Lecture Presentation Chapter 9 Solutions Karen C Timberlake

Lecture Presentation Chapter 9 Solutions Karen C. Timberlake General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Topics covered in this Chapter • 9. 1 Solutions • 9. 2 Electrolytes and Nonelectrolytes • 9. 3 Solubility • 9. 4 Solution Concentrations and Reaction • 9. 5 Dilution of Solutions • 9. 6 Properties of Solutions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 9 Solutions A dialysis nurse informs Michelle that • her side effects are due to her body’s inability to regulate the amount of water in her cells. • the amount of water in her body fluids is regulated by the concentration of electrolytes and the rate at which waste products are removed from her body. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 9 Readiness Key Math Skills • Calculating a Percentage (1. 4 C) • Solving Equations (1. 4 D) • Interpreting a Line Graph (1. 4 E) Core Chemistry Skills • Writing Conversion Factors from Conversion Equalities (2. 5) • Using Conversion Factors (2. 6) • Identifying Attractive Forces (6. 8) • Using Mole–Mole Factors (7. 6) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

9. 1 Solutions • are homogeneous mixtures of two or more substances. • form when there is sufficient attraction between the solute and solvent molecules. • have two components: the solvent, present in a larger amount, and the solute, present in a smaller amount. Learning Goal Identify the solute and solvent in a solution; describe the formation of a solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutes • may be a liquid, gas, or solid. • are spread evenly throughout the solution. • mix with solvents so the solute and solvent have the same physical state. • cannot be separated by filtration, but they can be separated by evaporation. • are not visible, but they can give a color to the solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
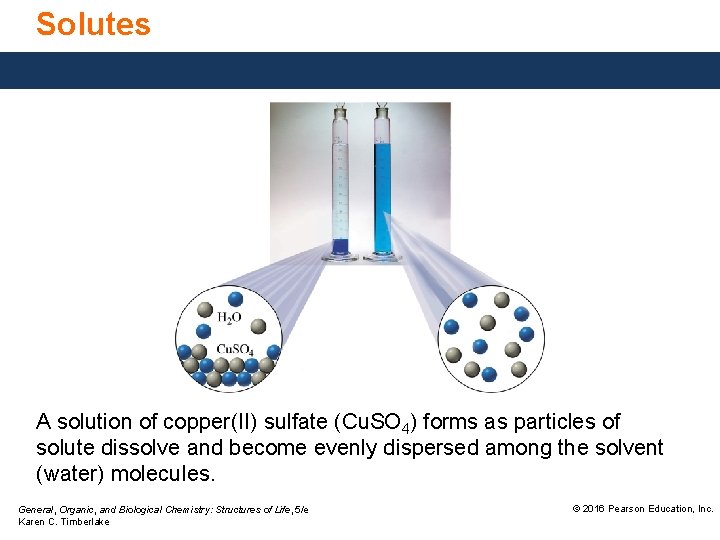
Solutes A solution of copper(II) sulfate (Cu. SO 4) forms as particles of solute dissolve and become evenly dispersed among the solvent (water) molecules. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
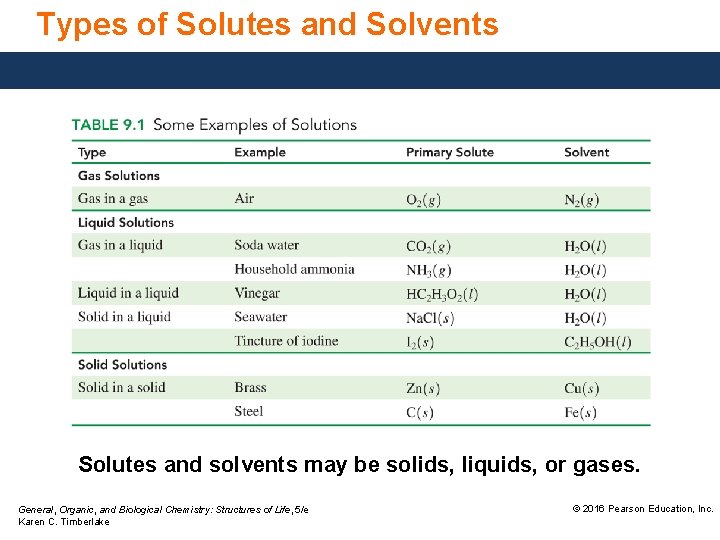
Types of Solutes and Solvents Solutes and solvents may be solids, liquids, or gases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Water as a Solvent Water • is one of the most common solvents in nature. • is a polar molecule due to polar O–H bonds. • molecules form hydrogen bonds important in many biological compounds. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Formation of Solutions form when the solute–solvent interactions are large enough to overcome the solute–solute interactions and the solvent–solvent interactions. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions, Like Dissolves Like Solutions will form when the solute and solvent have similar polarities: “like dissolves like. ” General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
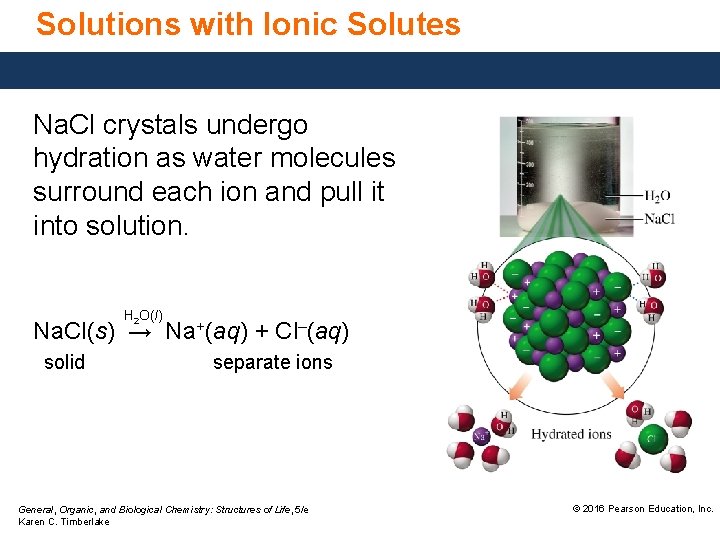
Solutions with Ionic Solutes Na. Cl crystals undergo hydration as water molecules surround each ion and pull it into solution. H 2 O(l) Na. Cl(s) → Na+(aq) + Cl–(aq) solid separate ions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions with Polar Solutes A polar molecular compound such as methanol, CH 3—OH, is soluble in water because methanol has a polar –OH group to form hydrogen bonds with water. Polar solutes require polar solvents for a solution to form. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions with Nonpolar Solutes Compounds containing nonpolar molecules, such as iodine (I 2), oil, or grease, do not dissolve in water because there are essentially no attractions between the particles of a nonpolar solute and the polar solvent. Nonpolar solutes require nonpolar solvents for a solution to form. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Identify the solute in each of the following solutions. A. 2 g of sugar and 100 m. L of water B. 60. 0 m. L of ethyl alcohol and 30. 0 m. L of methyl alcohol C. 55. 0 m. L of water and 1. 50 g of Na. Cl D. Air: 200 m. L of O 2 and 800 m. L of N 2 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check When solid Li. Cl is added to water it dissolves because A. the Li+ ions are attracted to the 1) oxygen atom ( −) of water. 2) hydrogen atom ( +) of water. B. the Cl− ions are attracted to the 1) oxygen atom ( −) of water. 2) hydrogen atom ( +) of water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Which of the following solutes will dissolve in water? Why? A. Na 2 SO 4 B. gasoline (nonpolar) C. I 2 D. HCl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

