LECTURE PRESENTATIONS For CAMPBELL BIOLOGY NINTH EDITION Jane

LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 9 Cellular Respiration and Fermentation Lectures by Erin Barley Kathleen Fitzpatrick © 2011 Pearson Education, Inc.

Harvesting stored energy • Energy is stored in organic molecules – carbohydrates, fats, proteins • Heterotrophs eat these organic molecules food – digest organic molecules to get… • raw materials for synthesis • fuels for energy – controlled release of energy – “burning” fuels in a series of step-by-step enzyme-controlled reactions
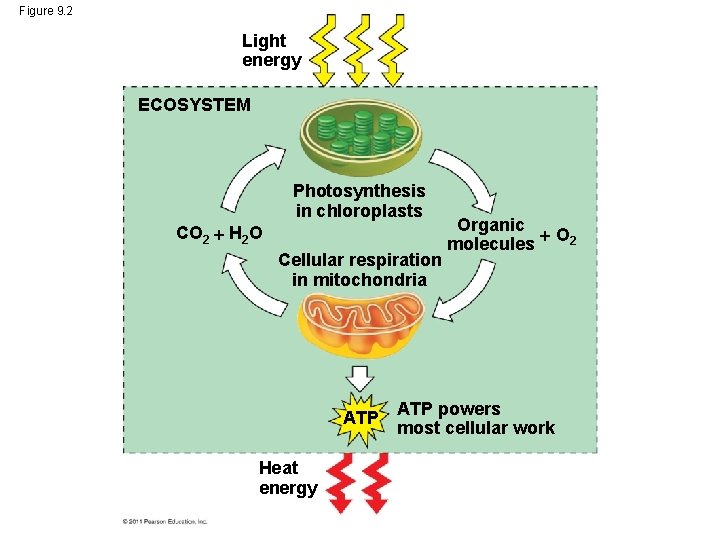
Figure 9. 2 Light energy ECOSYSTEM Photosynthesis in chloroplasts CO 2 H 2 O Cellular respiration in mitochondria ATP Heat energy Organic O 2 molecules ATP powers most cellular work

Living economy • Fueling the body’s economy – eat high energy organic molecules • food = carbohydrates, lipids, proteins, nucleic acids – break them down • digest = catabolism – capture released energy in a form the cell can use • Need an energy currency – a way to pass energy around – need a short term energy storage molecule Whoa! Hot stuff! ATP
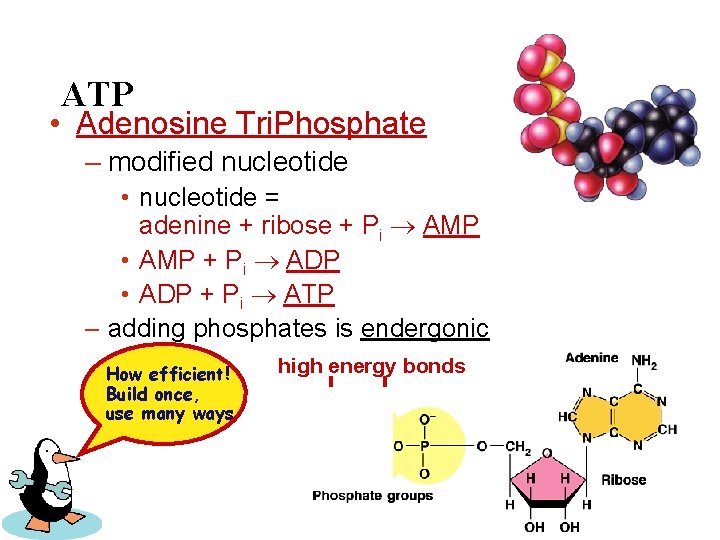
ATP • Adenosine Tri. Phosphate – modified nucleotide • nucleotide = adenine + ribose + Pi AMP • AMP + Pi ADP • ADP + Pi ATP – adding phosphates is endergonic How efficient! Build once, use many ways high energy bonds
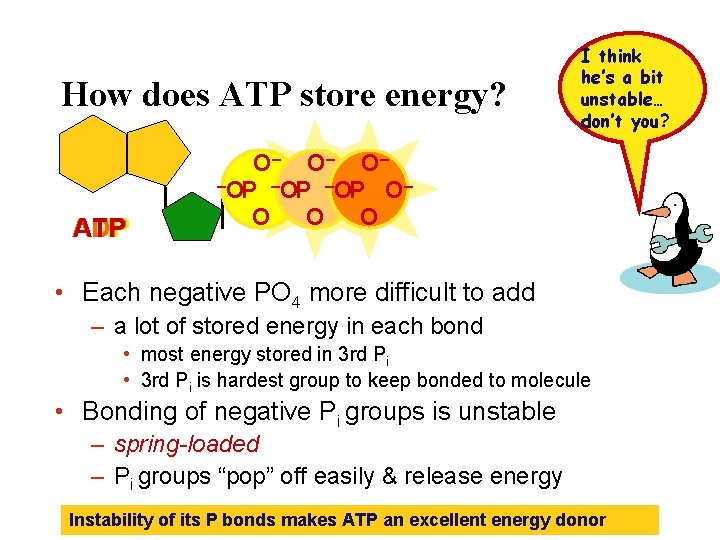
How does ATP store energy? ADP AMP ATP I think he’s a bit unstable… don’t you? O– O– O– –O – OP P –O OP OP O– O O O • Each negative PO 4 more difficult to add – a lot of stored energy in each bond • most energy stored in 3 rd Pi • 3 rd Pi is hardest group to keep bonded to molecule • Bonding of negative Pi groups is unstable – spring-loaded – Pi groups “pop” off easily & release energy Instability of its P bonds makes ATP an excellent energy donor
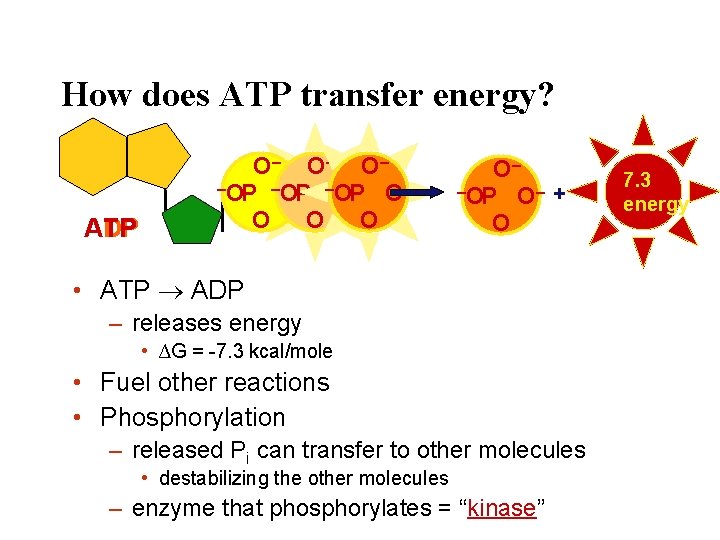
How does ATP transfer energy? ATP ADP O– O– O– –OP –O – OP OP O– O O– –OP O– + O • ATP ADP – releases energy • ∆G = -7. 3 kcal/mole • Fuel other reactions • Phosphorylation – released Pi can transfer to other molecules • destabilizing the other molecules – enzyme that phosphorylates = “kinase” 7. 3 energy
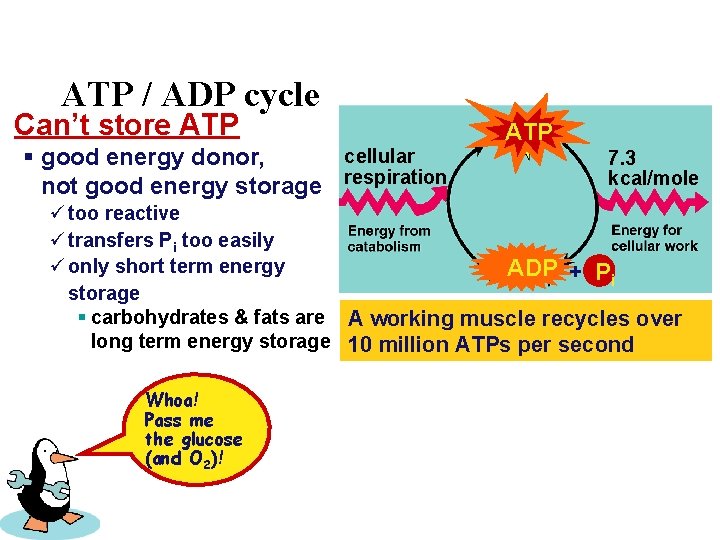
ATP / ADP cycle Can’t store ATP cellular § good energy donor, not good energy storage respiration ATP 7. 3 kcal/mole ü too reactive ü transfers Pi too easily ü only short term energy ADP + Pi storage § carbohydrates & fats are A working muscle recycles over long term energy storage 10 million ATPs per second Whoa! Pass me the glucose (and O 2)!
