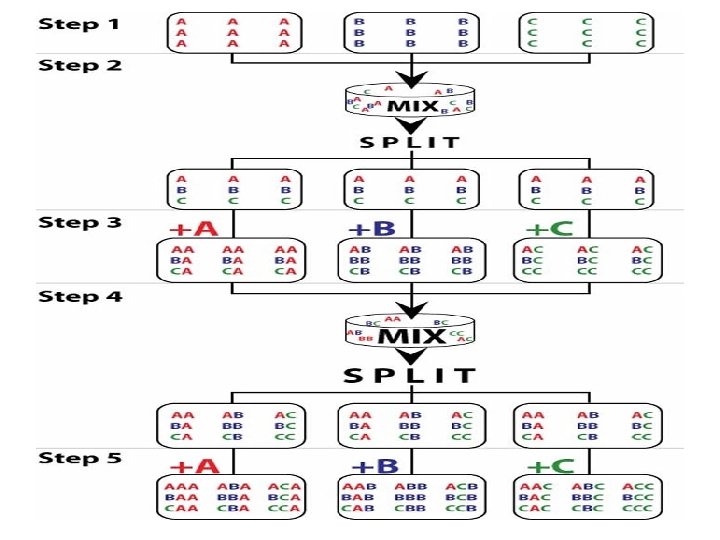
Combinatorial Chemistry • Involves simultaneous chemical synthesis • A large number of different but structurally related compounds (all possible combinations) from a small number of reactant molecules which are reacted with a variety of reactants, • Uses “mix-and-split” technique and resin beads • Screen each product for its biological activity, resulting in a “combinatorial library”. • All is automated and uses computers/robots

Solid-Phase Chemistry • A technique used in combinatorial chemistry • Synthesizes large volume of compounds • Reactions take place on the surface of resin beads • Each type of reactant molecule is bonded covalently onto a very small resin bead • Uses mix and split process


Advantage Of Solid Phase • When synthesis reactions are complete, the products are removed easily from the beads by filtering off the beads and washing them. • After that the products are tested “in vitro” and “in vivo” to find out their biological activity.

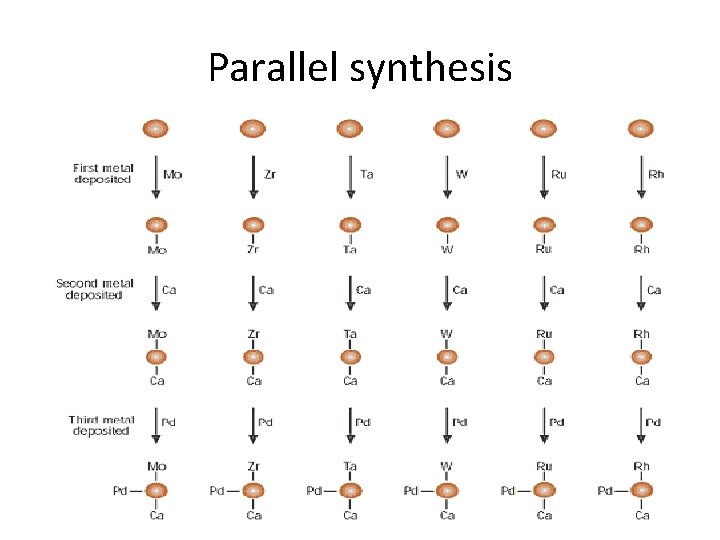
Parallel Chemistry • Parallel chemistry or parallel synthesis involves the synthesis of a smaller but selected group of compounds with a different compound in each reaction vessels. • In most combinatorial techniques the compounds are mixed and need to be separated; not necessary in parallel synthesis as multiple experiments run in parallel.

Parallel synthesis

Differences Combinatorial Synthesis Parallel Synthesis • Generates large, more diverse libraries “combinatorial library”. • Produces a ‘mixture’ of compounds in same reaction vessel. • Small focused libraries • Reactions performed in different reaction vessels • Produces a ‘single’ product in each reaction vessel.

Next Steps – Clinical Testing • Next, the pharmaceutical company must test their drugs to determine safety, efficacy, and dosing regimens • To do this, they must start clinical trials


Drug Development • https: //www. youtube. com/watch? v=wv. Dv. AE mq-c. M • https: //www. youtube. com/watch? v=v. YBtl. It. AT 3 c

Phase I • The first phase (known as Phase I) is carried out on a small number of healthy volunteers (usually fewer than 100) and its purpose is to nd the dose range of the drug that gives a therapeutic effect and also to identify any side effects.

Phase II • If the drug passes Phase I, it then enters Phase II clinical trials where it is tested on a small number of volunteer patients who have the disease or condition on which the drug acts. Phase II establishes whether or not the drug is effective in these patients and also identifies any side e ects. If deemed safe and effective, the drug then enters Phase III.

Phase III • In Phase III clinical trials, the drug is tested on a much larger group of volunteer patients. This phase confirms the effectiveness of the drug in the larger group and compares its activity with existing drug treatments or placebos.

Thalidomide • https: //www. youtube. com/watch? v=41 n 3 m. D o. Vbvk

Questions

Answers