LECTURE PRESENTATIONS For CAMPBELL BIOLOGY NINTH EDITION Jane

LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 10 Photosynthesis Lectures by Erin Barley Kathleen Fitzpatrick © 2011 Pearson Education, Inc.

Overview: The Process That Feeds the Biosphere • Photosynthesis is the process that converts solar energy into chemical energy • Directly or indirectly, photosynthesis nourishes almost the entire living world © 2011 Pearson Education, Inc.

• Autotrophs sustain themselves without eating anything derived from other organisms • Autotrophs are the producers of the biosphere, producing organic molecules from CO 2 and other inorganic molecules • Almost all plants are photoautotrophs, using the energy of sunlight to make organic molecules © 2011 Pearson Education, Inc.

Figure 10. 1

• Photosynthesis occurs in plants, algae, certain other protists, and some prokaryotes • These organisms feed not only themselves but also most of the living world Bio. Flix: Photosynthesis © 2011 Pearson Education, Inc.

Figure 10. 2 (b) Multicellular alga (a) Plants (d) Cyanobacteria (c) Unicellular protists 10 m (e) Purple sulfur 1 m bacteria 40 m

Figure 10. 2 a (a) Plants

Figure 10. 2 b (b) Multicellular alga
Figure 10. 2 c (c) Unicellular protists 10 m

Figure 10. 2 d (d) Cyanobacteria 40 m
Figure 10. 2 e (e) Purple sulfur bacteria 1 m

• Heterotrophs obtain their organic material from other organisms • Heterotrophs are the consumers of the biosphere • Almost all heterotrophs, including humans, depend on photoautotrophs for food and O 2 © 2011 Pearson Education, Inc.

• The Earth’s supply of fossil fuels was formed from the remains of organisms that died hundreds of millions of years ago • In a sense, fossil fuels represent stores of solar energy from the distant past © 2011 Pearson Education, Inc.

Figure 10. 3

Concept 10. 1: Photosynthesis converts light energy to the chemical energy of food • Chloroplasts are structurally similar to and likely evolved from photosynthetic bacteria • The structural organization of these cells allows for the chemical reactions of photosynthesis © 2011 Pearson Education, Inc.

Chloroplasts: The Sites of Photosynthesis in Plants • Leaves are the major locations of photosynthesis • Their green color is from chlorophyll, the green pigment within chloroplasts • Chloroplasts are found mainly in cells of the mesophyll, the interior tissue of the leaf • Each mesophyll cell contains 30– 40 chloroplasts © 2011 Pearson Education, Inc.

• CO 2 enters and O 2 exits the leaf through microscopic pores called stomata • The chlorophyll is in the membranes of thylakoids (connected sacs in the chloroplast); thylakoids may be stacked in columns called grana • Chloroplasts also contain stroma, a dense interior fluid © 2011 Pearson Education, Inc.
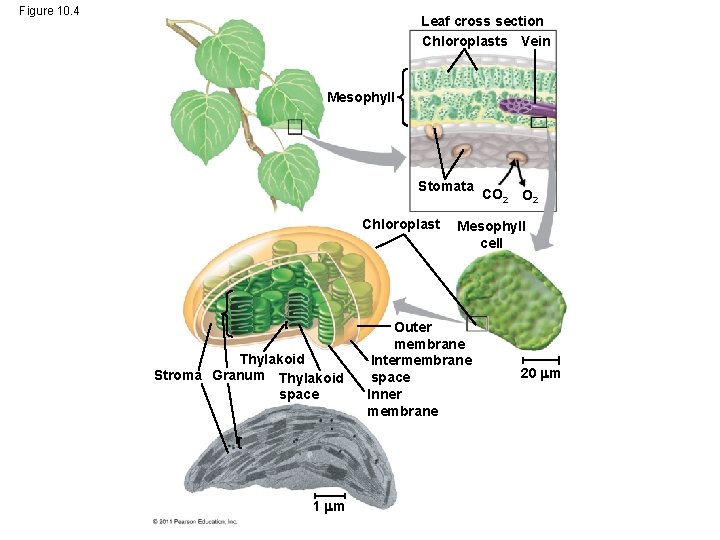
Figure 10. 4 Leaf cross section Chloroplasts Vein Mesophyll Stomata Chloroplast Thylakoid Stroma Granum Thylakoid space 1 m CO 2 Mesophyll cell Outer membrane Intermembrane space Inner membrane 20 m
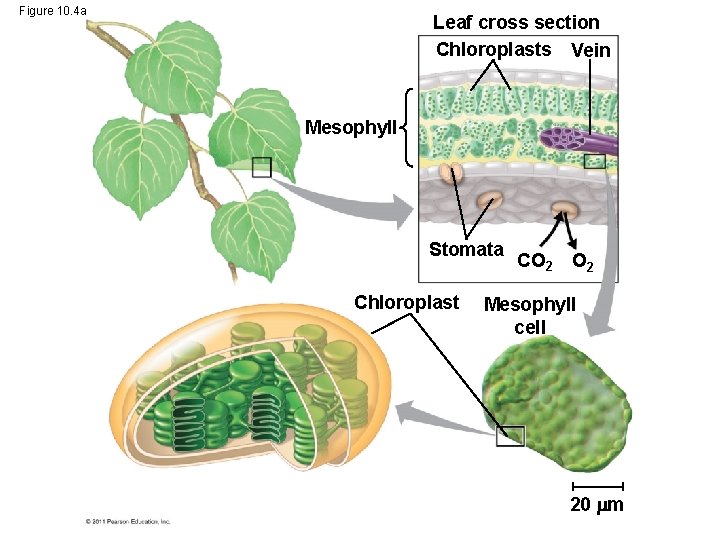
Figure 10. 4 a Leaf cross section Chloroplasts Vein Mesophyll Stomata Chloroplast CO 2 Mesophyll cell 20 m
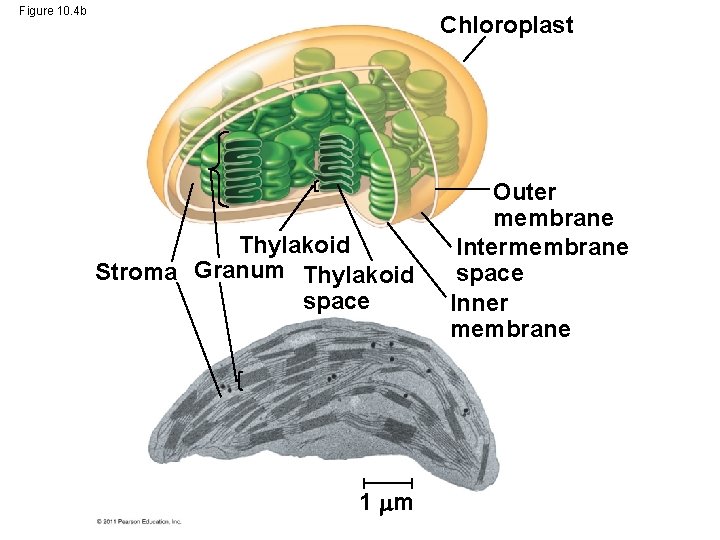
Figure 10. 4 b Chloroplast Thylakoid Stroma Granum Thylakoid space 1 m Outer membrane Intermembrane space Inner membrane
Figure 10. 4 c Mesophyll cell 20 m
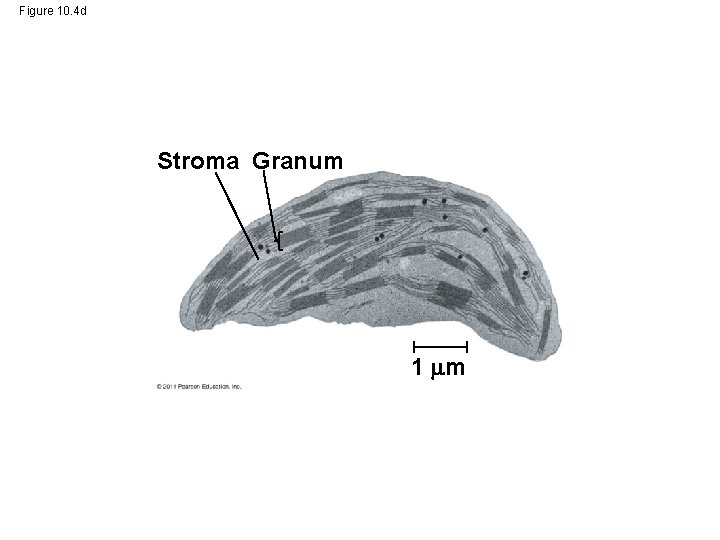
Figure 10. 4 d Stroma Granum 1 m

Tracking Atoms Through Photosynthesis: Scientific Inquiry • Photosynthesis is a complex series of reactions that can be summarized as the following equation: 6 CO 2 + 12 H 2 O + Light energy C 6 H 12 O 6 + 6 O 2 + 6 H 2 O © 2011 Pearson Education, Inc.

