Lecture Presentation Chapter 16 AcidBase Equilibria 2012 Pearson

Lecture Presentation Chapter 16 Acid–Base Equilibria © 2012 Pearson Education, Inc.

Some Definitions • Arrhenius – An acid is a substance that, when dissolved in water, increases the concentration of hydrogen ions. – A base is a substance that, when dissolved in water, increases the concentration of hydroxide ions. © 2012 Pearson Education, Inc. Acids and Bases

Some Definitions • Brønsted–Lowry – An acid is a proton donor. – A base is a proton acceptor. © 2012 Pearson Education, Inc. Acids and Bases

A Brønsted–Lowry acid… …must have a removable (acidic) proton. A Brønsted–Lowry base… …must have a pair of nonbonding electrons. © 2012 Pearson Education, Inc. Acids and Bases

If it can be either… …it is amphiprotic. HCO 3 HSO 4 H 2 O © 2012 Pearson Education, Inc. Acids and Bases

What Happens When an Acid Dissolves in Water? • Water acts as a Brønsted–Lowry base and abstracts a proton (H+) from the acid. • As a result, the conjugate base of the acid and a hydronium ion are formed. © 2012 Pearson Education, Inc. Acids and Bases
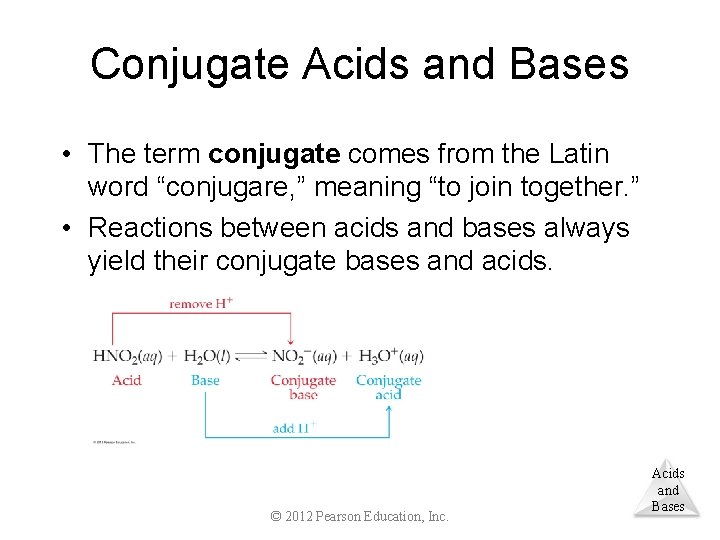
Conjugate Acids and Bases • The term conjugate comes from the Latin word “conjugare, ” meaning “to join together. ” • Reactions between acids and bases always yield their conjugate bases and acids. © 2012 Pearson Education, Inc. Acids and Bases

Acid and Base Strength • Strong acids are completely dissociated in water. – Their conjugate bases are quite weak. • Weak acids only dissociate partially in water. – Their conjugate bases are weak bases. © 2012 Pearson Education, Inc. Acids and Bases

Acid and Base Strength • Substances with negligible acidity do not dissociate in water. – Their conjugate bases are exceedingly strong. © 2012 Pearson Education, Inc. Acids and Bases

Acid and Base Strength • In any acid–base reaction, the equilibrium will favor the reaction that moves the proton to the stronger base. HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl (aq) • H 2 O is a much stronger base than Cl , so the equilibrium lies so far to the right that K is not measured (K >> 1). © 2012 Pearson Education, Inc. Acids and Bases

Acid and Base Strength • In any acid–base reaction, the equilibrium will favor the reaction that moves the proton to the stronger base: CH 3 CO 2 H(aq) + H 2 O(l) H 3 O+(aq) + CH 3 CO 2 (aq) • Acetate is a stronger base than H 2 O, so the equilibrium favors the left side (K < 1). © 2012 Pearson Education, Inc. Acids and Bases

Autoionization of Water • As we have seen, water is amphoteric. • In pure water, a few molecules act as bases and a few act as acids. • This is referred to as autoionization. © 2012 Pearson Education, Inc. Acids and Bases

