Lecture Presentation Chapter 10 Molecular Geometry and Bonding

Lecture Presentation Chapter 10 Molecular Geometry and Bonding Theories © 2015 Pearson Education, Inc. James F. Kirby Quinnipiac University Hamden, CT
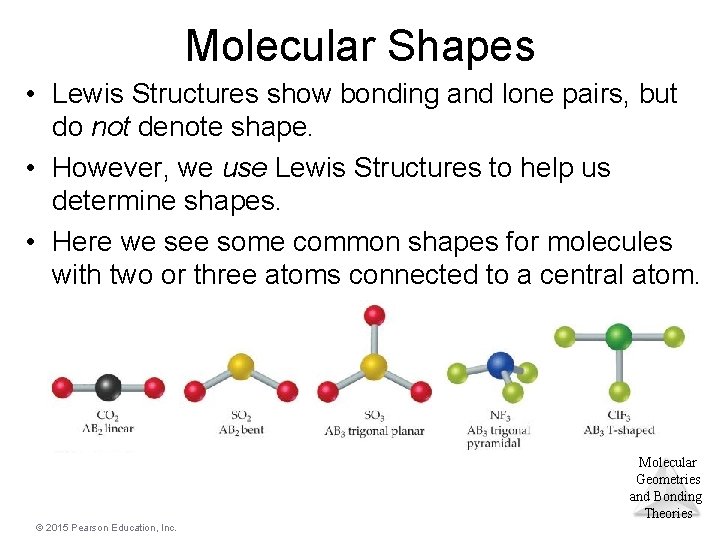
Molecular Shapes • Lewis Structures show bonding and lone pairs, but do not denote shape. • However, we use Lewis Structures to help us determine shapes. • Here we see some common shapes for molecules with two or three atoms connected to a central atom. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
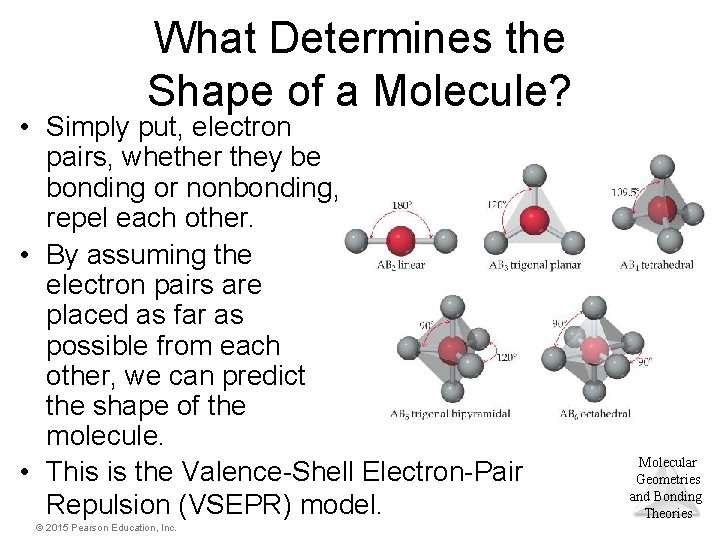
What Determines the Shape of a Molecule? • Simply put, electron pairs, whether they be bonding or nonbonding, repel each other. • By assuming the electron pairs are placed as far as possible from each other, we can predict the shape of the molecule. • This is the Valence-Shell Electron-Pair Repulsion (VSEPR) model. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories

VSEPR Model • VSEPR: Valence Shell Electron-Pair Repulsion. • The structure around a given atom is determined principally by minimizing electron pair repulsions. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 4
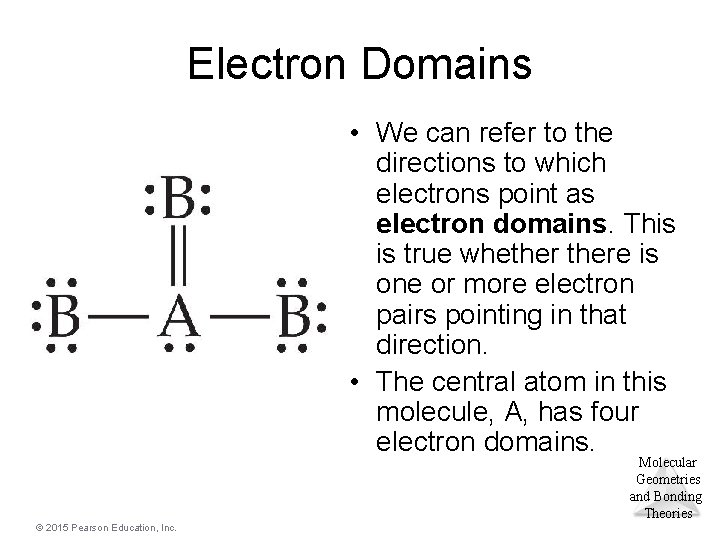
Electron Domains • We can refer to the directions to which electrons point as electron domains. This is true whethere is one or more electron pairs pointing in that direction. • The central atom in this molecule, A, has four electron domains. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Valence-Shell Electron-Pair Repulsion (VSEPR) Model “The best arrangement of a given number of electron domains is the one that minimizes the repulsions among them. ” (The balloon analogy in the figure to the left demonstrates the maximum distances, which minimize Molecular Geometries repulsions. ) and Bonding Theories © 2015 Pearson Education, Inc.

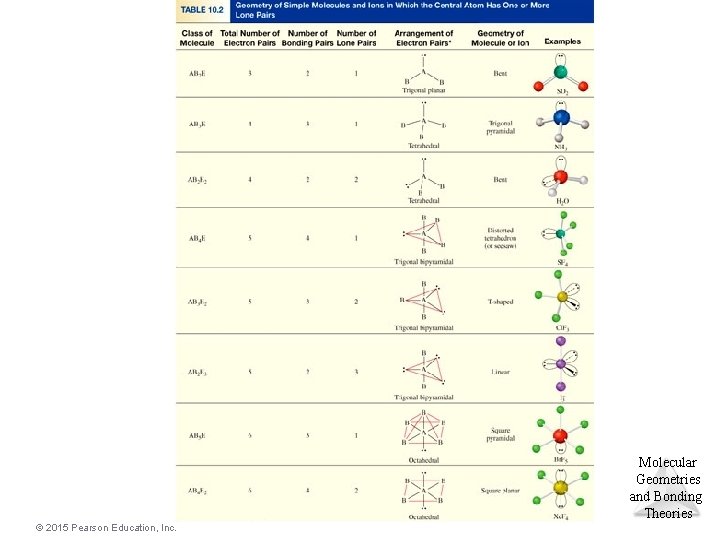
Electron-Domain Geometries • The Table shows the electron-domain geometries for two through six electron domains around a central atom. • To determine the electron-domain geometry, count the total number of lone pairs, single, double, and triple bonds on Molecular Geometries the central atom. and Bonding Theories © 2015 Pearson Education, Inc.
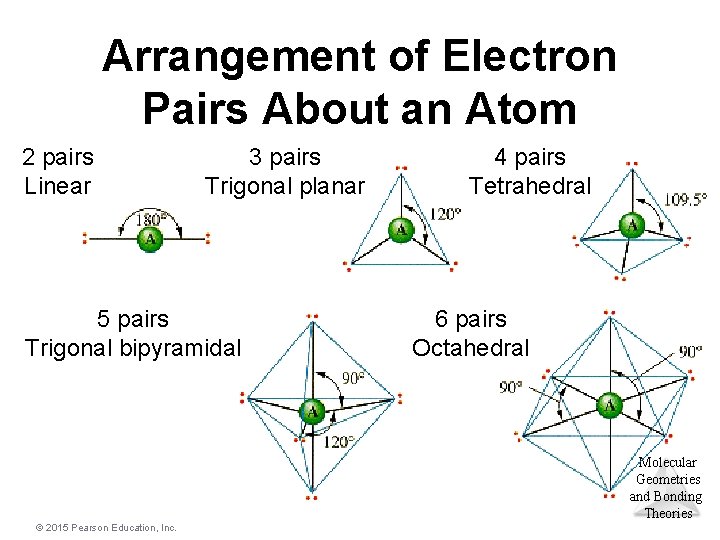
Arrangement of Electron Pairs About an Atom 2 pairs Linear 3 pairs Trigonal planar 5 pairs Trigonal bipyramidal 4 pairs Tetrahedral 6 pairs Octahedral Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Predicting Molecular Geometry • The following rules and figures will help discern electron pair arrangements. 1. Draw the Lewis structure 2. Determine how many electrons pairs are around the central atom. Count a multiple bond as one pair. 3. Arrange the electron pairs. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
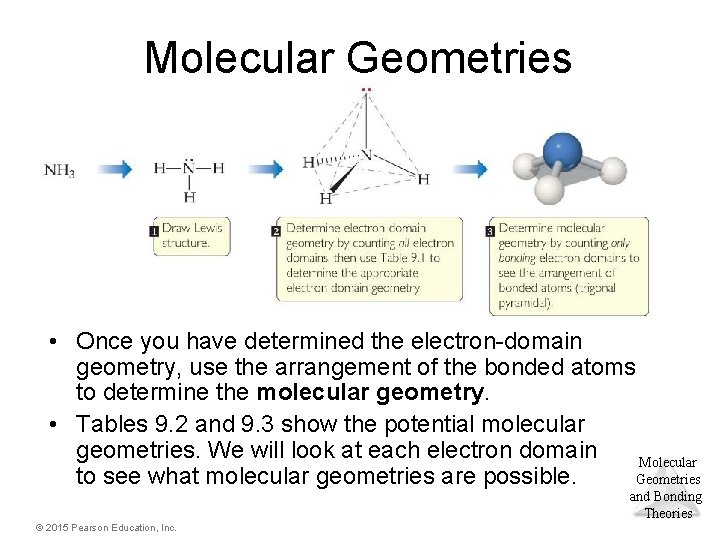
Molecular Geometries • Once you have determined the electron-domain geometry, use the arrangement of the bonded atoms to determine the molecular geometry. • Tables 9. 2 and 9. 3 show the potential molecular geometries. We will look at each electron domain Molecular Geometries to see what molecular geometries are possible. and Bonding Theories © 2015 Pearson Education, Inc.

