LECTURE PROCESS AND PROCESS VARIABLE 1 PROCESS A

LECTURE PROCESS AND PROCESS VARIABLE 1

PROCESS A process is any operation or series of operations by which a particular objective is accomplished. The operations cause a physical or chemical change in a substance or mixture of substances. The material that enters a process is referred as the INPUT or FEED, and that which leaves is the OUTPUT or PRODUCT. It is common for processes to consist of multiple steps, each of which is carried out in a process unit, and each process unit has associated with it a set of INPUT and OUTPUT process streams. 2
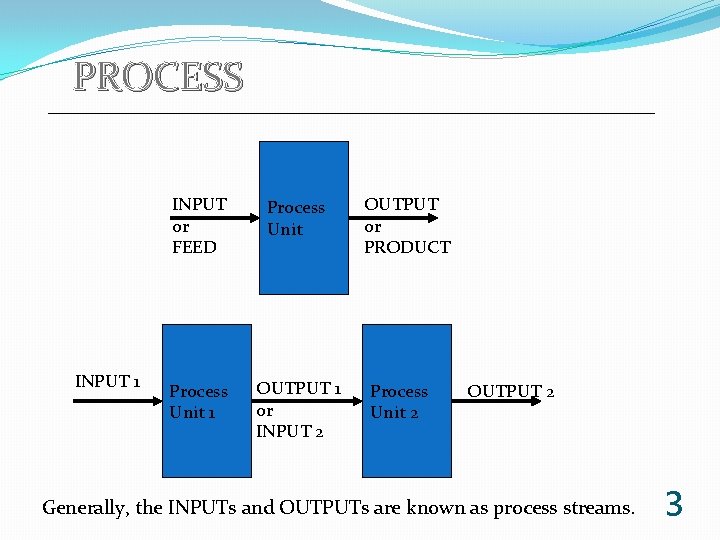
PROCESS INPUT 1 INPUT or FEED Process Unit 1 OUTPUT 1 or INPUT 2 OUTPUT or PRODUCT Process Unit 2 OUTPUT 2 Generally, the INPUTs and OUTPUTs are known as process streams. 3

PROCESS UNIT There a number of process unit. Each of the process unit is selected based on the individual function. Most of the process units are used for the physical separation particularly, • Distillation-separation of mixture liquids based on boiling point • Absorption-separation of mixture of gases based on gas solubility in liquid solvent • Adsorption-separation of mixture of liquids or gases based on affinity into solid adsorbent • Heat Exchanger-particularly for condenser, separation of mixture of gases by removing heat at lower temperature • Crystallization- separation of mixture of liquids by vaporizing the liquid component and remaining the dissolved solid component to form crystal. • Extraction- separation of mixture of liquid based on liquid affinity into liquid solvent. • Membrane-separation of mixture of liquids or gases or mixture solids & gases based on molecular sizes. • Dryer- separation of mixture of solid & liquid by removing the liquid component by heating. 4
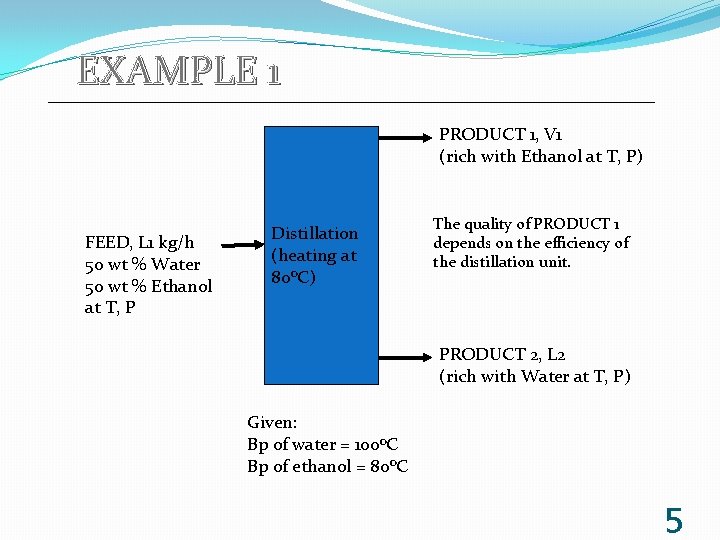
EXAMPLE 1 PRODUCT 1, V 1 (rich with Ethanol at T, P) FEED, L 1 kg/h 50 wt % Water 50 wt % Ethanol at T, P Distillation (heating at 80 o. C) The quality of PRODUCT 1 depends on the efficiency of the distillation unit. PRODUCT 2, L 2 (rich with Water at T, P) Given: Bp of water = 100 o. C Bp of ethanol = 80 o. C 5
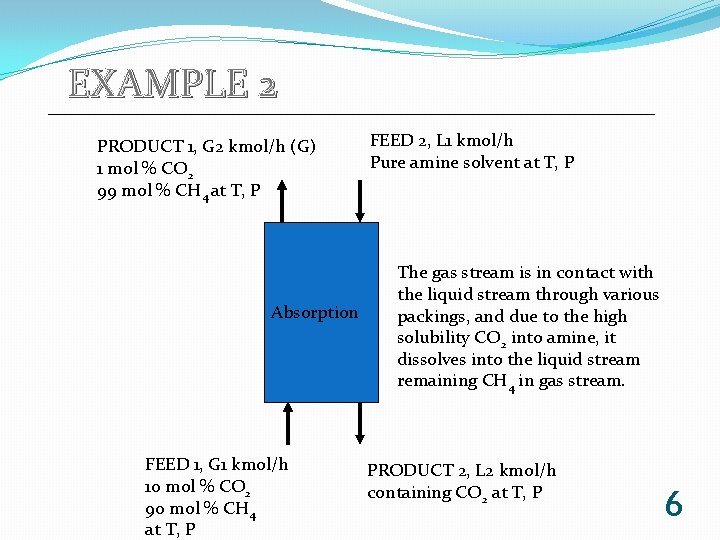
EXAMPLE 2 PRODUCT 1, G 2 kmol/h (G) 1 mol % CO 2 99 mol % CH 4 at T, P Absorption FEED 1, G 1 kmol/h 10 mol % CO 2 90 mol % CH 4 at T, P FEED 2, L 1 kmol/h Pure amine solvent at T, P The gas stream is in contact with the liquid stream through various packings, and due to the high solubility CO 2 into amine, it dissolves into the liquid stream remaining CH 4 in gas stream. PRODUCT 2, L 2 kmol/h containing CO 2 at T, P 6