The Splitting of Water • Chloroplasts split H 2 O into hydrogen and oxygen, incorporating the electrons of hydrogen into sugar molecules and releasing oxygen as a by-product © 2011 Pearson Education, Inc.
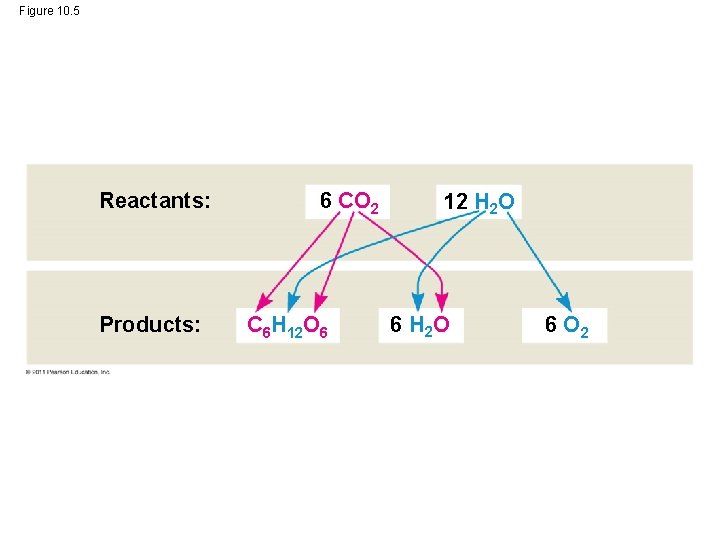
Figure 10. 5 Reactants: Products: 6 CO 2 C 6 H 12 O 6 12 H 2 O 6 O 2

Photosynthesis as a Redox Process • Photosynthesis reverses the direction of electron flow compared to respiration • Photosynthesis is a redox process in which H 2 O is oxidized and CO 2 is reduced • Photosynthesis is an endergonic process; the enery boost is provided by light © 2011 Pearson Education, Inc.
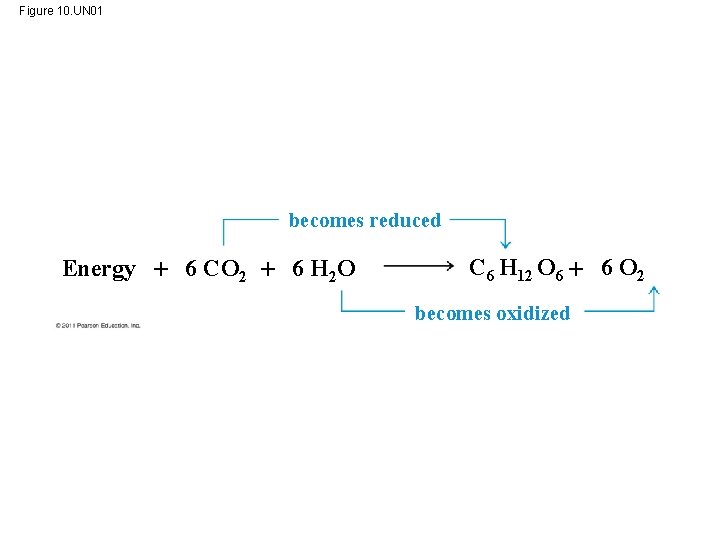
Figure 10. UN 01 becomes reduced Energy 6 CO 2 6 H 2 O C 6 H 12 O 6 6 O 2 becomes oxidized

The Two Stages of Photosynthesis: A Preview • Photosynthesis consists of the light reactions (the photo part) and Calvin cycle (the synthesis part) • The light reactions (in the thylakoids) – – Split H 2 O Release O 2 Reduce NADP+ to NADPH Generate ATP from ADP by photophosphorylation © 2011 Pearson Education, Inc.

• The Calvin cycle (in the stroma) forms sugar from CO 2, using ATP and NADPH • The Calvin cycle begins with carbon fixation, incorporating CO 2 into organic molecules © 2011 Pearson Education, Inc.
Figure 10. 6 -1 H 2 O Light NADP +Pi Light Reactions Chloroplast
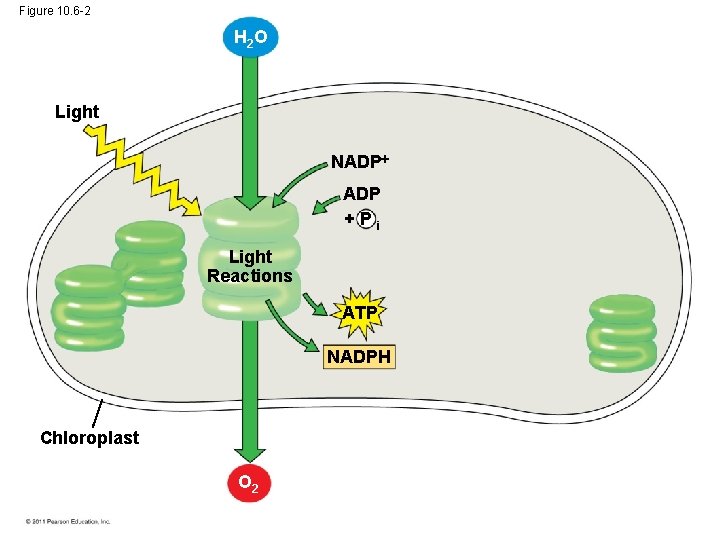
Figure 10. 6 -2 H 2 O Light NADP +Pi Light Reactions ATP NADPH Chloroplast O 2
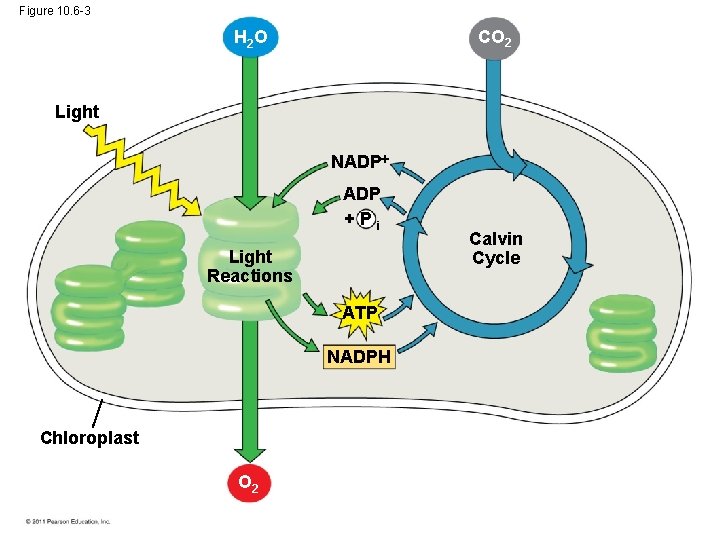
Figure 10. 6 -3 CO 2 H 2 O Light NADP +Pi Light Reactions ATP NADPH Chloroplast O 2 Calvin Cycle
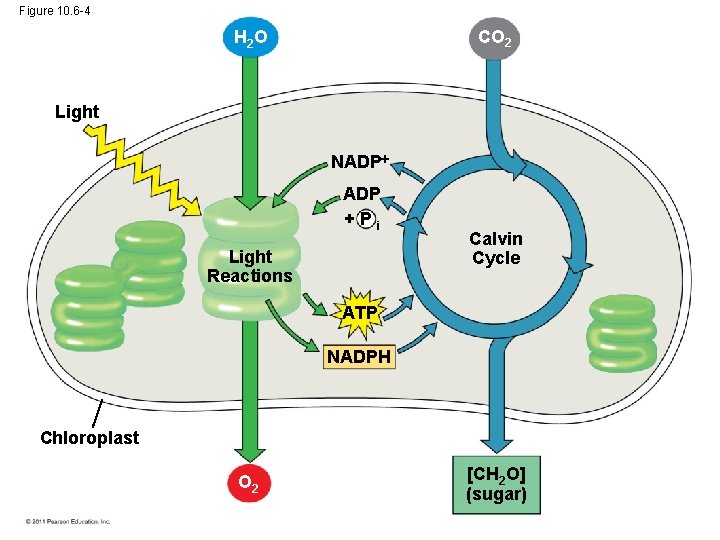
Figure 10. 6 -4 CO 2 H 2 O Light NADP +Pi Light Reactions Calvin Cycle ATP NADPH Chloroplast O 2 [CH 2 O] (sugar)

Concept 10. 2: The light reactions convert solar energy to the chemical energy of ATP and NADPH • Chloroplasts are solar-powered chemical factories • Their thylakoids transform light energy into the chemical energy of ATP and NADPH © 2011 Pearson Education, Inc.

The Nature of Sunlight • Light is a form of electromagnetic energy, also called electromagnetic radiation • Like other electromagnetic energy, light travels in rhythmic waves • Wavelength is the distance between crests of waves • Wavelength determines the type of electromagnetic energy © 2011 Pearson Education, Inc.

• The electromagnetic spectrum is the entire range of electromagnetic energy, or radiation • Visible light consists of wavelengths (including those that drive photosynthesis) that produce colors we can see • Light also behaves as though it consists of discrete particles, called photons © 2011 Pearson Education, Inc.
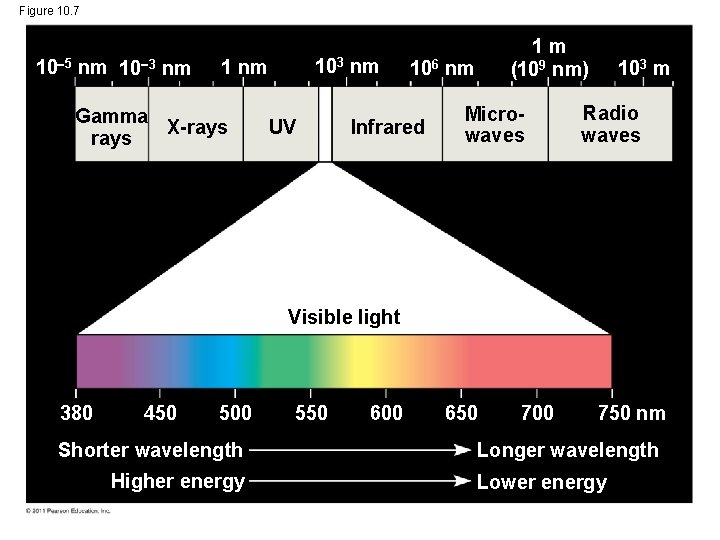
Figure 10. 7 10 5 nm 10 3 nm 103 1 nm Gamma X-rays UV nm 1 m (109 nm) 106 nm Infrared Microwaves 103 m Radio waves Visible light 380 450 500 Shorter wavelength Higher energy 550 600 650 700 750 nm Longer wavelength Lower energy

