Leech Heart Half Center Oscillator Control of Burst

Leech Heart Half. Center Oscillator: Control of Burst Duration by Low. Voltage Activated Calcium Current Olypher A, et al. (2006); Hill J, et al. (2001) Math 723: Mathematical Neuroscience Khaldoun Hamade June 7, 2007

Introduction n Half-center oscillators, also called central pattern generators (CPG), drive rhythmic behaviors n Burst Period = Burst Duration + Interburst Interval n Burst period varies depending on functional demand of activity (ex. Heart rate, breathing rate, locomotion speed…) n Bursting is maintained by slowly inactivating inward currents
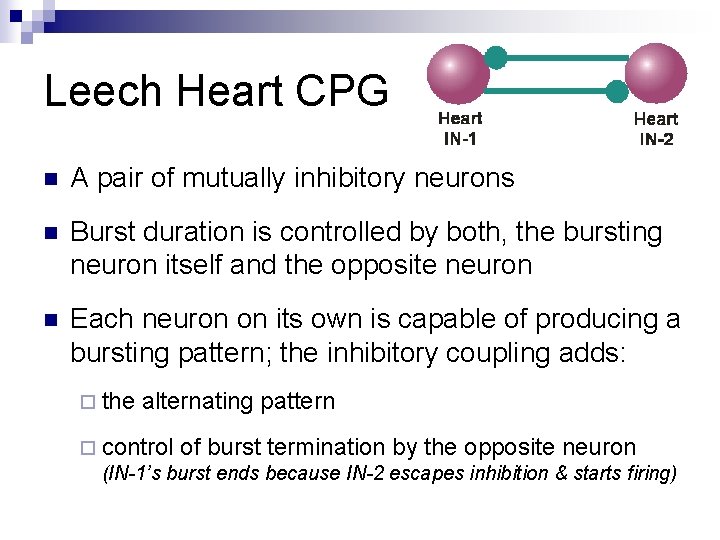
Leech Heart CPG n A pair of mutually inhibitory neurons n Burst duration is controlled by both, the bursting neuron itself and the opposite neuron n Each neuron on its own is capable of producing a bursting pattern; the inhibitory coupling adds: ¨ the alternating pattern ¨ control of burst termination by the opposite neuron (IN-1’s burst ends because IN-2 escapes inhibition & starts firing)
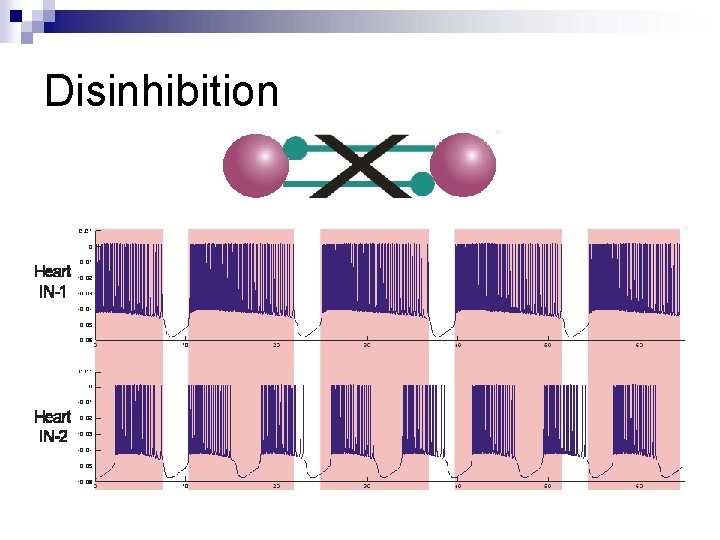
Disinhibition

Modeling of Leech Heart CPG (Hill et al. 2001) n n In Out One compartment model Ionic currents: ¨ INa : fast Na+ ¨ IP : persistent Na+ ¨ ICa. F : fast, low-threshold Ca 2+ ¨ ICa. S : slow, low-threshold Ca 2+ ¨ Ih : hyperpolarization-activated cation current ¨ IK 1 : ¨ IK 2 : delayed rectifier K+ persistent K+ ¨ IKA : fast, transient K+