Ion Product Constant • The equilibrium expression for this process is Kc = [H 3 O+] [OH ] • This special equilibrium constant is referred to as the ion product constant for water, Kw. • At 25 C, Kw = 1. 0 10 14 © 2012 Pearson Education, Inc. Acids and Bases

p. H is defined as the negative base-10 logarithm of the concentration of hydronium ion: p. H = log [H 3 O+] © 2012 Pearson Education, Inc. Acids and Bases
![p. H • In pure water, Kw = [H 3 O+] [OH ] = p. H • In pure water, Kw = [H 3 O+] [OH ] =](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-15.jpg)
p. H • In pure water, Kw = [H 3 O+] [OH ] = 1. 0 10 14 • Since in pure water, [H 3 O+] = [OH ], [H 3 O+] = 1. 0 10 14 = 1. 0 10 7 © 2012 Pearson Education, Inc. Acids and Bases

p. H • Therefore, in pure water, p. H = log (1. 0 10 7) = 7. 00 • An acid has a higher [H 3 O+] than pure water, so its p. H is <7. • A base has a lower [H 3 O+] than pure water, so its p. H is >7. © 2012 Pearson Education, Inc. Acids and Bases

p. H These are the p. H values for several common substances Fig. 16. 5. © 2012 Pearson Education, Inc. Acids and Bases

Other “p” Scales • The “p” in p. H tells us to take the negative base-10 logarithm of the quantity (in this case, hydronium ions). • Some similar examples are – p. OH: log [OH ] – p. Kw: log Kw © 2012 Pearson Education, Inc. Acids and Bases
![Watch This! Because [H 3 O+] [OH ] = Kw = 1. 0 10 Watch This! Because [H 3 O+] [OH ] = Kw = 1. 0 10](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-19.jpg)
Watch This! Because [H 3 O+] [OH ] = Kw = 1. 0 10 14 we know that log [H 3 O+] + log [OH ] = log Kw = 14. 00 or, in other words, p. H + p. OH = p. Kw = 14. 00 © 2012 Pearson Education, Inc. Acids and Bases

How Do We Measure p. H? • For less accurate measurements, one can use – Litmus paper • “Red” paper turns blue above ~p. H = 8 • “Blue” paper turns red below ~p. H = 5 – Or an indicator. © 2012 Pearson Education, Inc. Acids and Bases

How Do We Measure p. H? For more accurate measurements, one uses a p. H meter, which measures the voltage in the solution. © 2012 Pearson Education, Inc. Acids and Bases

Strong Acids • You will recall that the seven strong acids are HCl, HBr, HI, HNO 3, H 2 SO 4, HCl. O 3, and HCl. O 4. • These are, by definition, strong electrolytes and exist totally as ions in aqueous solution. • For the monoprotic strong acids, [H 3 O+] = [acid]. © 2012 Pearson Education, Inc. Acids and Bases

Strong Bases • Strong bases are the soluble hydroxides, which are the alkali metal and heavier alkaline earth metal hydroxides (Ca 2+, Sr 2+, and Ba 2+). • Again, these substances dissociate completely in aqueous solution. © 2012 Pearson Education, Inc. Acids and Bases

Dissociation Constants • For a generalized acid dissociation, HA(aq) + H 2 O(l) A (aq) + H 3 O+(aq) the equilibrium expression would be [H 3 O+] [A ] Kc = [HA] • This equilibrium constant is called the acid-dissociation constant, Ka. © 2012 Pearson Education, Inc. Acids and Bases

Dissociation Constants The greater the value of Ka, the stronger is the acid. © 2012 Pearson Education, Inc. Acids and Bases

Calculating Ka from the p. H The p. H of a 0. 10 M solution of formic acid, HCOOH, at 25 C is 2. 38. Calculate Ka formic acid at this temperature. We know that [H 3 O+] [HCOO ] Ka = [HCOOH] © 2012 Pearson Education, Inc. Acids and Bases