9. 2 Electrolytes and Nonelectrolytes Electrolytes in the body play an important role in maintaining the proper function of the cells and organs. Electrolytes such as sodium, potassium, chloride, bicarbonate can be measured in a blood test. Learning Goal Identify solutes as electrolytes or nonelectrolytes. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
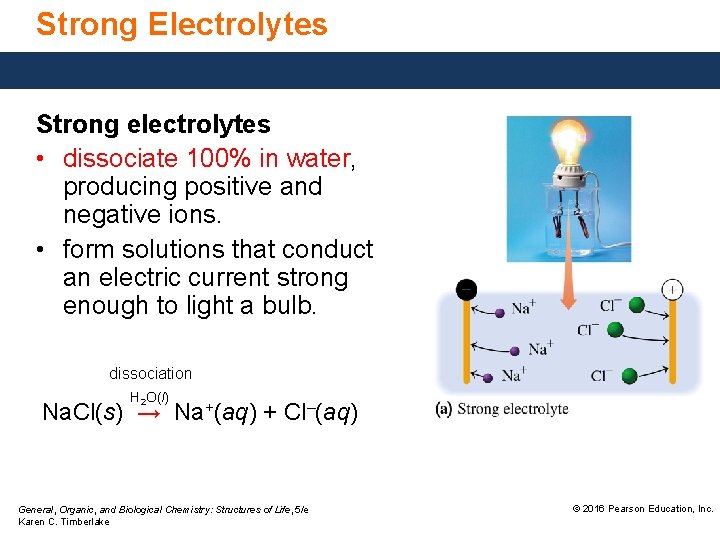
Strong Electrolytes Strong electrolytes • dissociate 100% in water, producing positive and negative ions. • form solutions that conduct an electric current strong enough to light a bulb. dissociation H 2 O(l) Na. Cl(s) → Na+(aq) + Cl–(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
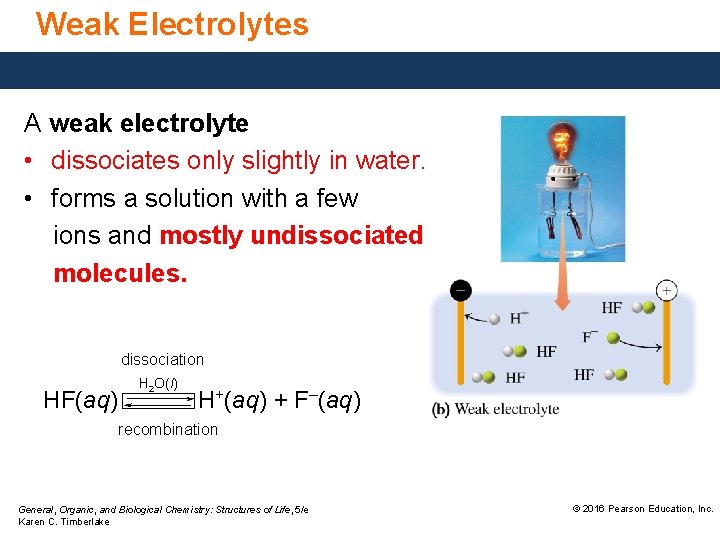
Weak Electrolytes A weak electrolyte • dissociates only slightly in water. • forms a solution with a few ions and mostly undissociated molecules. dissociation HF(aq) H 2 O(l) H+(aq) + F–(aq) recombination General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
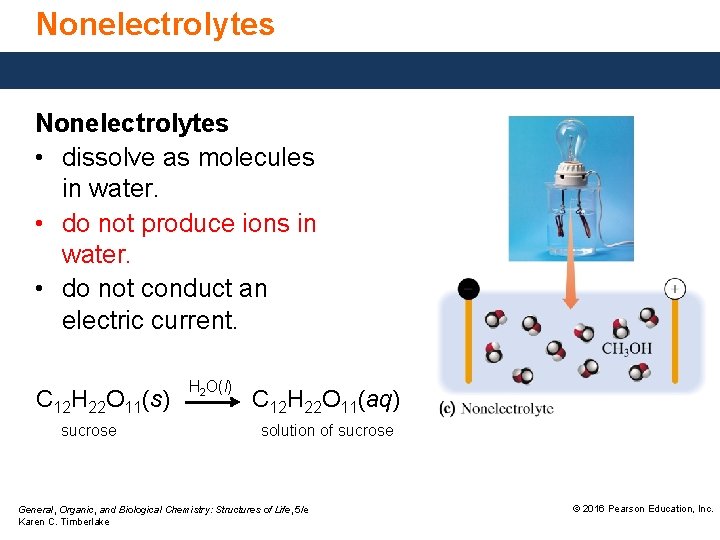
Nonelectrolytes • dissolve as molecules in water. • do not produce ions in water. • do not conduct an electric current. C 12 H 22 O 11(s) sucrose H 2 O(l) C 12 H 22 O 11(aq) solution of sucrose General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
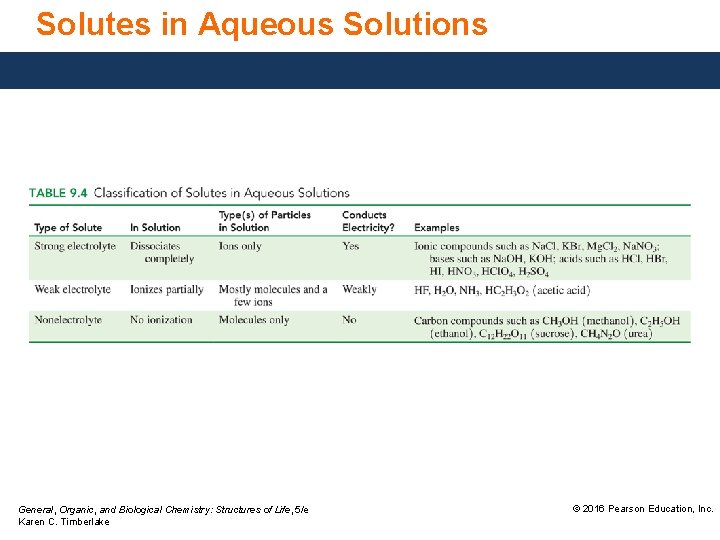
Solutes in Aqueous Solutions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Complete each for strong electrolytes in water. H 2 O(l) 1. Ca. Cl 2(s) _____ A. Ca. Cl 2(s) B. Ca 2+(aq) + Cl 2−(aq) C. Ca 2+(aq) + 2 Cl−(aq) H 2 O(l) 2. K 3 PO 4(s) ______ A. 3 K+(aq) + PO 43−(aq) B. K 3 PO 4(s) C. K 3+(aq) + P 3−(aq) + O 4−(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Which of the following reactions represents the dissociation of a strong electrolyte in water? A. NH 3(g) + H 2 O(l) NH 4 OH(aq) B. CH 3 OH(l) H 2 O(l) CH 3 OH(aq) C. Na 2 SO 4(s) H 2 O(l) 2 Na+(aq) + SO 42−(aq) D. C 2 H 5 OH(l) H 2 O(l) C 2 H 5 OH (aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Write the equation for the formation of a solution for each of the following: u The dissociation of K 2 Cr. O 4(s), a strong electrolyte, in water K 2 Cr. O 4(s) 2 K+(aq) + Cr. O 42−(aq) u The partial dissociation of the weak electrolyte H 3 PO 4(aq) in water H O(l) H 3 PO 4(s) H+(aq) + H 2 PO 4−(aq) u The dissolving of the solid sugar (C 12 H 22 O 11)(s) in water C 12 H 22 O 11(s) C 12 H 22 O 11(aq) 2 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Equivalents of Electrolytes An equivalent (Eq) is the amount of an electrolyte or an ion that provides 1 mole of electrical charge (+ or −). In solution, • the charge of the positive ions is always balanced by the charge of the negative ions. • the concentrations of electrolytes in intravenous fluids are expressed in milliequivalents per liter (m. Eq/L): 1 Eq = 1000 m. Eq General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Equivalents, Milliequivalenets For example, a solution containing • 25 m. Eq/L of Na+ and 4 m. Eq/L of K+ has a total positive charge of 29 m. Eq/L. • Cl− as the only anion must have a concentration of 29 m. Eq/ L. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Equivalents The laboratory tests for a patient indicate a blood calcium level of 8. 8 m. Eq/L. A. How many moles of calcium ion are in 0. 50 L of blood? × × We can then convert equivalents to moles (for Ca 2+ there are 2 Eq per mole). × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake mol © 2016 Pearson Education, Inc.