Burst duration n The low-voltage-activated (LVA) calcium current: ¨ ICa. F (Fast): contributes to burst initiation ¨ ICa. S (Slow): determines burst duration n The inactivation time constant of ICa. S (τh, Ca. S) determines the spike frequency decay rate n The spike frequency determines the amount of inhibition the opposite neuron is receiving n Once the spike frequency (inhibition) falls below a certain value (f. Final) the opposite neuron escapes inhibition and begins to burst

Burst duration (Continued) n Spike frequency is maximum shortly after burst initiation, and declines to f. Final at the end of burst ¨ Low τh, Ca. S correspond to fast inactivation, fast frequency decay, and shorter bursts ¨ High τh, Ca. S correspond to slow inactivation, slow frequency decay, and longer bursts n *** Maximal value of gh can control the length of the interburst duration; a higher value allows the neuron to escape inhibition earlier, when it is still higher
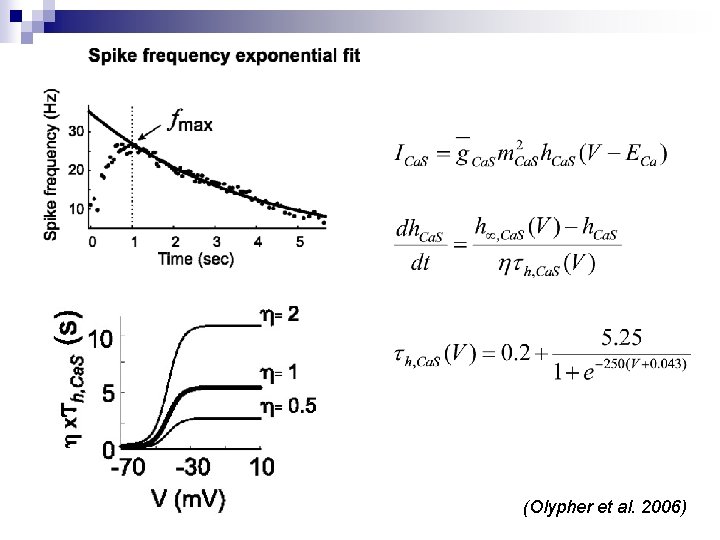
(Olypher et al. 2006)
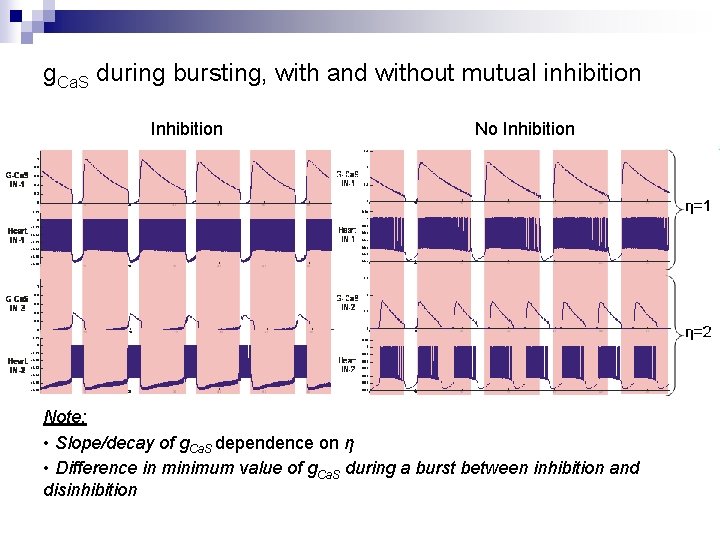
g. Ca. S during bursting, with and without mutual inhibition Inhibition Note: • Slope/decay of g. Ca. S dependence on η • Difference in minimum value of g. Ca. S during a burst between inhibition and disinhibition

Simulations n τh, Ca. S was varied unilaterally in m. HNv (constant in m. HNc, η=1) by varying the scaling factor η between 0. 25 & 4 n Period, burst duration, f. Final, and decay time constants of g. Ca. S and spike frequency were recorded