Photosynthetic Pigments: The Light Receptors • Pigments are substances that absorb visible light • Different pigments absorb different wavelengths • Wavelengths that are not absorbed are reflected or transmitted • Leaves appear green because chlorophyll reflects and transmits green light Animation: Light and Pigments © 2011 Pearson Education, Inc.
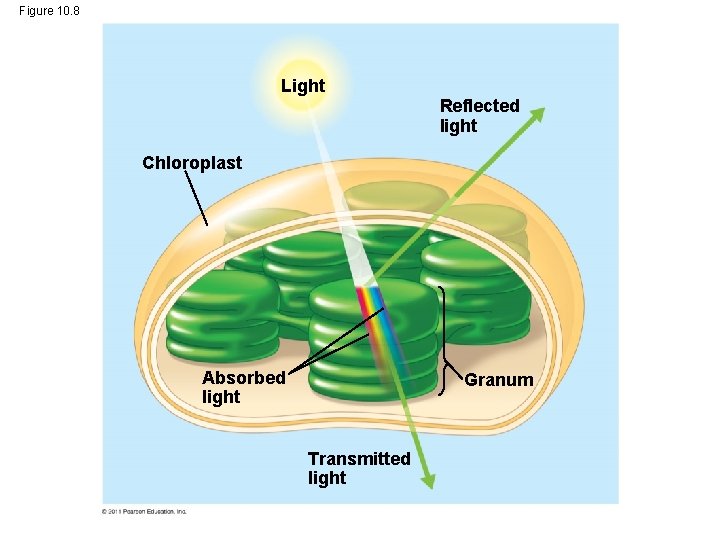
Figure 10. 8 Light Reflected light Chloroplast Absorbed light Granum Transmitted light

• A spectrophotometer measures a pigment’s ability to absorb various wavelengths • This machine sends light through pigments and measures the fraction of light transmitted at each wavelength © 2011 Pearson Education, Inc.
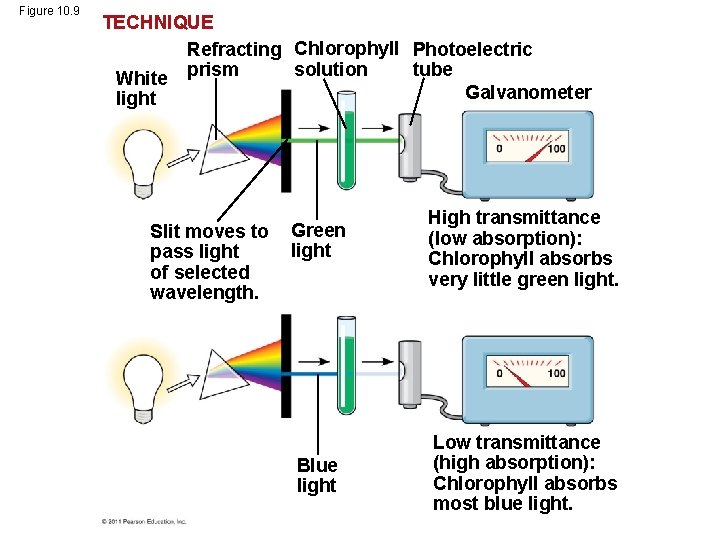
Figure 10. 9 TECHNIQUE Refracting Chlorophyll Photoelectric solution tube White prism Galvanometer light Slit moves to pass light of selected wavelength. Green light High transmittance (low absorption): Chlorophyll absorbs very little green light. Blue light Low transmittance (high absorption): Chlorophyll absorbs most blue light.

• An absorption spectrum is a graph plotting a pigment’s light absorption versus wavelength • The absorption spectrum of chlorophyll a suggests that violet-blue and red light work best for photosynthesis • An action spectrum profiles the relative effectiveness of different wavelengths of radiation in driving a process © 2011 Pearson Education, Inc.
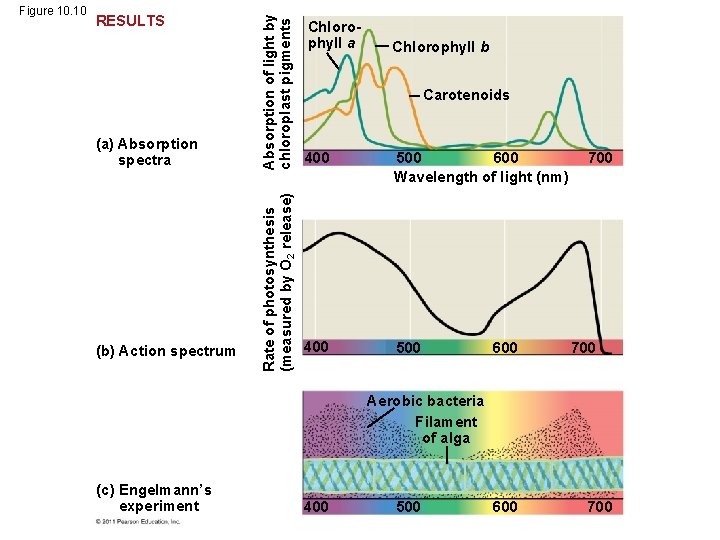
(a) Absorption spectra (b) Action spectrum Absorption of light by chloroplast pigments RESULTS Rate of photosynthesis (measured by O 2 release) Figure 10. 10 Chlorophyll a Chlorophyll b Carotenoids 400 500 600 Wavelength of light (nm) 400 500 600 700 Aerobic bacteria Filament of alga (c) Engelmann’s experiment 400 500 600 700

• The action spectrum of photosynthesis was first demonstrated in 1883 by Theodor W. Engelmann • In his experiment, he exposed different segments of a filamentous alga to different wavelengths • Areas receiving wavelengths favorable to photosynthesis produced excess O 2 • He used the growth of aerobic bacteria clustered along the alga as a measure of O 2 production © 2011 Pearson Education, Inc.

• Chlorophyll a is the main photosynthetic pigment • Accessory pigments, such as chlorophyll b, broaden the spectrum used for photosynthesis • Accessory pigments called carotenoids absorb excessive light that would damage chlorophyll © 2011 Pearson Education, Inc.
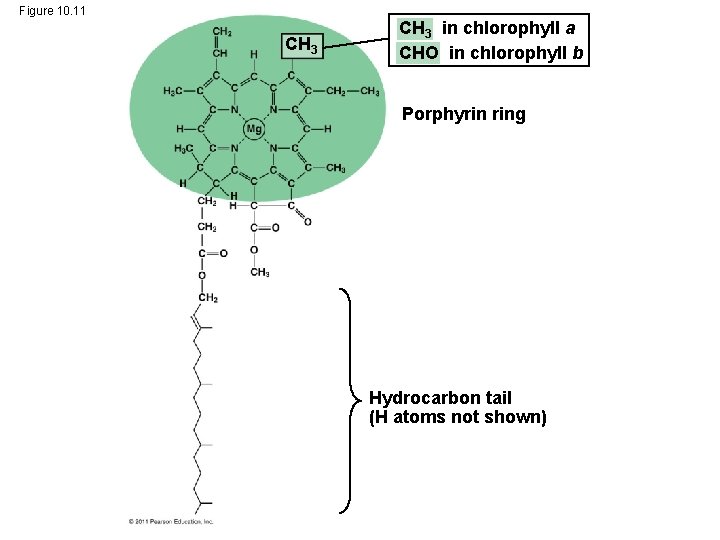
Figure 10. 11 CH 3 in chlorophyll a CHO in chlorophyll b Porphyrin ring Hydrocarbon tail (H atoms not shown)

Excitation of Chlorophyll by Light • When a pigment absorbs light, it goes from a ground state to an excited state, which is unstable • When excited electrons fall back to the ground state, photons are given off, an afterglow called fluorescence • If illuminated, an isolated solution of chlorophyll will fluoresce, giving off light and heat © 2011 Pearson Education, Inc.
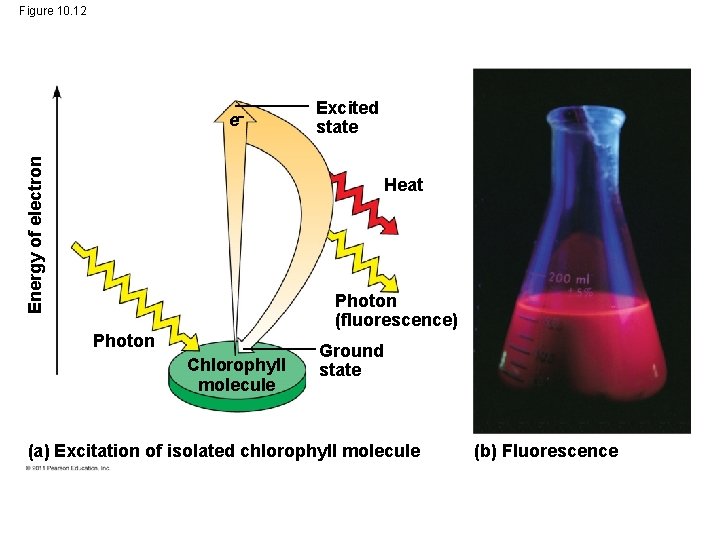
Figure 10. 12 Energy of electron e Excited state Heat Photon (fluorescence) Photon Chlorophyll molecule Ground state (a) Excitation of isolated chlorophyll molecule (b) Fluorescence