Calculating Ka from the p. H The p. H of a 0. 10 M solution of formic acid, HCOOH, at 25 C is 2. 38. Calculate Ka formic acid at this temperature. To calculate Ka, we need the equilibrium concentrations of all three things. We can find [H 3 O+], which is the same as [HCOO ], from the p. H. © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Ka from the p. H = log [H 3 O+] 2. 38 = Calculating Ka from the p. H = log [H 3 O+] 2. 38 =](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-28.jpg)
Calculating Ka from the p. H = log [H 3 O+] 2. 38 = log [H 3 O+] 10 2. 38 = 10 log [H 3 O+] = [H 3 O+] 4. 2 10 3 = [H 3 O+] = [HCOO ] © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Ka from p. H Now we can set up a table… Initially [HCOOH], Calculating Ka from p. H Now we can set up a table… Initially [HCOOH],](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-29.jpg)
Calculating Ka from p. H Now we can set up a table… Initially [HCOOH], M [H 3 O+], M [HCOO ], M 0. 10 0 0 Change At equilibrium © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Ka from p. H Now we can set up a table… [HCOOH], M Calculating Ka from p. H Now we can set up a table… [HCOOH], M](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-30.jpg)
Calculating Ka from p. H Now we can set up a table… [HCOOH], M [H 3 O+], M [HCOO ], M Initially 0. 10 0 0 Change 4. 2 10 3 +4. 2 10 3 At equilibrium © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Ka from p. H Now we can set up a table… [HCOOH], M Calculating Ka from p. H Now we can set up a table… [HCOOH], M](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-31.jpg)
Calculating Ka from p. H Now we can set up a table… [HCOOH], M [H 3 O+], M [HCOO ], M Initially 0. 10 0 0 Change 4. 2 10 3 +4. 2 10 3 0. 10 4. 2 10 3 = 0. 0958 = 0. 10 4. 2 10 3 At equilibrium © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Ka from p. H [4. 2 10 3] Ka = [0. 10] = Calculating Ka from p. H [4. 2 10 3] Ka = [0. 10] =](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-32.jpg)
Calculating Ka from p. H [4. 2 10 3] Ka = [0. 10] = 1. 8 10 4 © 2012 Pearson Education, Inc. Acids and Bases
![Calculating Percent Ionization [H 3 O+]eq • Percent ionization = [HA] 100 initial • Calculating Percent Ionization [H 3 O+]eq • Percent ionization = [HA] 100 initial •](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-33.jpg)
Calculating Percent Ionization [H 3 O+]eq • Percent ionization = [HA] 100 initial • In this example, [H 3 O+]eq = 4. 2 10 3 M [HCOOH]initial = 0. 10 M 4. 2 10 3 Percent ionization = 100 0. 10 = 4. 2% © 2012 Pearson Education, Inc. Acids and Bases