Calculating Equivalents The laboratory tests for a patient indicate a blood calcium level of 8. 8 m. Eq/L. B. If chloride ion is the only other ion present, what is its concentration in m. Eq/L? If the concentration of Ca 2+ is 8. 8 m. Eq/L, then the concentration of Cl− must be 8. 8 m. Eq/L to balance the charge. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chemistry Link to Health: Electrolytes in Body Fluids Typical concentrations of electrolytes in blood plasma • have a charge balance; the total number of positive charges is equal to the total number of negative charges. • varies due to the nutritional, electrolyte, and fluid needs of the patient. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
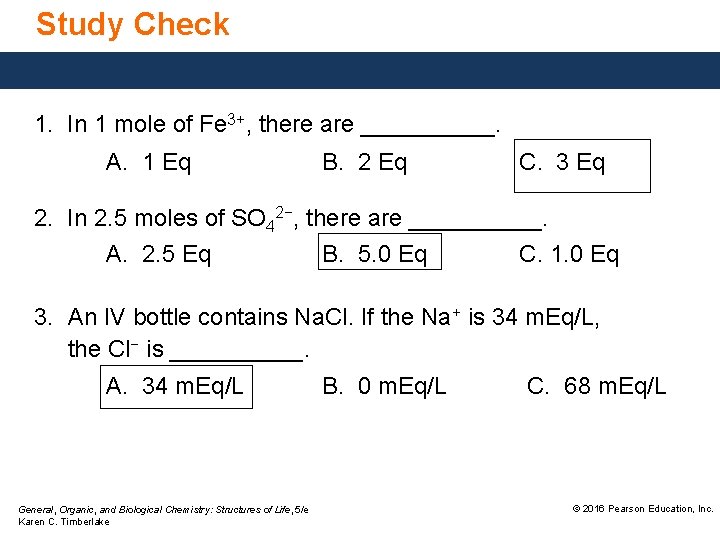
Study Check 1. In 1 mole of Fe 3+, there are _____. A. 1 Eq B. 2 Eq C. 3 Eq 2. In 2. 5 moles of SO 42−, there are _____. A. 2. 5 Eq B. 5. 0 Eq C. 1. 0 Eq 3. An IV bottle contains Na. Cl. If the Na+ is 34 m. Eq/L, the Cl− is _____. A. 34 m. Eq/L General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake B. 0 m. Eq/L C. 68 m. Eq/L © 2016 Pearson Education, Inc.
9. 3 Solubility Gout primarily affects adult men over the age of 40. Attacks of gout may occur when the concentration of uric acid in blood plasma exceeds its solubility of 7 mg/100 m. L of plasma at 37 °C. Learning Goal Define solubility; distinguish between an unsaturated and a saturated solution. Identify an ionic compound as soluble or insoluble. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solubility is • the maximum amount of solute that dissolves in a specific amount of solvent. • temperature sensitive for solutes. • expressed as grams of solute in 100 grams of solvent, usually water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Unsaturated Solution Unsaturated solutions • contain less than the maximum amount of solute. • can dissolve more solute. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Saturated Solution Saturated solutions • contain the maximum amount of solute that can dissolve. • have un-dissolved solute at the bottom of the container. • contain solute that dissolves as well as solute that recrystallizes in an equilibrium process. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Saturated Solution More solute can dissolve in an unsaturated solution but not in a saturated solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Identify each of the following solutions as saturated or unsaturated. A. Salt disappears when put in water. unsaturated B. Sugar added to a cup of water does not disappear, but sits at the bottom of the cup. saturated General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check At 40 C, the solubility of KBr is 80 g/100 g of H 2 O. Identify the following solutions as either saturated or unsaturated. Explain. A. 60 g KBr added to 100 g of water at 40 C unsaturated B. 200 g KBr added to 200 g of water at 40 C saturated C. 25 g KBr added to 50 g of water at 40 C unsaturated General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
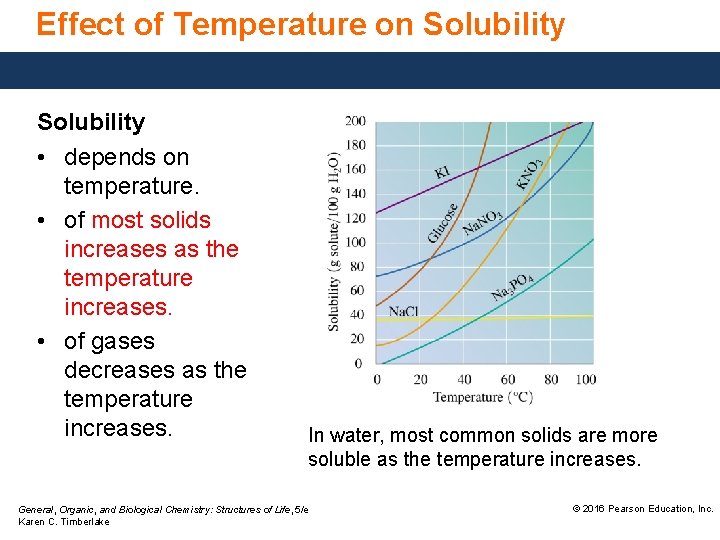
Effect of Temperature on Solubility • depends on temperature. • of most solids increases as the temperature increases. • of gases decreases as the temperature increases. In water, most common solids are more soluble as the temperature increases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
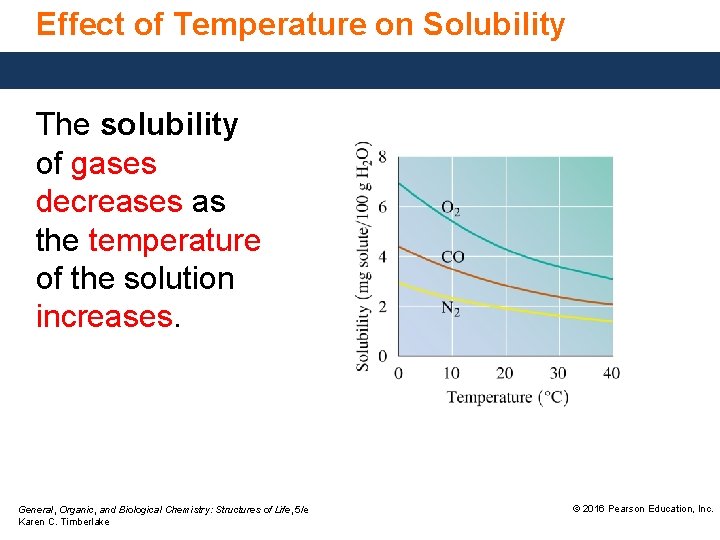
Effect of Temperature on Solubility The solubility of gases decreases as the temperature of the solution increases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check 1. Why could a bottle of carbonated drink possibly burst (explode) when it is left out in the hot sun? The pressure in a bottle increases as the gas leaves solution when it becomes less soluble at higher temperatures. As pressure increases, the bottle could burst 2. Why do fish die in water that is too warm? Because O 2 gas is less soluble in warm water, fish cannot obtain the amount of O 2 required for their survival. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solubility and Pressure Henry’s law states that • the solubility of a gas in a liquid is directly related to the pressure of that gas above the liquid. • at higher pressures, more gas molecules dissolve in the liquid. When the pressure of a gas above a solution decreases, the solubility of that gas in the solution also decreases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Soluble vs. Insoluble Ionic Compounds • Only ionic compounds that contain a soluble cation or anion are soluble in water. • In an insoluble ionic compound, the ionic bonds are too strong for the polar water molecules to break. We can use the solubility rules to predict whether an ionic compound would be expected to dissolve in water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Soluble vs. Insoluble Ionic Compounds Mixing certain aqueous solutions produces insoluble ionic compounds. If an ionic compound contains a combination of a cation and an anion that are not soluble, that ionic compound is insoluble. For example, combinations of cadmium and sulfide, iron and sulfide, lead and iodide, and nickel and hydroxide do not contain any soluble ions. Thus, they form insoluble ionic compounds. Core Chemistry Skill Using Solubility Rules General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Soluble vs. Insoluble Ionic Compounds Sulfates, SO 42−, are soluble unless combined with Ba 2+, Pb 2+, Ca 2+, Sr 2+, or Hg 22+. Barium sulfate, Ba. SO 4, an insoluble ionic compound, is used to enhance X-rays. Core Chemistry Skill Using Solubility Rules General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solubility Rules • Soluble salts typically contain at least one ion from Groups 1 A(1), NO 3−, NH 4+ or C 2 H 3 O 2− (acetate). • Most other combinations are insoluble. © 2013 Pearson Education, Inc. Chapter 8, Section 1 46