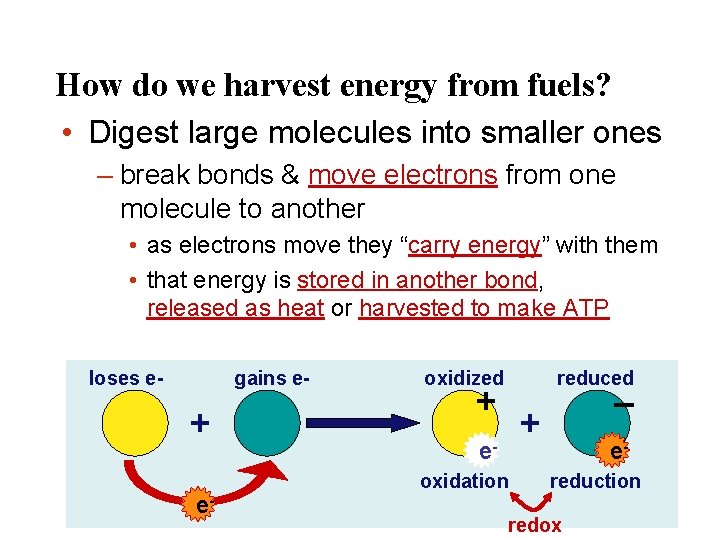
How do we harvest energy from fuels? • Digest large molecules into smaller ones – break bonds & move electrons from one molecule to another • as electrons move they “carry energy” with them • that energy is stored in another bond, released as heat or harvested to make ATP loses e- gains e- + e- oxidized reduced + – + eoxidation ereduction redox

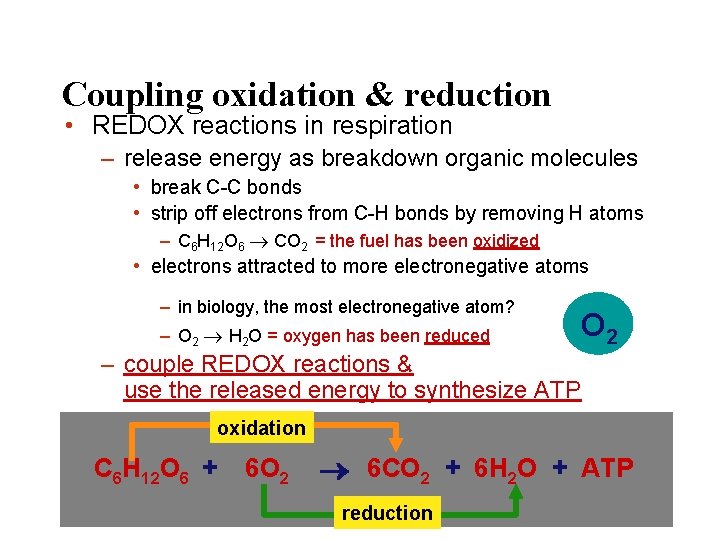
Coupling oxidation & reduction • REDOX reactions in respiration – release energy as breakdown organic molecules • break C-C bonds • strip off electrons from C-H bonds by removing H atoms – C 6 H 12 O 6 CO 2 = the fuel has been oxidized • electrons attracted to more electronegative atoms – in biology, the most electronegative atom? – O 2 H 2 O = oxygen has been reduced O 2 – couple REDOX reactions & use the released energy to synthesize ATP oxidation C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + ATP reduction
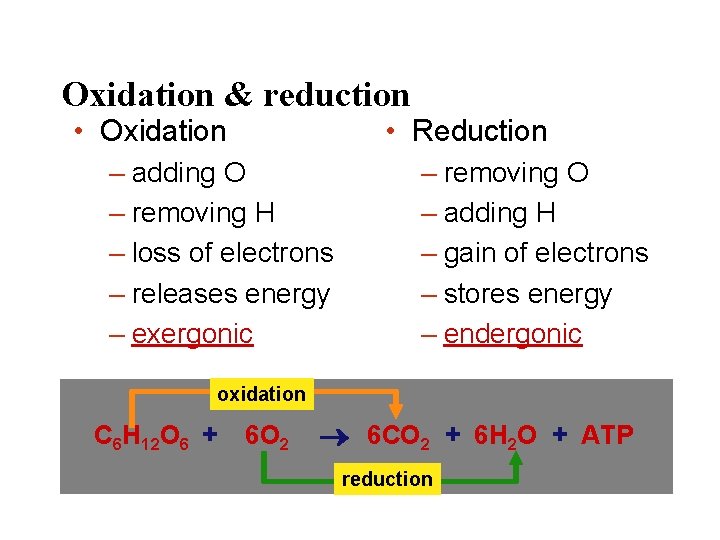
Oxidation & reduction • Oxidation • Reduction – adding O – removing H – loss of electrons – releases energy – exergonic – removing O – adding H – gain of electrons – stores energy – endergonic oxidation C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + ATP reduction
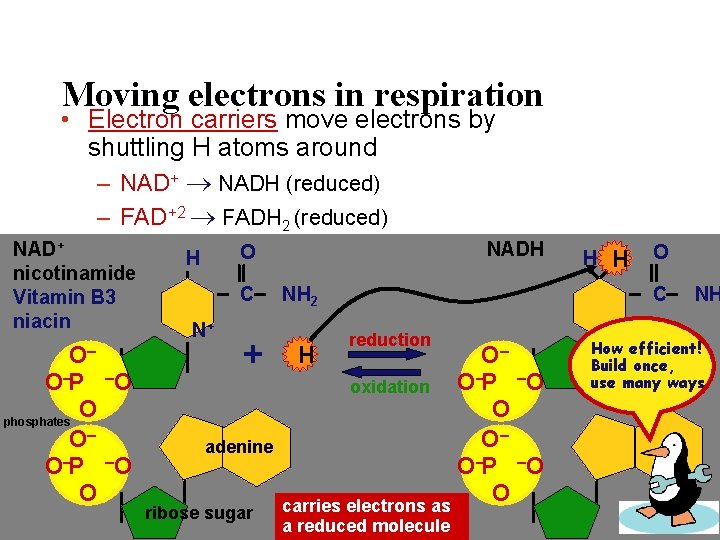
Moving electrons in respiration • Electron carriers move electrons by shuttling H atoms around – NAD+ NADH (reduced) – FAD+2 FADH 2 (reduced) NAD+ nicotinamide Vitamin B 3 niacin O– O–P –O O phosphates O– O–P –O O NADH O H C N+ + adenine ribose sugar H H O NH 2 H C reduction O– – –O oxidation O P O O– O–P –O O carries electrons as a reduced molecule N+ NH How efficient! Build once, use many ways
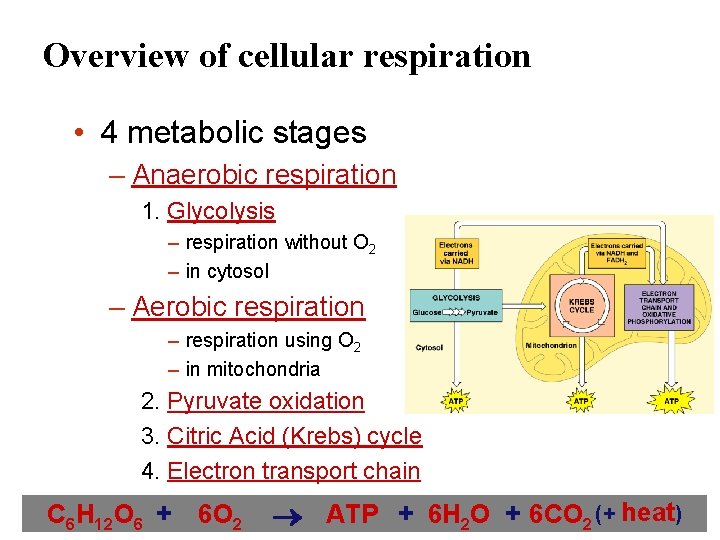
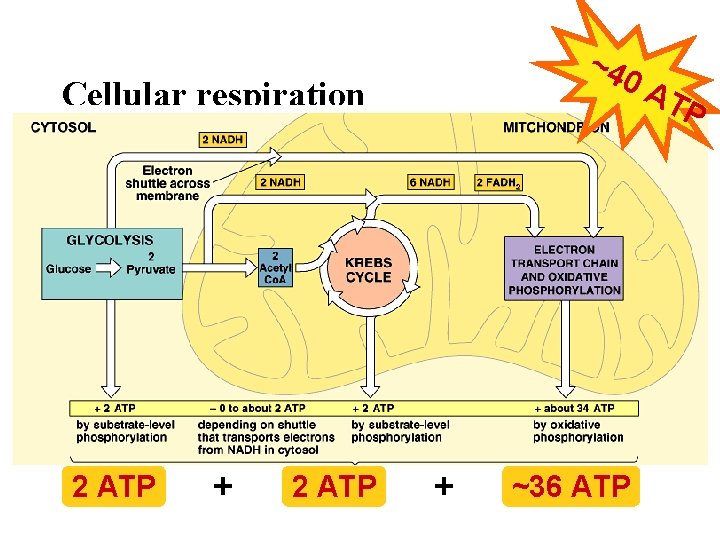
Overview of cellular respiration • 4 metabolic stages – Anaerobic respiration 1. Glycolysis – respiration without O 2 – in cytosol – Aerobic respiration – respiration using O 2 – in mitochondria 2. Pyruvate oxidation 3. Citric Acid (Krebs) cycle 4. Electron transport chain C 6 H 12 O 6 + 6 O 2 ATP + 6 H 2 O + 6 CO 2 (+ heat)

Concept 9. 2: Glycolysis harvests chemical energy by oxidizing glucose to pyruvate • Glycolysis (“splitting of sugar”) breaks down glucose into two molecules of pyruvate • Glycolysis occurs in the cytoplasm and has two major phases – Energy investment phase – Energy payoff phase • Glycolysis occurs whether or not O 2 is present © 2011 Pearson Education, Inc.