PROCESS VARIABLES MASS & VOLUME Density, ρ = mass / volume units: g/cm 3, kg/m 3, lbm/ft 3 Specific Gravity, SG, is ratio of density of a substance to density of a reference: SG = ρ/ ρ ref , usually water at 4 o. C = 1. 000 g/cm 3 7

PROCESS VARIABLES FLOW RATE - Fluid movement from a point to another • Mass Flow Rate m - mass (lbm, kg, g) per time (h, s, min) • Volumetric Flow Rate V - volume (ft 3, cm 3, liter) per time • Molar Flow Rate n - molar (mol, kmol, lb-mol) per time In industries, rotameter and orifice are used to measure the volumetric flowrate for any process stream, for gas and liquid process streams. In addition, mass flowmeter is used to measure the mass flowrate of any procees stream. Density is used to convert between mass and volumetric flow rates, � = m/v = m/V 8

EXAMPLE Given, m (hexane) = 6. 59 g/s , r =0. 659 g/cm 3 so, V = m/r = 6. 59 g cm 3 = 10 cm 3/s 0. 659 s g Given, V (CCl 4) = 100 cm 3/min, r =1. 595 g/cm 3 so, m = Vr = 100 × 1. 595 = 159. 5 g/min 9

EXAMPLE Calculate the volumetric and mass flowrate if a volume of 500 liters of water is collected for a period of 5 minutes. Given the density of water is 1000 kg/m 3. i. Volumetric flowrate = 500/5= 100 liters/min ii. Mass flowrate 100 liter 1 min 1 m 3 1000 kg 1000 liter 1 m 3 100 kg/min 10

PROCESS VARIABLES Molecular Weight • • molecular weight = sum of atomic weights basis: atomic weight of 12 C = 12 (exactly) atomic weight of naturally occurring C = 12. 01115 (back cover of text) e. g. atomic oxygen, O, MW = 16 molecular oxygen, O 2, MW = 32 • 1 mol = 6. 023 x 1023 molecules (Avogadro’s number) ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 11