Calculating p. H from Ka Calculate the p. H of a 0. 30 M solution of acetic acid, HC 2 H 3 O 2, at 25 C. HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2 (aq) Ka for acetic acid at 25 C is 1. 8 10 5. © 2012 Pearson Education, Inc. Acids and Bases
![Calculating p. H from Ka The equilibrium constant expression is [H 3 O+] [C Calculating p. H from Ka The equilibrium constant expression is [H 3 O+] [C](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-35.jpg)
Calculating p. H from Ka The equilibrium constant expression is [H 3 O+] [C 2 H 3 O 2 ] Ka = [HC 2 H 3 O 2] © 2012 Pearson Education, Inc. Acids and Bases
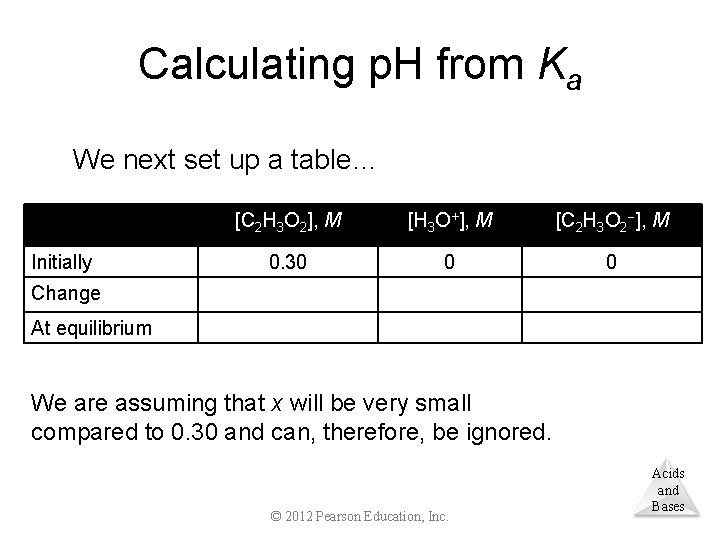
Calculating p. H from Ka We next set up a table… Initially [C 2 H 3 O 2], M [H 3 O+], M [C 2 H 3 O 2 ], M 0. 30 0 0 Change At equilibrium We are assuming that x will be very small compared to 0. 30 and can, therefore, be ignored. © 2012 Pearson Education, Inc. Acids and Bases
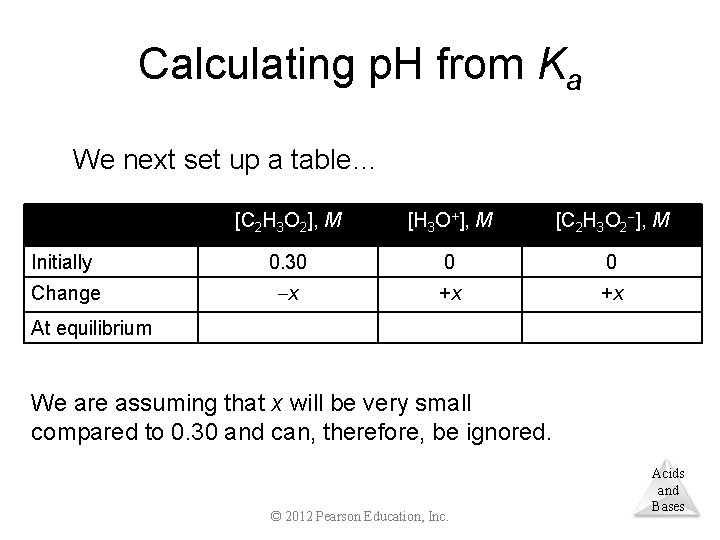
Calculating p. H from Ka We next set up a table… [C 2 H 3 O 2], M [H 3 O+], M [C 2 H 3 O 2 ], M Initially 0. 30 0 0 Change x +x +x At equilibrium We are assuming that x will be very small compared to 0. 30 and can, therefore, be ignored. © 2012 Pearson Education, Inc. Acids and Bases
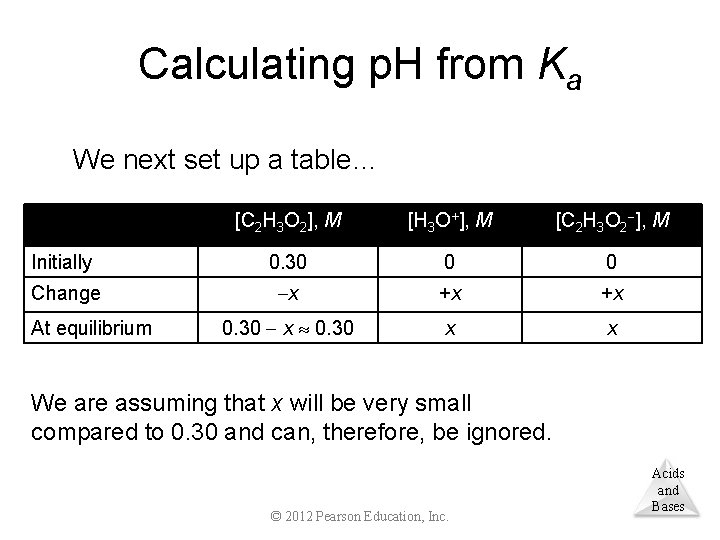
Calculating p. H from Ka We next set up a table… [C 2 H 3 O 2], M [H 3 O+], M [C 2 H 3 O 2 ], M Initially 0. 30 0 0 Change x +x +x 0. 30 x 0. 30 x x At equilibrium We are assuming that x will be very small compared to 0. 30 and can, therefore, be ignored. © 2012 Pearson Education, Inc. Acids and Bases

