Lecture Presentation Chapter 23 Metabolism and Energy Production

Lecture Presentation Chapter 23 Metabolism and Energy Production Karen C. Timberlake General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 23 Metabolism and Energy Production Exercise physiologists work with athletes as well as patients who have been diagnosed with diabetes, heart disease, pulmonary disease, or other chronic disabilities or diseases. Often these patients have been prescribed exercise as a form of treatment, and they have been referred to an exercise physiologist. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Chapter 23 Readiness Core Chemistry Skills • Writing Equations for Hydrogenation, Hydration, and Polymerization (12. 7) • Classifying Enzymes (20. 2) • Identifying Important Coenzymes in Metabolism (22. 2) • Identifying the Compounds in Glycolysis (22. 4) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

23. 1 The Citric Acid Cycle The citric acid cycle is a series of reactions that connects the intermediate acetyl Co. A from the catabolic pathways in stage 2 with electron transport and the synthesis of ATP in stage 3. Learning Goal Describe the oxidation of acetyl Co. A in the citric acid cycle. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

23. 1 The Citric Acid Cycle The citric acid cycle (stage 3) • operates under aerobic conditions. • oxidizes the two-carbon acetyl group in acetyl Co. A to CO 2. • produces reduced coenzymes NADH and FADH 2. • is named for the six-carbon citrate ion from citric acid (C 6 H 8 O 7), a tricarboxylic acid, formed in the first reaction. • is also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle. Core Chemistry Skill Describing the Reactions in the Citric Acid Cycle General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Citric Acid Cycle Overview In the citric acid cycle, • six carbons move through the citric acid cycle, producing oxaloacetate and 2 CO 2. • each turn contains four oxidation reactions producing the reduced coenzymes NADH and FADH 2. • one GTP (converted to ATP in the cell) is produced during the citric acid cycle. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