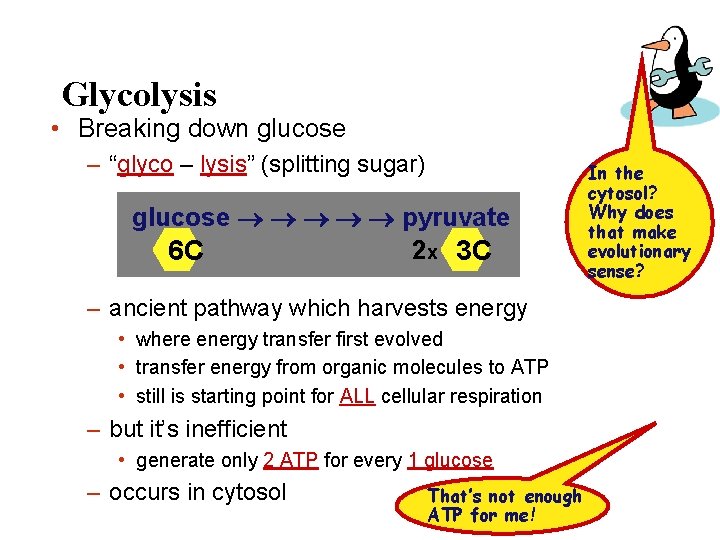
Glycolysis • Breaking down glucose – “glyco – lysis” (splitting sugar) glucose pyruvate 2 x 3 C 6 C – ancient pathway which harvests energy • where energy transfer first evolved • transfer energy from organic molecules to ATP • still is starting point for ALL cellular respiration – but it’s inefficient • generate only 2 ATP for every 1 glucose – occurs in cytosol That’s not enough ATP for me! In the cytosol? Why does that make evolutionary sense?

Evolutionary perspective • Prokaryotes – first cells had no organelles Enzymes of glycolysis are “well-conserved” • Anaerobic atmosphere – life on Earth first evolved without free oxygen (O 2) in atmosphere – energy had to be captured from organic molecules in absence of O 2 • Prokaryotes that evolved glycolysis are ancestors of all modern life – ALL cells still utilize glycolysis You mean we’re related? Do I have to invite them over for the holidays?
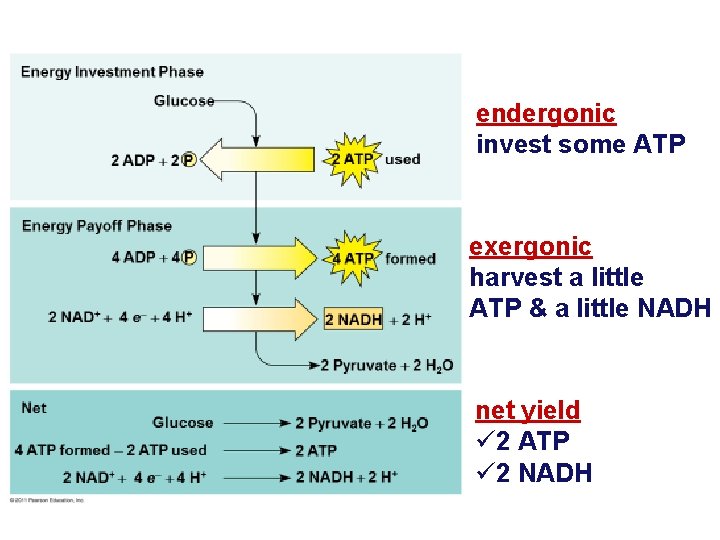
endergonic invest some ATP exergonic harvest a little ATP & a little NADH net yield ü 2 ATP ü 2 NADH
If you get bored……but you don’t need to know this!!!

The Evolutionary Significance of Glycolysis • Ancient prokaryotes are thought to have used glycolysis long before there was oxygen in the atmosphere • Very little O 2 was available in the atmosphere until about 2. 7 billion years ago, so early prokaryotes likely used only glycolysis to generate ATP • Glycolysis is a very ancient process © 2011 Pearson Education, Inc.
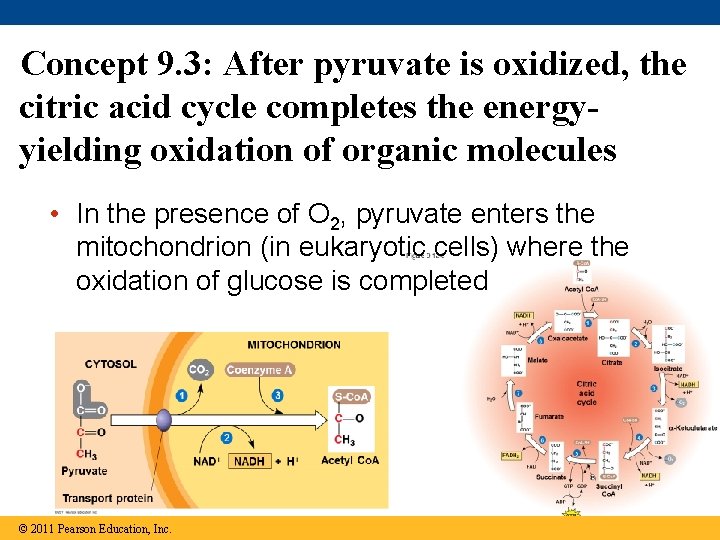
Concept 9. 3: After pyruvate is oxidized, the citric acid cycle completes the energyyielding oxidation of organic molecules • In the presence of O 2, pyruvate enters the mitochondrion (in eukaryotic cells) where the oxidation of glucose is completed © 2011 Pearson Education, Inc.
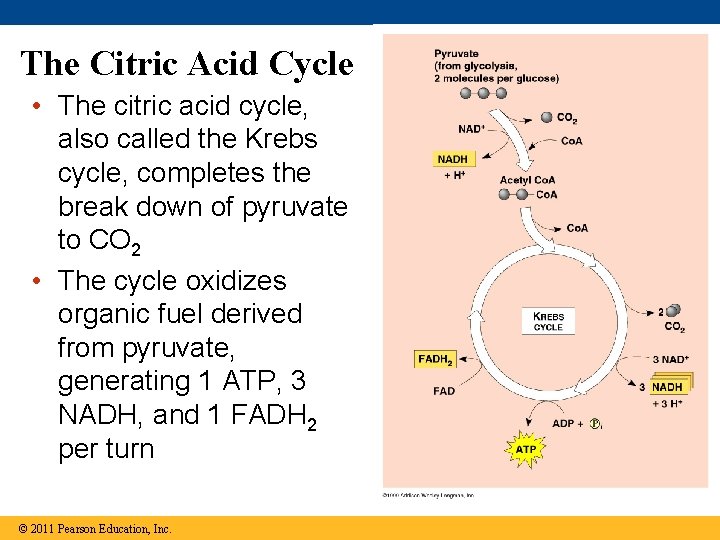
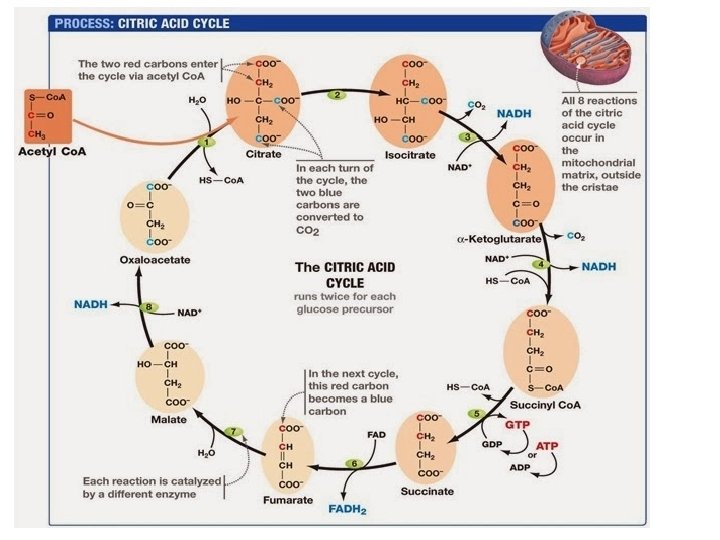
The Citric Acid Cycle • The citric acid cycle, also called the Krebs cycle, completes the break down of pyruvate to CO 2 • The cycle oxidizes organic fuel derived from pyruvate, generating 1 ATP, 3 NADH, and 1 FADH 2 per turn © 2011 Pearson Education, Inc.