Study Check Predict if the following compounds are soluble or insoluble. Explain why. A. Cd. S B. Na 2 SO 4 C. Pb. I 2 D. Ni(NO 3)2 Insoluble; S 2− compounds are generally insoluble. Soluble; Na+ compounds are always soluble. Insoluble; I− is soluble unless combined with Pb 2+ Soluble; NO 3− compounds are always soluble General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Writing an Equation for the Formation of a Solid General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check We can use solubility rules to predict whether a solid, called a precipitate, forms when two solutions of ionic compounds are mixed. What precipitate forms when solutions of Pb(NO 3)2 and K 2 SO 4 are mixed? STEP 1 Write the ions of the reactants. Reactants, initial combinations: Pb 2+(aq) NO 3−(aq) K+(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake SO 42−(aq) © 2016 Pearson Education, Inc.
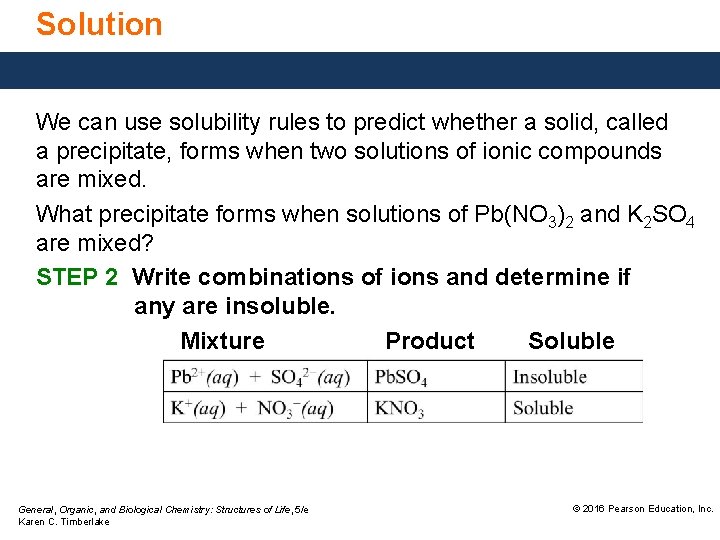
Solution We can use solubility rules to predict whether a solid, called a precipitate, forms when two solutions of ionic compounds are mixed. What precipitate forms when solutions of Pb(NO 3)2 and K 2 SO 4 are mixed? STEP 2 Write combinations of ions and determine if any are insoluble. Mixture Product Soluble General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution We can use solubility rules to predict whether a solid, called a precipitate, forms when two solutions of ionic compounds are mixed. What precipitate forms when solutions of Pb(NO 3)2 and K 2 SO 4 are mixed? STEP 3 Write the ionic equation including any solid. Pb 2+(aq) + SO 42−(aq) + 2 K+(aq) + 2 NO 3−(aq) Pb. SO 4(s) + 2 K+(aq) + 2 NO 3−(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution We can use solubility rules to predict whether a solid, called a precipitate, forms when two solutions of ionic compounds are mixed. What precipitate forms when solutions of Pb(NO 3)2 and K 2 SO 4 are mixed? STEP 4 Write the net ionic equation. Remove the spectator ions. Pb 2+(aq) + SO 42−(aq) + 2 K+(aq) + 2 NO 3−(aq) Pb. SO 4(s) + 2 K+(aq) + 2 NO 3−(aq) Pb 2+(aq) + SO 42−(aq) Pb. SO 4(s) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
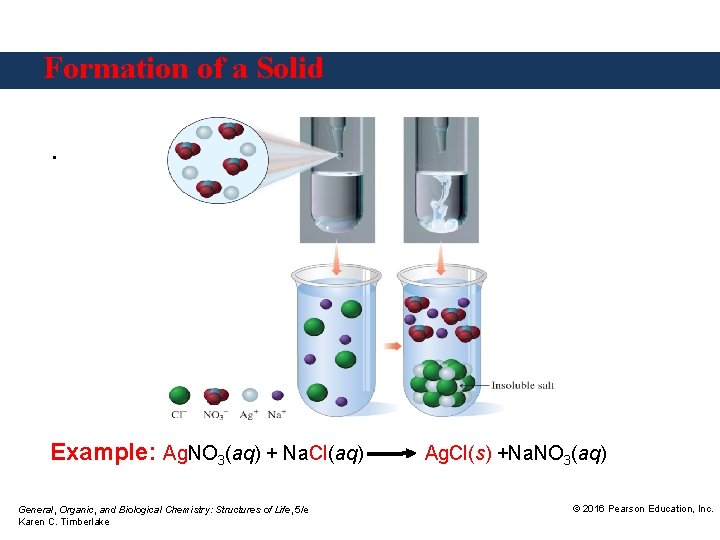
Formation of a Solid. Example: Ag. NO 3(aq) + Na. Cl(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Ag. Cl(s) +Na. NO 3(aq) © 2016 Pearson Education, Inc.

Study Check When solutions of Na 2 CO 3 and Ba(NO 3)2 are mixed, a white solid forms. Write the ionic and net ionic equations for the reaction. Step 1 Write the ions of the reactants. Ba 2+(aq) + 2 NO 3−(aq) 2 Na+(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake + CO 32−(aq) © 2016 Pearson Education, Inc.

