Lenalidomidemelphalandexamethasone in newly diagnosed patients with immunoglobulin light

Lenalidomide/melphalan/dexamethasone in newly diagnosed patients with immunoglobulin light chain amyloidosis: results of a prospective phase 2 study with long-term follow up by Ute Hegenbart, Tilmann Bochtler, Axel Benner, Natalia Becker, Christoph Kimmich, Arnt V. Kristen, Jörg Beimler, Ernst Hund, Markus Zorn, Anja Freiberger, Marianne Gawlik, Hartmut Goldschmidt, Dirk Hose, Anna Jauch, Anthony D. Ho, and Stefan O. Schönland haematol Volume 102(8): 1424 -1431 July 31, 2017 © 2017 by Ferrata Storti Foundation
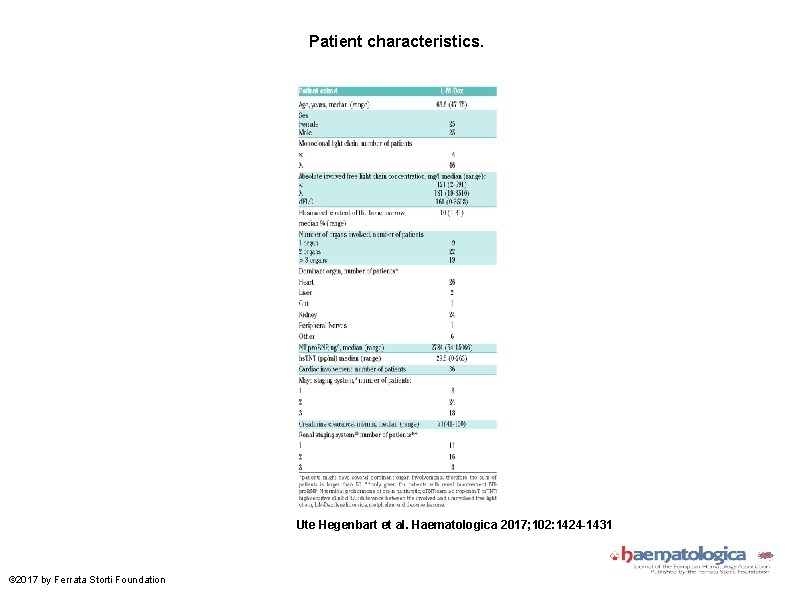
Patient characteristics. Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
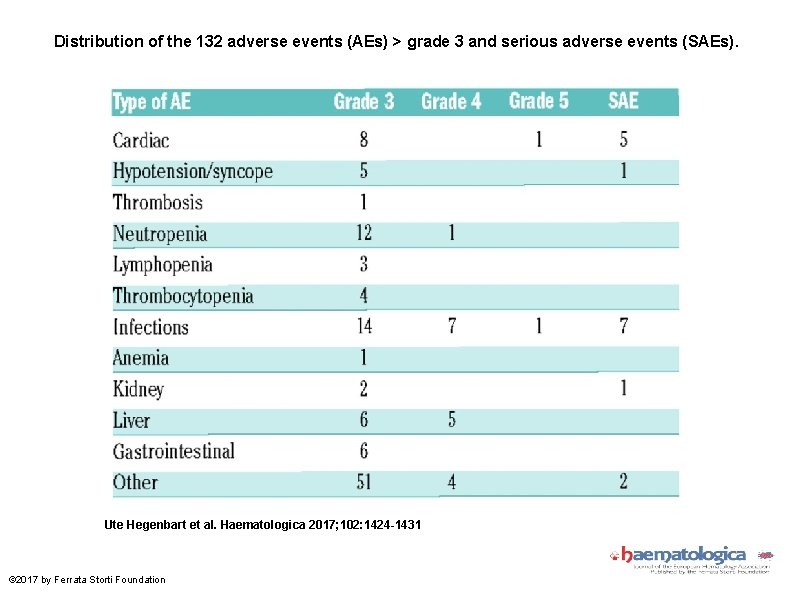