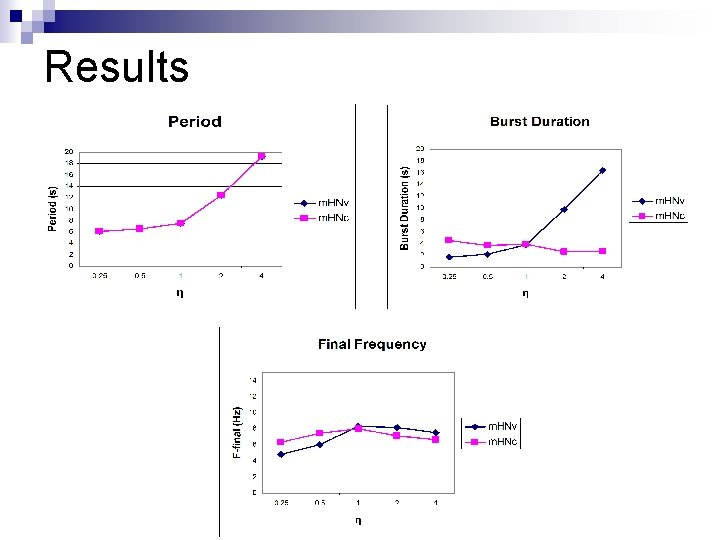
Results
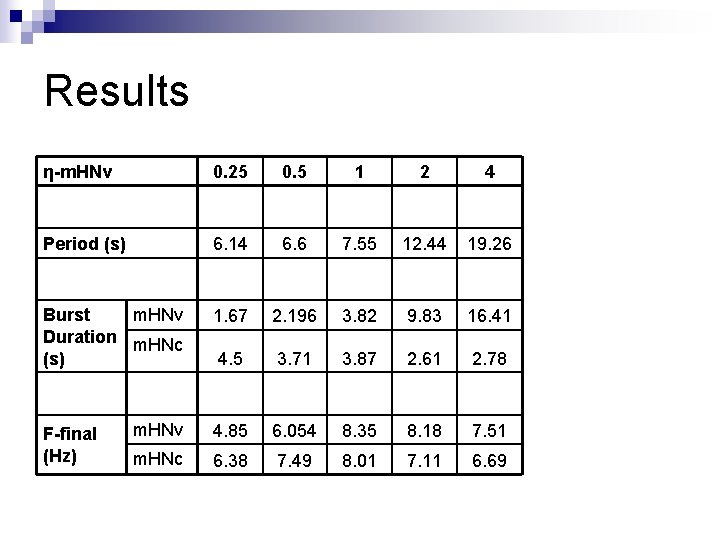
Results η-m. HNv 0. 25 0. 5 1 2 4 Period (s) 6. 14 6. 6 7. 55 12. 44 19. 26 Burst m. HNv Duration m. HNc (s) 1. 67 2. 196 3. 82 9. 83 16. 41 4. 5 3. 71 3. 87 2. 61 2. 78 m. HNv 4. 85 6. 054 8. 35 8. 18 7. 51 m. HNc 6. 38 7. 49 8. 01 7. 11 6. 69 F-final (Hz)
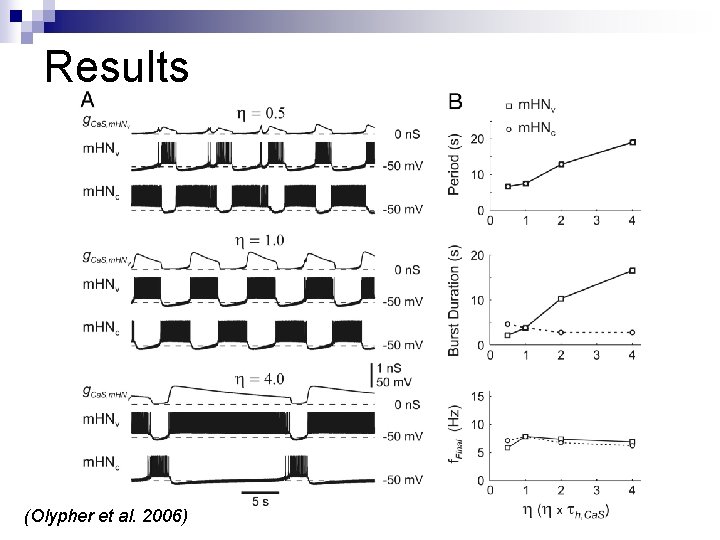
Results (Olypher et al. 2006)