Solution Step 2 Write the combinations of ions and determine if any are insoluble. Mixture Product Soluble 2 Na+(aq) + 2 NO 3−(aq) 2 Na. NO 3 (aq) yes Ba 2+(aq) + CO 32 -(aq) Ba. CO 3(s) no Step 3 Write the ionic equation, including any insoluble salt as a solid. Step 4 Write the net ionic equation deleting spectator ions. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

9. 4 Solution Concentrations and Reactions Suppose we prepare a solution from 8. 00 g of KI (solute) and 42. 00 g of water solvent). Learning Goal Calculate the concentration of a solute in a solution; use concentration units to calculate the amount of solute or solution. Given the volume and concentration of a solution, calculate the amount of another reactant or product in a reaction. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

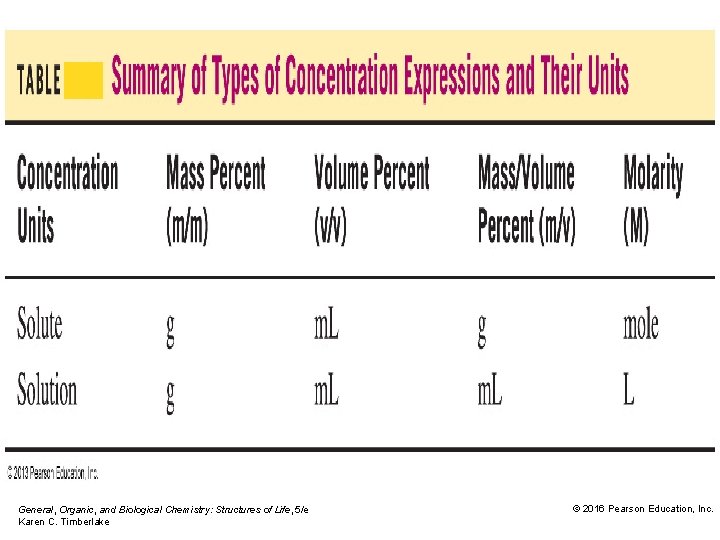
Solution Concentrations • The amount of a solute may be expressed in units of grams, milliliters, or moles. • The amount of a solution may be expressed in units of grams, milliliters, or liters. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Mass Percent (m/m) Concentration Mass percent (m/m) is • the concentration by mass of solute in mass of solution. × × • the grams of solute in 100 grams of solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Mass of Solute − Mass Solution When 42. 00 g of water is added to 8. 00 g of KCl, the mass percent concentration is 16. 0% (m/m). × × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculating Solution Concentration General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Mass Percent What is the mass percent of Na. OH in a solution prepared by dissolving 30. 0 g of Na. OH in 120. 0 g of H 2 O? STEP 1 State the given and needed quantities. ANALYZE Given THE 30. 0 g Na. OH solute PROBLEM 30. 0 g Na. OH + 120. 0 g H 2 O = 150. 0 g of Na. OH solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Needed mass percent (m/m) © 2016 Pearson Education, Inc.

Calculating Mass Percent What is the mass percent of Na. OH in a solution prepared by dissolving 30. 0 g of Na. OH in 120. 0 g of H 2 O? STEP 2 Write the concentration expression. × STEP 3 Substitute solute and solution quantities into the expression and calculate. × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check A solution is prepared by mixing 15. 0 g of Na 2 CO 3 and 235 g of H 2 O. Calculate the mass percent (m/m) of the solution. A. 15. 0% (m/m) Na 2 CO 3 solution B. 6. 38% (m/m) Na 2 CO 3 solution C. 6. 00% (m/m) Na 2 CO 3 solution ANALYZE Given Need THE 15. 0 g Na 2 CO 3 solute mass percent (m/m) PROBLEM 15. 0 g Na 2 CO 3 + 235 g H 2 O = 250. g solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Volume Percent (v/v) Concentration The volume percent (v/v) is the • percent volume (m. L) of solute (liquid) to volume (m. L) of solution. × • volume of solute (m. L) in 100 m. L of solution (conversion factor for volume percent) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Mass/Volume Percent The mass/volume percent (m/v) is the • percent mass (g) of solute to volume (m. L) of solution. × • mass of solute (g) in 100 m. L of solution. (conversion factor for mass/volume percent) Core Chemistry Skill Calculating Concentration General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Write two conversion factors for each solution. A. 8. 50% (m/m) Na. OH B. 5. 75% (v/v) ethanol C. 4. 8% (m/v) HCl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution A. 8. 50% (m/m) Na. OH B. 5. 75% (v/v) ethanol C. 4. 8% (m/v) HCl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Molarity (moles of solute/liter of solution) is defined as the moles of solute per volume (L) of solution. A 1. 0 M solution of Na. Cl is defined as General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Molarity Calculations What is the molarity of 0. 500 L of Na. OH solution if it contains 6. 00 g of Na. OH? STEP 1 State the given and needed quantities. ANALYZE Given THE 6. 00 g Na. OH solute PROBLEM 0. 500 L of Na. OH solution Need molarity (mole/L) 1 mole Na. OH = 40. 00 g Na. OH × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check What is the molarity of 0. 225 L of a KNO 3 solution containing 34. 8 g of KNO 3? A. 0. 344 M B. 1. 53 M C. 15. 5 M 34. 8 g KNO 3 x 1 mole KNO 3 = 0. 344 mole = 1. 53 M 101. 10 g KNO 3 0. 225 L General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
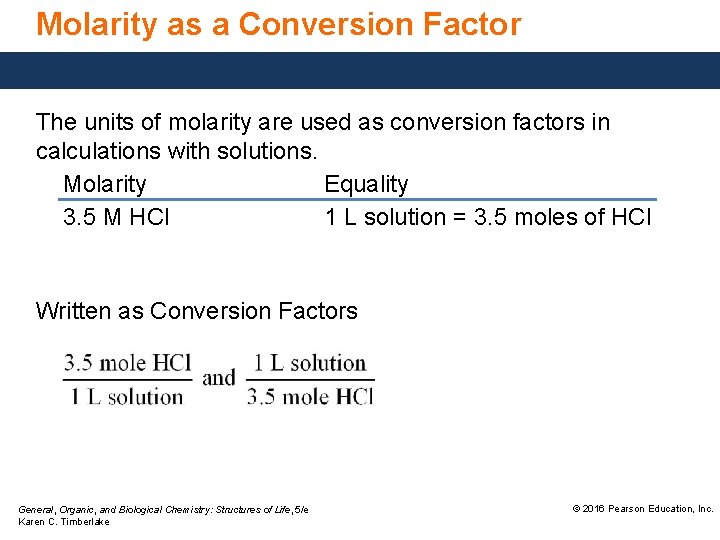
Molarity as a Conversion Factor The units of molarity are used as conversion factors in calculations with solutions. Molarity Equality 3. 5 M HCl 1 L solution = 3. 5 moles of HCl Written as Conversion Factors General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
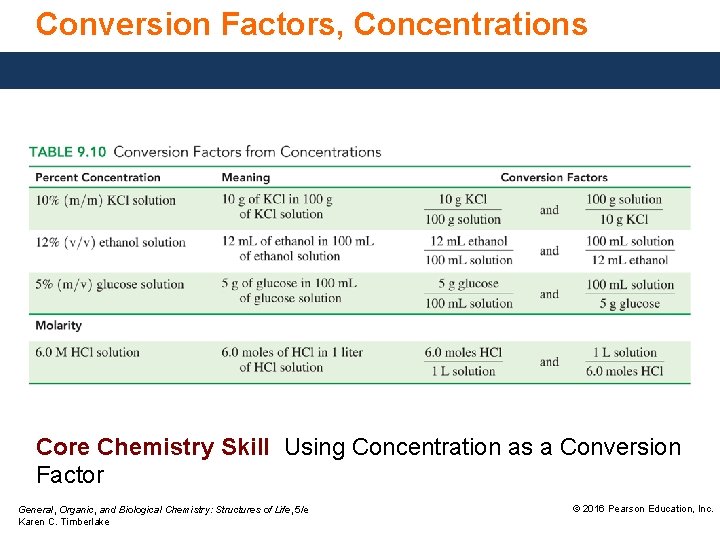
Conversion Factors, Concentrations Core Chemistry Skill Using Concentration as a Conversion Factor General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Using Concentration to Calculate Mass or Volume General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check How many grams of Na. OH are needed to prepare 75. 0 g of 14. 0% (m/m) Na. OH solution? A. 10. 5 g of Na. OH B. 75. 0 g of Na. OH C. 536 g of Na. OH General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check How many grams of Al. Cl 3 are needed to prepare 125 m. L of a 0. 150 M solution? A. 20. 0 g of Al. Cl 3 B. 16. 7 g of Al. Cl 3 C. 2. 50 g of Al. Cl 3 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution How many grams of Al. Cl 3 are needed to prepare 125 m. L of a 0. 150 M solution? Set up the problem to calculate mass or volume. × × Answer is C, 2. 50 g Al. Cl 3. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chemical Reactions in Solution When chemical reactions involve aqueous solutions, we use • the balanced chemical equation, • the molarity, and • the volume to determine the moles or grams of the reactants or products. Core Chemistry Skill Calculating the Quantity of a Reactant or Product for a Chemical Reaction in Solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculations Involving Solutions in Chemical Reactions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Zinc reacts with HCl to produce hydrogen gas, H 2 , and Zn. Cl 2. Zn(s) + 2 HCl(aq) H 2(g) + Zn. Cl 2(aq) How many liters of a 1. 50 M HCl solution completely react with 5. 32 g of zinc? General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Zinc reacts with HCl to produce hydrogen gas, H 2 , and Zn. Cl 2. Zn(s) + 2 HCl(aq) H 2(g) + Zn. Cl 2(aq) How many liters of a 1. 50 M HCl solution completely react with 5. 32 g of zinc? STEP 1 State the given and needed quantities. ANALYZE Given Need THE 5. 32 g Zn PROBLEM 1. 50 M HCl solution liters of HCl Equation solution Zn(s) + 2 HCl(aq) H 2(g) + Zn. Cl 2(aq) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Zinc reacts with HCl to produce hydrogen gas, H 2 , and Zn. Cl 2. Zn(s) + 2 HCl(aq) H 2(g) + Zn. Cl 2(aq) How many liters of a 1. 50 M HCl solution completely react with 5. 32 g of zinc? ( g Zn to mole Zn first) STEP 2 Write a plan to calculate the needed quantity. moles of zinc Mole–mole factor General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake moles of HCl Molarity volume of HCl © 2016 Pearson Education, Inc.