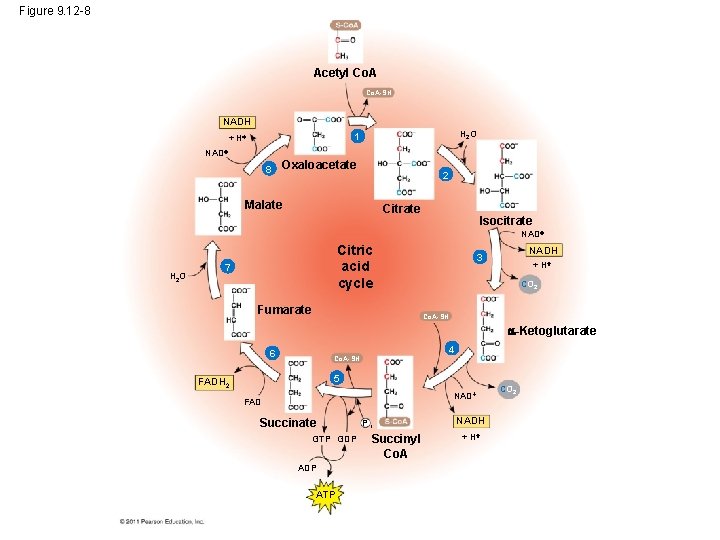
Figure 9. 12 -8 Acetyl Co. A-SH NADH H 2 O 1 + H NAD 8 Oxaloacetate 2 Malate Citrate Isocitrate NAD H 2 O Citric acid cycle 7 Fumarate NADH 3 + H CO 2 Co. A-SH -Ketoglutarate 6 4 Co. A-SH 5 FADH 2 NAD FAD Succinate GTP GDP ATP Pi Succinyl Co. A NADH + H CO 2
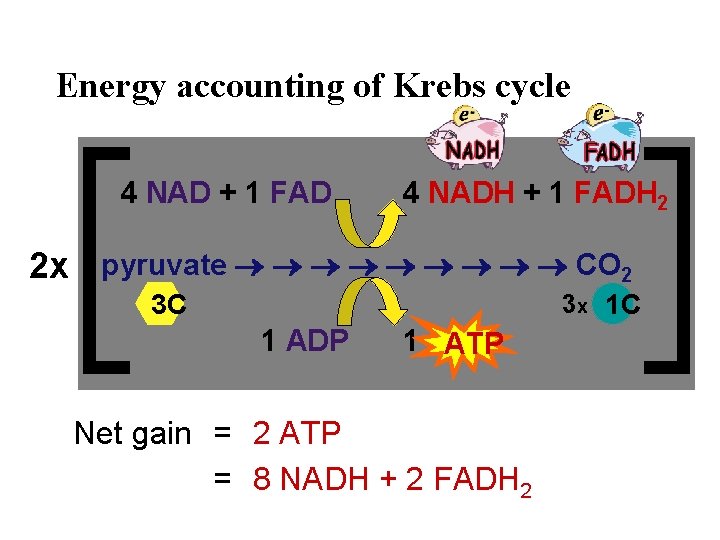
Energy accounting of Krebs cycle 4 NAD + 1 FAD 4 NADH + 1 FADH 2 2 x pyruvate CO 2 3 x 1 C 3 C 1 ADP 1 ATP Net gain = 2 ATP = 8 NADH + 2 FADH 2
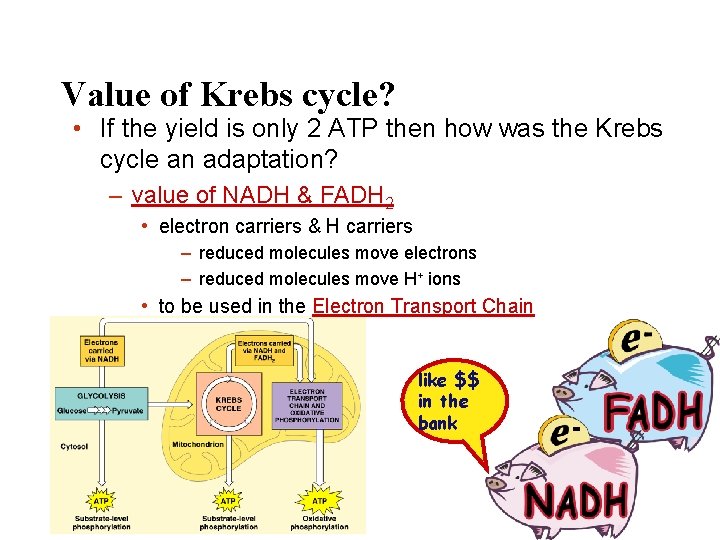
Value of Krebs cycle? • If the yield is only 2 ATP then how was the Krebs cycle an adaptation? – value of NADH & FADH 2 • electron carriers & H carriers – reduced molecules move electrons – reduced molecules move H+ ions • to be used in the Electron Transport Chain like $$ in the bank
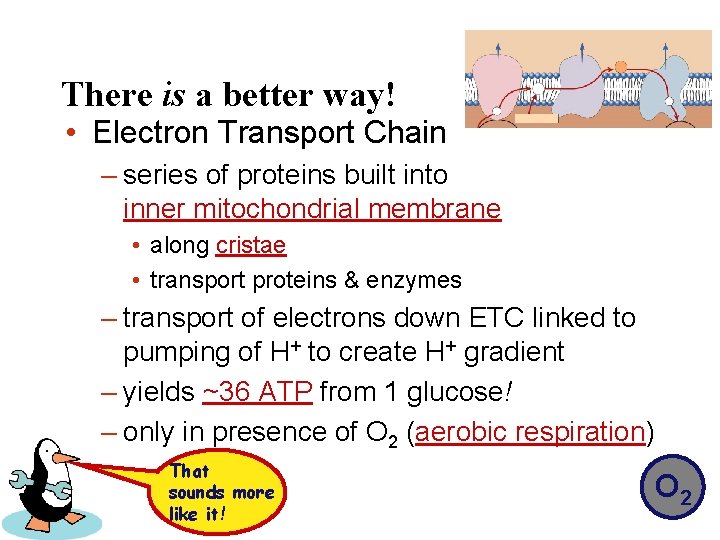
There is a better way! • Electron Transport Chain – series of proteins built into inner mitochondrial membrane • along cristae • transport proteins & enzymes – transport of electrons down ETC linked to pumping of H+ to create H+ gradient – yields ~36 ATP from 1 glucose! – only in presence of O 2 (aerobic respiration) That sounds more like it! O 2

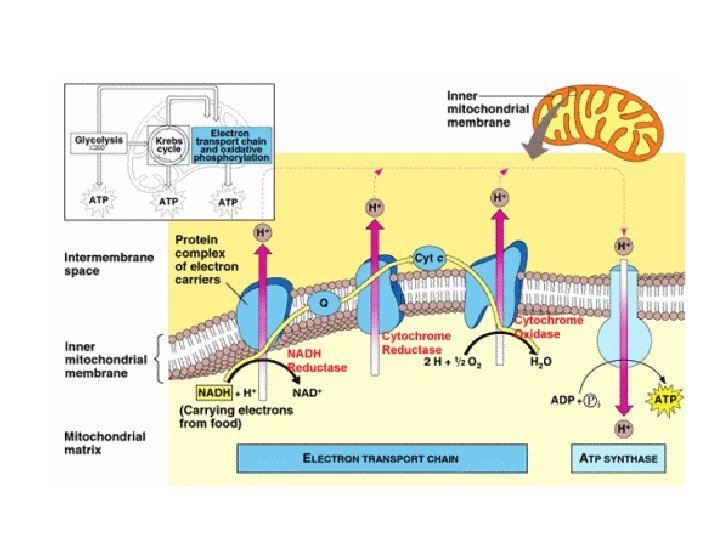
Concept 9. 4: During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis • NADH and FADH 2 account for most of the energy extracted from food • They donate electrons to the electron transport chain, • powers ATP synthesis via oxidative phosphorylation © 2011 Pearson Education, Inc.
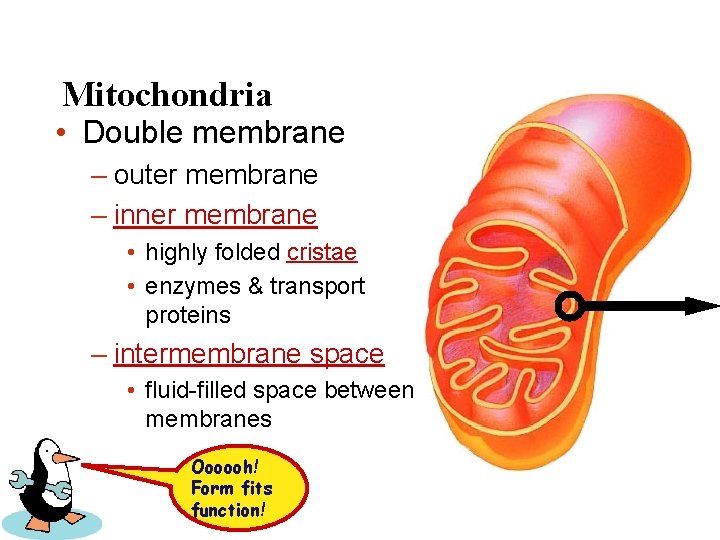
Mitochondria • Double membrane – outer membrane – inner membrane • highly folded cristae • enzymes & transport proteins – intermembrane space • fluid-filled space between membranes Oooooh! Form fits function!

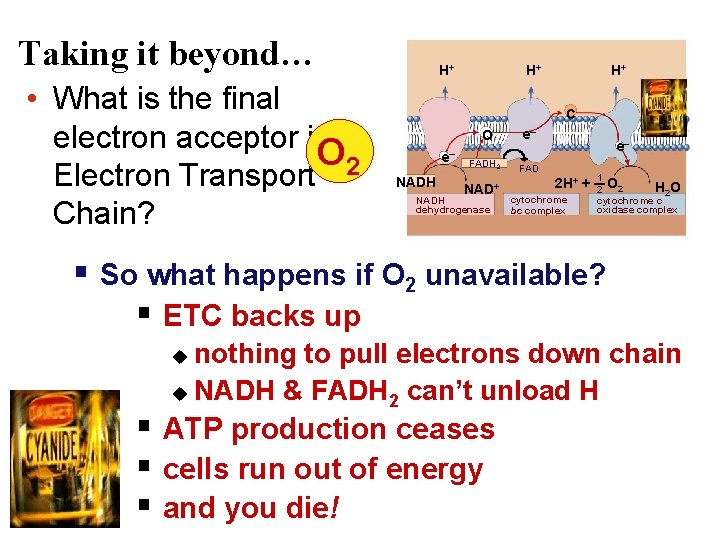
Electrons flow downhill • Electrons move in steps from carrier to carrier downhill to oxygen – each carrier (most are proteins – except Co. Q)more electronegative – controlled oxidation (alternate red/ox) – controlled release of energy make ATP instead of fire!
