Lecture Objectives Finish with Electric Energy Generation Learn

Lecture Objectives: • Finish with Electric Energy Generation • Learn about Sorption Cooling
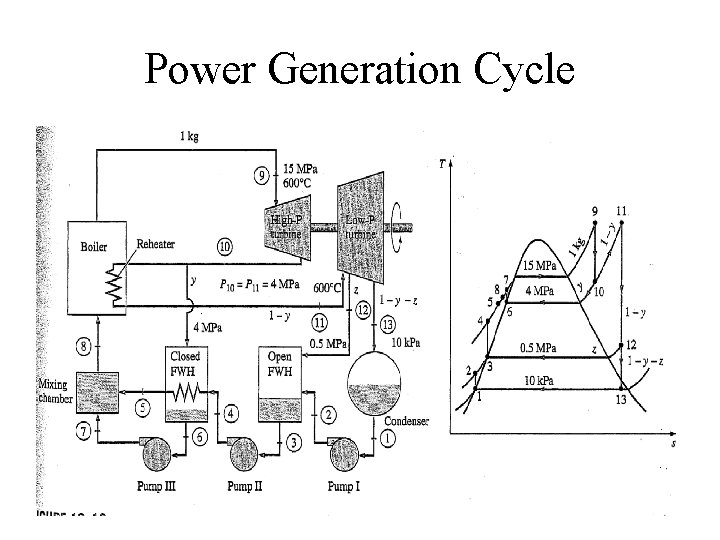
Power Generation Cycle
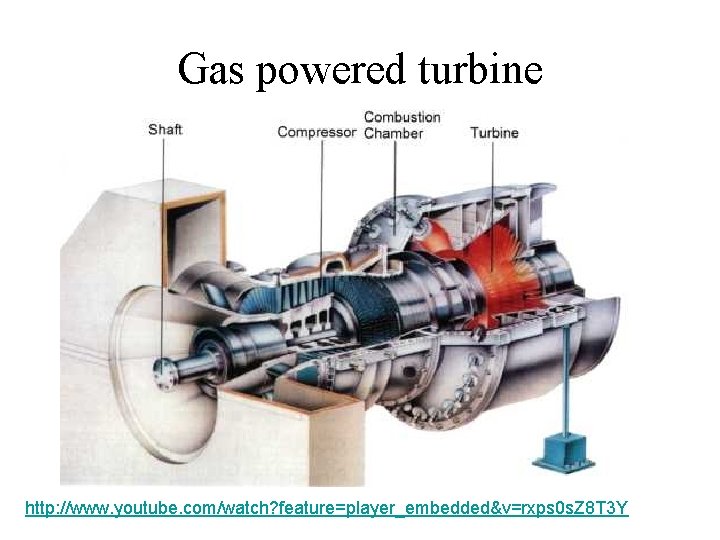
Gas powered turbine http: //www. youtube. com/watch? feature=player_embedded&v=rxps 0 s. Z 8 T 3 Y

Combustion product gas powered turbines • Limited to gas or oil as a major source of fuel • Approximately 55 to 65% of the power produced by the turbine is used for compressor. • Gas temperatures at the turbine inlet can be 1200ºC to 1400ºC • Because