Solution Zinc reacts with HCl to produce hydrogen gas, H 2, and Zn. Cl 2. Zn(s) + 2 HCl(aq) H 2(g) + Zn. Cl 2(aq) How many liters of a 1. 50 M HCl solution completely react with 5. 32 g of zinc? STEP 3 Set up the problem to calculate needed quantity. × × × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
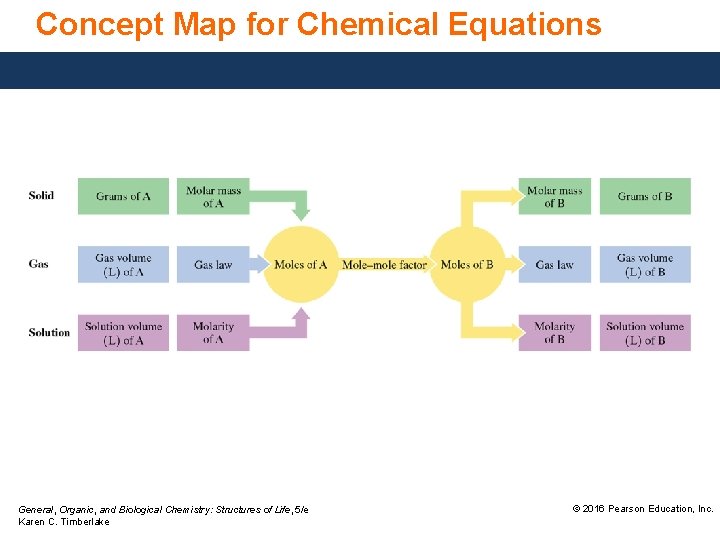
Concept Map for Chemical Equations General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

9. 5 Dilution of Solutions In a dilution, a solvent, usually water, is added to a solution, which increases its volume and decreases the concentration of the solution. Making orange juice from concentrate is an example of a dilution. Learning Goal Describe the dilution of a solution; calculate the unknown concentration or volume when a solution is diluted. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Dilution In a dilution, • water is added. • the volume of the solution increases. • concentration decreases. • the mass of solute in the solution remains the same. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solute Concentrations In the initial and diluted solution, • the moles of solute are the same. • the concentrations and volumes are related by the following equation: C 1 V 1 initial = C 2 V 2 diluted This equality is written in terms of the concentration, C, and the volume, V. The concentration, C, may be percent concentration or molarity. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Dilution of a Solution When water is added to a concentrated solution, there is no change in the number of particles. The solute particles spread out as the volume of the solution increases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculating Dilution Quantities General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Dilution: Molarity What is the final concentration when 0. 50 L of 6. 0 M HCl solution is diluted to a final volume of 1. 0 L? STEP 1 Prepare a table of the concentrations and volumes of the solutions. ANALYZE THE PROBLEM STEP 2 Given Need C 1 = 6. 0 M HCl C 2 = ? M HCl Know Predict C decreases V 1 = 0. 50 L V 2 = 1. 0 L V increases STEP 3 M 2 = 6. 0 M x 0. 50 L = 3. 0 M 1. 0 L General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check What volume of a 2. 00% (m/v) HCl solution can be prepared by diluting 25. 0 m. L of 14. 0% (m/v) HCl solution? C 1 = 14. 0% V 1 = 25. 0 m. L V 2 = C 1 V 1 C 2 = 2. 00% V 2 = ? = 14. 0% x 25. 0 m. L = 2. 00% General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake 175 m. L © 2016 Pearson Education, Inc.

Study Check What is the percent (% m/v) of a solution prepared by diluting 10. 0 m. L of 9. 00% Na. OH to 60. 0 m. L? Rearrange the dilution expression to solve for the unknown quantity. × Substitute the known quantities into the dilution expression and calculate. × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check What is the molarity (M) of a solution prepared by diluting 0. 180 L of 0. 600 M HNO 3 to 0. 540 L? ANALYZE THE PROBLEM increases Given M 1 = 0. 600 M V 1 = 0. 180 L V 2 = 0. 540 L General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Need Know Predict M 2 = ? M HNO 3 M decreases V © 2016 Pearson Education, Inc.