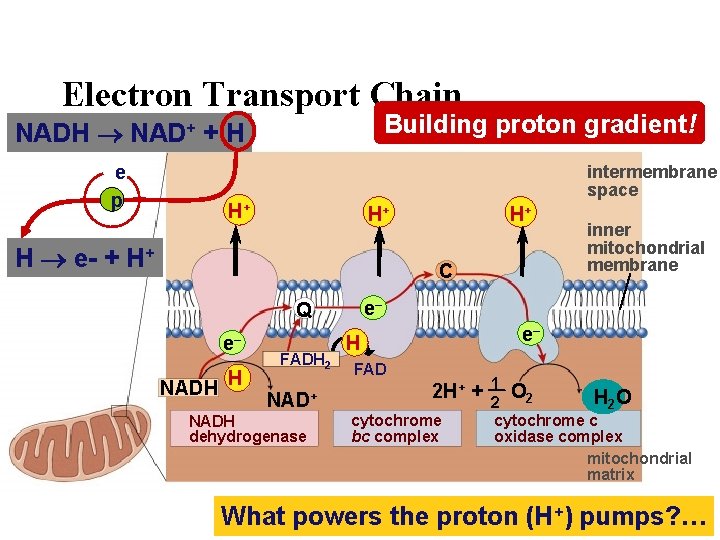
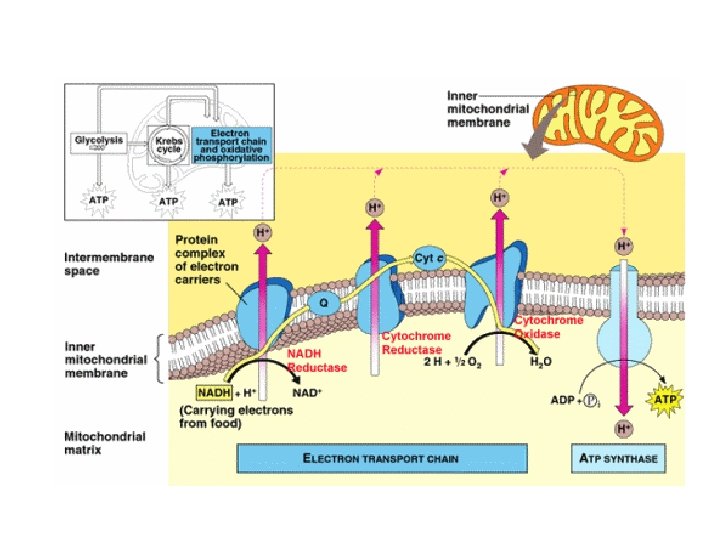
Electron Transport Chain Building proton gradient! NADH NAD+ + H e p intermembrane space H+ H+ H e- + H+ H+ C e– Q e– NADH H FADH 2 NAD+ NADH dehydrogenase inner mitochondrial membrane e– H FAD 2 H+ + cytochrome bc complex 1 O 2 H 2 O 2 cytochrome c oxidase complex mitochondrial matrix What powers the proton (H+) pumps? …
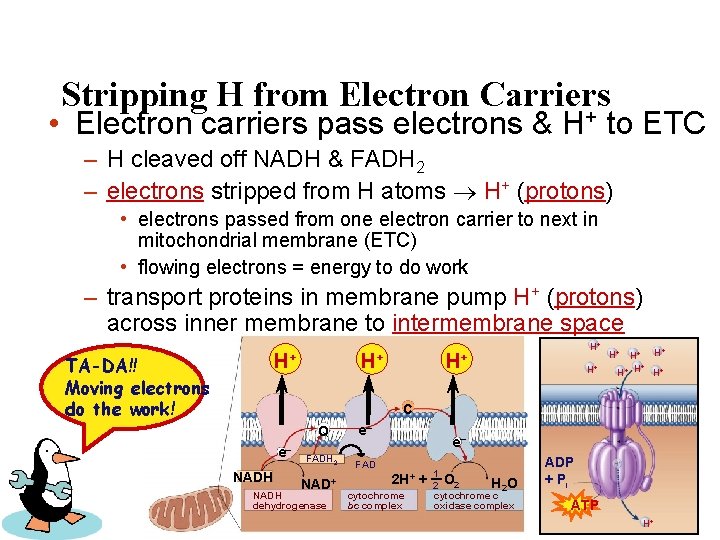
Stripping H from Electron Carriers + • Electron carriers pass electrons & H to ETC – H cleaved off NADH & FADH 2 – electrons stripped from H atoms H+ (protons) • electrons passed from one electron carrier to next in mitochondrial membrane (ETC) • flowing electrons = energy to do work – transport proteins in membrane pump H+ (protons) across inner membrane to intermembrane space + H H+ TA-DA!! Moving electrons do the work! + H H+ H+ H+ H H+ C e– NADH Q e– FADH 2 FAD NAD+ NADH dehydrogenase e– 2 H+ cytochrome bc complex + 1 H 2 O 2 cytochrome c oxidase complex ADP + Pi ATP H+
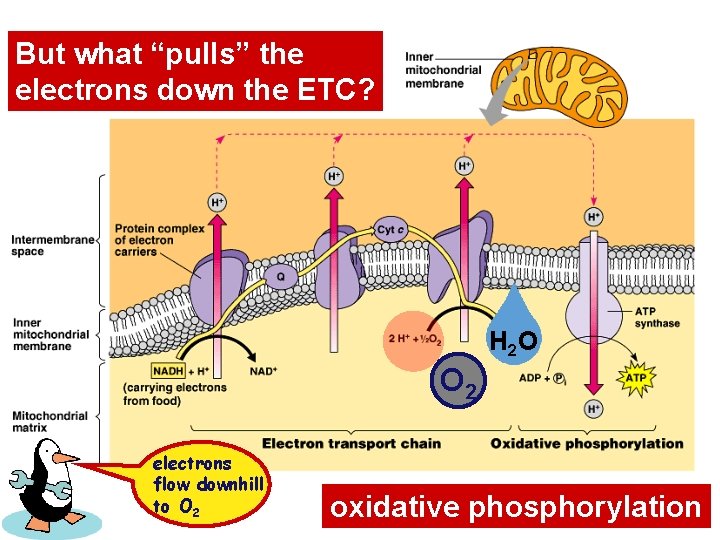
But what “pulls” the electrons down the ETC? H 2 O O 2 electrons flow downhill to O 2 oxidative phosphorylation
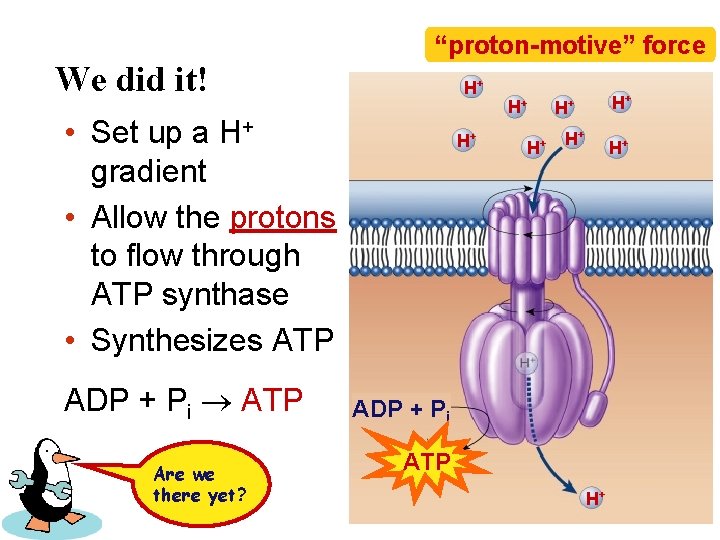
We did it! “proton-motive” force H+ • Set up a H+ gradient • Allow the protons to flow through ATP synthase • Synthesizes ATP ADP + Pi ATP Are we there yet? H+ H+ ADP + Pi ATP H+
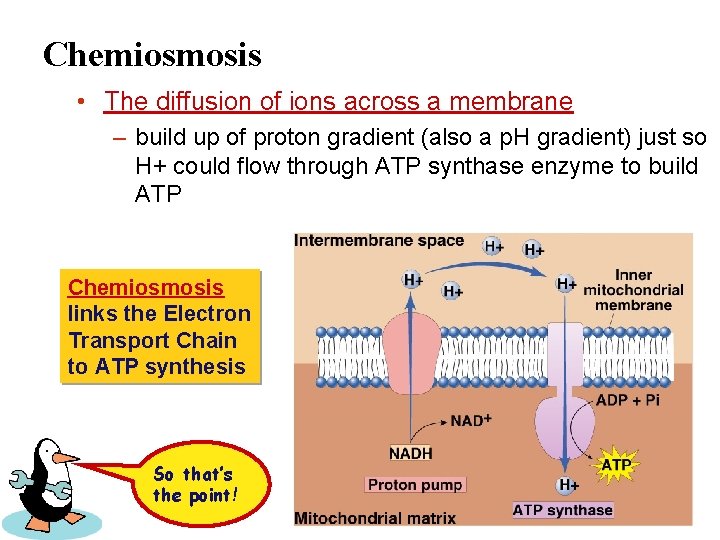
Chemiosmosis • The diffusion of ions across a membrane – build up of proton gradient (also a p. H gradient) just so H+ could flow through ATP synthase enzyme to build ATP Chemiosmosis links the Electron Transport Chain to ATP synthesis So that’s the point!