of the power required to drive the compressor, energy conversion efficiency for a simple cycle gas turbine plant is ~ 30%
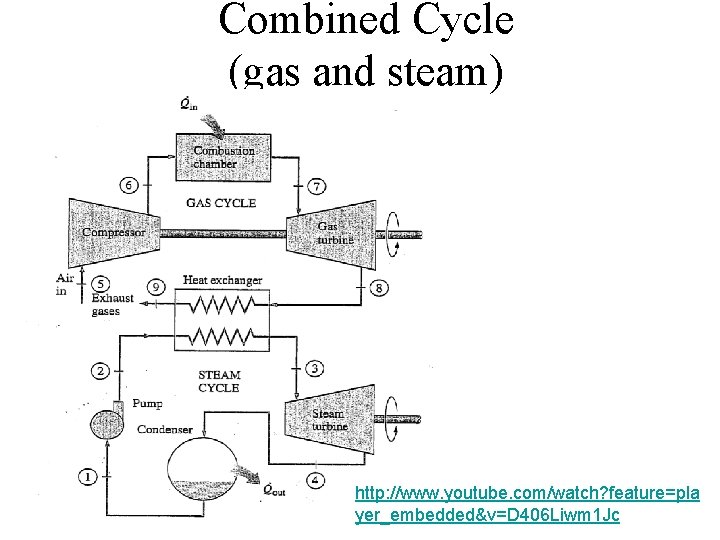
Combined Cycle (gas and steam) http: //www. youtube. com/watch? feature=pla yer_embedded&v=D 406 Liwm 1 Jc
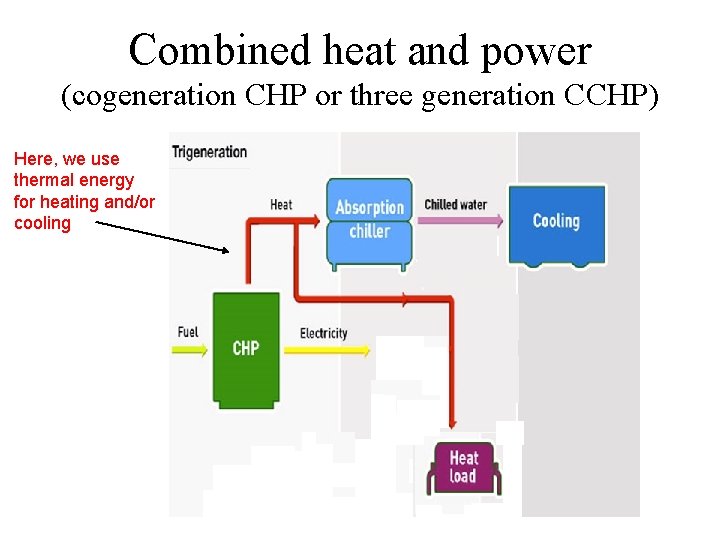
Combined heat and power (cogeneration CHP or three generation CCHP) Here, we use thermal energy for heating and/or cooling
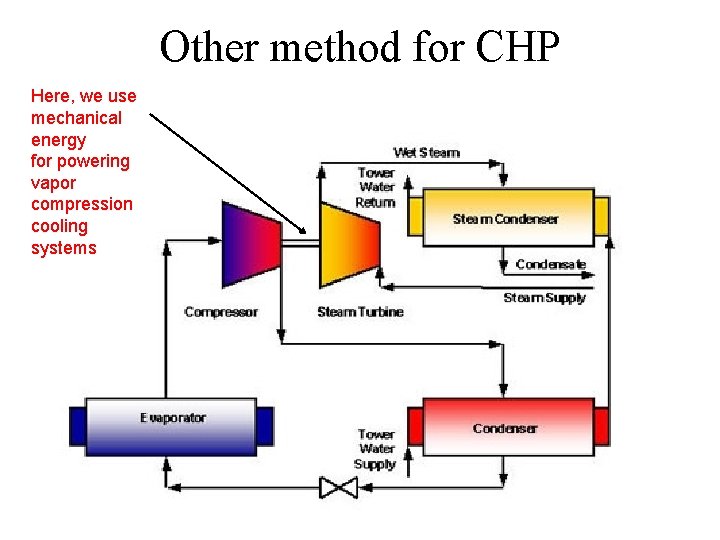
Other method for CHP Here, we use mechanical energy for powering vapor compression cooling systems
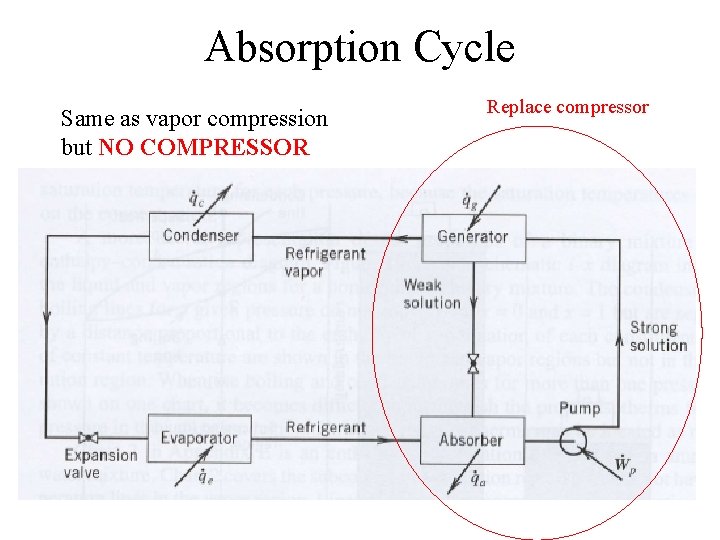