Figure 10. 12 a (b) Fluorescence

A Photosystem: A Reaction-Center Complex Associated with Light-Harvesting Complexes • A photosystem consists of a reaction-center complex (a type of protein complex) surrounded by light-harvesting complexes • The light-harvesting complexes (pigment molecules bound to proteins) transfer the energy of photons to the reaction center © 2011 Pearson Education, Inc.
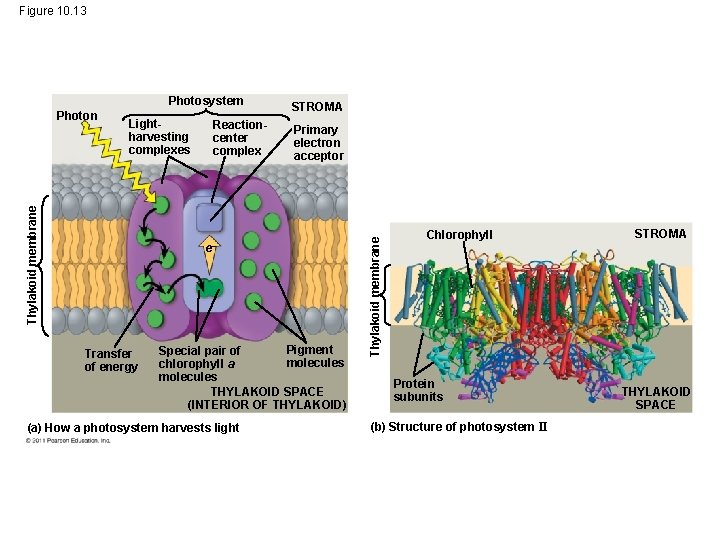
Figure 10. 13 Lightharvesting complexes Thylakoid membrane Photon Reactioncenter complex STROMA Primary electron acceptor e Transfer of energy Pigment Special pair of molecules chlorophyll a molecules THYLAKOID SPACE (INTERIOR OF THYLAKOID) (a) How a photosystem harvests light Thylakoid membrane Photosystem Chlorophyll Protein subunits (b) Structure of photosystem II STROMA THYLAKOID SPACE
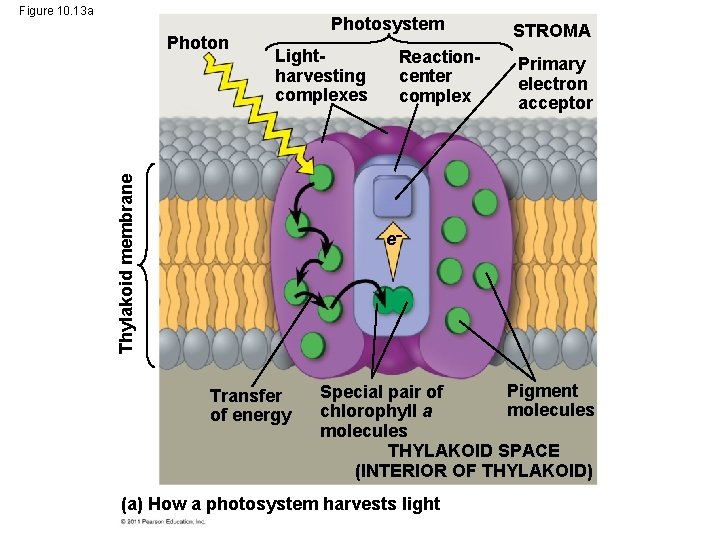
Figure 10. 13 a Lightharvesting complexes Thylakoid membrane Photon Photosystem Reactioncenter complex STROMA Primary electron acceptor e Transfer of energy Pigment Special pair of molecules chlorophyll a molecules THYLAKOID SPACE (INTERIOR OF THYLAKOID) (a) How a photosystem harvests light
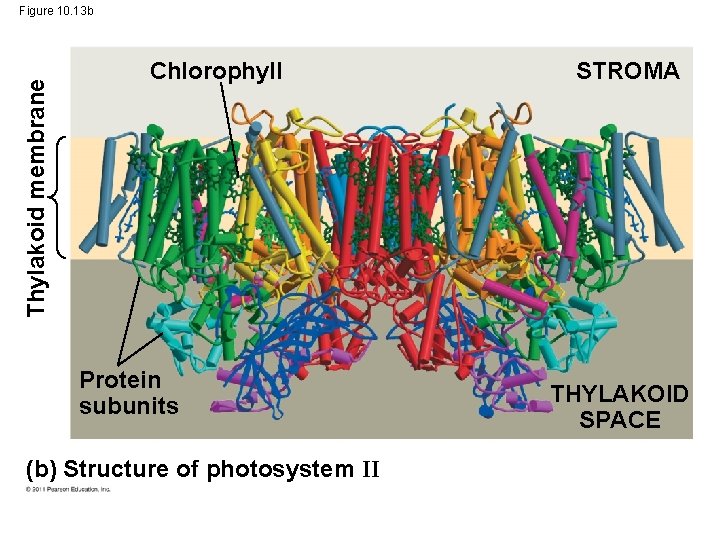
Thylakoid membrane Figure 10. 13 b Chlorophyll Protein subunits (b) Structure of photosystem II STROMA THYLAKOID SPACE

• A primary electron acceptor in the reaction center accepts excited electron and is reduced as a result • Solar-powered transfer of an electron from a chlorophyll a molecule to the primary electron acceptor is the first step of the light reactions © 2011 Pearson Education, Inc.

• There are two types of photosystems in the thylakoid membrane • Photosystem II (PS II) functions first (the numbers reflect order of discovery) and is best at absorbing a wavelength of 680 nm • The reaction-center chlorophyll a of PS II is called P 680 © 2011 Pearson Education, Inc.

• Photosystem I (PS I) is best at absorbing a wavelength of 700 nm • The reaction-center chlorophyll a of PS I is called P 700 © 2011 Pearson Education, Inc.

Linear Electron Flow • During the light reactions, there are two possible routes for electron flow: cyclic and linear • Linear electron flow, the primary pathway, involves both photosystems and produces ATP and NADPH using light energy © 2011 Pearson Education, Inc.

• A photon hits a pigment and its energy is passed among pigment molecules until it excites P 680 • An excited electron from P 680 is transferred to the primary electron acceptor (we now call it P 680+) © 2011 Pearson Education, Inc.
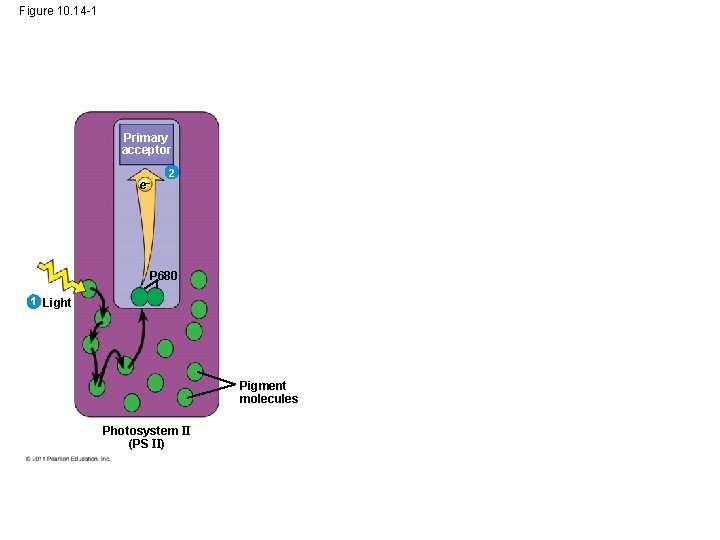
Figure 10. 14 -1 Primary acceptor e 2 P 680 1 Light Pigment molecules Photosystem II (PS II)

• P 680+ is a very strong oxidizing agent • H 2 O is split by enzymes, and the electrons are transferred from the hydrogen atoms to P 680+, thus reducing it to P 680 • O 2 is released as a by-product of this reaction © 2011 Pearson Education, Inc.
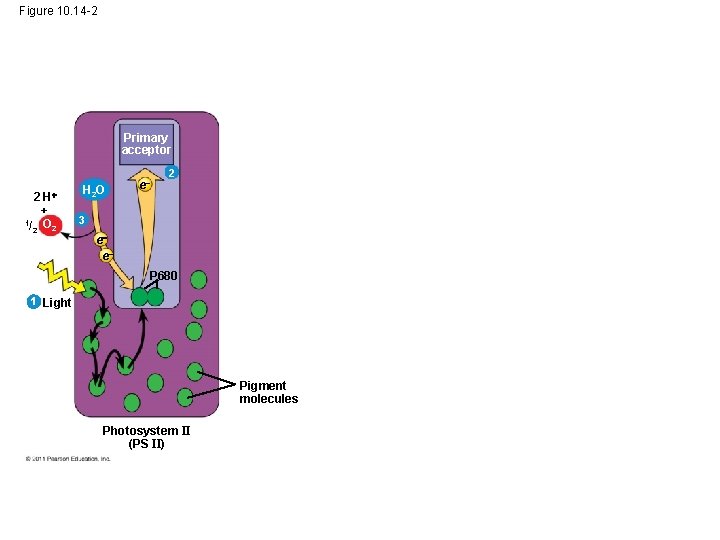
Figure 10. 14 -2 Primary acceptor 2 H + 1/ O 2 2 H 2 O e 2 3 e e P 680 1 Light Pigment molecules Photosystem II (PS II)