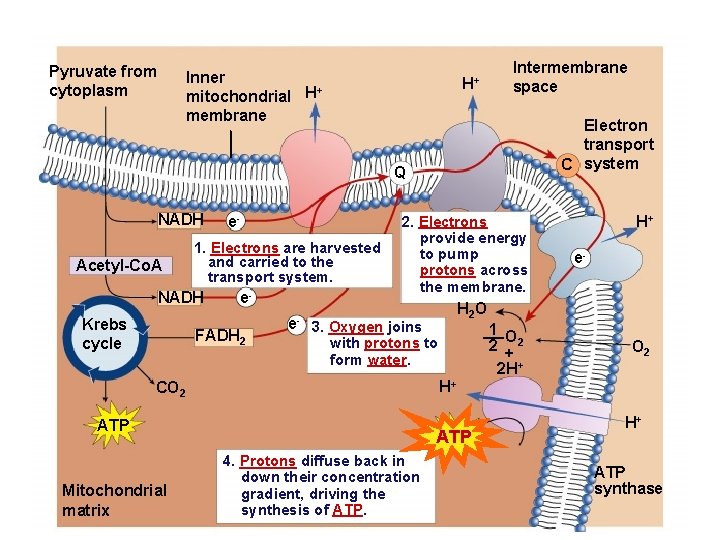
Pyruvate from cytoplasm Inner + mitochondrial H membrane H+ Intermembrane space Electron transport C system Q NADH Acetyl-Co. A 1. Electrons are harvested and carried to the transport system. NADH Krebs cycle e- e- FADH 2 e- 2. Electrons provide energy to pump protons across the membrane. e- H 2 O 3. Oxygen joins with protons to form water. 1 O 2 +2 2 H+ O 2 H+ CO 2 ATP Mitochondrial matrix H+ ATP 4. Protons diffuse back in down their concentration gradient, driving the synthesis of ATP. H+ ATP synthase
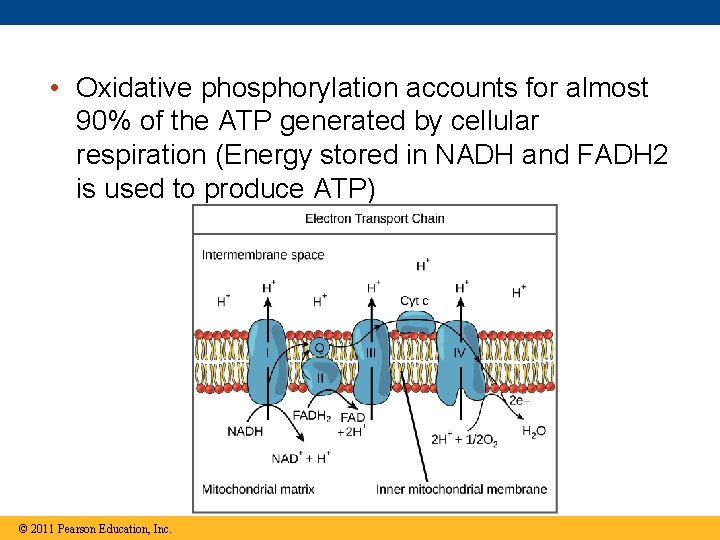
• Oxidative phosphorylation accounts for almost 90% of the ATP generated by cellular respiration (Energy stored in NADH and FADH 2 is used to produce ATP) © 2011 Pearson Education, Inc.
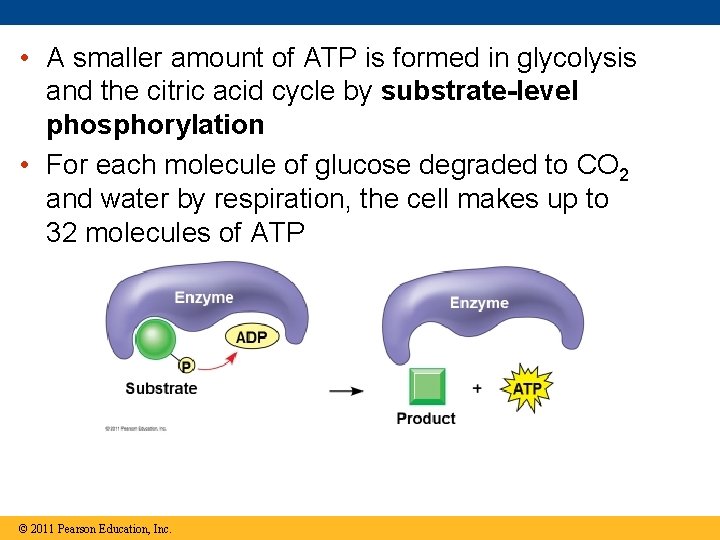
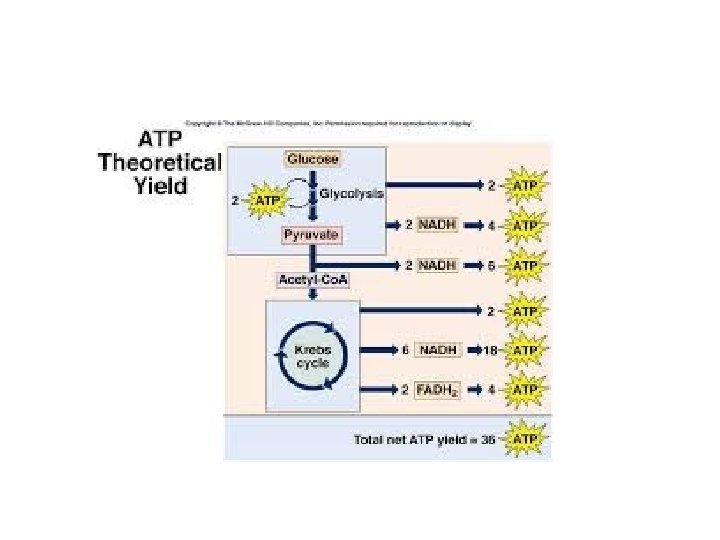
• A smaller amount of ATP is formed in glycolysis and the citric acid cycle by substrate-level phosphorylation • For each molecule of glucose degraded to CO 2 and water by respiration, the cell makes up to 32 molecules of ATP © 2011 Pearson Education, Inc.
~4 0 A Cellular respiration 2 ATP + ~36 ATP TP
Taking it beyond… • What is the final electron acceptor in O 2 Electron Transport Chain? H+ H+ H+ C e– NADH Q e– FADH 2 FAD NAD+ NADH dehydrogenase e– 2 H+ + cytochrome bc complex 1 H 2 O 2 cytochrome c oxidase complex § So what happens if O 2 unavailable? § ETC backs up nothing to pull electrons down chain u NADH & FADH 2 can’t unload H u § ATP production ceases § cells run out of energy § and you die!

An Accounting of ATP Production by Cellular Respiration • During cellular respiration, most energy flows in this sequence: glucose NADH electron transport chain proton-motive force ATP • About 34% of the energy in a glucose molecule is transferred to ATP during cellular respiration, making about 32 ATP © 2011 Pearson Education, Inc.
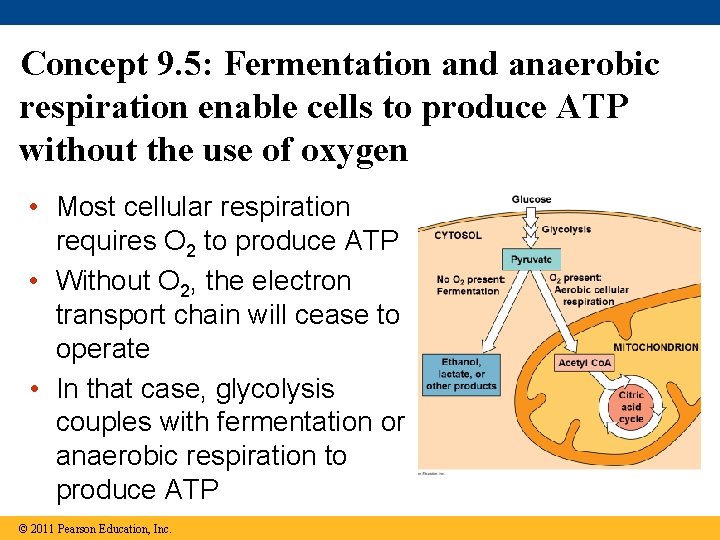
Concept 9. 5: Fermentation and anaerobic respiration enable cells to produce ATP without the use of oxygen • Most cellular respiration requires O 2 to produce ATP • Without O 2, the electron transport chain will cease to operate • In that case, glycolysis couples with fermentation or anaerobic respiration to produce ATP © 2011 Pearson Education, Inc.
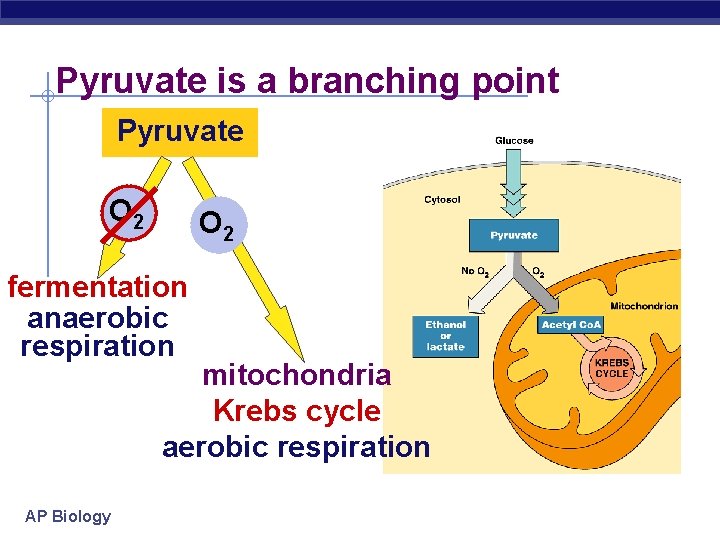
Pyruvate is a branching point Pyruvate O 2 fermentation anaerobic respiration mitochondria Krebs cycle aerobic respiration AP Biology
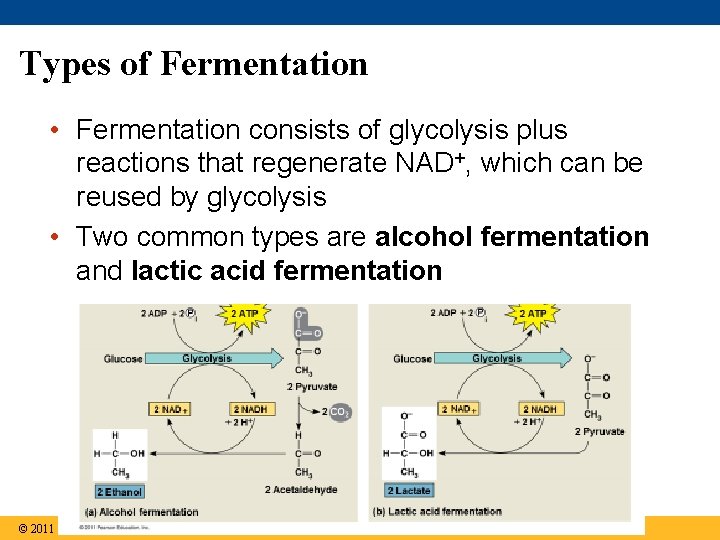
Types of Fermentation • Fermentation consists of glycolysis plus reactions that regenerate NAD+, which can be reused by glycolysis • Two common types are alcohol fermentation and lactic acid fermentation © 2011 Pearson Education, Inc.