Linear Electron Domain • In the linear domain, there is only one molecular geometry: linear. • NOTE: If there are only two atoms in the molecule, the molecule will be linear no matter what the electron domain is. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
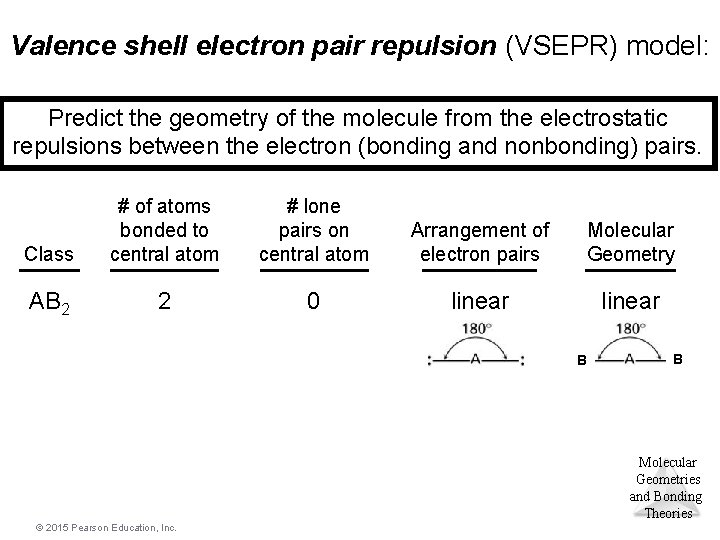
Valence shell electron pair repulsion (VSEPR) model: Predict the geometry of the molecule from the electrostatic repulsions between the electron (bonding and nonbonding) pairs. Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear B B Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Trigonal Planar Electron Domain • There are two molecular geometries: – trigonal planar, if all electron domains are bonding, and – bent, if one of the domains is a nonbonding pair. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
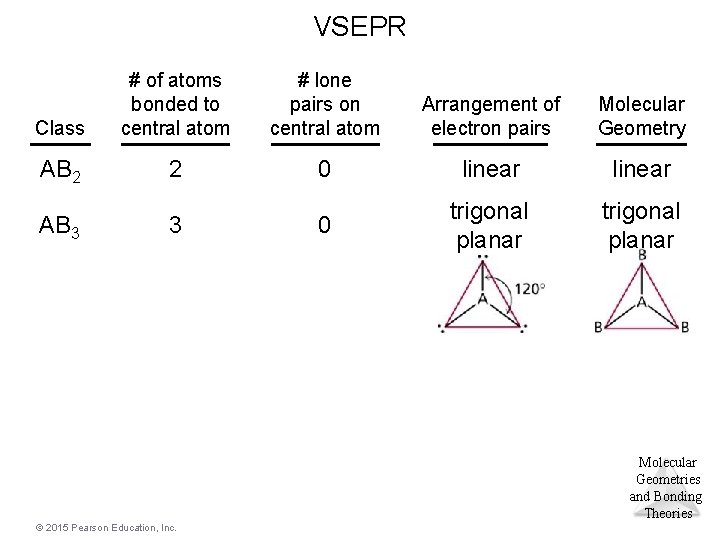
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear 0 trigonal planar AB 3 3 Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Tetrahedral Electron Domain • There are three molecular geometries: – tetrahedral, if all are bonding pairs, – trigonal pyramidal, if one is a nonbonding pair, and – bent, if there are two nonbonding pairs. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
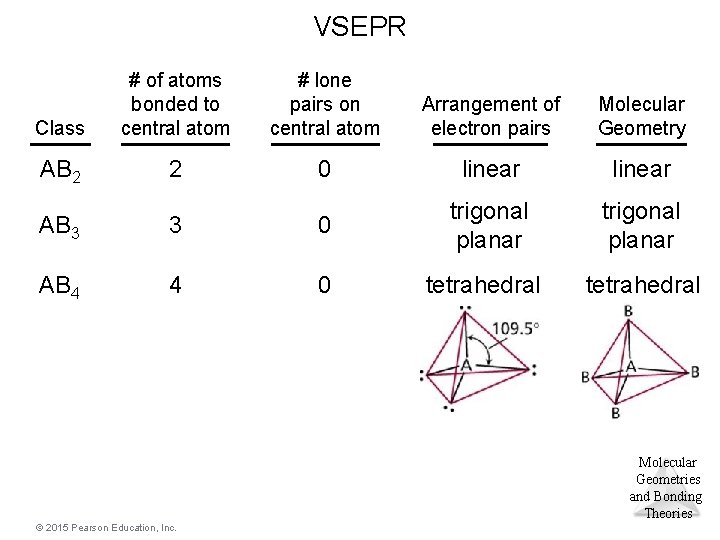
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear trigonal planar tetrahedral AB 3 3 0 trigonal planar AB 4 4 0 tetrahedral Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
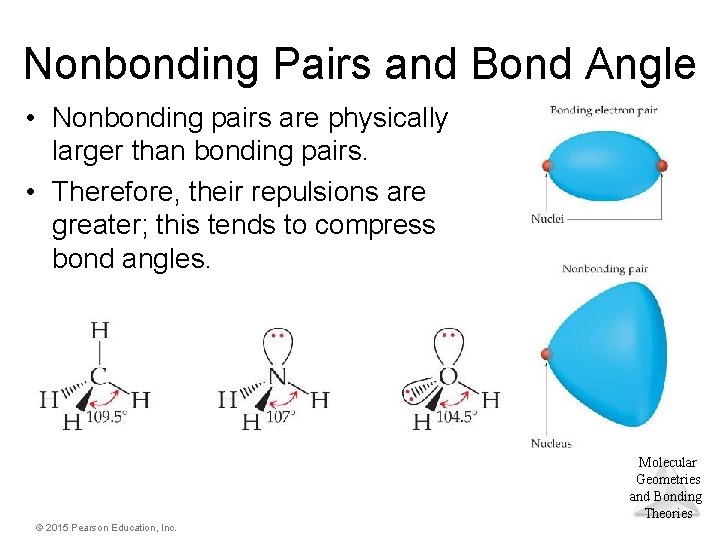
Nonbonding Pairs and Bond Angle • Nonbonding pairs are physically larger than bonding pairs. • Therefore, their repulsions are greater; this tends to compress bond angles. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
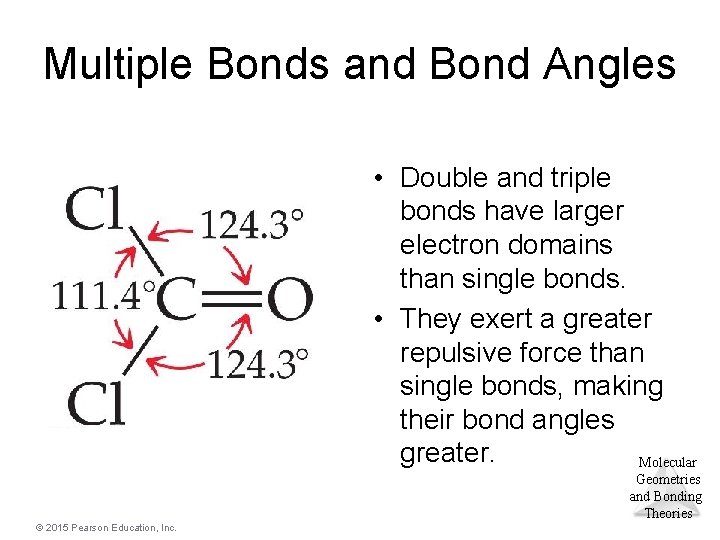
Multiple Bonds and Bond Angles • Double and triple bonds have larger electron domains than single bonds. • They exert a greater repulsive force than single bonds, making their bond angles greater. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Expanding beyond the Octet Rule • Remember that some elements can break the octet rule and make more than four bonds (or have more than four electron domains). • The result is two more possible electron domains: five = trigonal bipyramidal; six = octahedral (as was seen in the slide on electron-domain geometries). Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
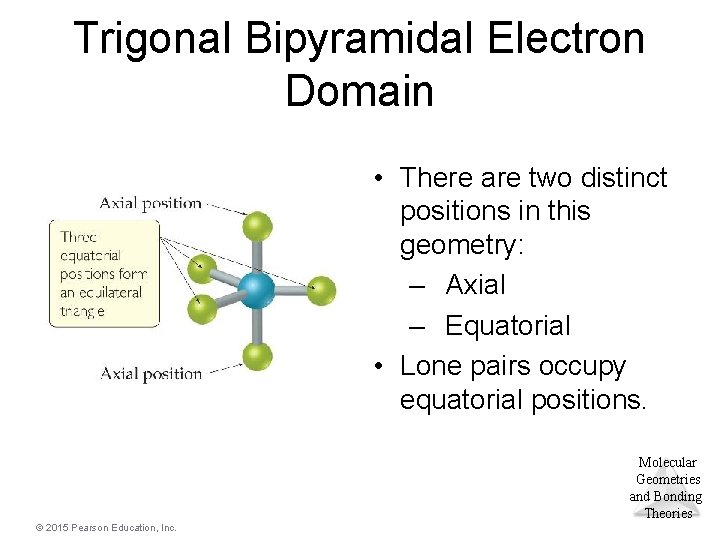
Trigonal Bipyramidal Electron Domain • There are two distinct positions in this geometry: – Axial – Equatorial • Lone pairs occupy equatorial positions. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Trigonal Bipyramidal Electron Domain • There are four distinct molecular geometries in this domain: – Trigonal bipyramidal – Seesaw – T-shaped – Linear © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
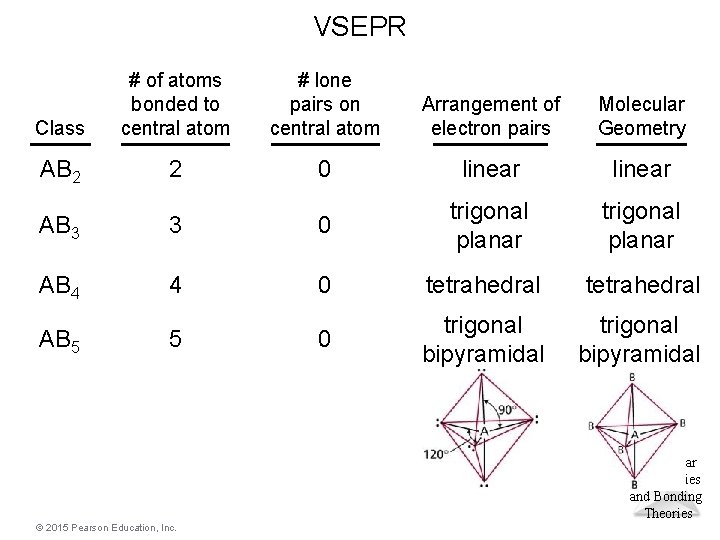
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear trigonal planar AB 3 3 0 trigonal planar AB 4 4 0 tetrahedral AB 5 5 0 trigonal bipyramidal Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Octahedral Electron Domain • All positions are equivalent in the octahedral domain. • There are three molecular geometries: – Octahedral – Square pyramidal – Square planar Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
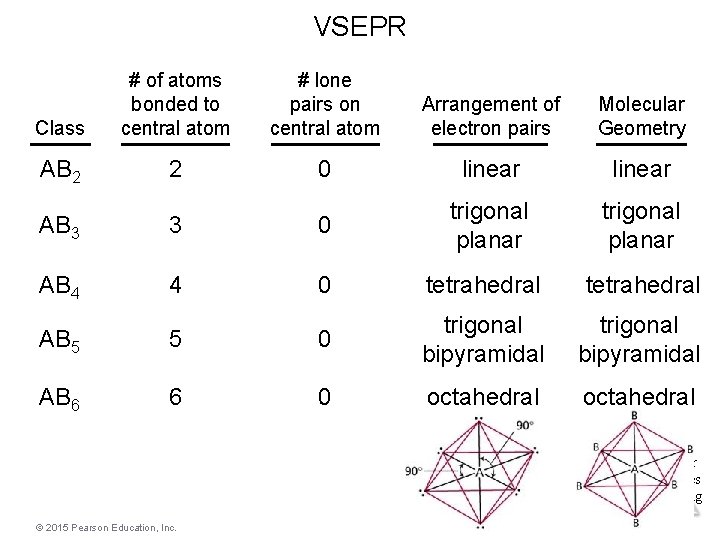
VSEPR Class # of atoms bonded to central atom # lone pairs on central atom Arrangement of electron pairs Molecular Geometry AB 2 2 0 linear trigonal planar AB 3 3 0 trigonal planar AB 4 4 0 tetrahedral AB 5 5 0 trigonal bipyramidal AB 6 6 0 octahedral Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
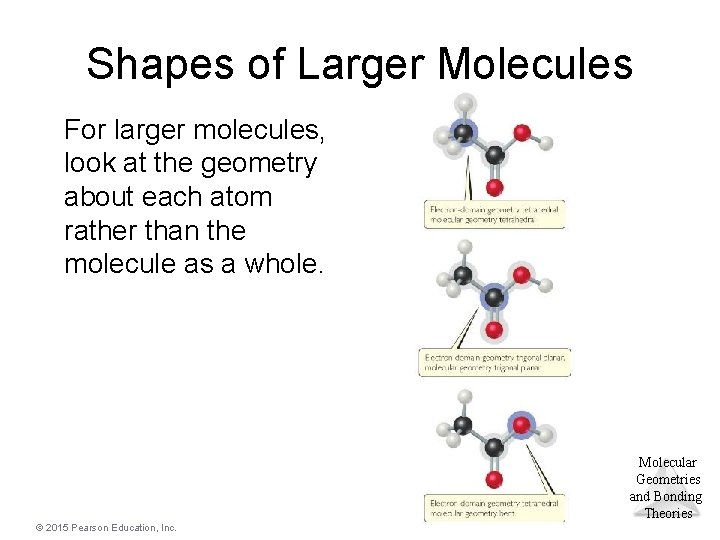
Shapes of Larger Molecules For larger molecules, look at the geometry about each atom rather than the molecule as a whole. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Polarity of Molecules Ask yourself: COVALENT or IONIC? If COVALENT: Are the BONDS polar? a. NO: The molecule is NONPOLAR! b. YES: Continue—Do the AVERAGE position of δ+ and δ– coincide? 1) YES: The molecule is NONPOLAR. 2) NO: The molecule is POLAR. NOTE: Different atoms attached to the central Molecular Geometries atom have different polarity of bonds. and Bonding Theories © 2015 Pearson Education, Inc.