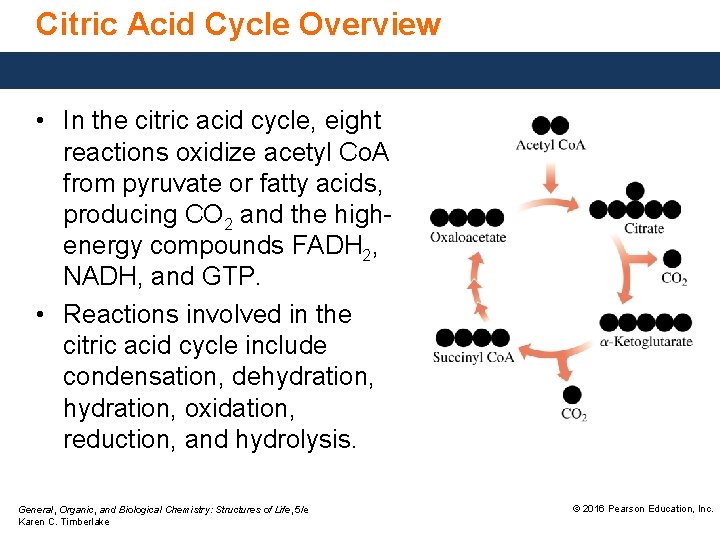
Citric Acid Cycle Overview • In the citric acid cycle, eight reactions oxidize acetyl Co. A from pyruvate or fatty acids, producing CO 2 and the highenergy compounds FADH 2, NADH, and GTP. • Reactions involved in the citric acid cycle include condensation, dehydration, oxidation, reduction, and hydrolysis. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
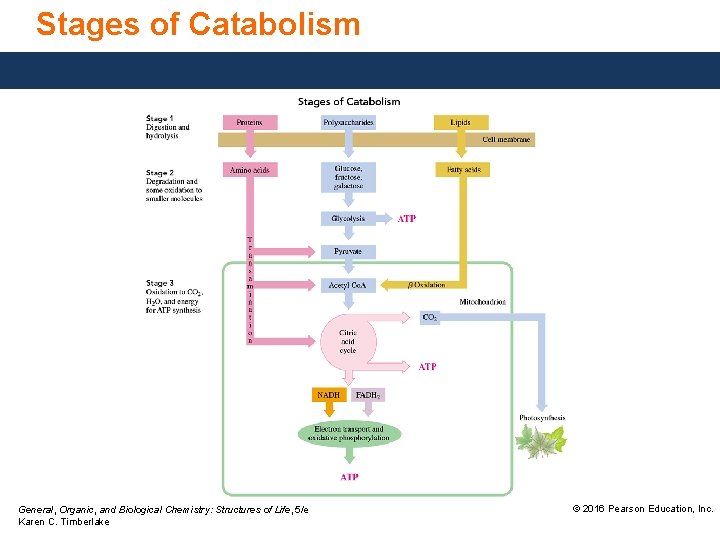
Stages of Catabolism General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Reaction 1: Formation of Citrate In the first reaction of the citric acid cycle, • citrate synthase catalyzes the condensation of an acetyl group (2 C) from acetyl Co. A with oxaloacetate (4 C) to yield citrate (6 C) and coenzyme A. • the energy to form citrate is provided by the hydrolysis of the high-energy thioester bond in acetyl Co. A. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
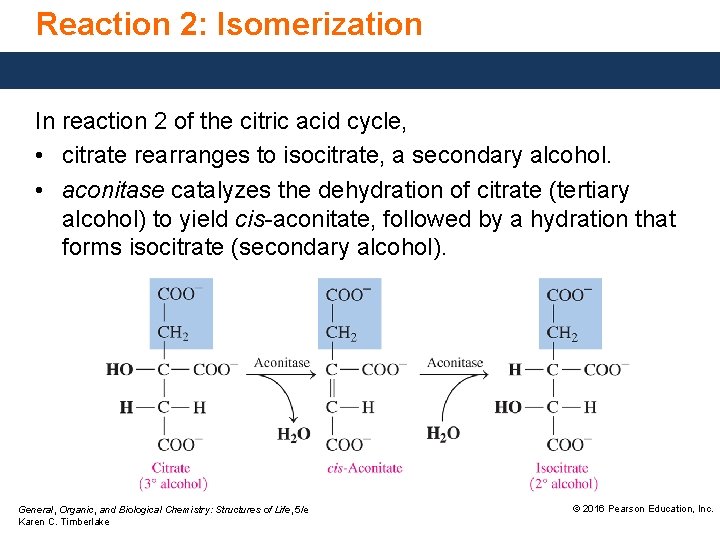
Reaction 2: Isomerization In reaction 2 of the citric acid cycle, • citrate rearranges to isocitrate, a secondary alcohol. • aconitase catalyzes the dehydration of citrate (tertiary alcohol) to yield cis-aconitate, followed by a hydration that forms isocitrate (secondary alcohol). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Reaction 3: Oxidation, Decarboxylation In reaction 3, isocitrate undergoes decarboxylation by isocitrate dehydrogenase. • One carbon is removed by converting a carboxylate group (COO−) to CO 2. • The dehydrogenase removes hydrogen ions and electrons, used to reduce NAD+ to NADH and H+. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
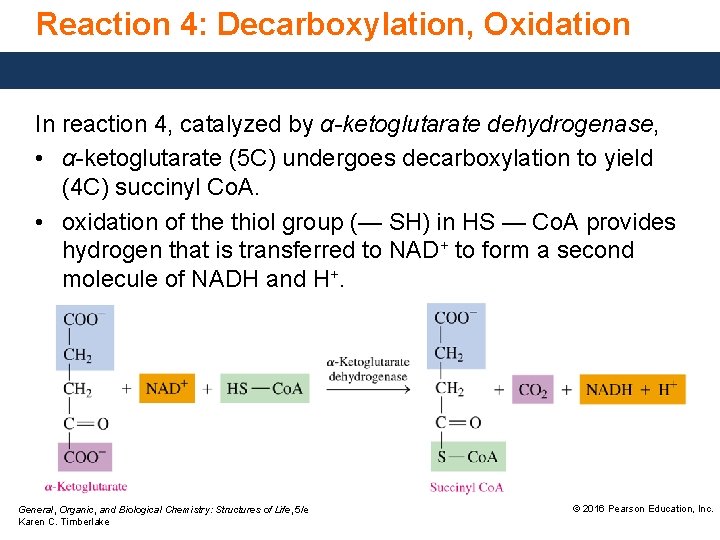
Reaction 4: Decarboxylation, Oxidation In reaction 4, catalyzed by α-ketoglutarate dehydrogenase, • α-ketoglutarate (5 C) undergoes decarboxylation to yield (4 C) succinyl Co. A. • oxidation of the thiol group (— SH) in HS — Co. A provides hydrogen that is transferred to NAD+ to form a second molecule of NADH and H+. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Reaction 5: Hydrolysis In reaction 5, catalyzed by succinyl Co. A synthetase, • hydrolysis of the thioester bond in succinyl Co. A yields succinate and HS — Co. A. • energy from hydrolysis is transferred to the condensation of phosphate and GDP forming GTP, a high-energy compound similar to ATP. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