Results n Decay time constants for ICas, g. Ca. S, & h. Ca. S were measured for a representative burst (η=1) n Decay time constants for ICas & g. Ca. S were found to be equal, while that of h. Ca. S was different; this was attributed to a voltage decline during the burst n Decay time constant of g. Ca. S was chosen as the benchmark n For each simulation the decay time constants for g. Ca. S & spike frequency were calculated and compared
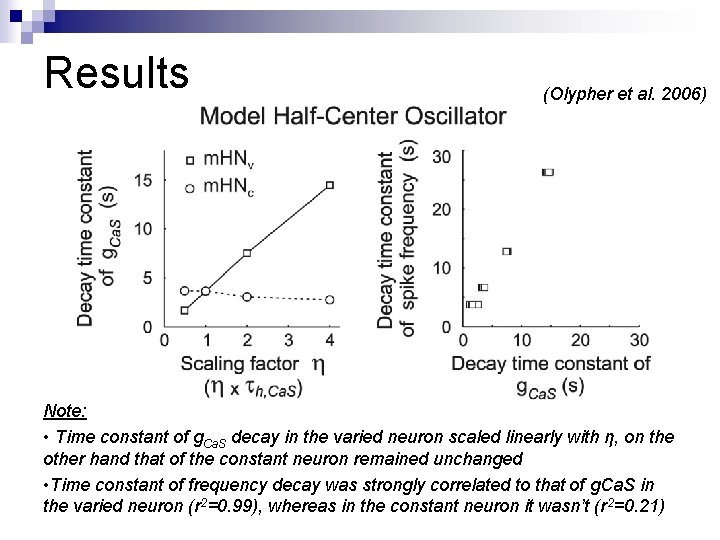
Results (Olypher et al. 2006) Note: • Time constant of g. Ca. S decay in the varied neuron scaled linearly with η, on the other hand that of the constant neuron remained unchanged • Time constant of frequency decay was strongly correlated to that of g. Ca. S in the varied neuron (r 2=0. 99), whereas in the constant neuron it wasn’t (r 2=0. 21)
Hybrid system n The hybrid system was constructed from a model neuron running in real time and a chemically isolated living heart neuron, with inhibitory coupling through a dynamic clamp n ICas time constant of inactivation was varied unilaterally, once in the model neuron and once in the living heart neuron n Results were similar to those obtained in the model system

Conclusion n Burst duration is controlled by inactivation of ICas n Scaling τh, Ca. S through η, scales the decay time constant of g. Ca. S & ICas equally n Decay of g. Ca. S is correlated with a parallel decay in spike frequency n f. Final n does not vary with (ηxτh, Ca. S) The escape point (from inhibition) of the opposite neuron is not affected by τh, Ca. S

Conclusion (Continued) n In living systems τh, Ca. S is not usually modulated n Varying maximal value of g. Ca. S modifies the burst duration, but also affect the output signal of the premotor CPG (strength and spike frequency) n Modulation of the maximal value of gh varies the period without affecting the signal output n gh is modulated in living systems n So why should we care about the affect of τh, Ca. S ?

Conclusion (Continued) n τh, Ca. S sets the baseline period of the CPG n τh, Ca. S sets the dynamic range over which modulation of gh max. can regulate the period of the heart half-center oscillator n gh max sets f. Final (the escapable inhibition) and thus the period n τh, Ca. S sets how long it will take for a burst to reach f. Final

References n Olypher A, Cymbalyuk G, Calabrese RL. Hybrid systems analysis of the control of burst duration by lowvoltage-activated calcium current in leech heart interneurons, J Neurophysiol. 2006 Dec; 96(6): 2857 -67 Model: n Hill AA, Lu J, Masino MA, Olsen OH, Calabrese RL. A model of a segmental oscillator in the leech heartbeat neuronal network. J Comput Neurosci. 2001 May-Jun; 10(3): 281 -302

- Slides: 23