Dipole Moment • Property of a molecule whose charge distribution can be represented by a center of positive charge and a center of negative charge. • Use an arrow to represent a dipole moment. – Point to the negative charge center with the tail of the arrow indicating the positive center of charge. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 28
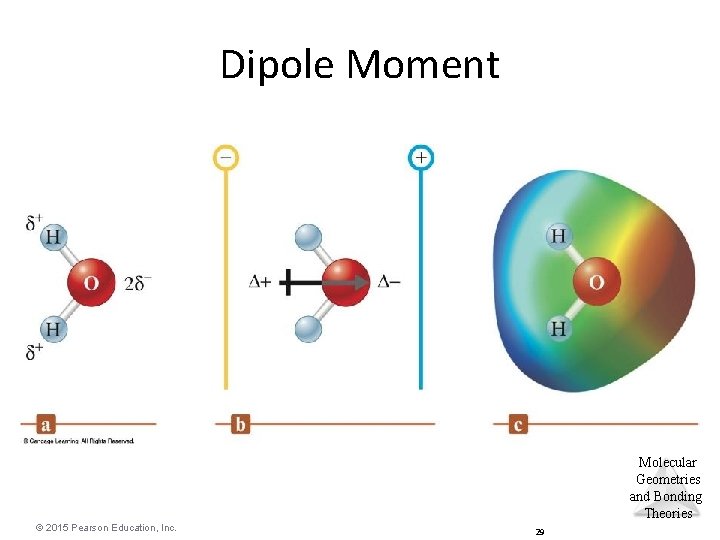
Dipole Moment Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 29
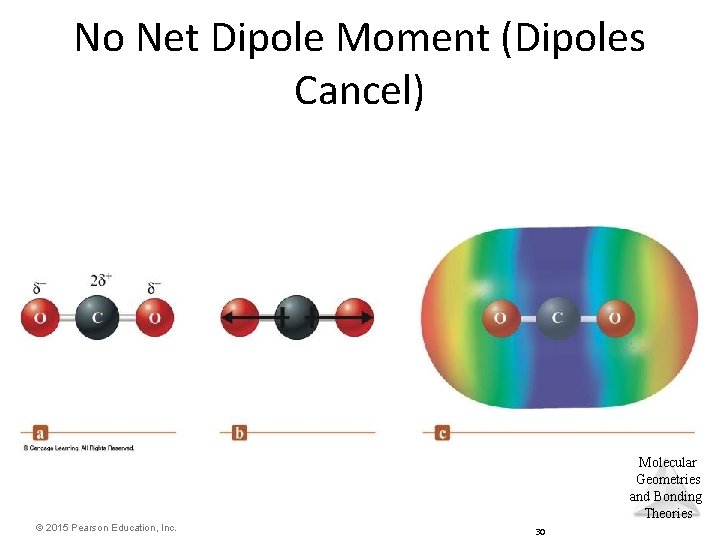
No Net Dipole Moment (Dipoles Cancel) Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 30
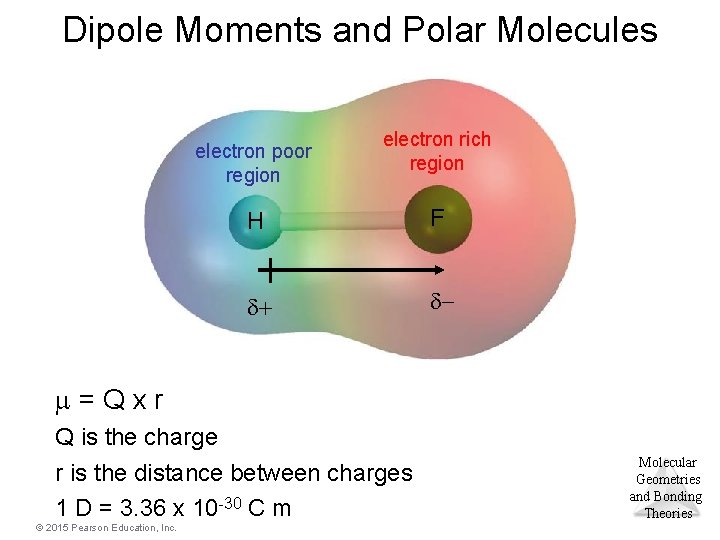
Dipole Moments and Polar Molecules electron poor region electron rich region H F d+ d- m=Qxr Q is the charge r is the distance between charges 1 D = 3. 36 x 10 -30 C m © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
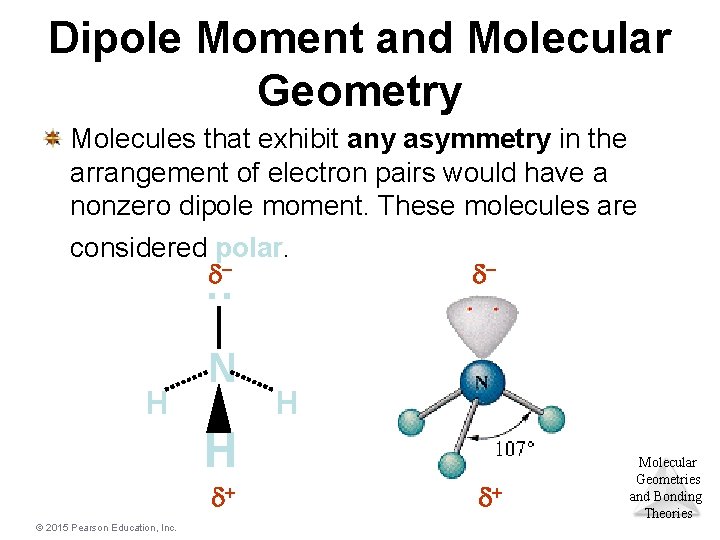
Dipole Moment and Molecular Geometry : Molecules that exhibit any asymmetry in the arrangement of electron pairs would have a nonzero dipole moment. These molecules are considered polar. dd- H N H H d+ © 2015 Pearson Education, Inc. d+ Molecular Geometries and Bonding Theories
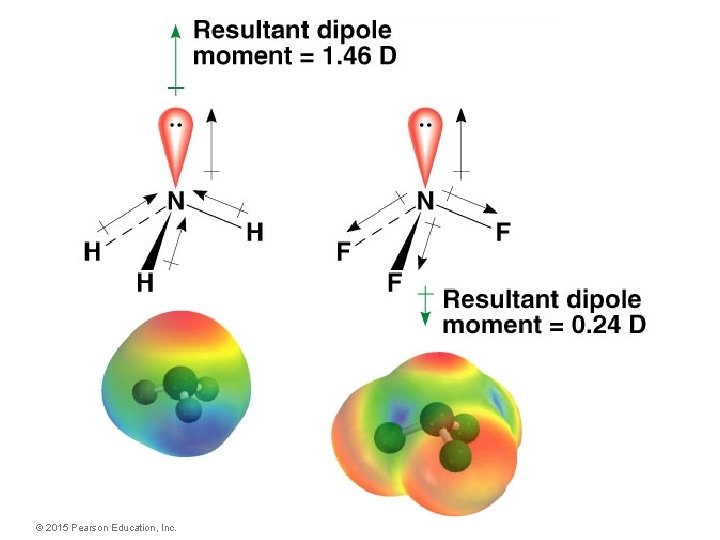
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
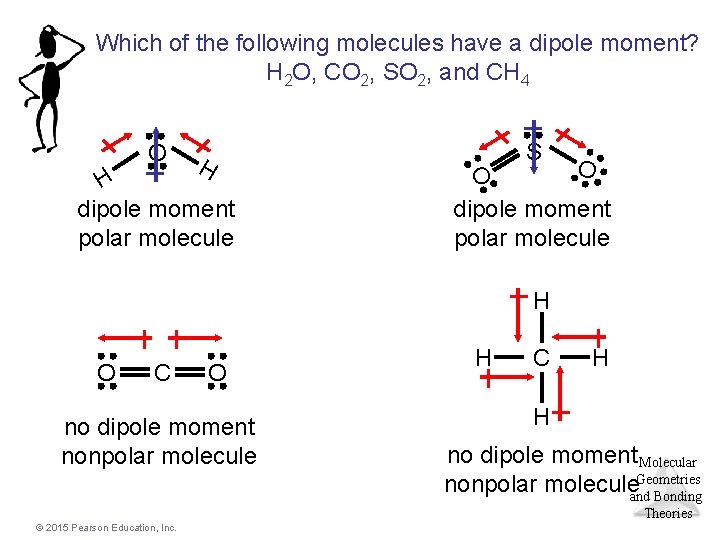
Which of the following molecules have a dipole moment? H 2 O, CO 2, SO 2, and CH 4 O H H dipole moment polar molecule S O O dipole moment polar molecule H O C O no dipole moment nonpolar molecule H C H H no dipole moment Molecular Geometries nonpolar moleculeand Bonding Theories © 2015 Pearson Education, Inc.
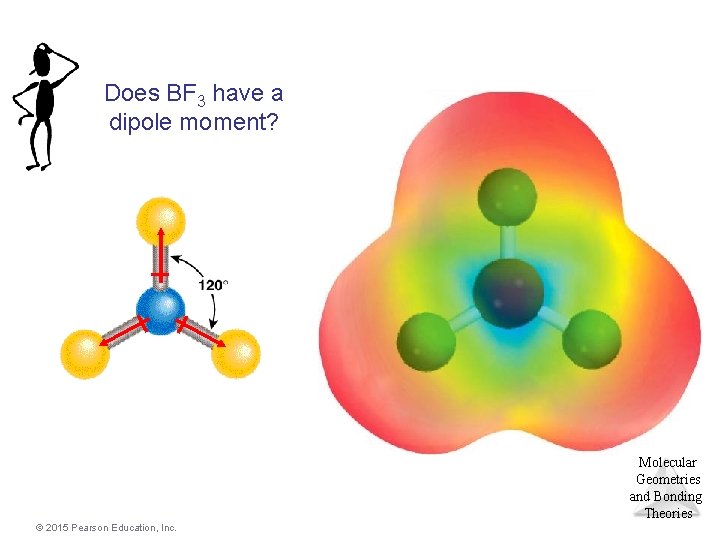
Does BF 3 have a dipole moment? Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
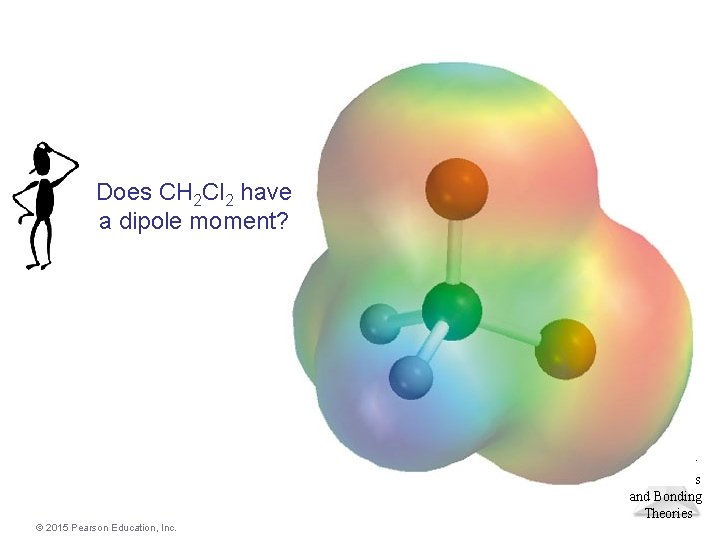
Does CH 2 Cl 2 have a dipole moment? Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Comparison of the Polarity of Two Molecules A NONPOLAR molecule A POLAR molecule Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
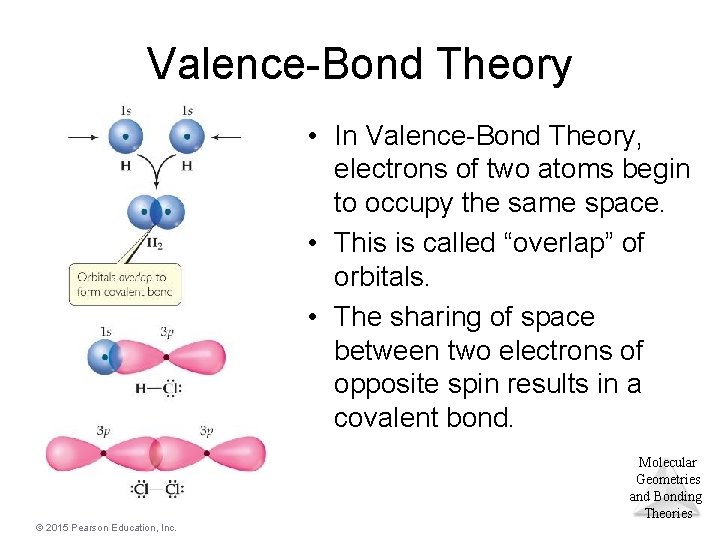
Valence-Bond Theory • In Valence-Bond Theory, electrons of two atoms begin to occupy the same space. • This is called “overlap” of orbitals. • The sharing of space between two electrons of opposite spin results in a covalent bond. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
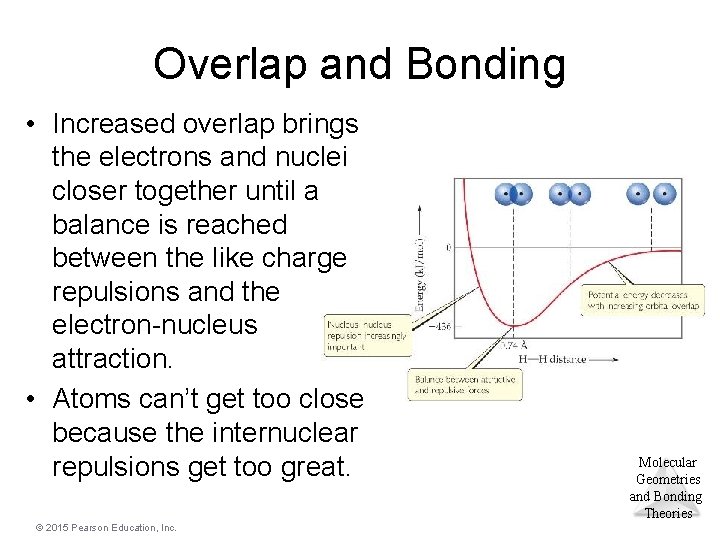
Overlap and Bonding • Increased overlap brings the electrons and nuclei closer together until a balance is reached between the like charge repulsions and the electron-nucleus attraction. • Atoms can’t get too close because the internuclear repulsions get too great. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories

VSEPR and Hybrid Orbitals • • VSEPR predicts shapes of molecules very well. How does that fit with orbitals? Let’s use CH 4 as an example: If we look at carbon, its electron configuration is 1 s 22 p 2. If it shares electrons with hydrogen it should have two different kind of interaction 2 s(H)-1 s(C) to form the four covalent bonds. • Is it what observed? Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

EXERCISE! Draw the Lewis structure for methane, CH 4. – What is the shape of a methane molecule? tetrahedral – What are the bond angles? 109. 5 o Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 42

CONCEPT CHECK! What is the valence electron configuration of a carbon atom? s 2 p 2 Why can’t the bonding orbitals for methane be formed by an overlap of atomic orbitals? Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 43

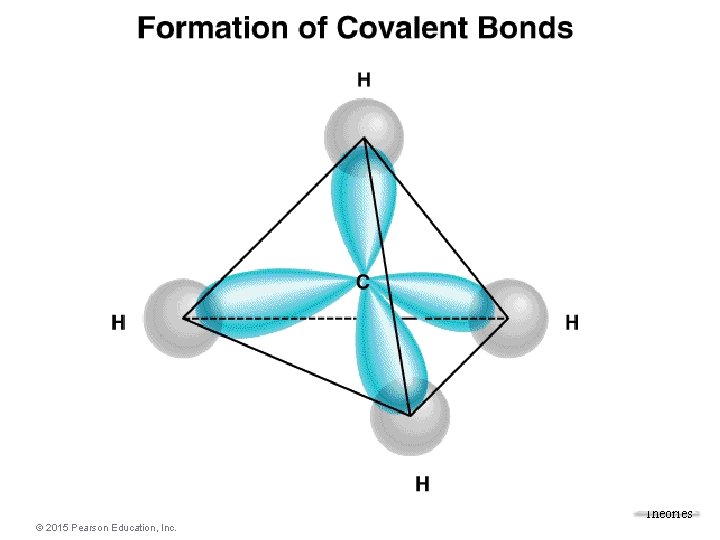
Bonding in Methane The carbon atom has four equivalent atomic orbitals, arranged tetrahedrally. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 44

Hybridization • Mixing of the native atomic orbitals to form special orbitals for bonding. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 45

sp 3 Hybridization • Combination of one s and three p orbitals. • Whenever a set of equivalent tetrahedral atomic orbitals is required by an atom, the localized electron model assumes that the atom adopts a set of sp 3 orbitals; the atom becomes sp 3 hybridized. • The four orbitals are identical in shape. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 46

Hybridization – mixing of two or more atomic orbitals to form a new set of hybrid orbitals. 1. Mix at least 2 nonequivalent atomic orbitals (e. g. s and p). Hybrid orbitals have very different shape from original atomic orbitals. 2. Number of hybrid orbitals is equal to number of pure atomic orbitals used in the hybridization process. 3. Covalent bonds are formed by: a. Overlap of hybrid orbitals with atomic orbitals Molecular b. Overlap of hybrid orbitals with other hybrid Geometries and Bonding orbitals Theories © 2015 Pearson Education, Inc.

Hybrid Orbitals • Hybrid orbitals are orbitals used to describe bonding that are obtained by taking combinations of atomic orbitals of an isolated atom. In this case, a set of hybrids are constructed from one “s” orbital and three “p” orbitals, so they are called sp 3 hybrid orbitals. The four sp 3 hybrid orbitals take the shape. Molecular Geometries of a tetrahedron. and Bonding Theories © 2015 Pearson Education, Inc.
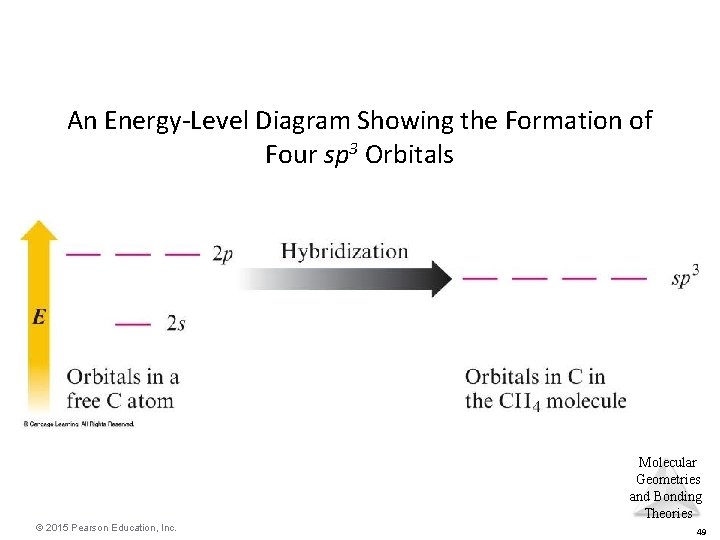
An Energy-Level Diagram Showing the Formation of Four sp 3 Orbitals Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 49
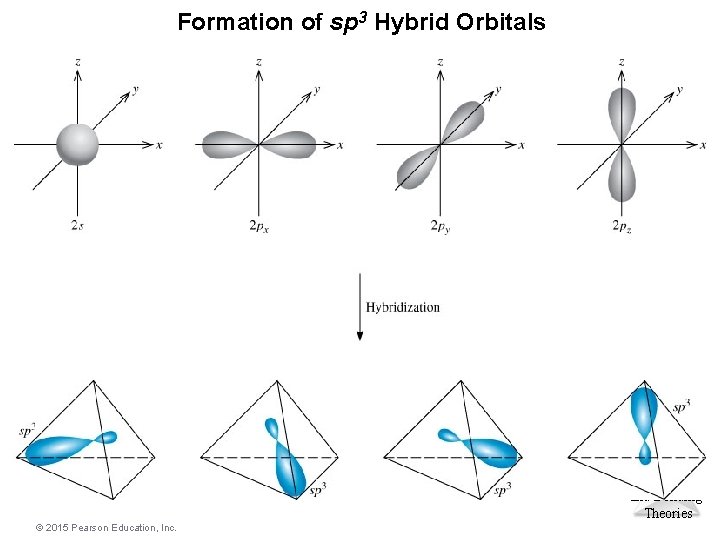
Formation of sp 3 Hybrid Orbitals Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

SP 3 Hybridization https: //www. khanacademy. org/science/or ganic-chemistry/gen-chemreview/hybridization/v/sp 3 -hybridizedorbitals-and-sigma-bonds Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Hybrid Orbitals • Hybrid orbitals form by “mixing” of atomic orbitals to create new orbitals of equal energy, called degenerate orbitals. • When two orbitals “mix” they create two orbitals; when three orbitals mix, they create three orbitals; etc. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Be—sp hybridization • When we look at the orbital diagram for beryllium (Be), we see that there are only paired electrons in full sub-levels. • Be makes electron deficient compounds with two bonds for Be. Why? sp hybridization (mixing of one s orbital and one p orbital) © 2015 Pearson Education, Inc.
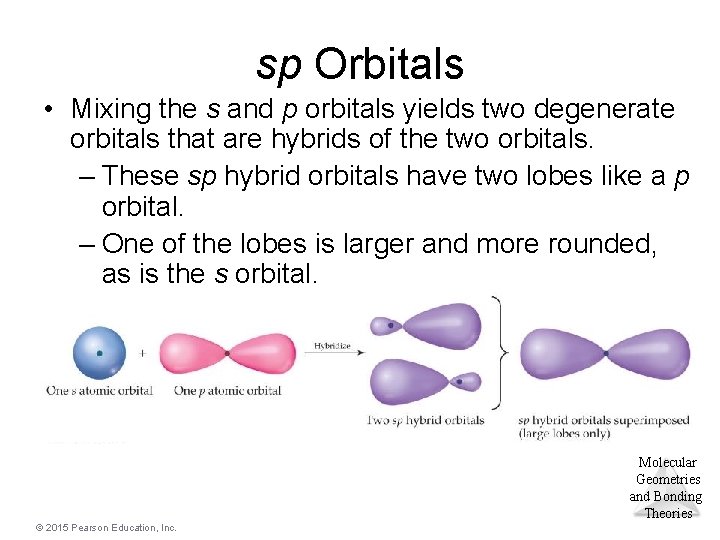
sp Orbitals • Mixing the s and p orbitals yields two degenerate orbitals that are hybrids of the two orbitals. – These sp hybrid orbitals have two lobes like a p orbital. – One of the lobes is larger and more rounded, as is the s orbital. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
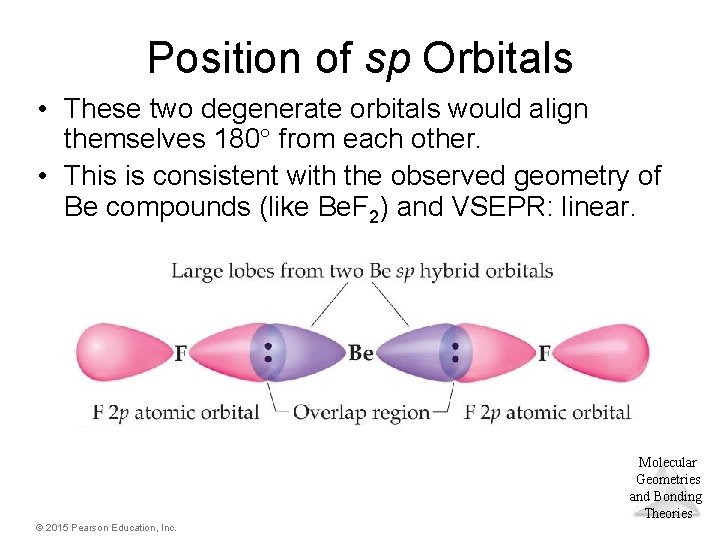
Position of sp Orbitals • These two degenerate orbitals would align themselves 180 from each other. • This is consistent with the observed geometry of Be compounds (like Be. F 2) and VSEPR: linear. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
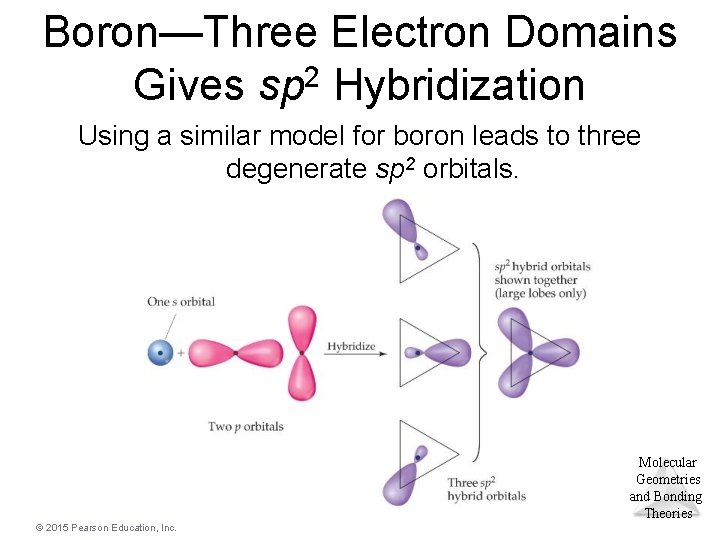
Boron—Three Electron Domains Gives sp 2 Hybridization Using a similar model for boron leads to three degenerate sp 2 orbitals. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
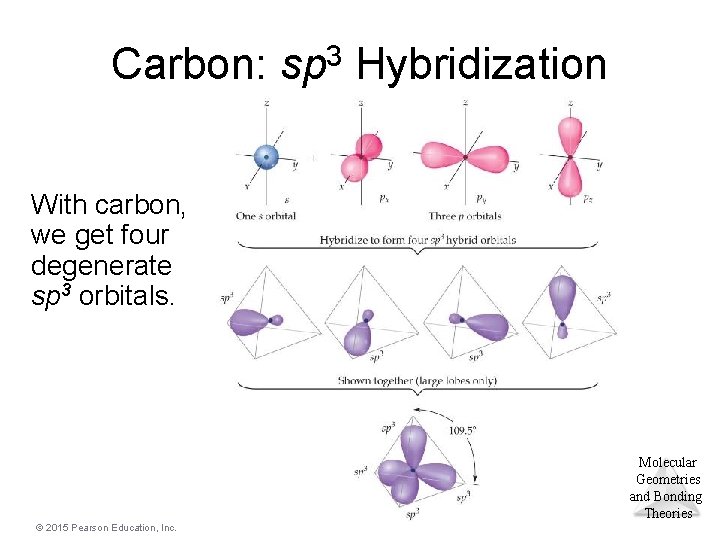
Carbon: sp 3 Hybridization With carbon, we get four degenerate sp 3 orbitals. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Hypervalent Molecules • The elements which have more than an octet • Valence-Bond model would use d orbitals to make more than four bonds. • This view works for period 3 and below. • Theoretical studies suggest that the energy needed would be too great for this. • A more detailed bonding view is needed Molecular than we will use in this course. Geometries and Bonding Theories © 2015 Pearson Education, Inc.