• Each electron “falls” down an electron transport chain from the primary electron acceptor of PS II to PS I • Energy released by the fall drives the creation of a proton gradient across the thylakoid membrane • Diffusion of H+ (protons) across the membrane drives ATP synthesis © 2011 Pearson Education, Inc.
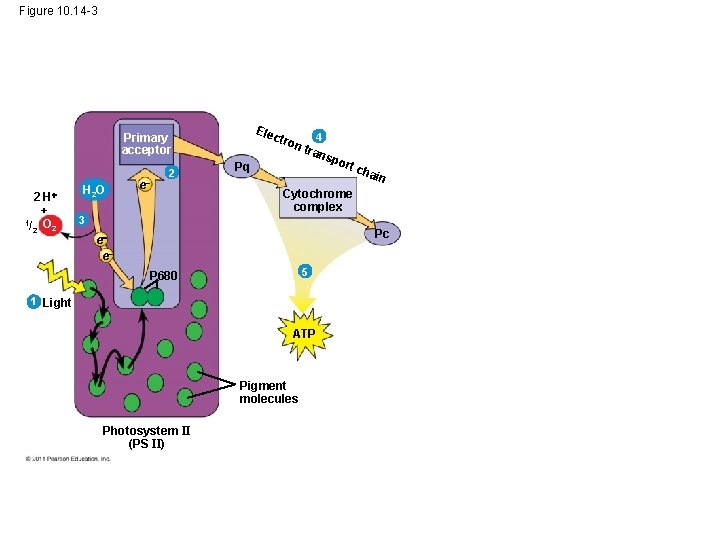
Figure 10. 14 -3 Ele ct Primary acceptor 2 H + 1/ O 2 2 H 2 O e 2 ron Pq 4 tran spo rt c hai n Cytochrome complex 3 Pc e e 5 P 680 1 Light ATP Pigment molecules Photosystem II (PS II)

• In PS I (like PS II), transferred light energy excites P 700, which loses an electron to an electron acceptor • P 700+ (P 700 that is missing an electron) accepts an electron passed down from PS II via the electron transport chain © 2011 Pearson Education, Inc.
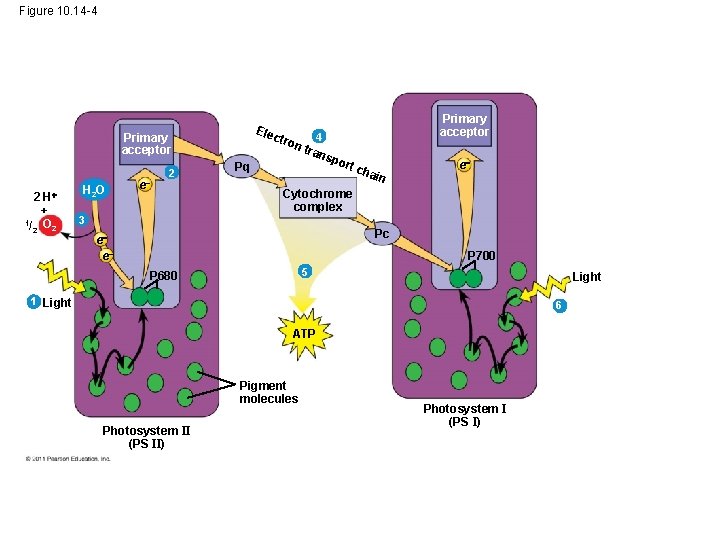
Figure 10. 14 -4 Ele ct Primary acceptor 1/ 2 H 2 + O 2 H 2 O e 2 ron Pq Primary acceptor 4 tran spo rt c hai n e Cytochrome complex 3 Pc e e P 700 5 P 680 Light 1 Light 6 ATP Pigment molecules Photosystem II (PS II) Photosystem I (PS I)

• Each electron “falls” down an electron transport chain from the primary electron acceptor of PS I to the protein ferredoxin (Fd) • The electrons are then transferred to NADP+ and reduce it to NADPH • The electrons of NADPH are available for the reactions of the Calvin cycle • This process also removes an H+ from the stroma © 2011 Pearson Education, Inc.
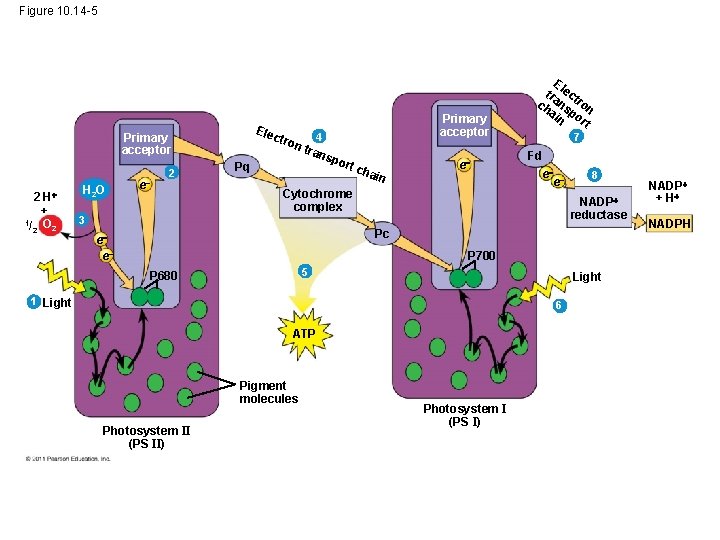
Figure 10. 14 -5 Ele ct Primary acceptor 2 H + 1/ O 2 2 H 2 O e 2 ron Pq Primary acceptor 4 tran spo rt c hai n e Cytochrome complex E tra lect ch ns ron ai po n rt 7 Fd e e 8 NADP reductase 3 Pc e e P 700 5 P 680 Light 1 Light 6 ATP Pigment molecules Photosystem II (PS II) Photosystem I (PS I) NADP + H NADPH
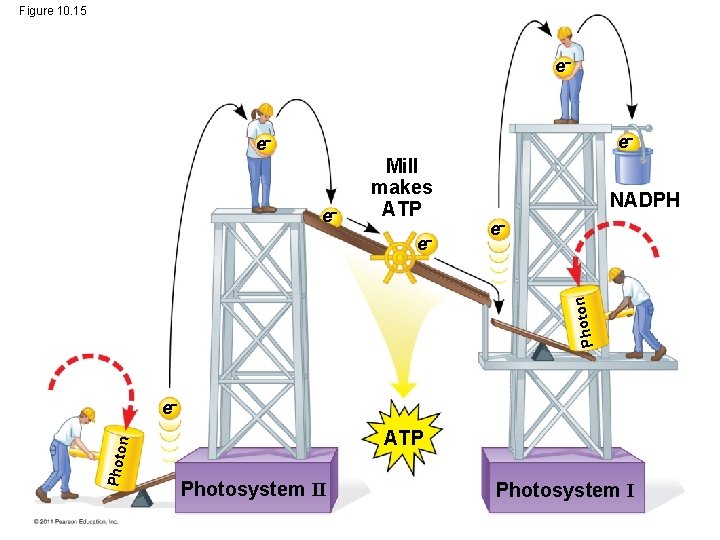
Figure 10. 15 e e Mill makes ATP e Photo n e NADPH e Photo n ATP Photosystem II Photosystem I

Cyclic Electron Flow • Cyclic electron flow uses only photosystem I and produces ATP, but not NADPH • No oxygen is released • Cyclic electron flow generates surplus ATP, satisfying the higher demand in the Calvin cycle © 2011 Pearson Education, Inc.
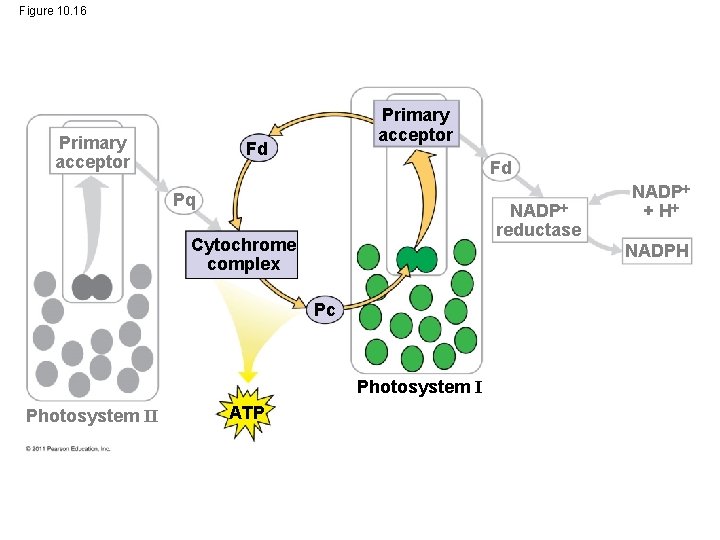
Figure 10. 16 Primary acceptor Fd Fd Pq NADP reductase Cytochrome complex NADPH Pc Photosystem II ATP NADP + H

• Some organisms such as purple sulfur bacteria have PS I but not PS II • Cyclic electron flow is thought to have evolved before linear electron flow • Cyclic electron flow may protect cells from light-induced damage © 2011 Pearson Education, Inc.

A Comparison of Chemiosmosis in Chloroplasts and Mitochondria • Chloroplasts and mitochondria generate ATP by chemiosmosis, but use different sources of energy • Mitochondria transfer chemical energy from food to ATP; chloroplasts transform light energy into the chemical energy of ATP • Spatial organization of chemiosmosis differs between chloroplasts and mitochondria but also shows similarities © 2011 Pearson Education, Inc.