Absorption Cycle Same as vapor compression but NO COMPRESSOR Replace compressor
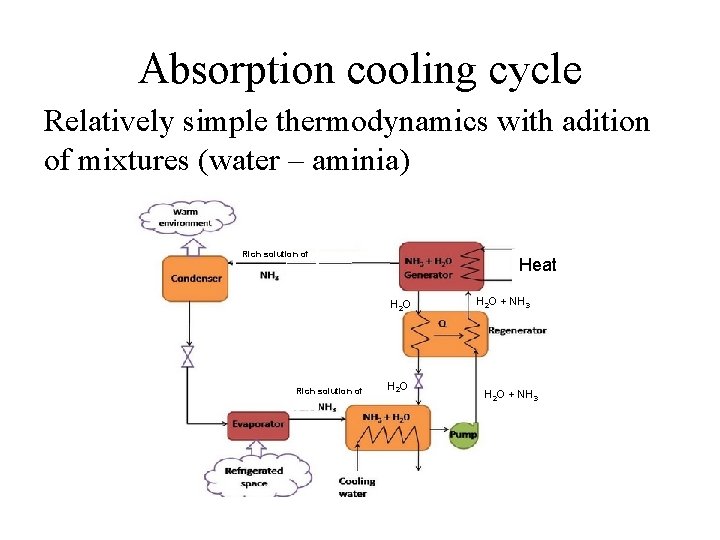
Absorption cooling cycle Relatively simple thermodynamics with adition of mixtures (water – aminia) Rich solution of Heat H 2 O Rich solution of H 2 O + NH 3
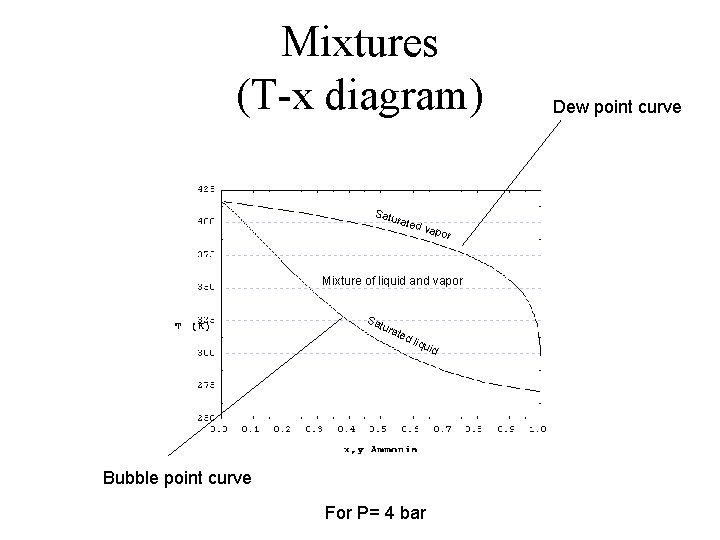
Mixtures (T-x diagram) Satu rate d va por Mixture of liquid and vapor Sa tura ted liqu id Bubble point curve For P= 4 bar Dew point curve
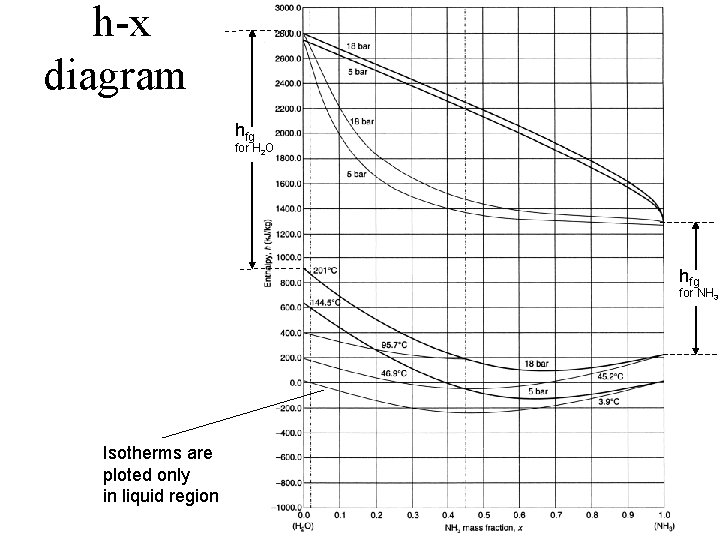
h-x diagram hfg for H 2 O hfg for NH 3 Isotherms are ploted only in liquid region