9. 6 Properties of Solutions Ethylene glycol is added to a radiator to form an aqueous solution that has a lower freezing point than water. Learning Goal Identify a mixture as a solution, a colloid, or a suspension. Describe how the number of particles in a solution affects the freezing point, the boiling point, and the osmotic pressure of a solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions or Colloids Solutions • are transparent. • do not separate. • contain small particles, ions, or molecules that cannot be filtered and pass through semipermeable membranes. Colloids • have medium-sized particles. • cannot be filtered. • can be separated by semipermeable membranes. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
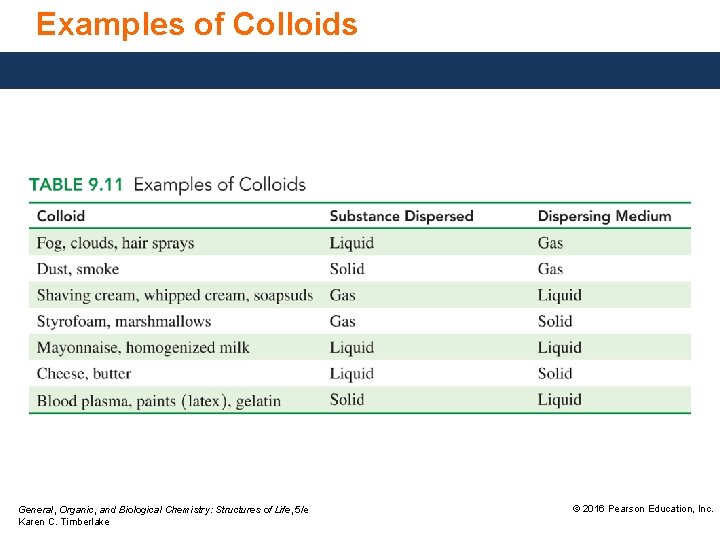
Examples of Colloids General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Suspensions • are heterogeneous, non-uniform mixtures. • have very large particles that settle out of solution. • can be filtered. • must be stirred to stay suspended. Examples include blood platelets, muddy water, and calamine lotion. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions, Colloids, and Suspensions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
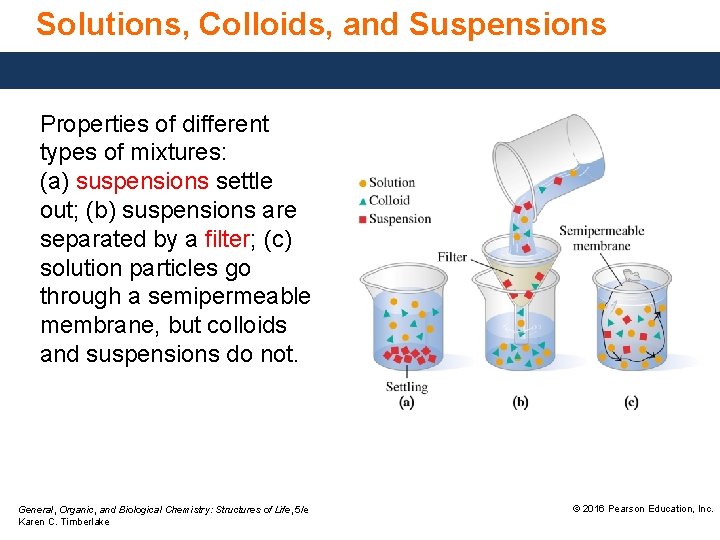
Solutions, Colloids, and Suspensions Properties of different types of mixtures: (a) suspensions settle out; (b) suspensions are separated by a filter; (c) solution particles go through a semipermeable membrane, but colloids and suspensions do not. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check A mixture that has solute particles that do not settle out but are too large to pass through a semipermeable membrane is called a _______. A. solution B. colloid C. suspension General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Freezing Point Lowering, Boiling Point Elevation When a solute is added to water, the physical properties change and the • vapor pressure above the solution decreases. • boiling point of the solution increases. • freezing point of the solution decreases. These types of properties are called colligative properties; they depend only on the concentration of solute particles in the solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Vapor Pressure Lowering Increasing the concentration of solute particles in the solution • decreases the number of solvent particles at the surface of the solution. • prevents some of the solvent particles from leaving the solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Boiling Point Elevation Increasing the concentration of nonvolatile solute particles in the solution raises the boiling point of the solution, and • more solute particles in the solution lowers the vapor pressure. • the solution boils at a higher temperature than the normal boiling point. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Freezing Point Lowering Adding salt to an icy road when temperatures drop below freezing • allows the particles of salt to mix with the water. • lowers the freezing point of the ice. Adding ethylene glycol to water increases the number of hydrogen bonds that form in the solution, lowering the freezing point and raising the boiling point of the solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Particles in Solution A solute that is a nonelectrolyte dissolves as molecules, whereas a solute that is a strong electrolyte dissolves entirely as ions. • The solute in antifreeze, which is ethylene glycol, C 2 H 6 O 2, is a nonelectrolyte and dissolves as molecules in water. 1 mole C 2 H 6 O 2(l) = 1 mole C 2 H 6 O 2(aq) • The solute Na. Cl, a strong electrolyte, dissolves as ions in water. Na. Cl(s) Na+(aq) + Cl− (aq) 1 mole Na. Cl(s) = 1 mole Na+(aq) + 1 mole Cl− (aq) 1 mole Na. Cl(s) = 2 moles of particles in solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Particles in Solution The effect of solute particles on boiling points and freezing points is summarized below: General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculating Boiling Point Elevation, Freezing Point Lowering Core Chemistry Skill Calculating the Boiling Point/Freezing Point of a Solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Freezing Point Lowering In the northeastern United States during freezing temperatures, Ca. Cl 2 is spread on icy highways to melt the ice. Calculate the freezing point of a solution containing 0. 50 mole of Ca. Cl 2 in 1 kg of water. STEP 1 State the given and needed quantities. ANALYZE Given THE 0. 50 mole of Ca. Cl 2 PROBLEM 1 kg of water General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Needed freezing point of solution © 2016 Pearson Education, Inc.