What Happens with Water? • We start this discussion with H 2 O and the angle question: Why is it 104. 5° instead of 90°? • Oxygen has two bonds and two lone pairs— four electron domains. • The result is sp 3 hybridization! © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
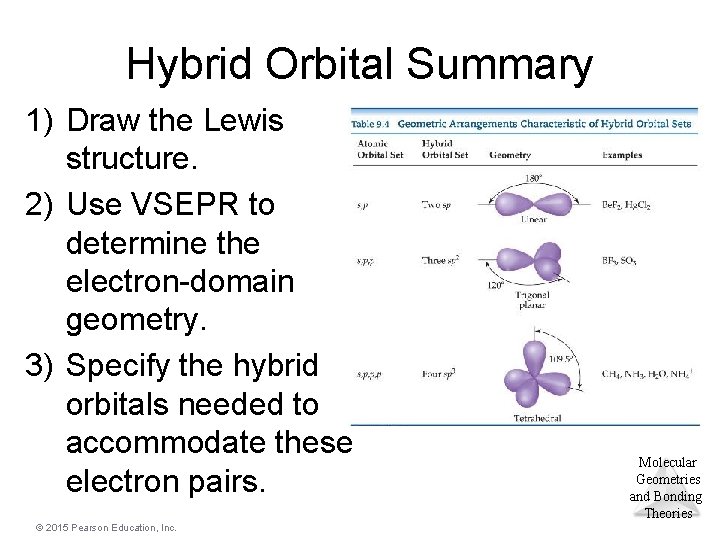
Hybrid Orbital Summary 1) Draw the Lewis structure. 2) Use VSEPR to determine the electron-domain geometry. 3) Specify the hybrid orbitals needed to accommodate these electron pairs. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories

Types of Bonds • How does a double or triple bond form? • It can’t, if we only use hybridized orbitals. • However, if we use the orbitals which are not hybridized, we can have a “side-ways” overlap. • Two types of bonds: • Sigma (σ) bond • Pi (π) bond Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
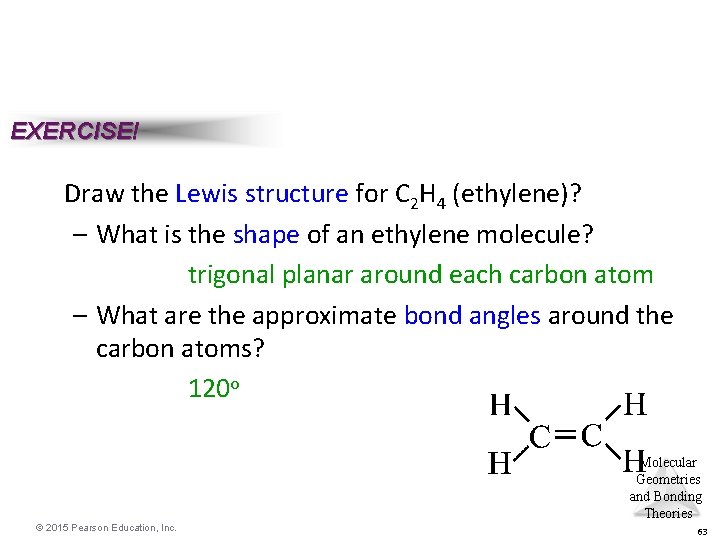
EXERCISE! Draw the Lewis structure for C 2 H 4 (ethylene)? – What is the shape of an ethylene molecule? trigonal planar around each carbon atom – What are the approximate bond angles around the carbon atoms? 120 o Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 63

CONCEPT CHECK! Why can’t sp 3 hybridization account for the ethylene molecule? Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 64

sp 2 Hybridization • Combination of one s and two p orbitals. • Gives a trigonal planar arrangement of atomic orbitals. • One p orbital is not used. – Oriented perpendicular to the plane of the sp 2 orbitals. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 65
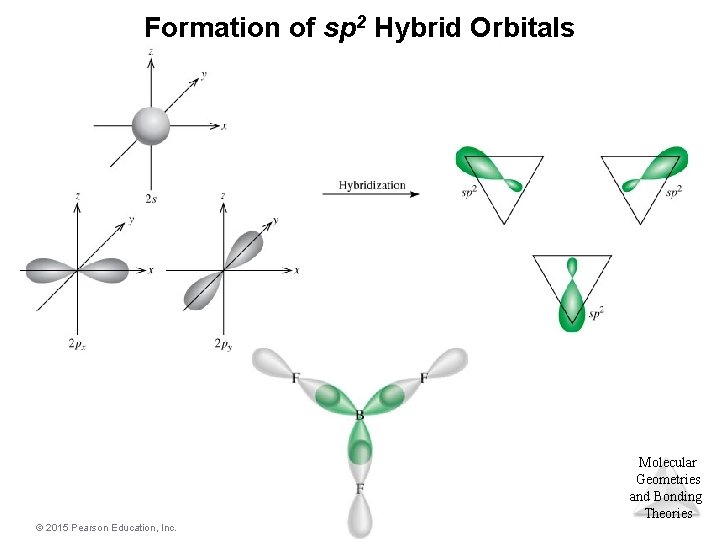
Formation of sp 2 Hybrid Orbitals Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Sigma (Σ) Bond • Electron pair is shared in an area centered on a line running between the atoms. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 67

Pi (Π) Bond • Forms double and triple bonds by sharing electron pair(s) in the space above and below the σ bond. • Uses the unhybridized p orbitals. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 68
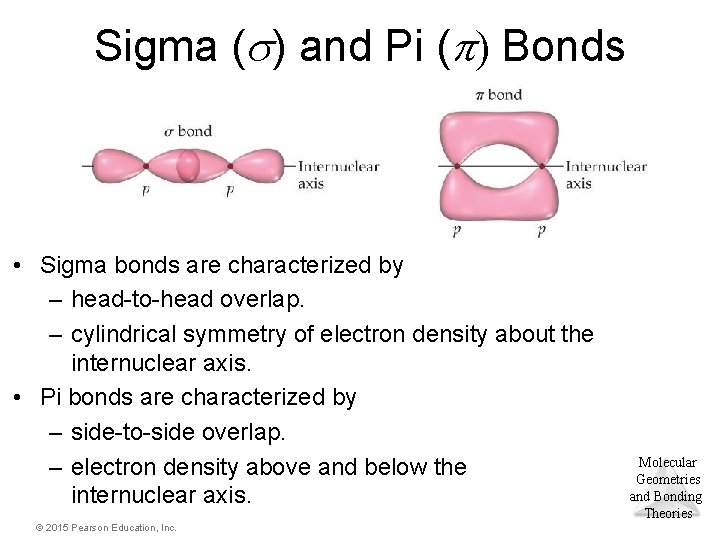
Sigma ( ) and Pi ( ) Bonds • Sigma bonds are characterized by – head-to-head overlap. – cylindrical symmetry of electron density about the internuclear axis. • Pi bonds are characterized by – side-to-side overlap. – electron density above and below the internuclear axis. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories

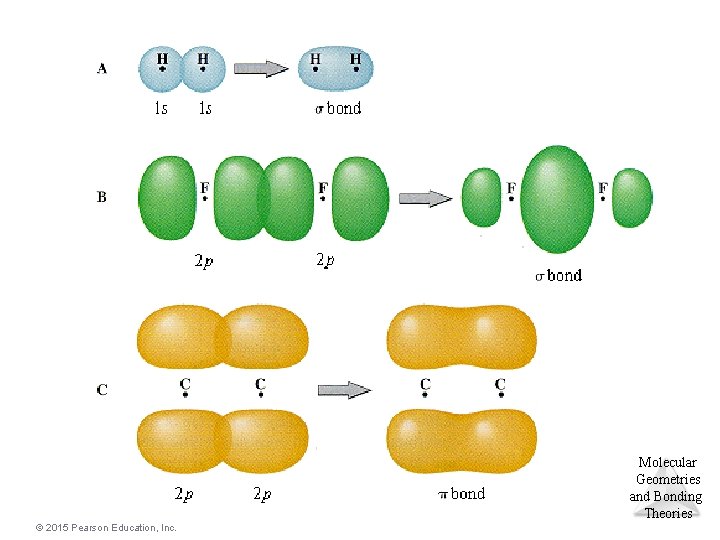
Multiple Bonding • To describe the multiple bonding in ethene, we must first distinguish between two kinds of bonds. A s (sigma) bond is a “head-to-head” overlap of orbitals with a cylindrical shape about the bond axis. This occurs when two “s” orbitals overlap or “p” orbitals overlap along their axis. A p (pi) bond is a “side-to-side” overlap of parallel “p” orbitals, creating an electron distribution above and below the bond axis. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
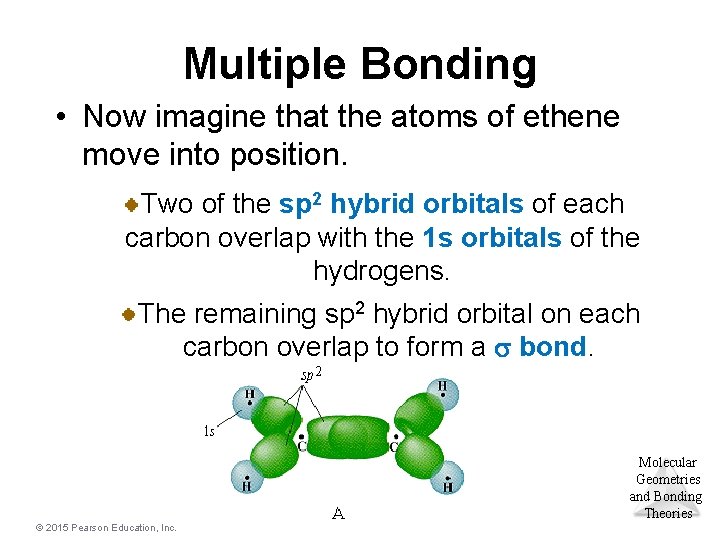
Multiple Bonding • Now imagine that the atoms of ethene move into position. Two of the sp 2 hybrid orbitals of each carbon overlap with the 1 s orbitals of the hydrogens. The remaining sp 2 hybrid orbital on each carbon overlap to form a s bond. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
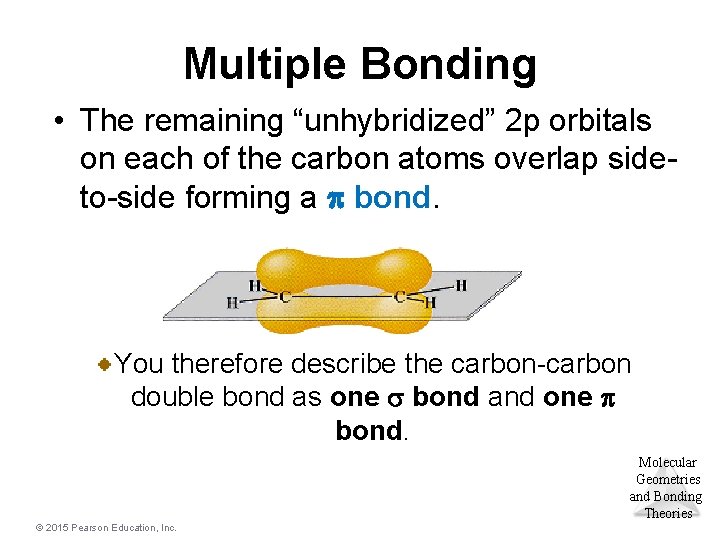
Multiple Bonding • The remaining “unhybridized” 2 p orbitals on each of the carbon atoms overlap sideto-side forming a p bond. You therefore describe the carbon-carbon double bond as one s bond and one p bond. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
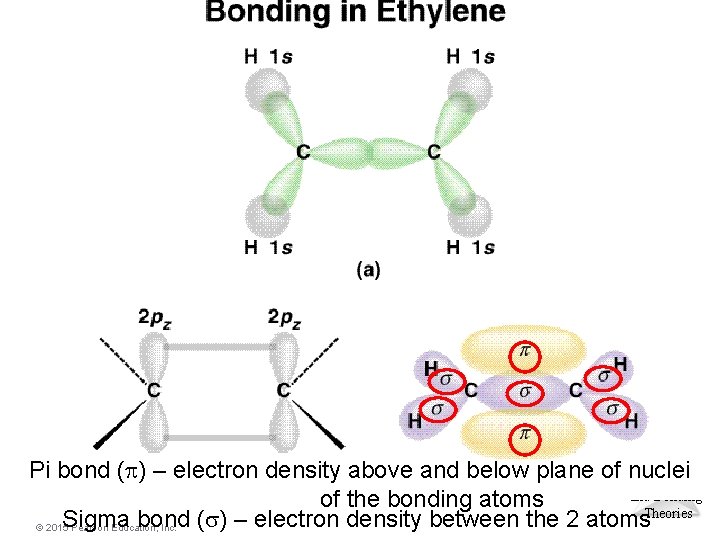
Molecular Pi bond (p) – electron density above and below plane of nuclei Geometries and Bonding of the bonding atoms Sigma bond (s) – electron density between the 2 atoms. Theories © 2015 Pearson Education, Inc.
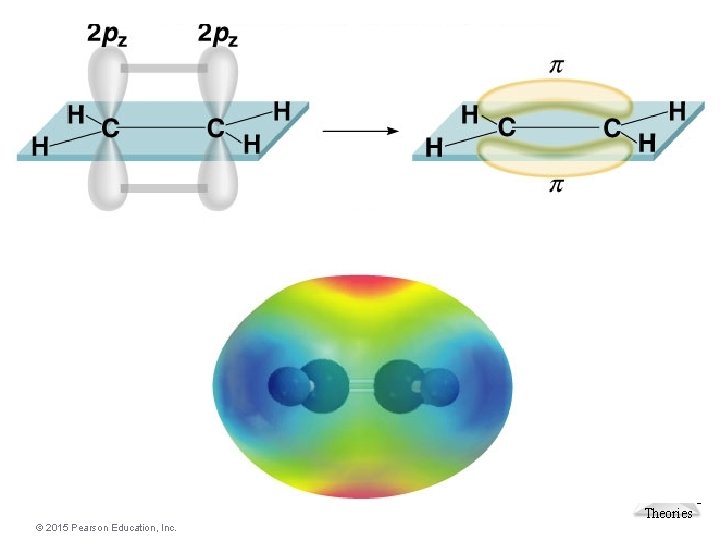
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
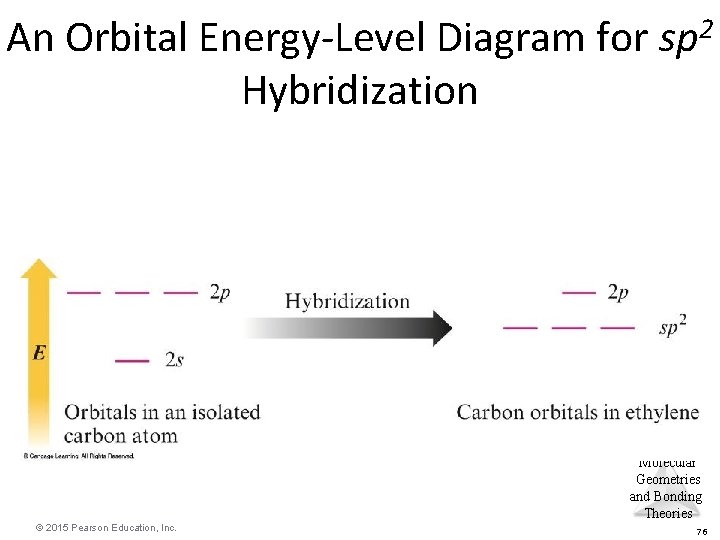
An Orbital Energy-Level Diagram for sp 2 Hybridization Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 76
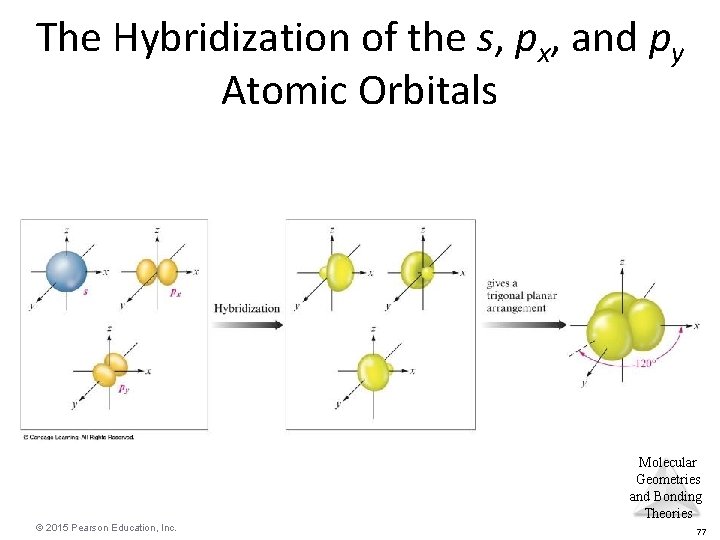
The Hybridization of the s, px, and py Atomic Orbitals Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 77
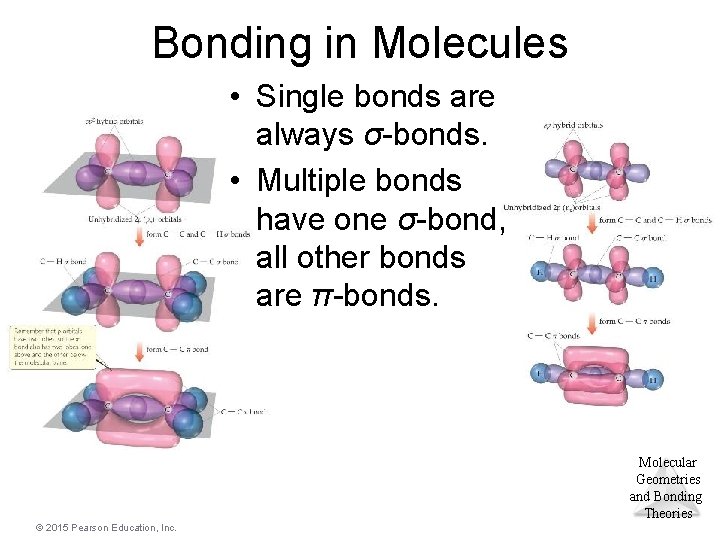
Bonding in Molecules • Single bonds are always σ-bonds. • Multiple bonds have one σ-bond, all other bonds are π-bonds. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

EXERCISE! Draw the Lewis structure for CO 2. – What is the shape of a carbon dioxide molecule? linear – What are the bond angles? 180 o Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 79
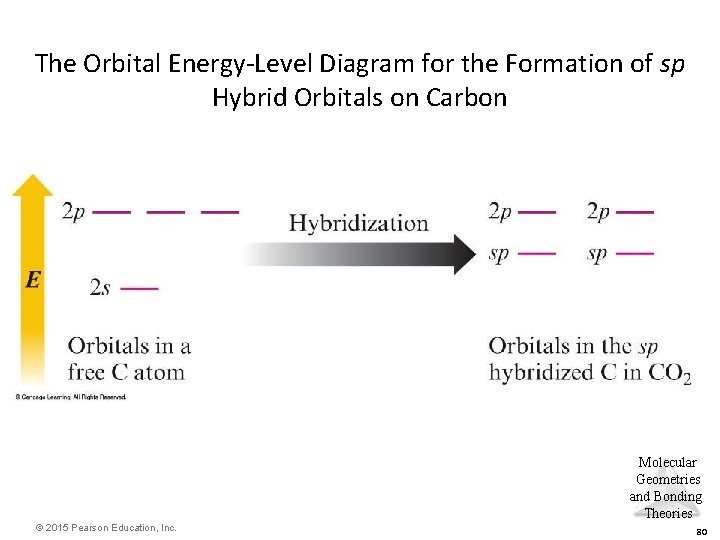
The Orbital Energy-Level Diagram for the Formation of sp Hybrid Orbitals on Carbon Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 80
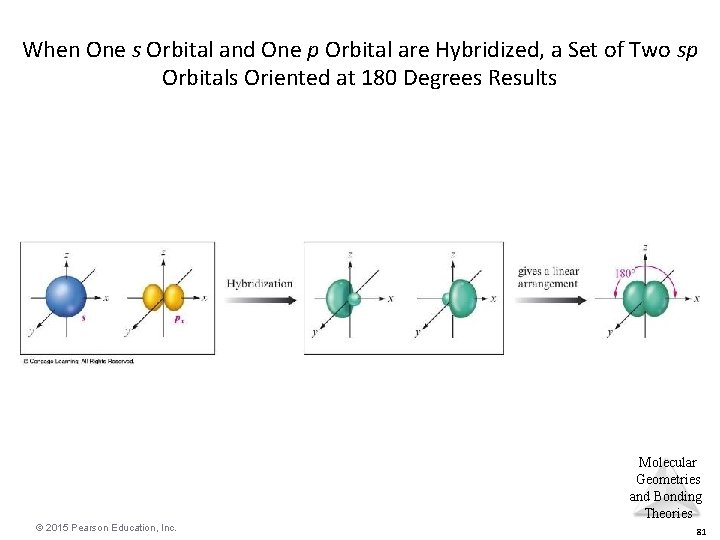
When One s Orbital and One p Orbital are Hybridized, a Set of Two sp Orbitals Oriented at 180 Degrees Results Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 81
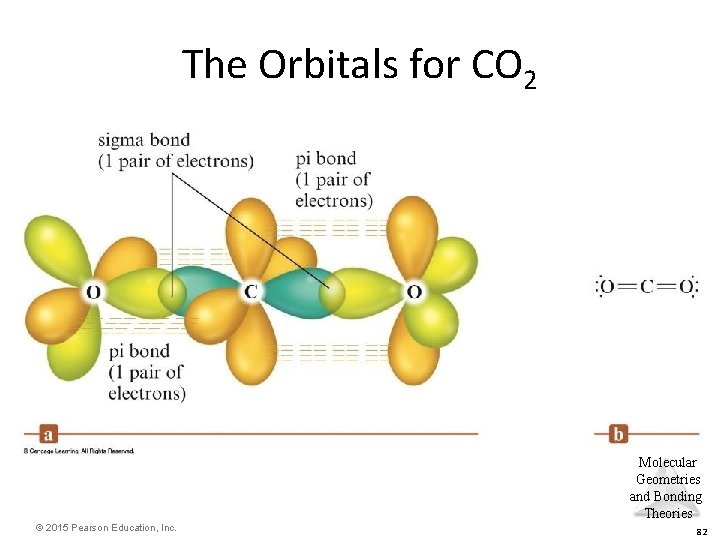
The Orbitals for CO 2 Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc. 82

