Lecture Presentation Chapter 9 Solutions Karen C Timberlake

Lecture Presentation Chapter 9 Solutions Karen C. Timberlake General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 9 Solutions A dialysis nurse informs Michelle that • her side effects are due to her body’s inability to regulate the amount of water in her cells. • the amount of water in her body fluids is regulated by the concentration of electrolytes and the rate at which waste products are removed from her body. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 9 Readiness Key Math Skills • Calculating a Percentage (1. 4 C) • Solving Equations (1. 4 D) • Interpreting a Line Graph (1. 4 E) Core Chemistry Skills • Writing Conversion Factors from Conversion Equalities (2. 5) • Using Conversion Factors (2. 6) • Identifying Attractive Forces (6. 8) • Using Mole–Mole Factors (7. 6) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

9. 1 Solutions • are homogeneous mixtures of two or more substances. • form when there is sufficient attraction between the solute and solvent molecules. • have two components: the solvent, present in a larger amount, and the solute, present in a smaller amount. Learning Goal Identify the solute and solvent in a solution; describe the formation of a solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutes • may be a liquid, gas, or solid. • are spread evenly throughout the solution. • mix with solvents so the solute and solvent have the same physical state. • cannot be separated by filtration, but they can be separated by evaporation. • are not visible, but they can give a color to the solution. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
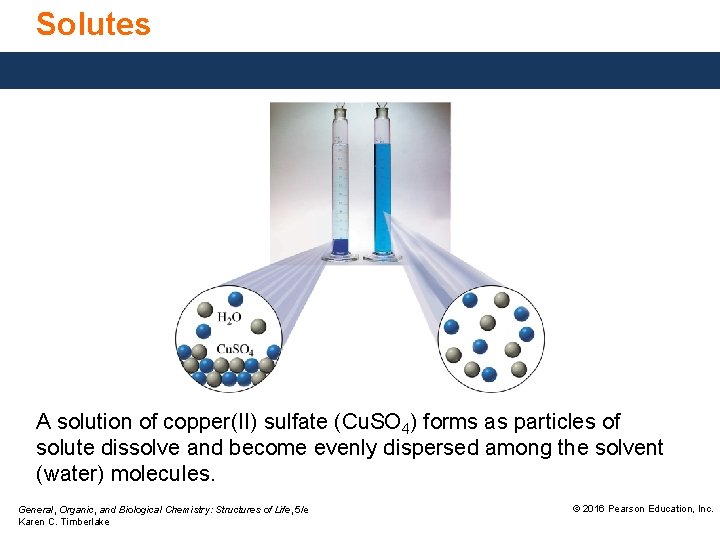
Solutes A solution of copper(II) sulfate (Cu. SO 4) forms as particles of solute dissolve and become evenly dispersed among the solvent (water) molecules. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
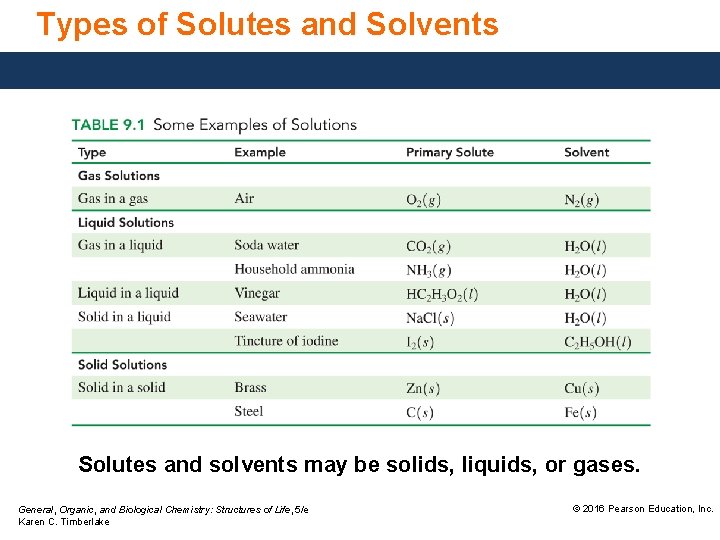
Types of Solutes and Solvents Solutes and solvents may be solids, liquids, or gases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Water as a Solvent Water • is one of the most common solvents in nature. • is a polar molecule due to polar O–H bonds. • molecules form hydrogen bonds important in many biological compounds. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Formation of Solutions form when the solute–solvent interactions are large enough to overcome the solute–solute interactions and the solvent–solvent interactions. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions, Like Dissolves Like Solutions will form when the solute and solvent have similar polarities: “like dissolves like. ” General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