• In mitochondria, protons are pumped to the intermembrane space and drive ATP synthesis as they diffuse back into the mitochondrial matrix • In chloroplasts, protons are pumped into the thylakoid space and drive ATP synthesis as they diffuse back into the stroma © 2011 Pearson Education, Inc.
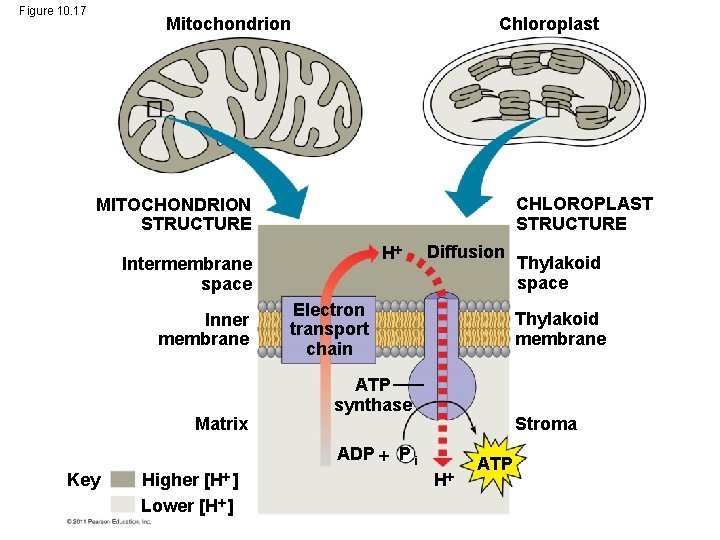
Figure 10. 17 Chloroplast Mitochondrion CHLOROPLAST STRUCTURE MITOCHONDRION STRUCTURE H Intermembrane space Inner membrane Matrix Diffusion Electron transport chain Thylakoid membrane ATP synthase Stroma ADP P i Key [H ] Higher Lower [H ] Thylakoid space H ATP

• ATP and NADPH are produced on the side facing the stroma, where the Calvin cycle takes place • In summary, light reactions generate ATP and increase the potential energy of electrons by moving them from H 2 O to NADPH © 2011 Pearson Education, Inc.
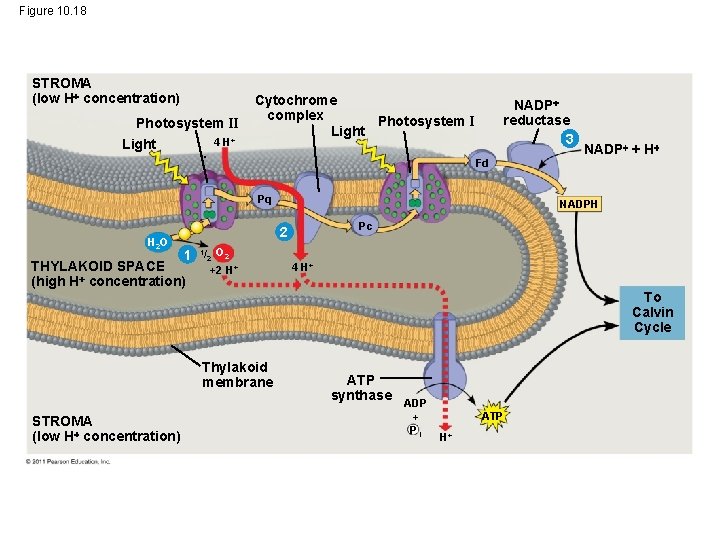
Figure 10. 18 STROMA (low H concentration) Photosystem II Light 4 H+ Cytochrome complex Photosystem I Light NADP reductase 3 Fd Pq H 2 O NADPH Pc 2 1 THYLAKOID SPACE (high H concentration) 1/ 2 NADP + H O 2 +2 H+ 4 H+ To Calvin Cycle Thylakoid membrane STROMA (low H concentration) ATP synthase ADP + Pi ATP H+

Concept 10. 3: The Calvin cycle uses the chemical energy of ATP and NADPH to reduce CO 2 to sugar • The Calvin cycle, like the citric acid cycle, regenerates its starting material after molecules enter and leave the cycle • The cycle builds sugar from smaller molecules by using ATP and the reducing power of electrons carried by NADPH © 2011 Pearson Education, Inc.

• Carbon enters the cycle as CO 2 and leaves as a sugar named glyceraldehyde 3 -phospate (G 3 P) • For net synthesis of 1 G 3 P, the cycle must take place three times, fixing 3 molecules of CO 2 • The Calvin cycle has three phases – Carbon fixation (catalyzed by rubisco) – Reduction – Regeneration of the CO 2 acceptor (Ru. BP) © 2011 Pearson Education, Inc.
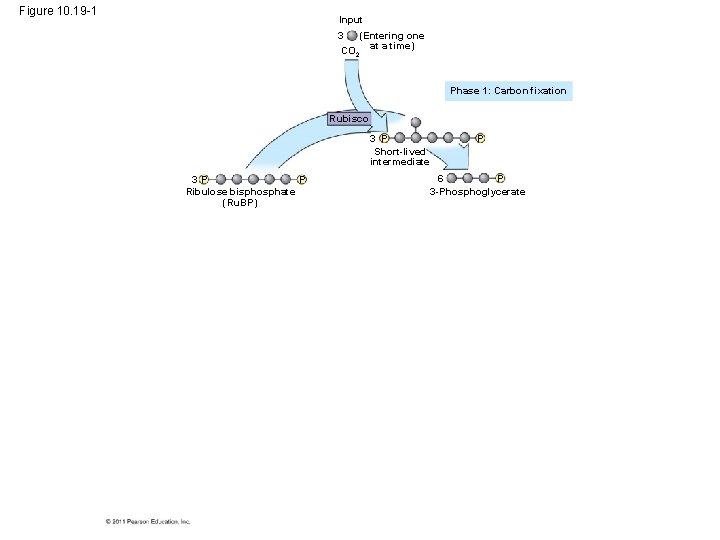
Figure 10. 19 -1 Input (Entering one CO 2 at a time) 3 Phase 1: Carbon fixation Rubisco 3 P Short-lived intermediate P 3 P Ribulose bisphosphate (Ru. BP) P 6 P 3 -Phosphoglycerate
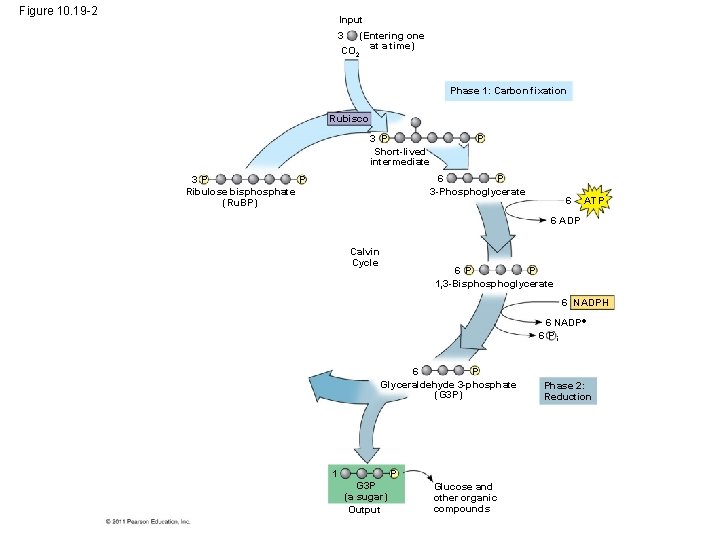
Figure 10. 19 -2 Input (Entering one CO 2 at a time) 3 Phase 1: Carbon fixation Rubisco 3 P Short-lived intermediate P 6 P 3 -Phosphoglycerate P 3 P Ribulose bisphosphate (Ru. BP) 6 ATP 6 ADP Calvin Cycle 6 P P 1, 3 -Bisphoglycerate 6 NADPH 6 NADP 6 Pi 6 P Glyceraldehyde 3 -phosphate (G 3 P) 1 P G 3 P (a sugar) Output Glucose and other organic compounds Phase 2: Reduction
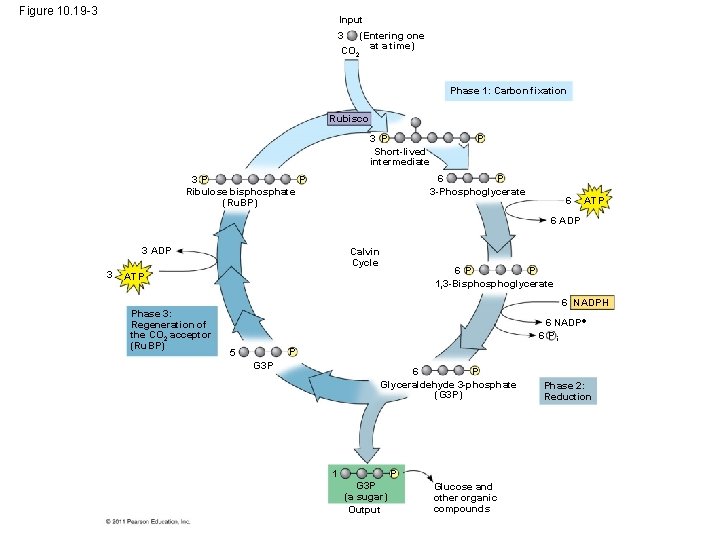
Figure 10. 19 -3 Input (Entering one CO 2 at a time) 3 Phase 1: Carbon fixation Rubisco 3 P Short-lived intermediate P 6 P 3 -Phosphoglycerate P 3 P Ribulose bisphosphate (Ru. BP) 6 ATP 6 ADP 3 Calvin Cycle 6 P P 1, 3 -Bisphoglycerate ATP Phase 3: Regeneration of the CO 2 acceptor (Ru. BP) 6 NADPH 6 NADP 6 Pi P 5 G 3 P 6 P Glyceraldehyde 3 -phosphate (G 3 P) 1 P G 3 P (a sugar) Output Glucose and other organic compounds Phase 2: Reduction

Concept 10. 4: Alternative mechanisms of carbon fixation have evolved in hot, arid climates • Dehydration is a problem for plants, sometimes requiring trade-offs with other metabolic processes, especially photosynthesis • On hot, dry days, plants close stomata, which conserves H 2 O but also limits photosynthesis • The closing of stomata reduces access to CO 2 and causes O 2 to build up • These conditions favor an apparently wasteful process called photorespiration © 2011 Pearson Education, Inc.