Localized or Delocalized Electrons • Bonding electrons (σ or π) that are specifically shared between two atoms are called localized electrons. • In many molecules, we can’t describe all electrons that way (resonance); the other electrons (shared by multiple atoms) are called delocalized electrons. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
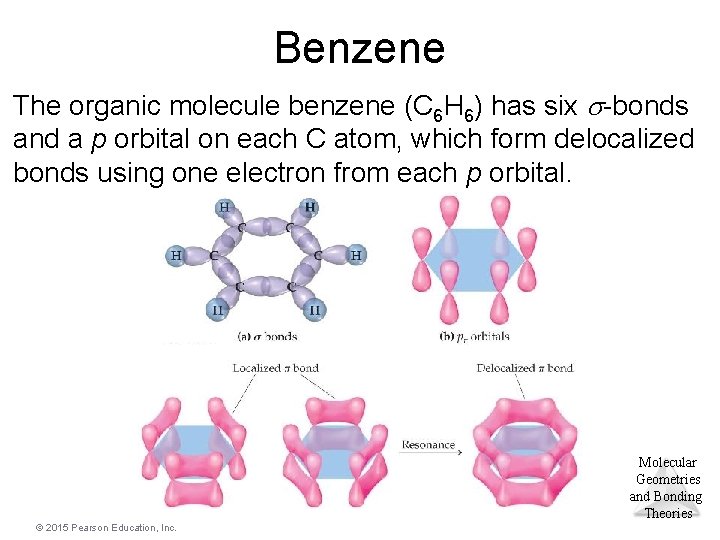
Benzene The organic molecule benzene (C 6 H 6) has six -bonds and a p orbital on each C atom, which form delocalized bonds using one electron from each p orbital. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Molecular Orbital (MO) Theory • Wave properties are used to describe the energy of the electrons in a molecule. • Molecular orbitals have many characteristics like atomic orbitals: – maximum of two electrons per orbital – Electrons in the same orbital have opposite spin. – Definite energy of orbital – Can visualize electron density by a contour diagram Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

More on MO Theory • They differ from atomic orbitals because they represent the entire molecule, not a single atom. • Whenever two atomic orbitals overlap, two molecular orbitals are formed: one bonding, one antibonding. • Bonding orbitals are constructive combinations of atomic orbitals. • Antibonding orbitals are destructive combinations of atomic orbitals. They have a new feature unseen before: A nodal plane occurs where electron density equals zero. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
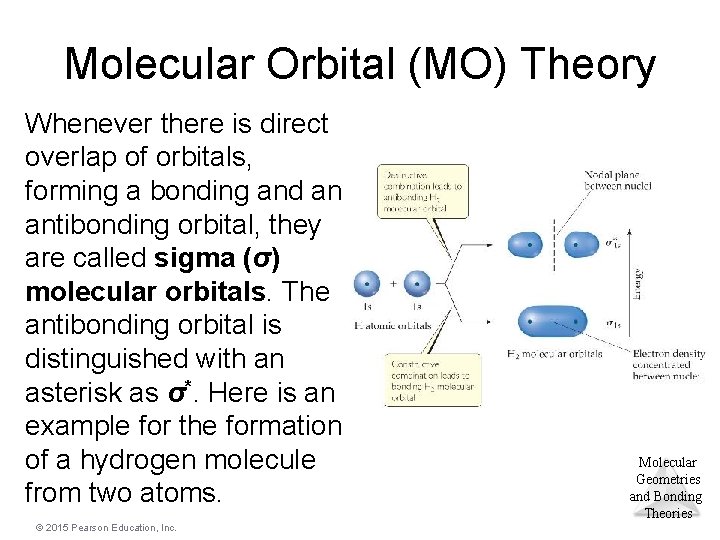
Molecular Orbital (MO) Theory Whenever there is direct overlap of orbitals, forming a bonding and an antibonding orbital, they are called sigma (σ) molecular orbitals. The antibonding orbital is distinguished with an asterisk as σ*. Here is an example for the formation of a hydrogen molecule from two atoms. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
Molecular Orbital Theory • Molecular orbital theory is a theory of the electronic structure of molecules in terms of molecular orbitals, which may spread over several atoms or the entire molecule. As atoms approach each other and their atomic orbitals overlap, molecular orbitals are formed. In the quantum mechanical view, both a Molecular bonding and an antibonding molecular Geometries and Bonding orbital are formed. Theories © 2015 Pearson Education, Inc.

Molecular Orbital Theory • https: //www. khanacademy. org/science/or ganic-chemistry/conjugation-diels-aldermo-theory/molecular-orbital-theory/v/introto-molecular-orbital-mo-theory Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Molecular Orbital Theory • For example, when two hydrogen atoms bond, a s 1 s (bonding) molecular orbital is formed as well as a s 1 s* (antibonding) molecular orbital. The following slide illustrates the relative energies of the molecular orbitals compared to the original atomic orbitals. Because the energy of the two electrons is lower than the energy of the individual atoms, Molecular Geometries the molecule is stable. and Bonding Theories © 2015 Pearson Education, Inc.