Calculating Freezing Point Lowering In the northeastern United States during freezing temperatures, Ca. Cl 2 is spread on icy highways to melt the ice. Calculate the freezing point of a solution containing 0. 50 mole of Ca. Cl 2 in 1 kg of water. STEP 2 Determine the number of moles of solute particles. Ca. Cl 2(s) Ca 2+(aq) + 2 Cl−(aq) 1 mole of Ca. Cl 2 = 3 moles of solute particles General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
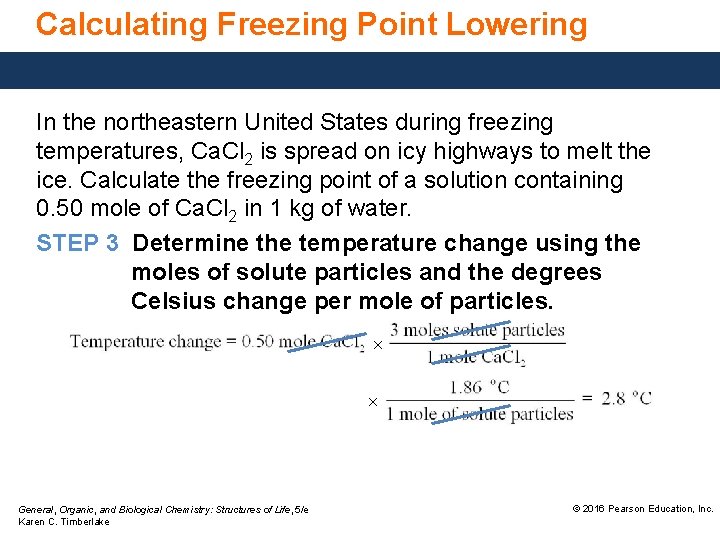
Calculating Freezing Point Lowering In the northeastern United States during freezing temperatures, Ca. Cl 2 is spread on icy highways to melt the ice. Calculate the freezing point of a solution containing 0. 50 mole of Ca. Cl 2 in 1 kg of water. STEP 3 Determine the temperature change using the moles of solute particles and the degrees Celsius change per mole of particles. × × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Osmosis In osmosis, • water (solvent) flows from a lower to a higher solute concentration. • the level of the solution with the higher solute concentration rises. • the concentrations of the two solutions become equal with time. Water flows into the solution with a higher solute concentration until the flow of water becomes equal in both directions. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
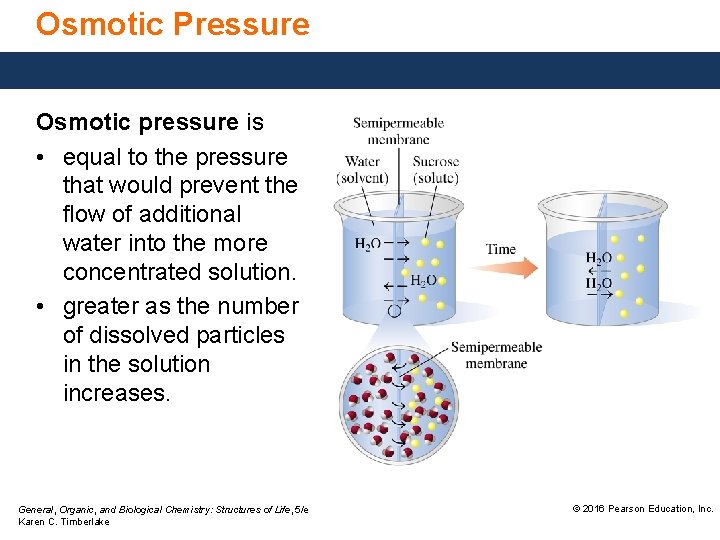
Osmotic Pressure Osmotic pressure is • equal to the pressure that would prevent the flow of additional water into the more concentrated solution. • greater as the number of dissolved particles in the solution increases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Reverse Osmosis In a process called reverse osmosis, • a pressure greater than the osmotic pressure is applied to a solution, forcing it through a purification membrane. • the flow of water is reversed because water flows from an area of lower to higher water concentration, leaving behind the molecules and ions in solution. Reverse osmosis, used in desalination plants to obtain pure water from sea (salt) water, requires a large amount of energy. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Isotonic Solutions Because cell membranes in biological systems are semipermeable, osmosis is an ongoing process. The solutes in body solutions such as blood, tissue fluids, lymph, and plasma all exert osmotic pressure. Most IV solutions used in hospitals are isotonic solutions, which • exert the same osmotic pressure as body fluids such as red blood cells (RBCs). • include a 5. 0% (m/v) glucose or 0. 90% (m/v) Na. Cl isotonic solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
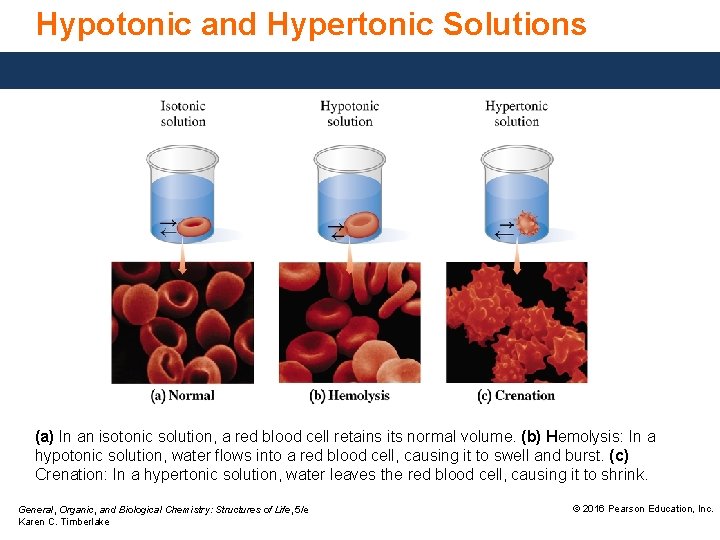
Hypotonic and Hypertonic Solutions (a) In an isotonic solution, a red blood cell retains its normal volume. (b) Hemolysis: In a hypotonic solution, water flows into a red blood cell, causing it to swell and burst. (c) Crenation: In a hypertonic solution, water leaves the red blood cell, causing it to shrink. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Hypotonic Solution A hypotonic solution • has a lower solute concentration than red blood cells. • means water flows into cells by osmosis. The increase in fluid causes the cells to swell and burst, a condition called hemolysis. (b) Hemolysis: In a hypotonic solution, water flows into a red blood cell, causing it to swell and burst. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Hypertonic Solution A hypertonic solution • has a higher solute concentration than RBCs. • Involves water going out of the cells by osmosis. • causes crenation: RBCs shrink in size. (c) Crenation: In a hypertonic solution, water leaves the red blood cell, causing it to shrink. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
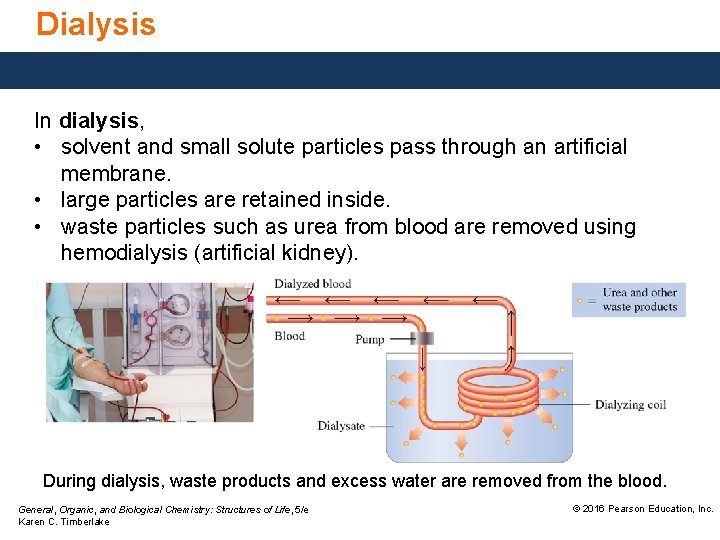
Dialysis In dialysis, • solvent and small solute particles pass through an artificial membrane. • large particles are retained inside. • waste particles such as urea from blood are removed using hemodialysis (artificial kidney). During dialysis, waste products and excess water are removed from the blood. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Indicate if each of the following solutions is A. isotonic. B. hypotonic. C. hypertonic. Hyper 2% Na. Cl solution 1. ____ Hypo 2. ____ 1% glucose solution Hypo 0. 5% Na. Cl solution 3. ____ Isotonic 4. ____ 5% glucose solution General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check When placed in each of the following, indicate if a red blood cell will A. not change. B. undergo hemolysis. C. undergo crenation. 1. ____ 5% glucose solution A B 2. ____ 1% glucose solution B 3. ____ 0. 5% Na. Cl solution 4. ____ 2% Na. Cl solution C General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
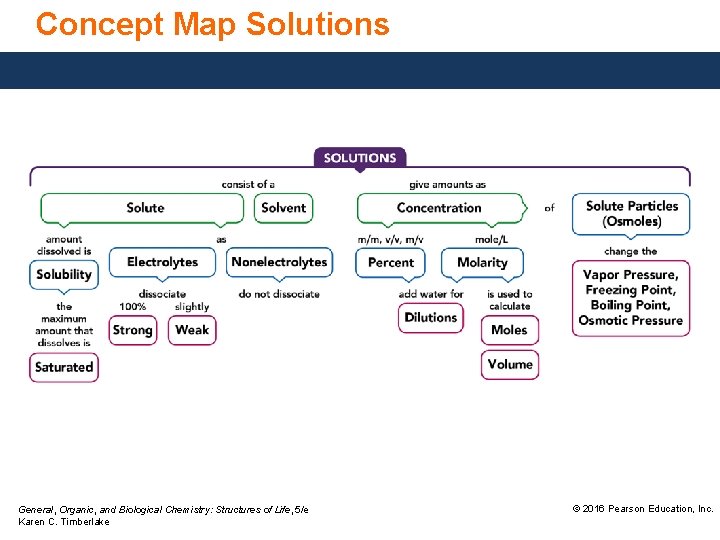
Concept Map Solutions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

End of Chapter 9 Any Questions? General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake 122 © 2016 Pearson Education, Inc.
- Slides: 122