Calculating p. H from Ka Now, 2 (x) 1. 8 10 5 = (0. 30) (1. 8 10 5) (0. 30) = x 2 5. 4 10 6 = x 2 2. 3 10 3 = x © 2012 Pearson Education, Inc. Acids and Bases
![Calculating p. H from Ka p. H = log [H 3 O+] p. H Calculating p. H from Ka p. H = log [H 3 O+] p. H](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-40.jpg)
Calculating p. H from Ka p. H = log [H 3 O+] p. H = log (2. 3 10 3) p. H = 2. 64 © 2012 Pearson Education, Inc. Acids and Bases

Polyprotic Acids Polyprotic acids have more than one acidic proton. If the difference between the Ka for the first dissociation and subsequent Ka values is 103 or more, the p. H generally depends only on the first dissociation. © 2012 Pearson Education, Inc. Acids and Bases

Weak Bases react with water to produce hydroxide ion. © 2012 Pearson Education, Inc. Acids and Bases
![Weak Bases The equilibrium constant expression for this reaction is [HB] [OH ] Kb Weak Bases The equilibrium constant expression for this reaction is [HB] [OH ] Kb](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-43.jpg)
Weak Bases The equilibrium constant expression for this reaction is [HB] [OH ] Kb = [B ] where Kb is the base-dissociation constant. © 2012 Pearson Education, Inc. Acids and Bases
![Weak Bases Kb can be used to find [OH ] and, through it, p. Weak Bases Kb can be used to find [OH ] and, through it, p.](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-44.jpg)
Weak Bases Kb can be used to find [OH ] and, through it, p. H. © 2012 Pearson Education, Inc. Acids and Bases
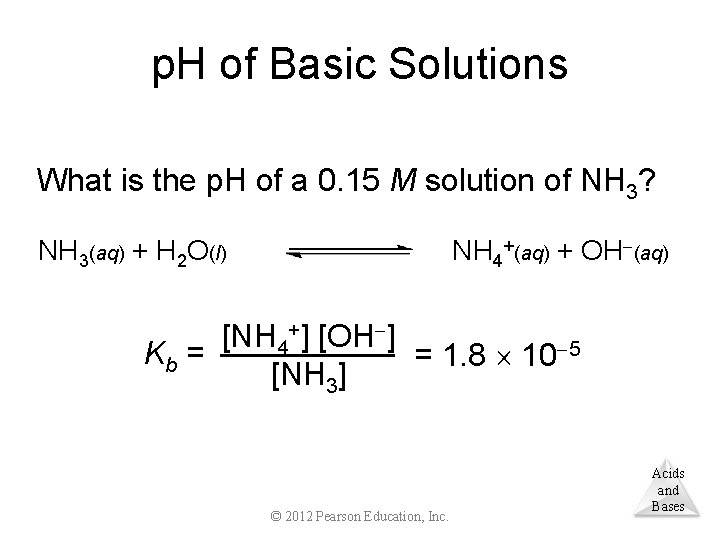
p. H of Basic Solutions What is the p. H of a 0. 15 M solution of NH 3? NH 4+(aq) + OH (aq) NH 3(aq) + H 2 O(l) [NH 4+] [OH ] Kb = = 1. 8 10 5 [NH 3] © 2012 Pearson Education, Inc. Acids and Bases
![p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-46.jpg)
p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M [NH 4+], M [OH ], M 0. 15 0 0 © 2012 Pearson Education, Inc. Acids and Bases
![p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-47.jpg)
p. H of Basic Solutions Tabulate the data. Initially At equilibrium [NH 3], M [NH 4+], M [OH ], M 0. 15 x 0. 15 0 x © 2012 Pearson Education, Inc. Acids and Bases

p. H of Basic Solutions 1. 8 10 5 = (x)2 (0. 15) (1. 8 10 5) (0. 15) = x 2 2. 7 10 6 = x 2 1. 6 10 3 = x 2 © 2012 Pearson Education, Inc. Acids and Bases
![p. H of Basic Solutions Therefore, [OH ] = 1. 6 10 3 M p. H of Basic Solutions Therefore, [OH ] = 1. 6 10 3 M](http://slidetodoc.com/presentation_image_h2/ac033743c2cfdc7e96920cae1c323831/image-49.jpg)
p. H of Basic Solutions Therefore, [OH ] = 1. 6 10 3 M p. OH = log (1. 6 10 3) p. OH = 2. 80 p. H = 14. 00 2. 80 p. H = 11. 20 © 2012 Pearson Education, Inc. Acids and Bases