H atom H 2 molecule H atom s 1 s* 1 s 1 s © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
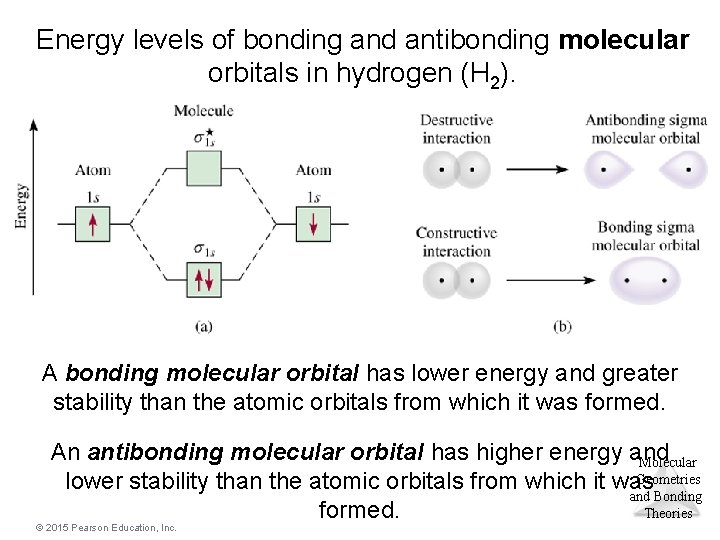
Energy levels of bonding and antibonding molecular orbitals in hydrogen (H 2). A bonding molecular orbital has lower energy and greater stability than the atomic orbitals from which it was formed. An antibonding molecular orbital has higher energy and Molecular Geometries lower stability than the atomic orbitals from which it was and Bonding Theories formed. © 2015 Pearson Education, Inc.
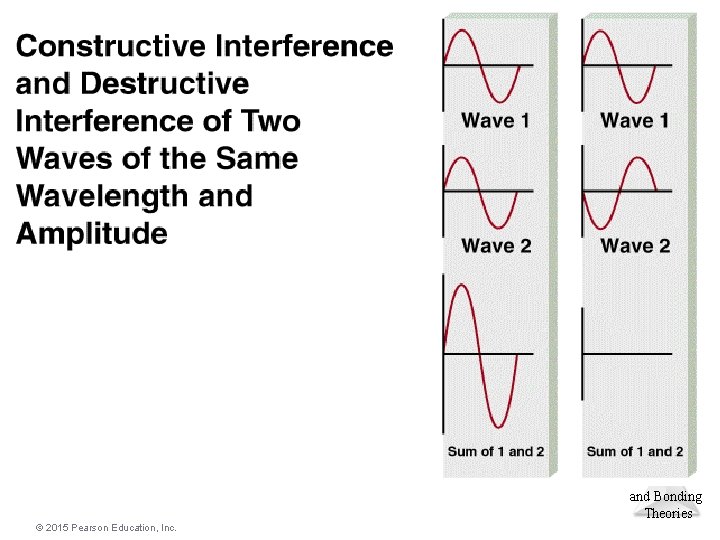
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
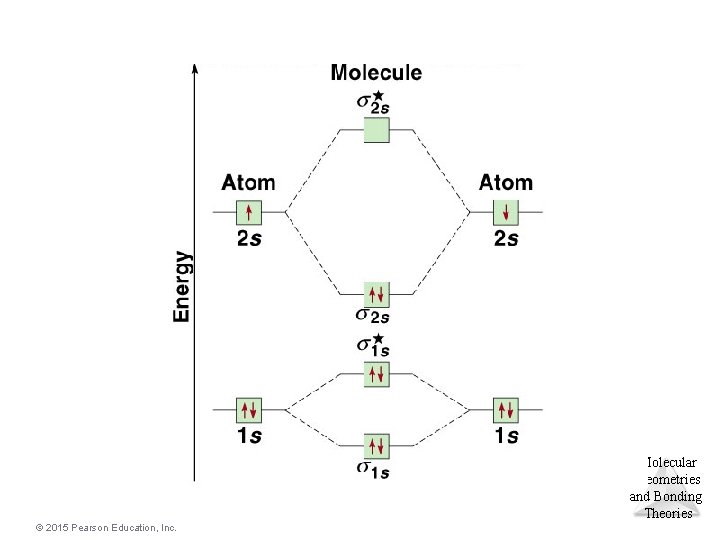
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
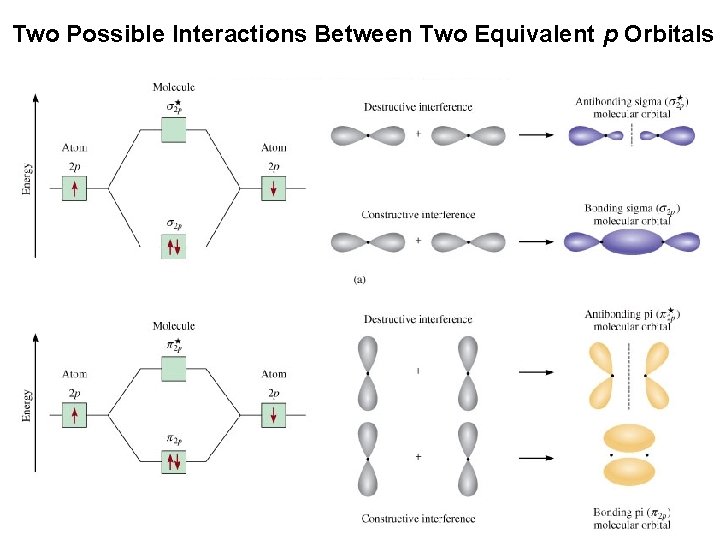
Two Possible Interactions Between Two Equivalent p Orbitals Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
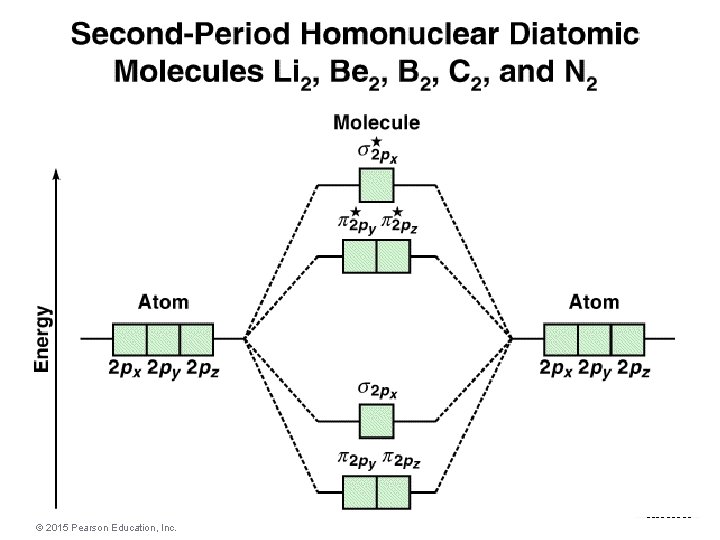
Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
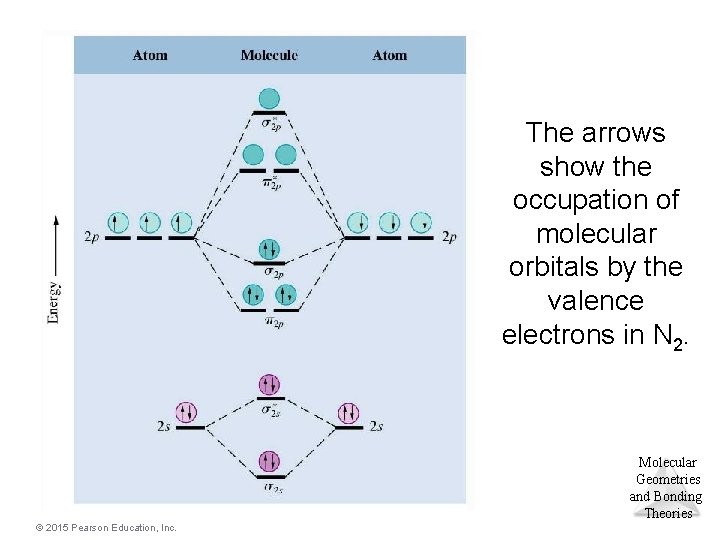
The arrows show the occupation of molecular orbitals by the valence electrons in N 2. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Molecular Orbital (MO) Configurations 1. The number of molecular orbitals (MOs) formed is always equal to the number of atomic orbitals combined. 2. The more stable the bonding MO, the less stable the corresponding antibonding MO. 3. The filling of MOs proceeds from low to high energies. 4. Each MO can accommodate up to two electrons. 5. Use Hund’s rule when adding electrons to MOs of the same energy. 6. The number of electrons in the MOs is equal to the sum of all the electrons on the bonding atoms. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
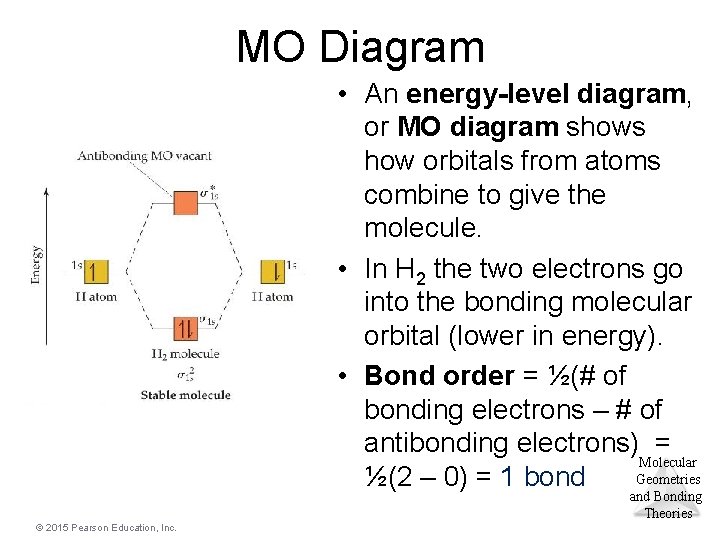
MO Diagram • An energy-level diagram, or MO diagram shows how orbitals from atoms combine to give the molecule. • In H 2 the two electrons go into the bonding molecular orbital (lower in energy). • Bond order = ½(# of bonding electrons – # of antibonding electrons) = Molecular Geometries ½(2 – 0) = 1 bond and Bonding Theories © 2015 Pearson Education, Inc.
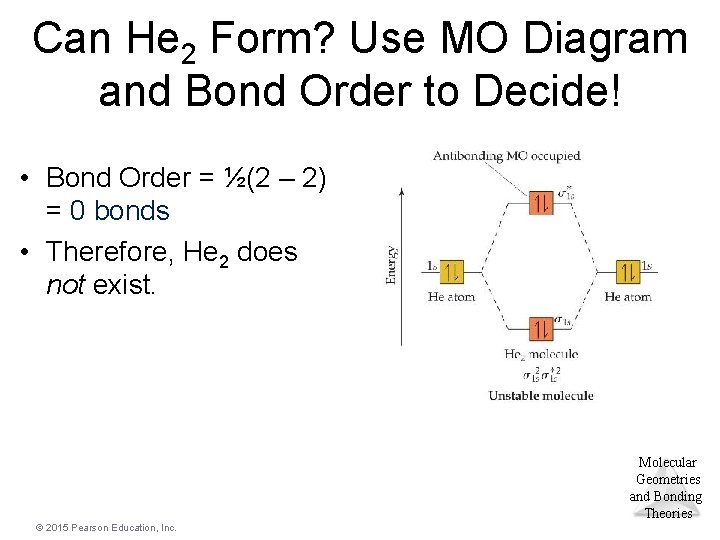
Can He 2 Form? Use MO Diagram and Bond Order to Decide! • Bond Order = ½(2 – 2) = 0 bonds • Therefore, He 2 does not exist. Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
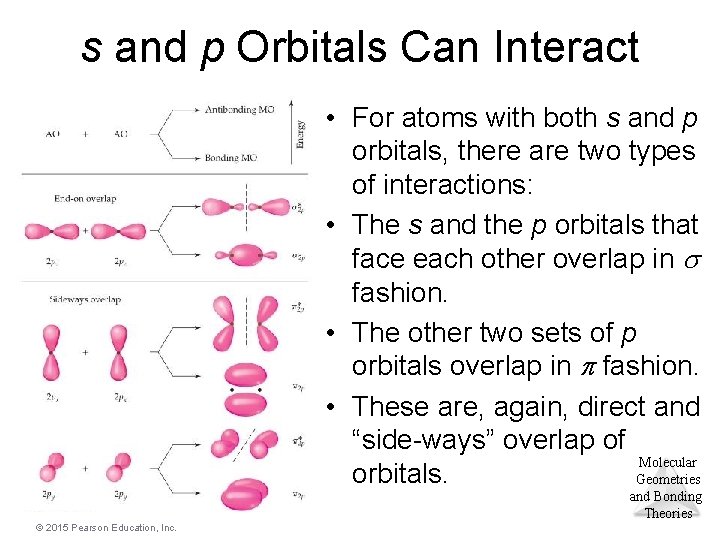
s and p Orbitals Can Interact • For atoms with both s and p orbitals, there are two types of interactions: • The s and the p orbitals that face each other overlap in fashion. • The other two sets of p orbitals overlap in fashion. • These are, again, direct and “side-ways” overlap of Molecular orbitals. Geometries and Bonding Theories © 2015 Pearson Education, Inc.
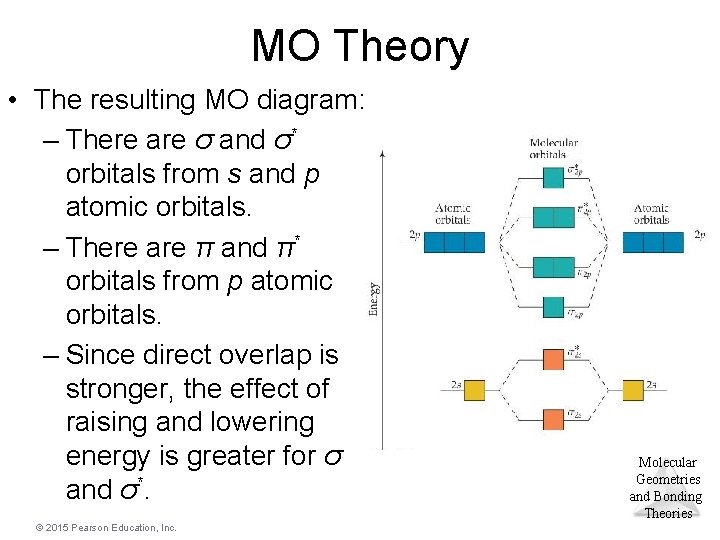
MO Theory • The resulting MO diagram: – There are σ and σ* orbitals from s and p atomic orbitals. – There are π and π* orbitals from p atomic orbitals. – Since direct overlap is stronger, the effect of raising and lowering energy is greater for σ and σ*. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
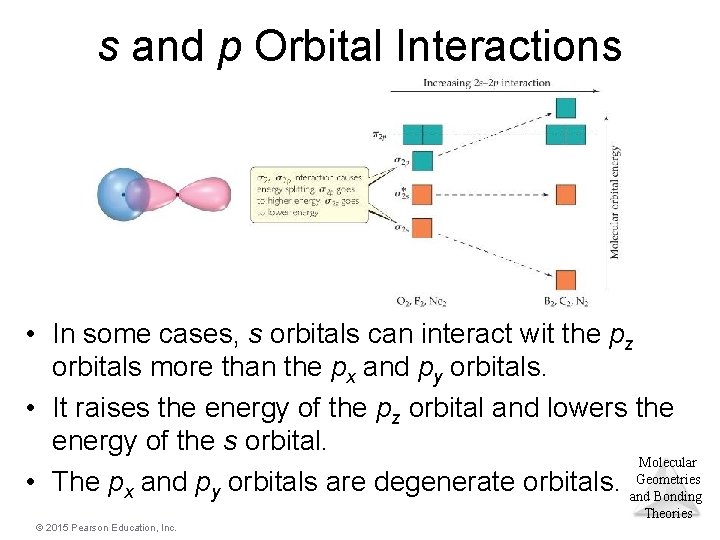
s and p Orbital Interactions • In some cases, s orbitals can interact wit the pz orbitals more than the px and py orbitals. • It raises the energy of the pz orbital and lowers the energy of the s orbital. Molecular Geometries • The px and py orbitals are degenerate orbitals. and Bonding Theories © 2015 Pearson Education, Inc.
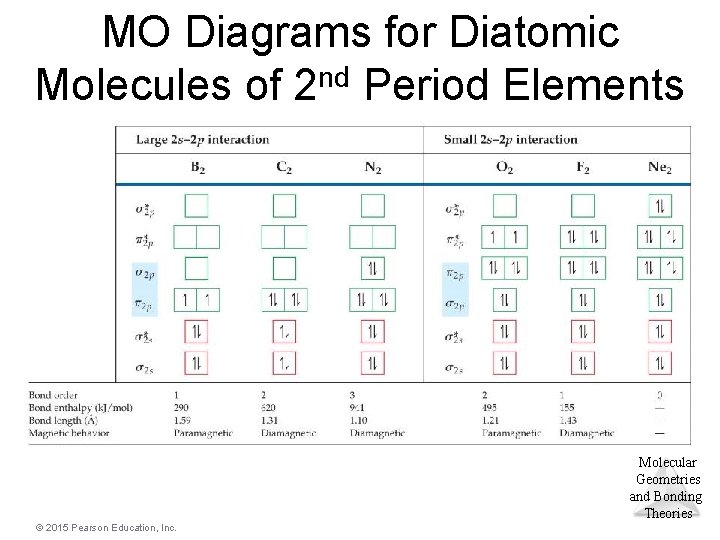
MO Diagrams for Diatomic Molecules of 2 nd Period Elements Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.

MO Diagrams and Magnetism • Diamagnetism is the result of all electrons in every orbital being spin paired. These substances are weakly repelled by a magnetic field. • Paramagnetism is the result of the presence of one or more unpaired electrons in an orbital. • Is oxygen (O 2) paramagnetic or diamagnetic? Look back at the MO Molecular diagram! It is paramagnetic. Geometries and Bonding Theories © 2015 Pearson Education, Inc.

Paramagnetism of Oxygen • Lewis structures would not predict that O 2 is paramagnetic. • The MO diagram clearly shows that O 2 is paramagnetic. • Both show a double bond (bond order = 2). Molecular Geometries and Bonding Theories © 2015 Pearson Education, Inc.
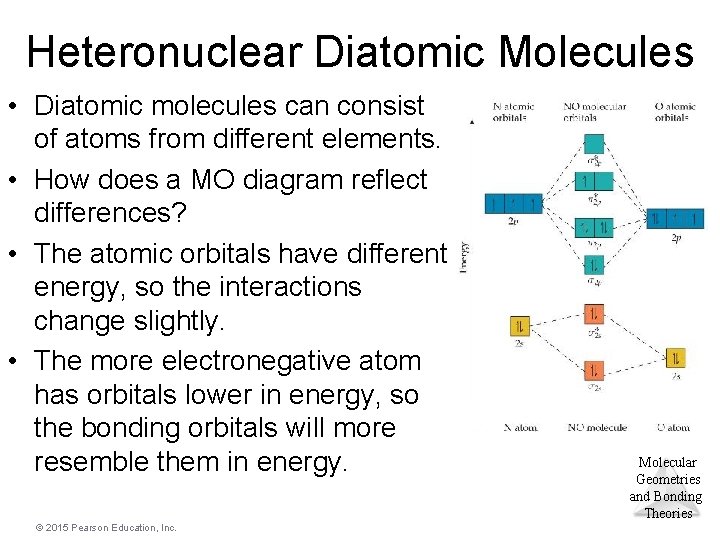
Heteronuclear Diatomic Molecules • Diatomic molecules can consist of atoms from different elements. • How does a MO diagram reflect differences? • The atomic orbitals have different energy, so the interactions change slightly. • The more electronegative atom has orbitals lower in energy, so the bonding orbitals will more resemble them in energy. © 2015 Pearson Education, Inc. Molecular Geometries and Bonding Theories
- Slides: 107