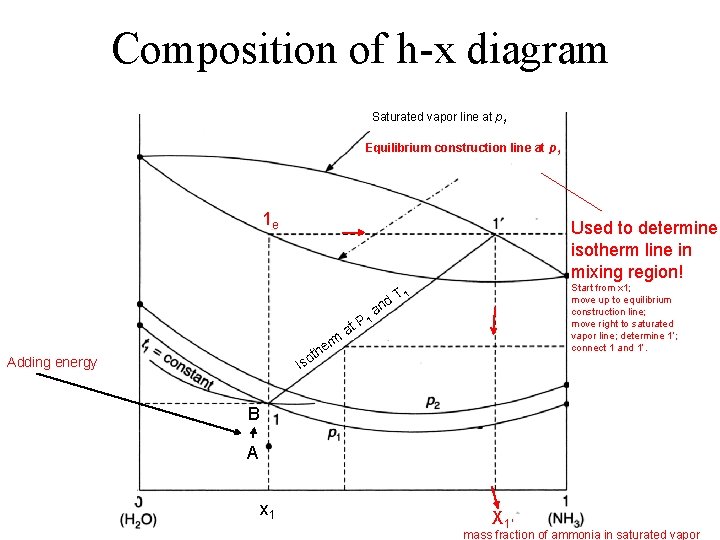
Composition of h-x diagram Saturated vapor line at p 1 Equilibrium construction line at p 1 1 e Used to determine isotherm line in mixing region! Start from x 1; move up to equilibrium construction line; move right to saturated vapor line; determine 1’; connect 1 and 1’. 1 d. T rm at P 1 an e oth Adding energy Is B A x 1 X 1’ mass fraction of ammonia in saturated vapor
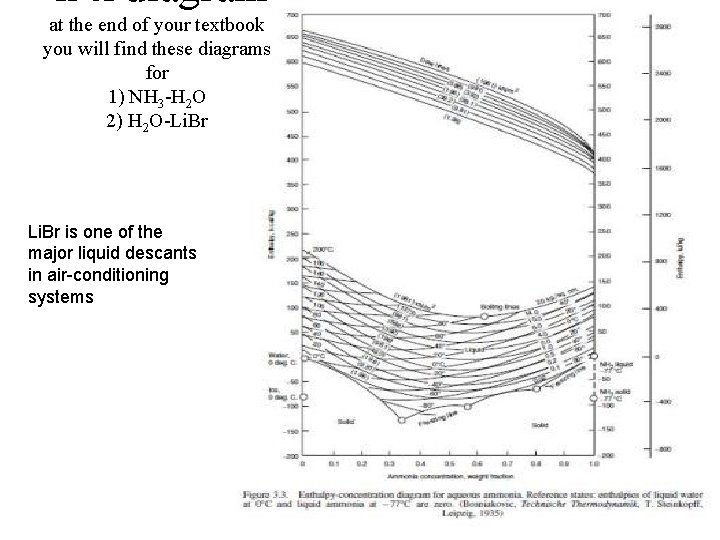
h-x diagram at the end of your textbook you will find these diagrams for 1) NH 3 -H 2 O 2) H 2 O-Li. Br is one of the major liquid descants in air-conditioning systems