PROCESS VARIABLES Mole and Mass Fraction mole fraction = (moles of a species)/(total moles) mass fraction = (mass of a species)/(total mass) = weight fraction ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 12
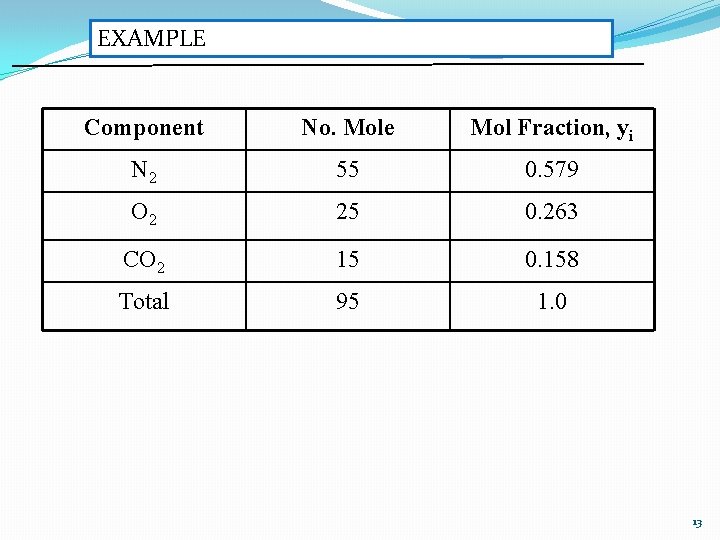
EXAMPLE Component No. Mole Mol Fraction, yi N 2 55 0. 579 O 2 25 0. 263 CO 2 15 0. 158 Total 95 1. 0 13
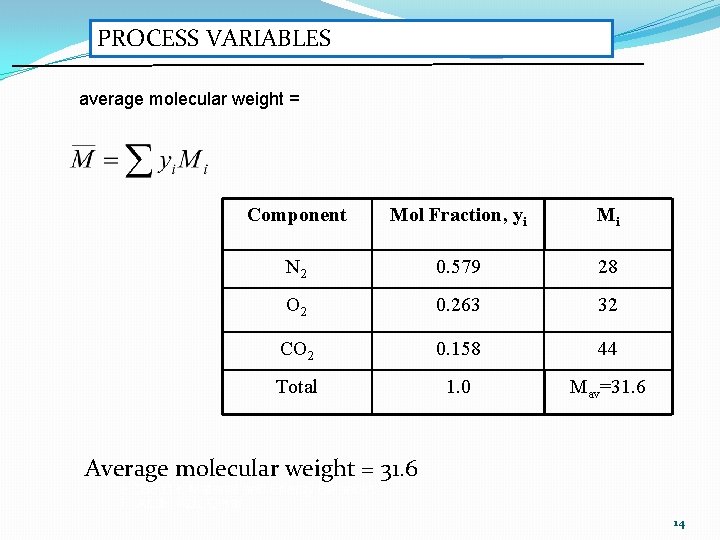
PROCESS VARIABLES average molecular weight = Component Mol Fraction, yi Mi N 2 0. 579 28 O 2 0. 263 32 CO 2 0. 158 44 Total 1. 0 Mav=31. 6 Average molecular weight = 31. 6 ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 14

PROCESS VARIABLES CONCENTRATION • molar concentration = moles of component per unit volume of solution • mass concentration = mass of component per unit volume of solution parts per million = moles (or mass) of component per million moles (or mass units) of mixture • 15

PROCESS VARIABLES PRESSURE • • force per unit area a fluid exerts a pressure on the wall of its container due to molecules bouncing off the wall measured by determining force and area Note: This is often written with a gc in the denominator 16

PROCESS VARIABLES Hydrostatic Pressure -- pressure difference between bottom and top of a column of fluid of height h -- based on Newton's law of gravity barometer = an instrument for measuring atmospheric pressure 1 atm = 760. 000 mm. Hg(0 o. C, std. go) Pressure may be measured as: • Atmospheric pressure • Absolute Pressure • Gauge Pressure 17

PROCESS VARIABLES Atmospheric Pressure measured at sea-level or at base level 760 mm. Hg 1 atm 1. 013 bar 101. 3 k. Pa 14. 7 psi (lb/sq. in. ) Absolute Pressure • sum of atmospheric pressure and gauge pressure Pabs = Pgauge + Patm 18

Gauge Pressure • • • Reading from the gauge Relative to atmospheric pressure zero means abs pressure = atmospheric pressure from: Pabs = Pgauge + Patm 0 Pgauge = Pabs- Patm 19

PROCESS VARIABLES TEMPERATURE • temperature is a property that tells us if two systems are in thermal equilibrium • measuring devices are systems that can be brought to thermal equilibrium with the system of interest (hopefully without disturbing this system) examples are liquid in glass thermometers, thermocouples, resistance thermometers, bimetallic thermometers, radiation pyrometers, etc. 20

PROCESS VARIABLES The unit temperature is given in o. C, o. F, o. R or K and related to each other using the following equations. T(K) = T(o. C) + 273. 15 T(o. R) = T(o. F) + 459. 67 T(o. R) = 1. 8 T(K) T(o. F) = 1. 8 T(o. C) + 32 ECB 3013: Material and Energy Balances Ir. Abdul Aziz Omar 21
- Slides: 21