Ka and Kb are related in this way: Ka Kb = Kw Therefore, if you know one of them, you can calculate the other. © 2012 Pearson Education, Inc. Acids and Bases

Reactions of Anions with Water • Anions are bases. • As such, they can react with water in a hydrolysis reaction to form OH and the conjugate acid: X (aq) + H 2 O(l) HX(aq) + OH (aq) © 2012 Pearson Education, Inc. Acids and Bases
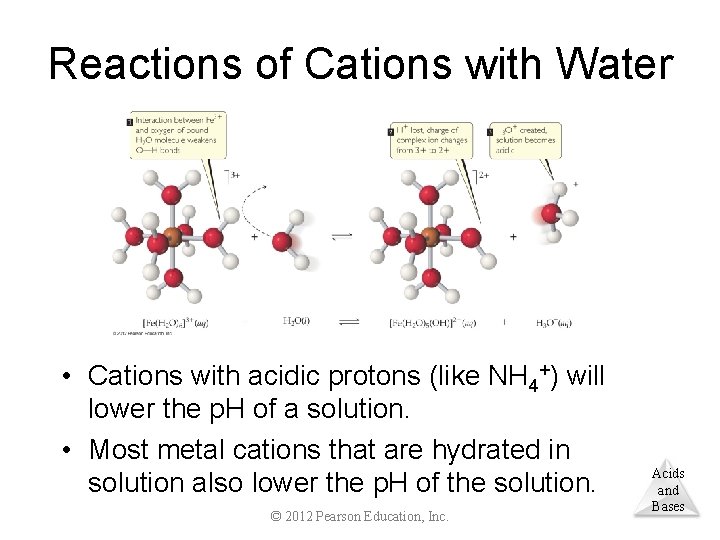
Reactions of Cations with Water • Cations with acidic protons (like NH 4+) will lower the p. H of a solution. • Most metal cations that are hydrated in solution also lower the p. H of the solution. © 2012 Pearson Education, Inc. Acids and Bases

Reactions of Cations with Water • Attraction between nonbonding electrons on oxygen and the metal causes a shift of the electron density in water. • This makes the O–H bond more polar and the water Acids and more acidic. Bases © 2012 Pearson Education, Inc.

Reactions of Cations with Water • Greater charge and smaller size make a cation more acidic. © 2012 Pearson Education, Inc. Acids and Bases

Effect of Cations and Anions 1. An anion that is the conjugate base of a strong acid will not affect the p. H. 2. An anion that is the conjugate base of a weak acid will increase the p. H. 3. A cation that is the conjugate acid of a weak base will decrease the p. H. © 2012 Pearson Education, Inc. Acids and Bases

Effect of Cations and Anions 4. Cations of the strong Arrhenius bases will not affect the p. H. 5. Other metal ions will cause a decrease in p. H. 6. When a solution contains both a weak acid and a weak base, the affect on p. H depends on the Ka and Kb values. © 2012 Pearson Education, Inc. Acids and Bases

Factors Affecting Acid Strength • The more polar the H –X bond and/or the weaker the H–X bond, the more acidic the compound. • So acidity increases from left to right across a row and from top to bottom down a group. © 2012 Pearson Education, Inc. Acids and Bases

Factors Affecting Acid Strength In oxyacids, in which an –OH is bonded to another atom, Y, the more electronegative Y is, the more acidic the acid. © 2012 Pearson Education, Inc. Acids and Bases