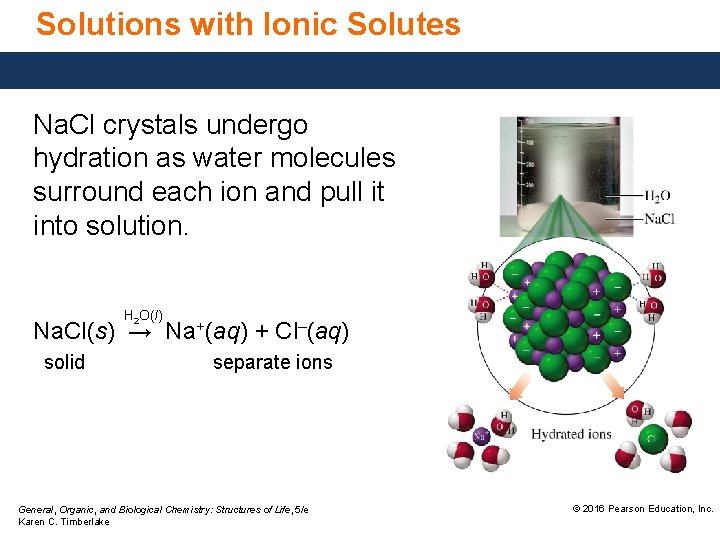
Solutions with Ionic Solutes Na. Cl crystals undergo hydration as water molecules surround each ion and pull it into solution. H 2 O(l) Na. Cl(s) → Na+(aq) + Cl–(aq) solid separate ions General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions with Polar Solutes A polar molecular compound such as methanol, CH 3—OH, is soluble in water because methanol has a polar –OH group to form hydrogen bonds with water. Polar solutes require polar solvents for a solution to form. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solutions with Nonpolar Solutes Compounds containing nonpolar molecules, such as iodine (I 2), oil, or grease, do not dissolve in water because there are essentially no attractions between the particles of a nonpolar solute and the polar solvent. Nonpolar solutes require nonpolar solvents for a solution to form. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Identify the solute in each of the following solutions. A. 2 g of sugar and 100 m. L of water B. 60. 0 m. L of ethyl alcohol and 30. 0 m. L of methyl alcohol C. 55. 0 m. L of water and 1. 50 g of Na. Cl D. Air: 200 m. L of O 2 and 800 m. L of N 2 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Identify the solute in each of the following solutions. A. 2 g of sugar and 100 m. L of water The solute is sugar. B. 60. 0 m. L of ethyl alcohol and 30. 0 m. L of methyl alcohol The solute is methyl alcohol. C. 55. 0 m. L of water and 1. 50 g of Na. Cl The solute is Na. Cl. D. Air: 200 m. L of O 2 and 800 m. L of N 2 The solute is O 2. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check When solid Li. Cl is added to water it dissolves because A. the Li+ ions are attracted to the 1) oxygen atom ( −) of water. 2) hydrogen atom ( +) of water. B. the Cl− ions are attracted to the 1) oxygen atom ( −) of water. 2) hydrogen atom ( +) of water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution When solid Li. Cl is added to water it dissolves because A. the Li+ ions are attracted to the 1) oxygen atom (σ −) of water. B. the Cl− ions are attracted to the 2) hydrogen atom (σ +) of water. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Which of the following solutes will dissolve in water? Why? A. Na 2 SO 4 B. gasoline (nonpolar) C. I 2 D. HCl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Which of the following solutes will dissolve in water? Why? Water is a polar solvent that can dissolve ionic and polar substances but nonpolar solutes. A. Na 2 SO 4 will dissolve, ionic B. gasoline (nonpolar)will not dissolve, nonpolar C. I 2 will not dissolve, nonpolar D. HCl will dissolve, polar General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 19