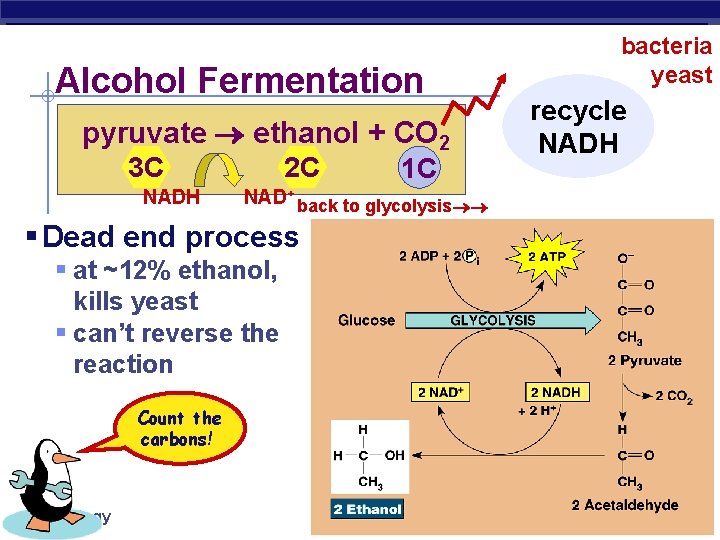
Alcohol Fermentation pyruvate ethanol + CO 2 3 C NADH 2 C NAD+ back to glycolysis § Dead end process § at ~12% ethanol, kills yeast § can’t reverse the reaction Count the carbons! AP Biology 1 C bacteria yeast recycle NADH
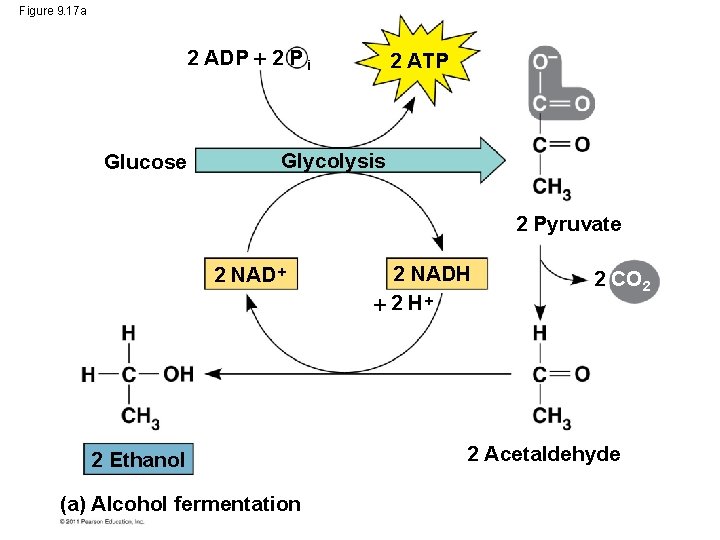
Figure 9. 17 a 2 ADP 2 P i Glucose 2 ATP Glycolysis 2 Pyruvate 2 NAD 2 Ethanol (a) Alcohol fermentation 2 NADH 2 CO 2 2 Acetaldehyde
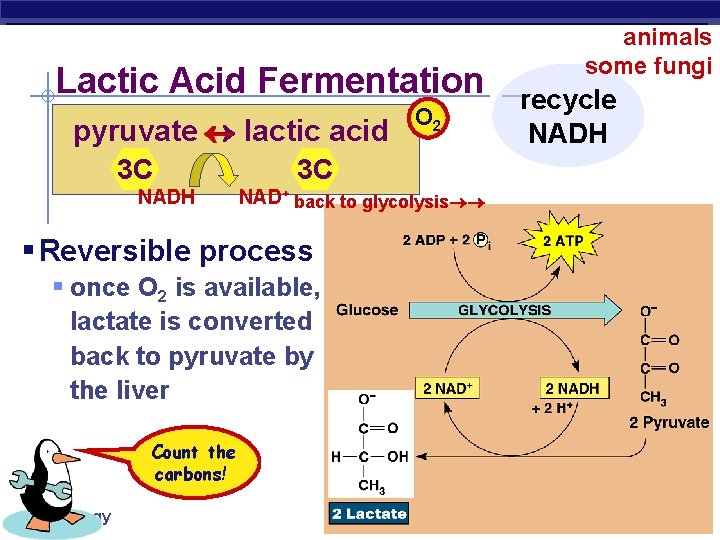
Lactic Acid Fermentation pyruvate lactic acid 3 C NADH 3 C NAD+ back to glycolysis § Reversible process § once O 2 is available, lactate is converted back to pyruvate by the liver Count the carbons! AP Biology O 2 animals some fungi recycle NADH
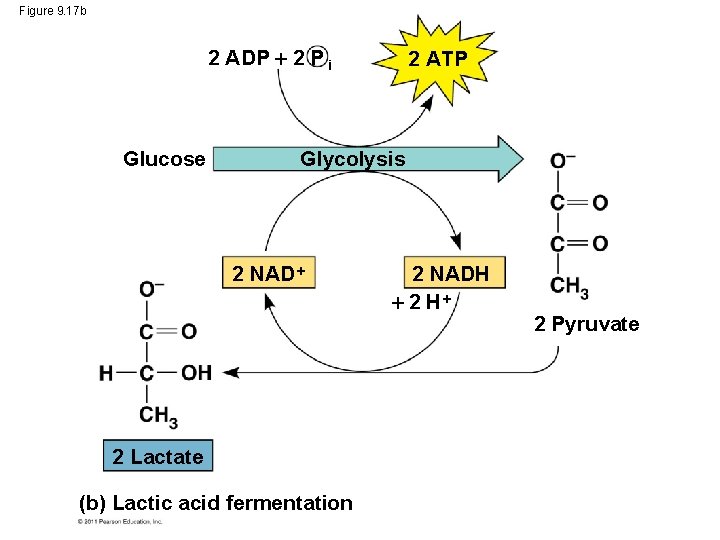
Figure 9. 17 b 2 ADP 2 P i Glucose 2 ATP Glycolysis 2 NAD 2 Lactate (b) Lactic acid fermentation 2 NADH 2 Pyruvate

• In lactic acid fermentation, pyruvate is reduced to NADH, forming lactate as an end product, with no release of CO 2 • Lactic acid fermentation by some fungi and bacteria is used to make cheese and yogurt • Human muscle cells use lactic acid fermentation to generate ATP when O 2 is scarce © 2011 Pearson Education, Inc.

• Anaerobic respiration uses an electron transport chain with a final electron acceptor other than O 2, for example sulfate • Fermentation uses substrate-level phosphorylation instead of an electron transport chain to generate ATP © 2011 Pearson Education, Inc.
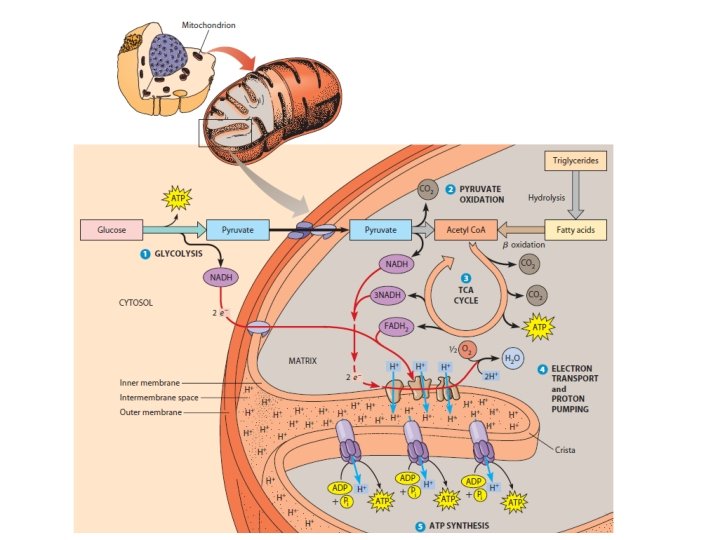

Comparing Fermentation with Anaerobic and Aerobic Respiration • All use glycolysis (net ATP = 2) to oxidize glucose and harvest chemical energy of food • In all three, NAD+ is the oxidizing agent that accepts electrons during glycolysis • The processes have different final electron acceptors: an organic molecule (such as pyruvate or acetaldehyde) in fermentation and O 2 in cellular respiration • Cellular respiration produces 32 ATP per glucose molecule; fermentation produces 2 ATP per glucose molecule © 2011 Pearson Education, Inc.

• Obligate anaerobes carry out fermentation or anaerobic respiration and cannot survive in the presence of O 2 • Yeast and many bacteria are facultative anaerobes, meaning that they can survive using either fermentation or cellular respiration • In a facultative anaerobe, pyruvate is a fork in the metabolic road that leads to two alternative catabolic routes © 2011 Pearson Education, Inc.