Factors Affecting Acid Strength For a series of oxyacids, acidity increases with the number of oxygens. © 2012 Pearson Education, Inc. Acids and Bases
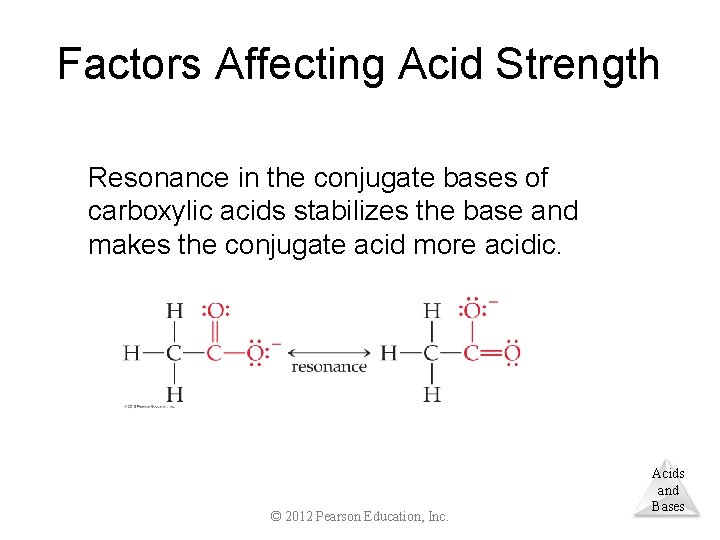
Factors Affecting Acid Strength Resonance in the conjugate bases of carboxylic acids stabilizes the base and makes the conjugate acid more acidic. © 2012 Pearson Education, Inc. Acids and Bases
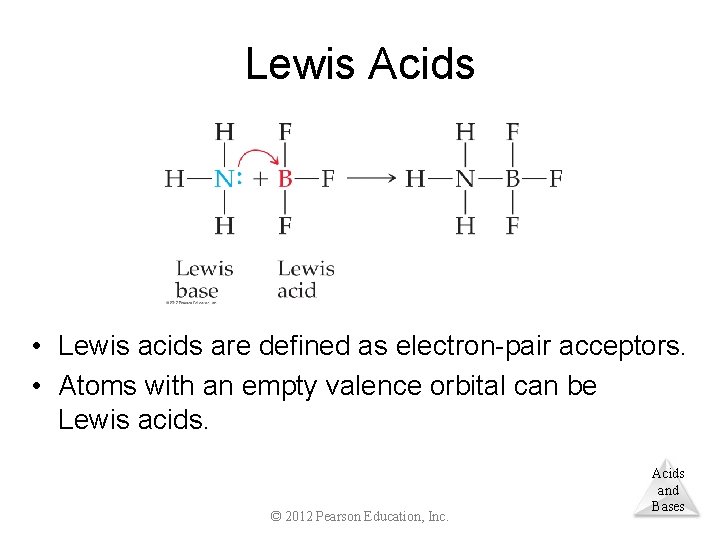
Lewis Acids • Lewis acids are defined as electron-pair acceptors. • Atoms with an empty valence orbital can be Lewis acids. © 2012 Pearson Education, Inc. Acids and Bases
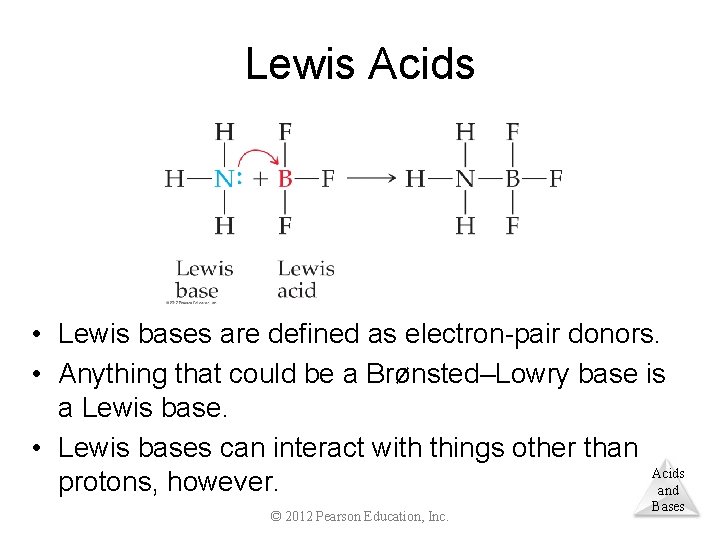
Lewis Acids • Lewis bases are defined as electron-pair donors. • Anything that could be a Brønsted–Lowry base is a Lewis base. • Lewis bases can interact with things other than Acids protons, however. and © 2012 Pearson Education, Inc. Bases
- Slides: 62