Lecturer Dr Hairul Nazirah Abdul Halim Pseudosteadystatehypothesis PSSH

Lecturer: Dr. Hairul Nazirah Abdul Halim

Pseudo-steady-state-hypothesis (PSSH) Chain reaction Reaction pathways Enzymatic reactions

Involve a number of elementary reactions and at least one active intermediate. Active intermediate is a high-energy molecule that reacts virtually as fast as it is formed.

Reactive intermediate reacts virtually as fast as it is formed, The net rate of formation of an active intermediate is zero; This condition is referred to PSSH. If active intermediate appears in n reactions:

Gas-phase decomposition of azomethane (AZO) to give ethane and nitrogen: From experimental observations; - At high concentration (pressure > 1 atm) - At low concentration;
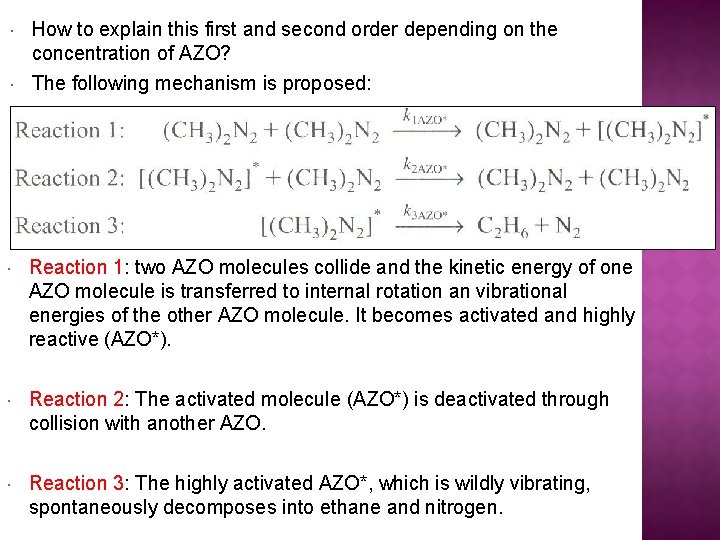
How to explain this first and second order depending on the concentration of AZO? The following mechanism is proposed: Reaction 1: two AZO molecules collide and the kinetic energy of one AZO molecule is transferred to internal rotation an vibrational energies of the other AZO molecule. It becomes activated and highly reactive (AZO*). Reaction 2: The activated molecule (AZO*) is deactivated through collision with another AZO. Reaction 3: The highly activated AZO*, which is wildly vibrating, spontaneously decomposes into ethane and nitrogen.

The following mechanism is proposed: Each of the reaction steps is elementary. The rate laws for the active intermediate AZO*: However, the concentration of AZO* is not readily measurable.

Use PSSH to obtain a rate law in terms of measurable concentrations The rate of formation of product: The net rate of AZO*, (Eq. 7 -6) (Eq. 7 -7) Solving for CAZO* : (Eq. 7 -8)

At low AZO concentrations: Obtain second order rate law:
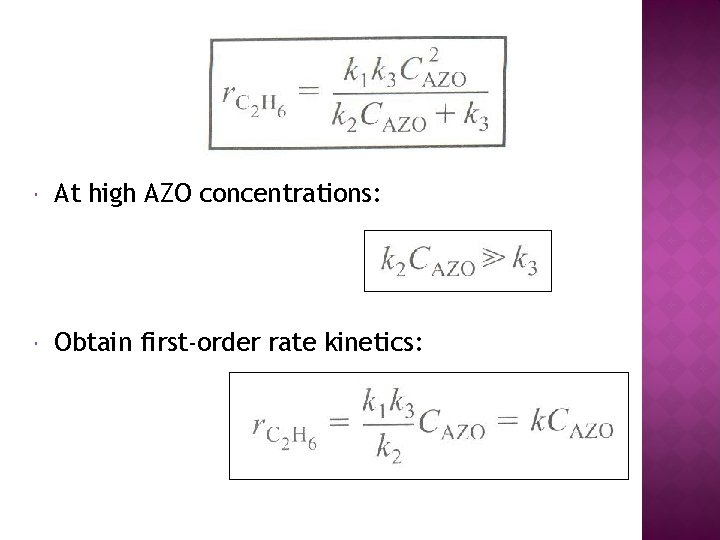
At high AZO concentrations: Obtain first-order rate kinetics:

A chain reaction consists of the following sequence: 1. Initiation: - formation of an active intermediate 2. Propagation or chain transfer: - interaction of an active intermediate with the reactant or product to produce another active intermediate 3. Termination: - deactivation of the active intermediate to form products

Example 7 -2 The thermal decomposition of ethane to ethylene, methane, butane and hydrogen is believed to proceed in the following sequence:

E 7 -2. 1 E 7 -2. 2

E 7 -2. 3 E 7 -2. 4 E 7 -2. 5 E 7 -2. 6

E 7 -2. 7 E 7 -2. 8

E 7 -2. 9 E 7 -2. 3 E 7 -2. 10

E 7 -2. 11 E 7 -2. 12
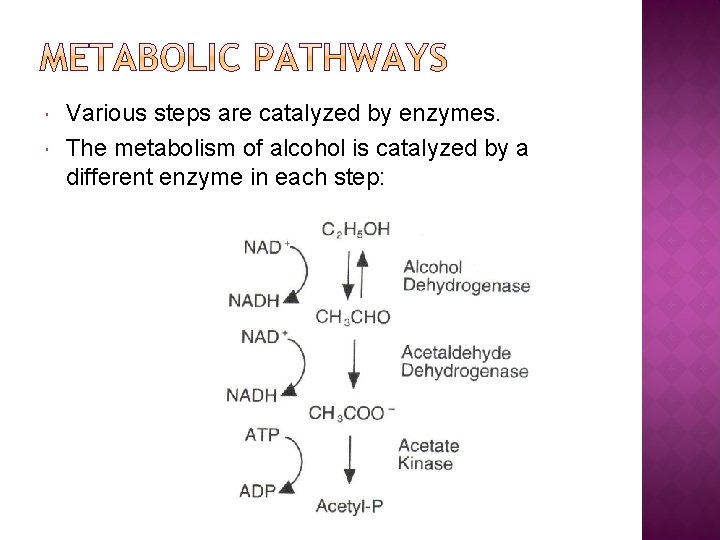
Various steps are catalyzed by enzymes. The metabolism of alcohol is catalyzed by a different enzyme in each step:

Enzyme is a high-molecular-weight protein, which accelerate reaction 103 to 1017 times faster than the uncatalyzed rate. For a reaction of a substrate (S) to form a product (P): Catalyzed reaction pathway: where is an active intermediate
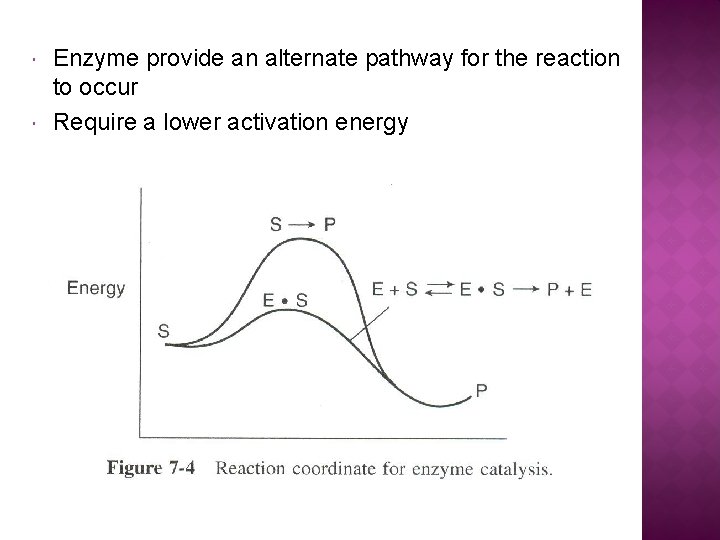
Enzyme provide an alternate pathway for the reaction to occur Require a lower activation energy

Mechanisms of Urea decomposition using urease into CO 2 and ammonia: 1. Urease (E) reacts with the substrate urea (S) to form enzyme-substrate complex ( ). 2. The decompose back to urea (S) and urease (E): 3. Or react with H 2 O to give NH 3 and CO 2:

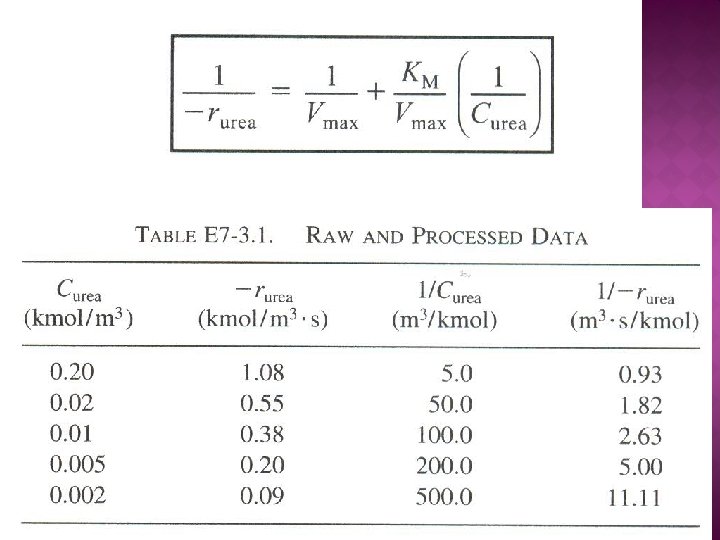
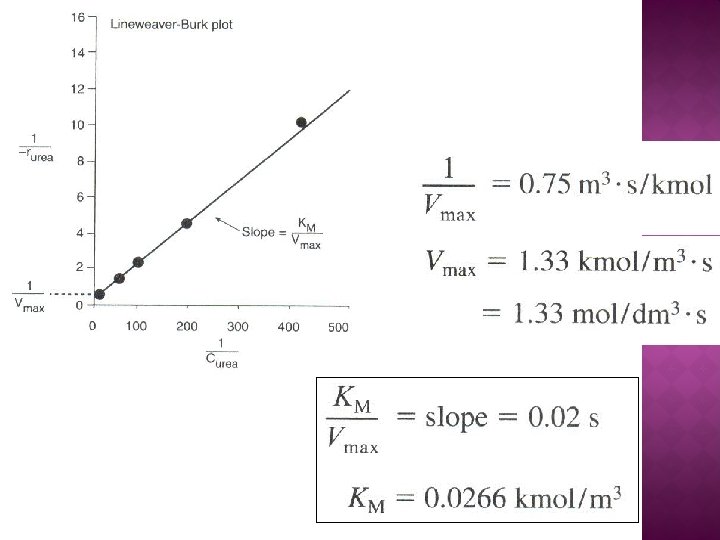
Rate law for Reaction (7 -16, 7 -17 and 7 -18): The net rate of disappearance of the substrate, S:

The net rate formation of the enzyme-substrate complex: Using PSSH, Substitute , into eq. 7 -19:

Total concentration of the enzyme in the system, Et E = unbounded enzyme Substitute for , Solving for (E): Rate law for substrate consumption:
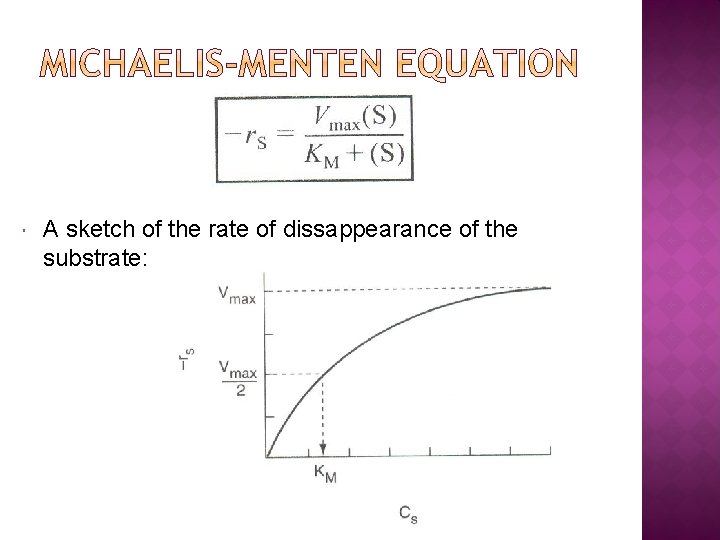
A sketch of the rate of dissappearance of the substrate:


Three most common types: 1. Competitive - the substrate and inhibitors compete for the same site on the enzyme. 2. Uncompetitive - the inhibitor deactivates the enzyme-substrate complex 3. Noncompetitive - occurs with enzyme containing at least two different types of sites - the substrate attaches only to one type of site - the inhibitor attached only to the other side

- Slides: 30