Distribution of the 132 adverse events (AEs) > grade 3 and serious adverse events (SAEs). Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
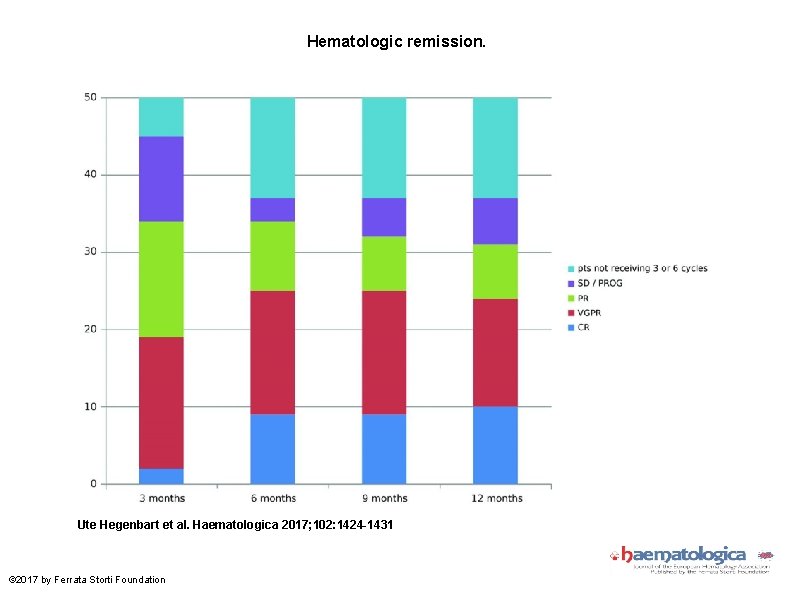
Hematologic remission. Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
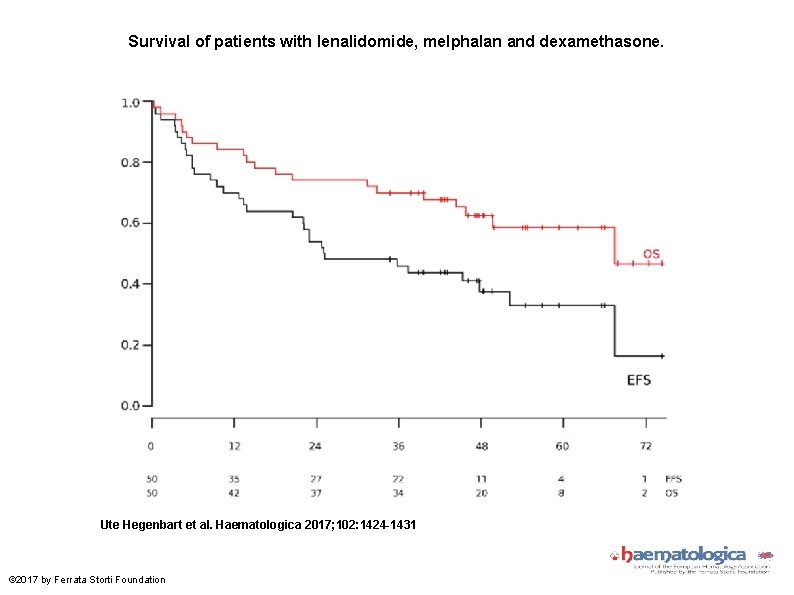
Survival of patients with lenalidomide, melphalan and dexamethasone. Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
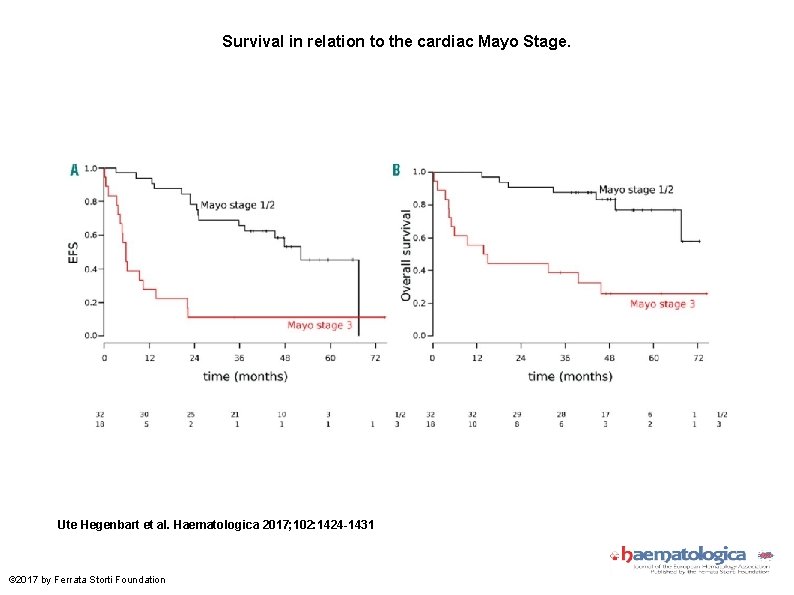
Survival in relation to the cardiac Mayo Stage. Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
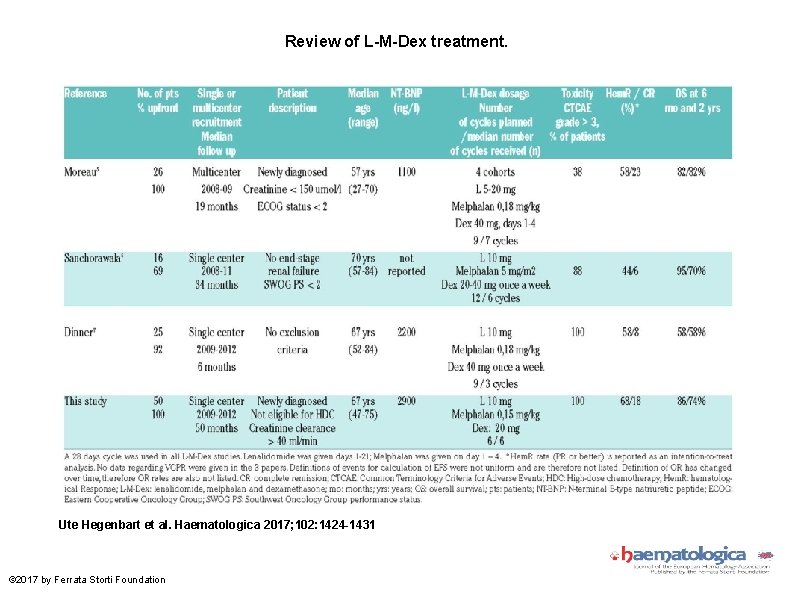
Review of L-M-Dex treatment. Ute Hegenbart et al. Haematologica 2017; 102: 1424 -1431 © 2017 by Ferrata Storti Foundation
- Slides: 7