Photorespiration: An Evolutionary Relic? • In most plants (C 3 plants), initial fixation of CO 2, via rubisco, forms a three-carbon compound (3 phosphoglycerate) • In photorespiration, rubisco adds O 2 instead of CO 2 in the Calvin cycle, producing a two-carbon compound • Photorespiration consumes O 2 and organic fuel and releases CO 2 without producing ATP or sugar © 2011 Pearson Education, Inc.

• Photorespiration may be an evolutionary relic because rubisco first evolved at a time when the atmosphere had far less O 2 and more CO 2 • Photorespiration limits damaging products of light reactions that build up in the absence of the Calvin cycle • In many plants, photorespiration is a problem because on a hot, dry day it can drain as much as 50% of the carbon fixed by the Calvin cycle © 2011 Pearson Education, Inc.

C 4 Plants • C 4 plants minimize the cost of photorespiration by incorporating CO 2 into four-carbon compounds in mesophyll cells • This step requires the enzyme PEP carboxylase • PEP carboxylase has a higher affinity for CO 2 than rubisco does; it can fix CO 2 even when CO 2 concentrations are low • These four-carbon compounds are exported to bundle-sheath cells, where they release CO 2 that is then used in the Calvin cycle © 2011 Pearson Education, Inc.
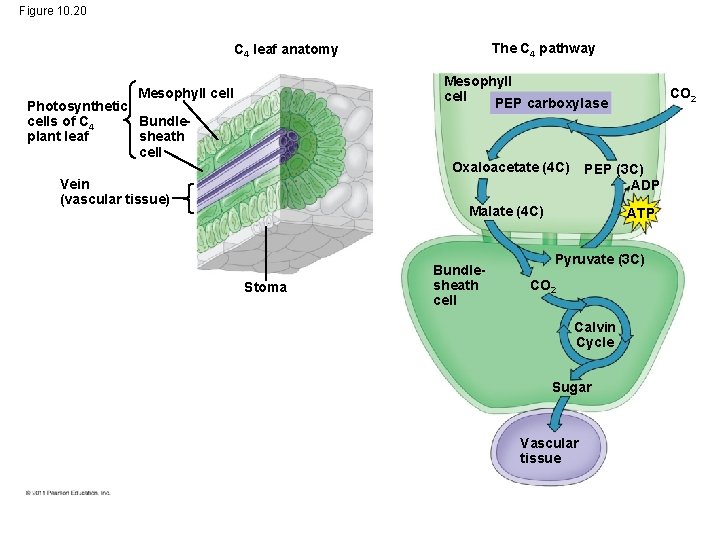
Figure 10. 20 The C 4 pathway C 4 leaf anatomy Mesophyll cell PEP carboxylase Mesophyll cell Photosynthetic cells of C 4 Bundleplant leaf sheath cell Oxaloacetate (4 C) Vein (vascular tissue) PEP (3 C) ADP Malate (4 C) Stoma Bundlesheath cell CO 2 ATP Pyruvate (3 C) CO 2 Calvin Cycle Sugar Vascular tissue
Figure 10. 20 a C 4 leaf anatomy Photosynthetic cells of C 4 plant leaf Mesophyll cell Bundlesheath cell Vein (vascular tissue) Stoma
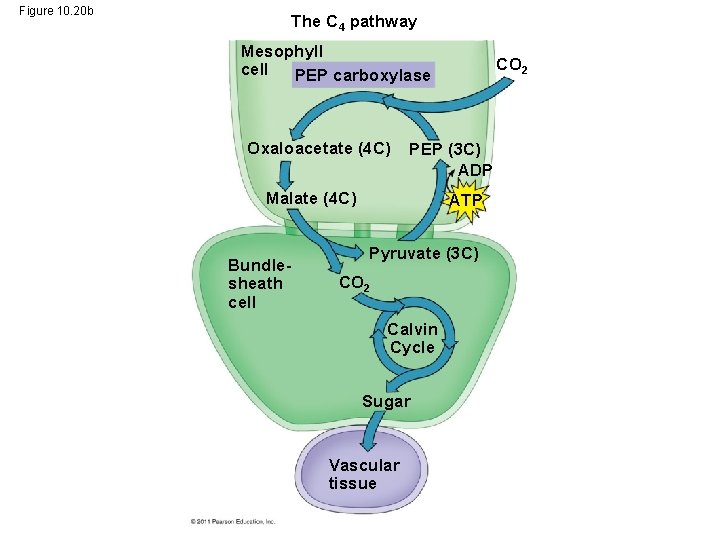
Figure 10. 20 b The C 4 pathway Mesophyll cell PEP carboxylase Oxaloacetate (4 C) PEP (3 C) ADP Malate (4 C) Bundlesheath cell CO 2 ATP Pyruvate (3 C) CO 2 Calvin Cycle Sugar Vascular tissue

• In the last 150 years since the Industrial Revolution, CO 2 levels have risen gratly • Increasing levels of CO 2 may affect C 3 and C 4 plants differently, perhaps changing relative abundance of these species • The effects of such changes are unpredictable and a cause for concern © 2011 Pearson Education, Inc.

CAM Plants • Some plants, including succulents, use crassulacean acid metabolism (CAM) to fix carbon • CAM plants open their stomata at night, incorporating CO 2 into organic acids • Stomata close during the day, and CO 2 is released from organic acids and used in the Calvin cycle © 2011 Pearson Education, Inc.
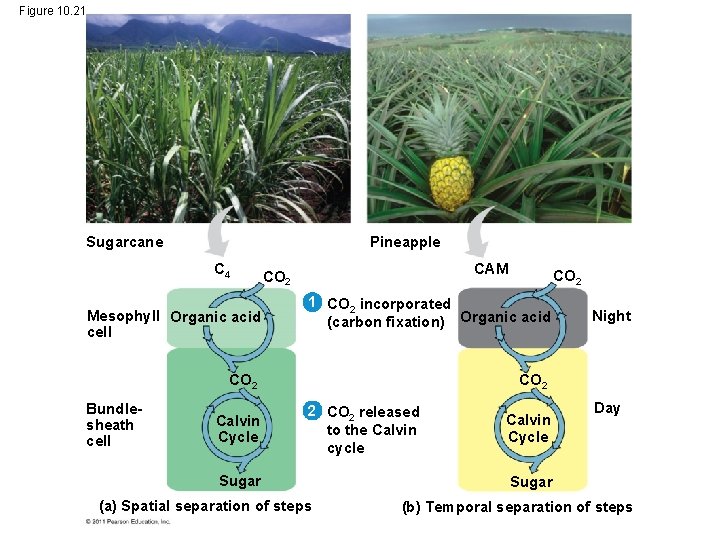
Figure 10. 21 Sugarcane Pineapple C 4 Mesophyll Organic acid cell CAM CO 2 1 CO 2 incorporated (carbon fixation) Organic acid Calvin Cycle Night CO 2 Bundlesheath cell CO 2 2 CO 2 released to the Calvin cycle Sugar (a) Spatial separation of steps Calvin Cycle Day Sugar (b) Temporal separation of steps