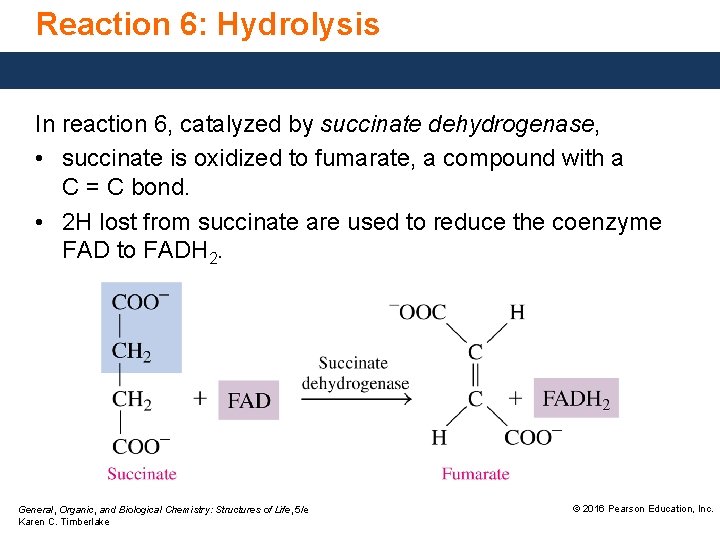
Reaction 6: Hydrolysis In reaction 6, catalyzed by succinate dehydrogenase, • succinate is oxidized to fumarate, a compound with a C = C bond. • 2 H lost from succinate are used to reduce the coenzyme FAD to FADH 2. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
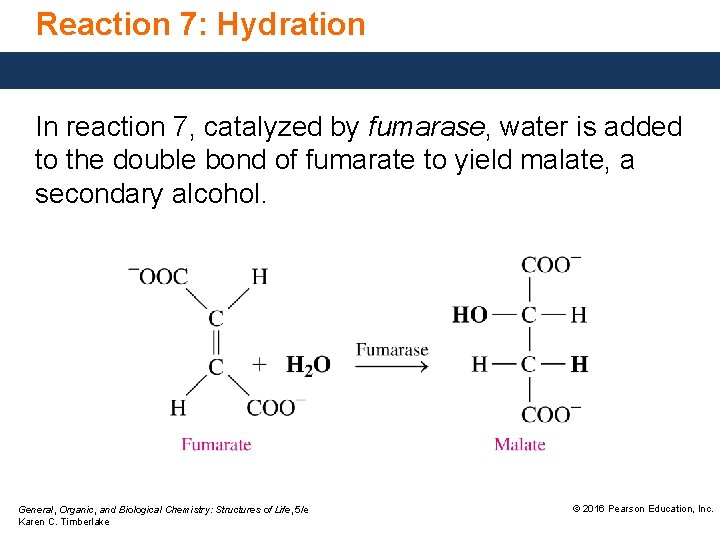
Reaction 7: Hydration In reaction 7, catalyzed by fumarase, water is added to the double bond of fumarate to yield malate, a secondary alcohol. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
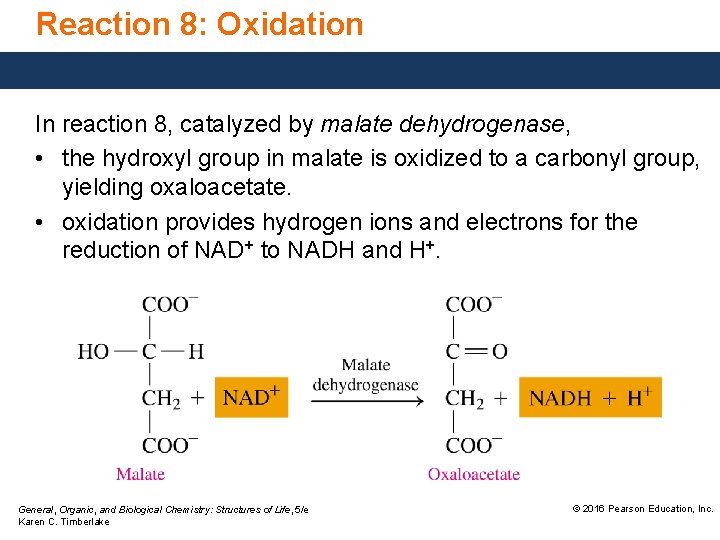
Reaction 8: Oxidation In reaction 8, catalyzed by malate dehydrogenase, • the hydroxyl group in malate is oxidized to a carbonyl group, yielding oxaloacetate. • oxidation provides hydrogen ions and electrons for the reduction of NAD+ to NADH and H+. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Summary, Citric Acid Cycle In the citric acid cycle, • an acetyl group bonds with oxaloacetate to form citrate. • two decarboxylations remove two carbons as two CO 2. • four oxidations provide hydrogen for three NADH and one FADH 2. • a direct phosphorylation forms GTP (ATP). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Regulation of the Citric Acid Cycle The reaction rate for the citric acid cycle • increases when low levels of ATP or NAD+ activate isocitrate dehydrogenase. • decreases when high levels of ATP or NADH inhibit citrate synthetase (first step in cycle). General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check How many of each of the following are produced in one turn of the citric acid cycle? A. _ CO 2 B. _ NADH C. _ FADH 2 D. _ GTP General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution How many of each of the following are produced in one turn of the citric acid cycle? A. 2 CO 2 B. 3 NADH C. 1 FADH 2 D. 1 GTP General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 20