Adiabatic mixing in h-x diagram (Water – Ammonia) From the textbook (Thermal Environmental Eng. ; Kuehen et al)
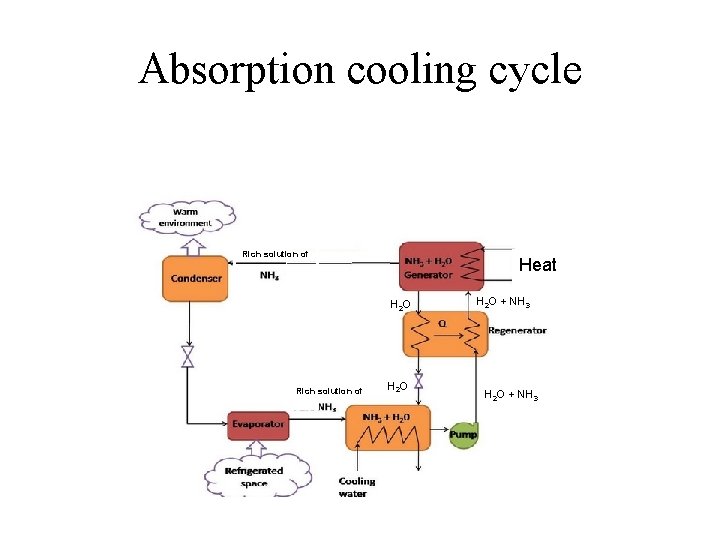
Absorption cooling cycle Rich solution of Heat H 2 O Rich solution of H 2 O + NH 3
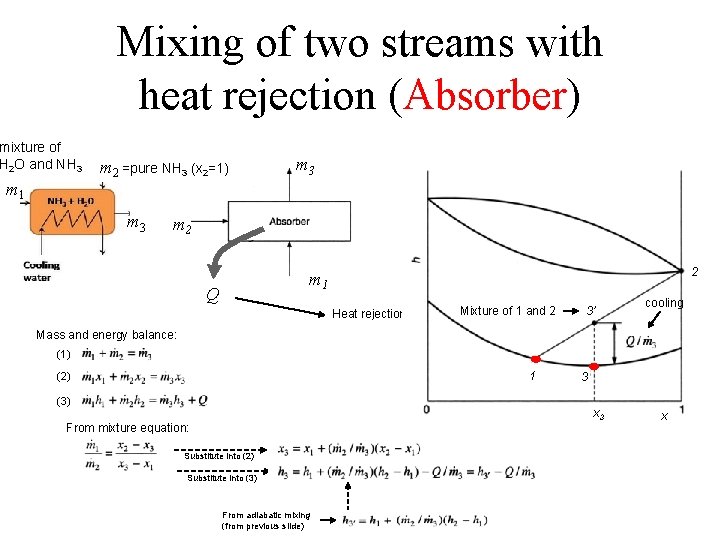
Mixing of two streams with heat rejection (Absorber) mixture of H 2 O and NH 3 m 1 m 2 =pure NH 3 (x 2=1) m 3 m 2 2 m 1 Q Heat rejection Mixture of 1 and 2 3’ cooling Mass and energy balance: (1) 1 (2) (3) 3 x 3 From mixture equation: Substitute into (2) Substitute into (3) From adiabatic mixing (from previous slide) x

- Slides: 17