Figure 10. 21 a Sugarcane

Figure 10. 21 b Pineapple

The Importance of Photosynthesis: A Review • The energy entering chloroplasts as sunlight gets stored as chemical energy in organic compounds • Sugar made in the chloroplasts supplies chemical energy and carbon skeletons to synthesize the organic molecules of cells • Plants store excess sugar as starch in structures such as roots, tubers, seeds, and fruits • In addition to food production, photosynthesis produces the O 2 in our atmosphere © 2011 Pearson Education, Inc.
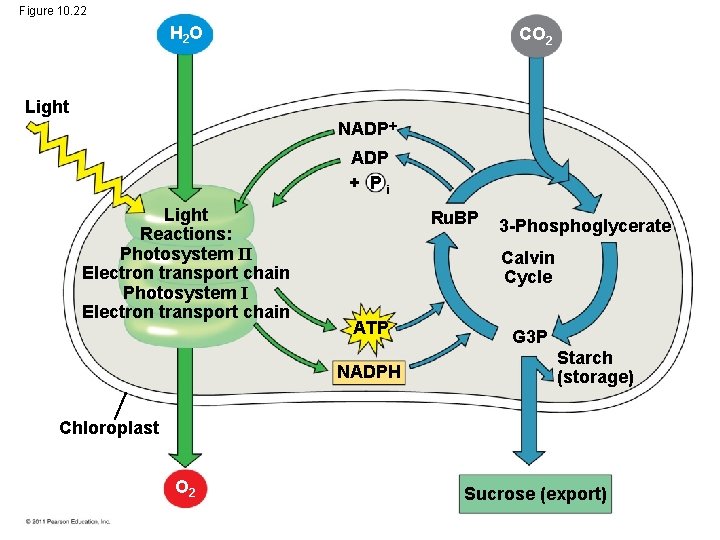
Figure 10. 22 H 2 O CO 2 Light NADP + Pi Light Reactions: Photosystem II Electron transport chain Photosystem I Electron transport chain Ru. BP 3 -Phosphoglycerate Calvin Cycle ATP NADPH G 3 P Starch (storage) Chloroplast O 2 Sucrose (export)
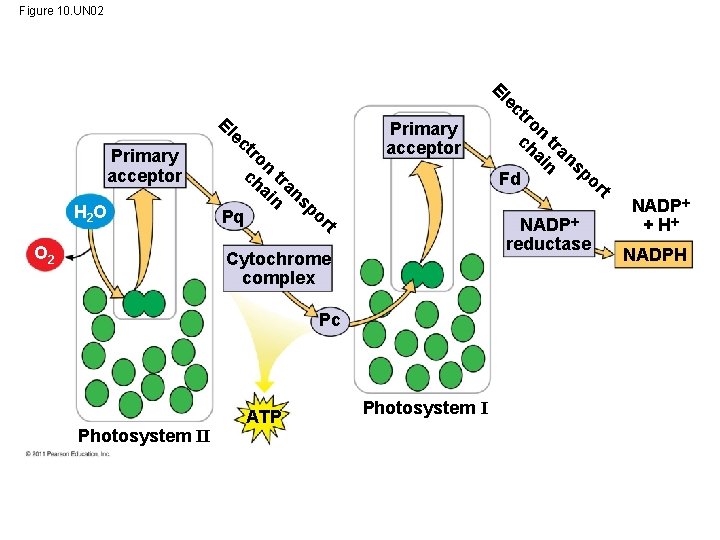
Figure 10. UN 02 El El Primary acceptor H 2 O O 2 ec tr on ch tr ai an n sp or Pq t Primary acceptor t NADP reductase Cytochrome complex Pc Photosystem II ATP ec tr on ch tr ai an n sp or Fd Photosystem I NADP + H NADPH
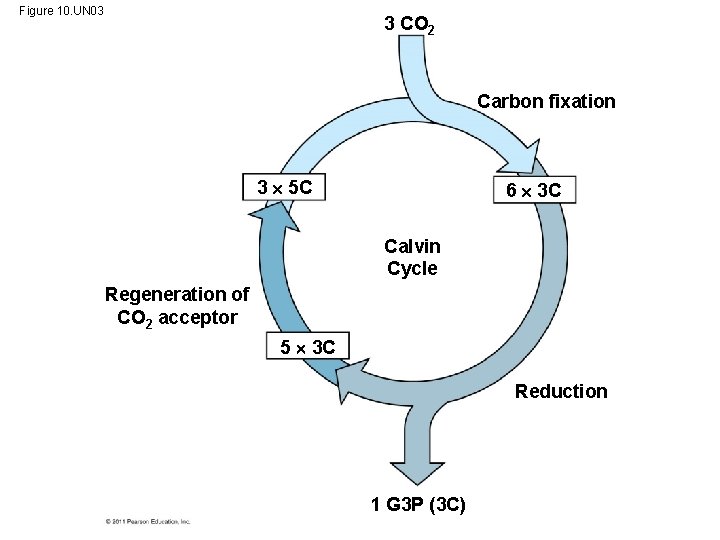
Figure 10. UN 03 3 CO 2 Carbon fixation 3 5 C 6 3 C Calvin Cycle Regeneration of CO 2 acceptor 5 3 C Reduction 1 G 3 P (3 C)
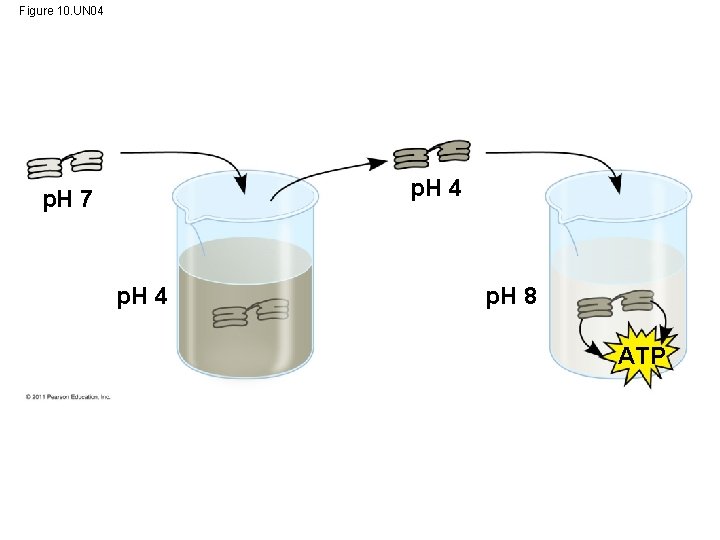
Figure 10. UN 04 p. H 7 p. H 4 p. H 8 ATP
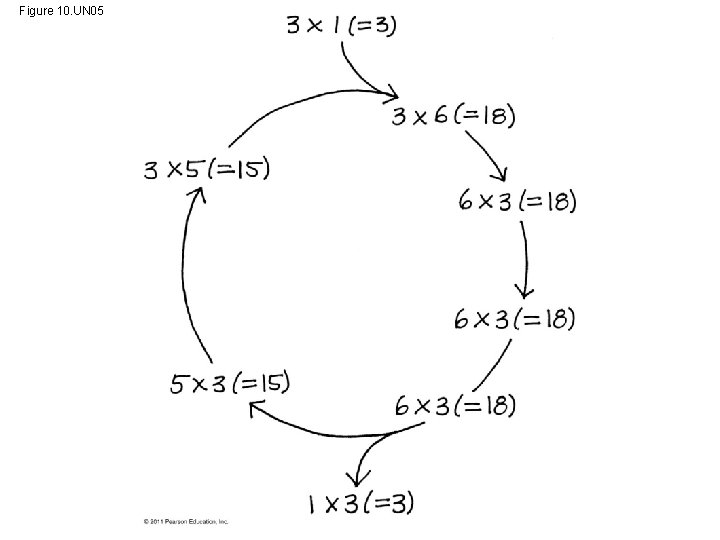
Figure 10. UN 05
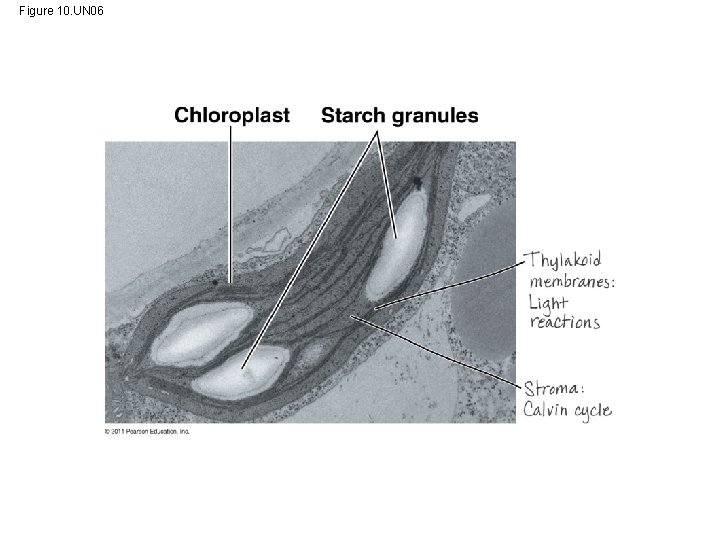
Figure 10. UN 06
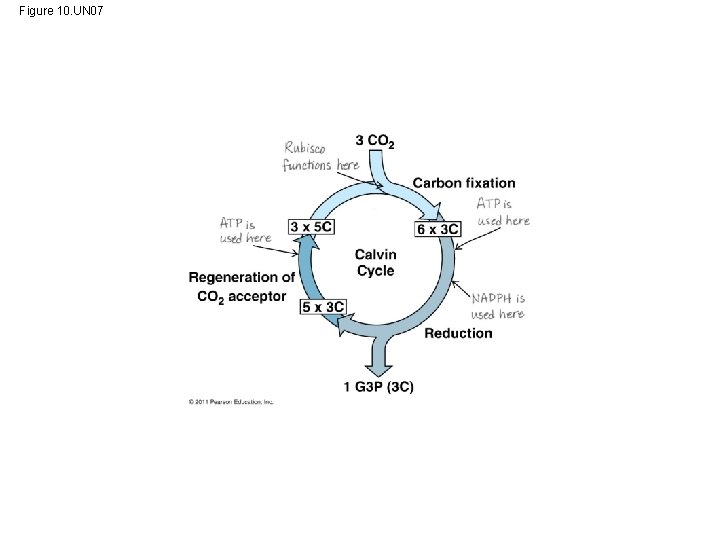
Figure 10. UN 07
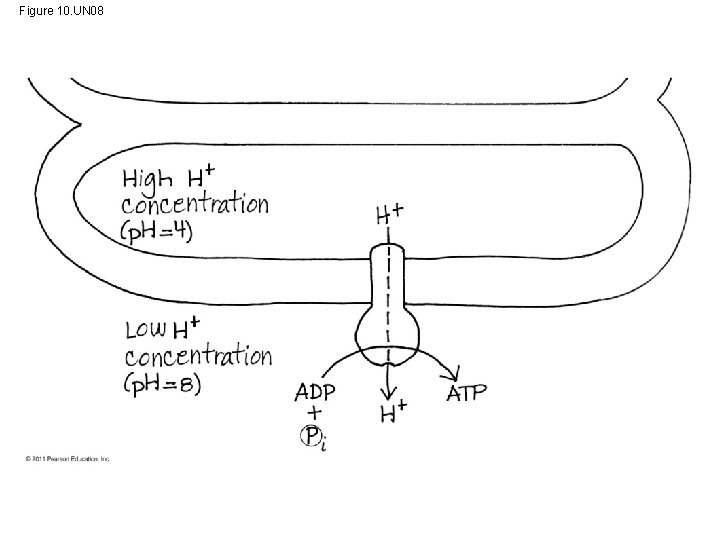
Figure 10. UN 08
- Slides: 102