Lecture Presentation Chapter 8 Gases Karen C Timberlake

Lecture Presentation Chapter 8 Gases Karen C. Timberlake General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 8 Gases Respiratory therapists assess and treat a range of patients. They perform a variety of diagnostic tests, including measuring • breathing capacity. • concentrations of oxygen and carbon dioxide in a patient’s blood, as well as blood p. H. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 8 Readiness Key Math Skills • Solving Equations (1. 4 D) Core Chemistry Skills • Using Significant Figures in Calculations (2. 3) • Writing Conversion Factors from Conversion Equalities (2. 5) • Using Conversion Factors (2. 6) • Using Molar Mass as a Conversion Factor (7. 5) • Using Mole–Mole Factors (7. 6) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

8. 1 Properties of Gases Generally molecules with fewer than five atoms from the first two periods in the periodic table are gases at room temperature. In addition, the following are also gases: • H 2, N 2, O 2, F 2, and Cl 2 • oxides of the nonmetals on the upper-right corner of the periodic table: CO, CO 2, NO 2, SO 2, and SO 3 • noble gases Learning Goal Describe the kinetic molecular theory of gases and the units of measurement used for gases. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Kinetic Molecular Theory A gas consists of small particles that 1. move randomly with high velocities. 2. have very small attractive (or repulsive) forces between molecules. 3. occupy a much larger volume than the volume of the molecules alone. 4. are in constant motion, moving rapidly in straight lines. 5. have a Kelvin temperature proportionate to the average kinetic energy of the molecules. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Gas particles which move in straight lines within a container, exert pressure when they collide with the walls of the container. © 2016 Pearson Education, Inc.

Properties That Describe a Gases are described in terms of four properties: pressure (P), volume (V), temperature (T), and amount (n). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Volume The volume of a gas • is the same as the volume of the container it occupies. • is usually measured in liters or milliliters. • increases with an increase in temperature at a constant pressure. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Gas particles which move in straight lines within a container, exert pressure when they collide with the walls of the container. © 2016 Pearson Education, Inc.

Temperature The temperature of a gas relates to the average kinetic energy of the molecules and is measured in the Kelvin (K) temperature scale. When the temperature of a gas is • decreased, the molecules have fewer collisions. • increased, the molecules have more collisions. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Pressure is a measure of the gas particle collisions with sides of a container and is measured in units of • millimeters of mercury, mm. Hg or torr. • atmospheres, atm. • pascals, Pa, or kilopascals, k. Pa. • pounds per square inch, psi. Gas particles in the air exert pressure on us called atmospheric pressure. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Barometers Measure Pressure A barometer • measures the pressure exerted by the gases in the atmosphere. • indicates atmospheric pressure as the height in mm of the mercury column. 760 mm. Hg = 1 atm = 760 Torr The barometer was invented by Evangelista Torricelli, at exactly 1 atm the barometer tube measures exactly 760 mm high. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
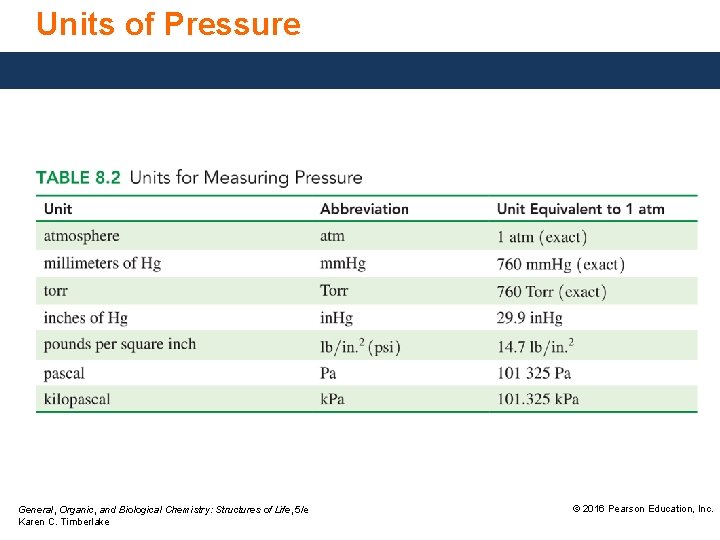
Units of Pressure General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Atmospheric Pressure Atmospheric pressure • is the pressure exerted by a column of air from the top of the atmosphere to the surface of Earth. • decreases as altitude increases. • about 1 atm at sea level. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
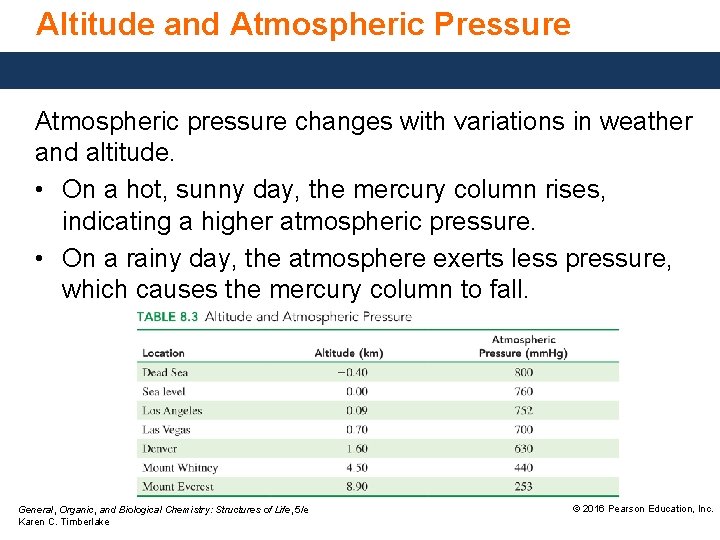
Altitude and Atmospheric Pressure Atmospheric pressure changes with variations in weather and altitude. • On a hot, sunny day, the mercury column rises, indicating a higher atmospheric pressure. • On a rainy day, the atmosphere exerts less pressure, which causes the mercury column to fall. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check 1. What is 475 mm. Hg expressed in atm? A. 475 atm B. 0. 625 atm C. 3. 61 105 atm 2. The pressure in a tire is 2. 00 atm. What is this pressure in mm. Hg? A. 2. 00 mm. Hg B. 1520 mm. Hg C. 22 300 mm. Hg General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution 1. What is 475 mm. Hg expressed in atm? The answer is B, 0. 625 atm. × 2. The pressure in a tire is 2. 00 atm. What is this pressure in mm. Hg? The answer is B, 1520 mm. Hg. × General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check 1. The downward pressure on the Hg in a barometer is _____ the pressure of the atmosphere. A. greater than B. less than C. the same as 2. A water barometer is 13. 6 times taller than an Hg barometer (d. Hg = 13. 6 g/m. L) because A. H 2 O is less dense than mercury. B. H 2 O is heavier than mercury. C. air is more dense than H 2 O. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution 1. The downward pressure on the Hg in a barometer is _____ the pressure of the atmosphere. The answer is C, the same as. 2. A water barometer is 13. 6 times taller than an Hg barometer (d. Hg = 13. 6 g/m. L) because The answer is A, H 2 O is less dense than mercury. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 17