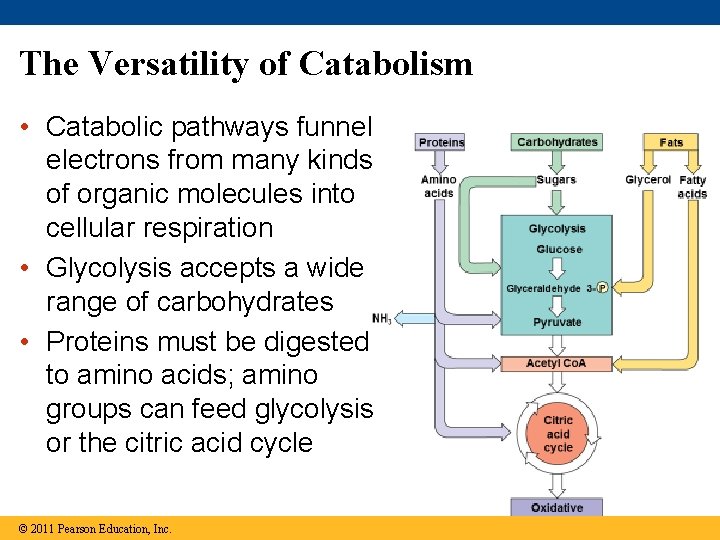
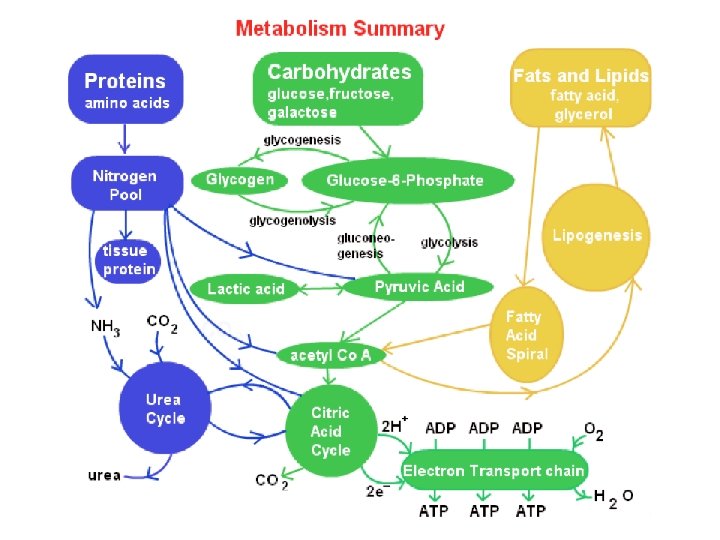
The Versatility of Catabolism • Catabolic pathways funnel electrons from many kinds of organic molecules into cellular respiration • Glycolysis accepts a wide range of carbohydrates • Proteins must be digested to amino acids; amino groups can feed glycolysis or the citric acid cycle © 2011 Pearson Education, Inc.
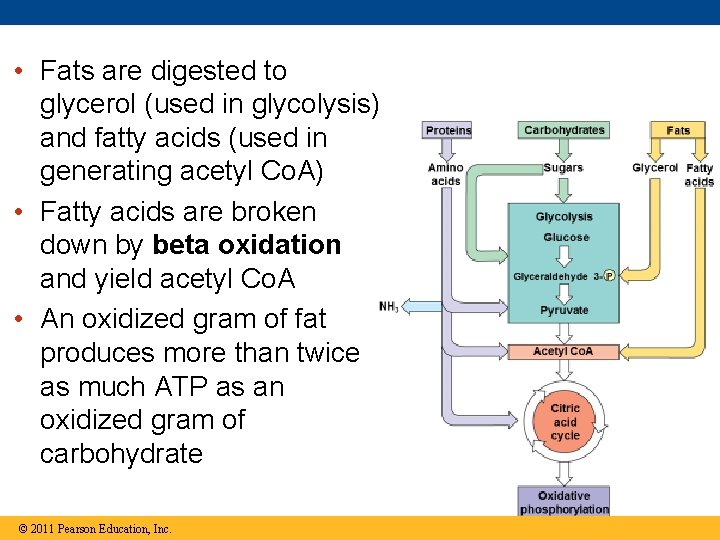
• Fats are digested to glycerol (used in glycolysis) and fatty acids (used in generating acetyl Co. A) • Fatty acids are broken down by beta oxidation and yield acetyl Co. A • An oxidized gram of fat produces more than twice as much ATP as an oxidized gram of carbohydrate © 2011 Pearson Education, Inc.
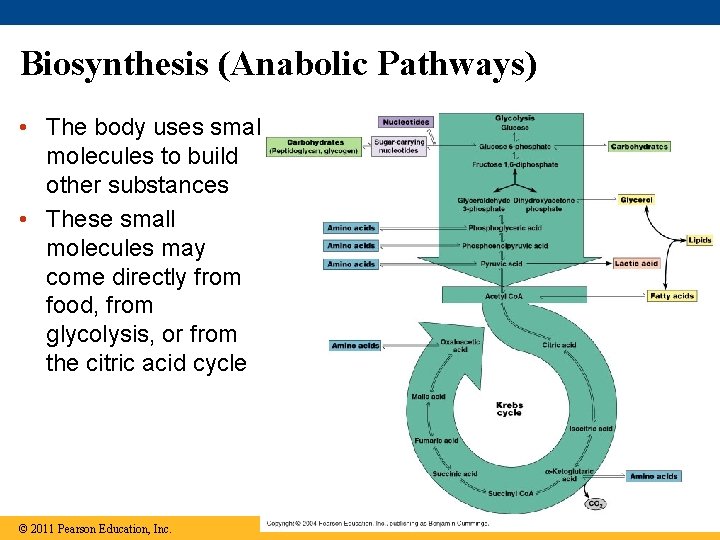
Biosynthesis (Anabolic Pathways) • The body uses small molecules to build other substances • These small molecules may come directly from food, from glycolysis, or from the citric acid cycle © 2011 Pearson Education, Inc.
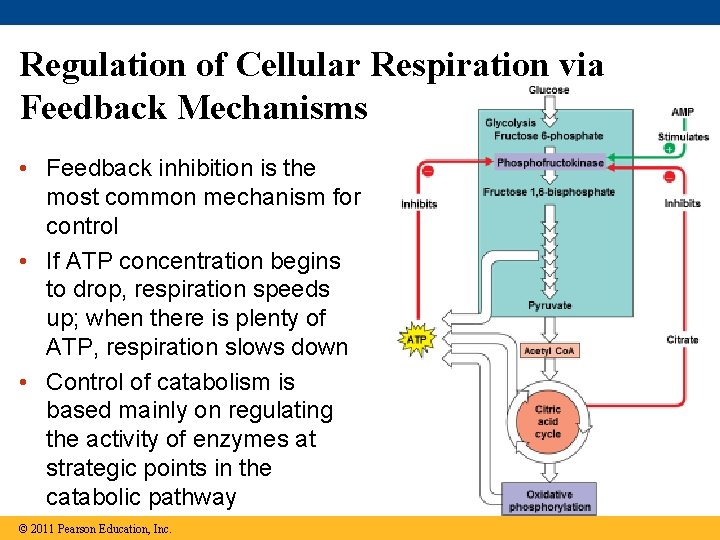
Regulation of Cellular Respiration via Feedback Mechanisms • Feedback inhibition is the most common mechanism for control • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway © 2011 Pearson Education, Inc.
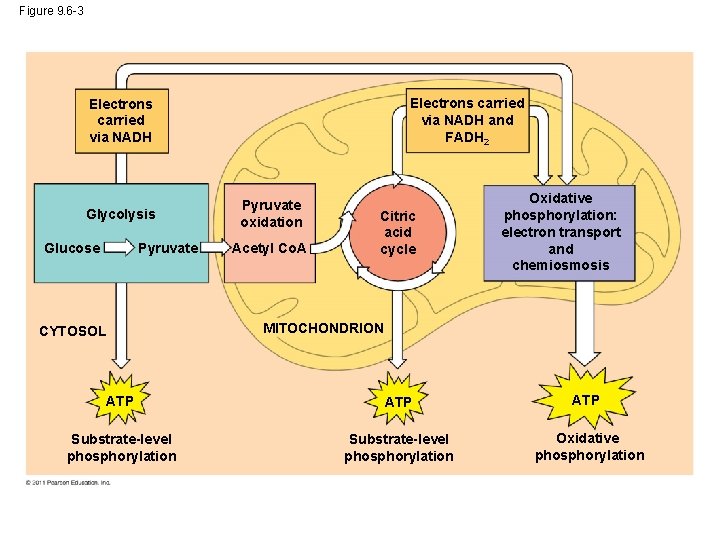
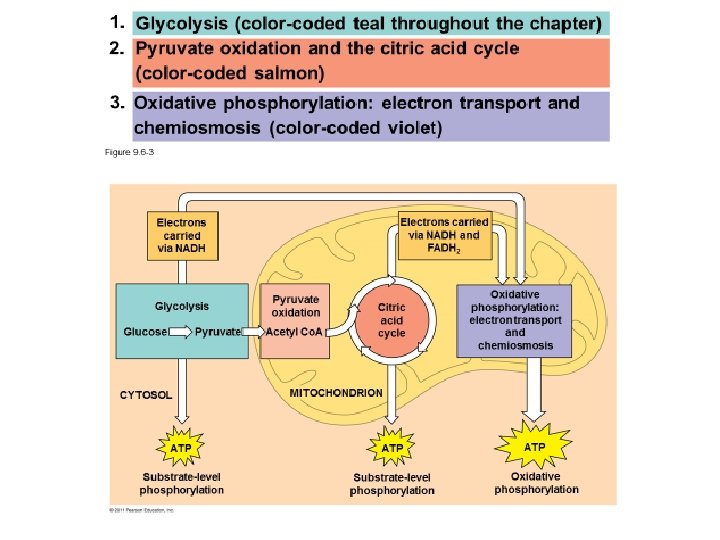
Figure 9. 6 -3 Electrons carried via NADH and FADH 2 Electrons carried via NADH Glycolysis Glucose Pyruvate CYTOSOL Pyruvate oxidation Acetyl Co. A Citric acid cycle Oxidative phosphorylation: electron transport and chemiosmosis MITOCHONDRION ATP ATP Substrate-level phosphorylation Oxidative phosphorylation
- Slides: 63