Lecture No 1 Carbohydrates Chemistry Dr Obeid Shanab

Lecture No. 1 Carbohydrates Chemistry Dr. Obeid Shanab. Assistant professor of Biochemistry, Clinical Biochemistry and Cancer Biology

Syllabus Ø Ø • • Introduction Classification of CHO Monosaccharides Disaccharides Trisaccharides Polysaccharides

Carbohydrates Ø The old definition: they are substances containing carbon, hydrogen and oxygen with the general formula Cn. H 2 n. On.

Ø This old definition is inaccurate because: • There are substances other than carbohydrates but have the formula Cn. H 2 n. On, e. g. , acetic acid CH 3 COOH. • There are some carbohydrates, but do not obey this formula, e. g. , rhamnose, amino sugars and deoxy sugars. Ø The new definition: • Carbohydrates are aldehyde (CHO) or ketone (C=O) derivatives of polyhydric alcohols (have more than one OH group) or compounds which derived from them.

v Importance of carbohydrates: Ø In plants: it is produced by photosynthesis and include: • Cellulose: the framework of plants. • Starch: the stored form of carbohydrates in plants. • Pectin: the main source of dietary fiber.

Ø In animals: CHO have diverse functions e. g. : • Glucose and glycogen: are sources of energy. • Ribose: enters in the structure of nucleic acids. • Galactose: enters in the structure of lactose of milk. • Glycoproteins and glycolipids: are combinations of CHO with proteins or lipids that enter in the structure of cell membrane and form the ground substances between tissues.

Ø Classification of carbohydrates v According to the number of sugar units: I. Monosaccharides: contain one sugar unit. II. Disaccharides: contain two sugar units. III. Oligosaccharides: contain 3 -10 sugar units. IV. Polysaccharides: contain more than 10 sugar units.

I. Monosaccharides q Types monosaccharides: v According to the presence of CHO or C=O group: 1) Aldoses: contain aldehyde group (-CHO). 2) Ketoses: contain ketone group (-C=O).
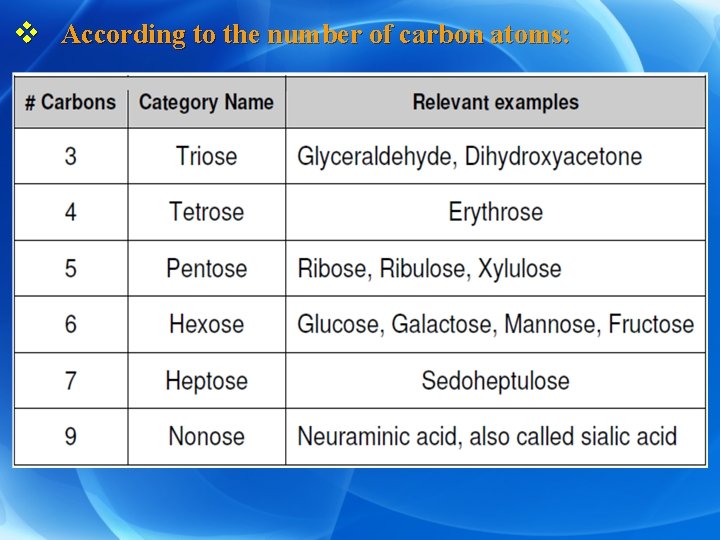
v According to the number of carbon atoms:

v According to both presence of aldehyde or ketone groups and number of carbon atoms: 1) Aldotrioses and ketotrioses. 2) Aldotetroses and ketotetroses. 3) Aldopentoses and ketopentoses. 4) Aldohexoses and ketohexoses.

Ø Examples: 1) Trioses: monosaccharides containing 3 carbon atoms including: a. Aldotrioses e. g. : b. Ketotriose: e. g. :
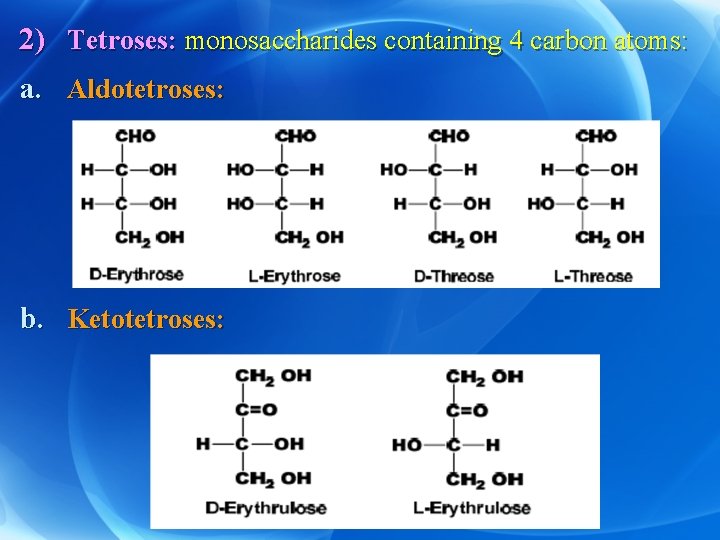
2) Tetroses: monosaccharides containing 4 carbon atoms: a. Aldotetroses: b. Ketotetroses:

3) Pentoses: monosaccharides containing 5 carbon atoms. 1. Aldopentoses: 2. Ketopentoses:

Ø Functions of pentoses : • Ribose enters in the structure of RNA, NADP, flavoproteins, ATP and other high energy phosphate compounds. • Deoxyribose enters in the structure of DNA. • Ribose phosphate and ribulose phosphate are intermediates in HMP shunt (a minor pathway of glucose oxidation).

4) Hexoses: monosaccharides containing 6 carbon atoms. a) Aldohexoses: i. Glucose: is called grape sugar, blood sugar or Dextrose because it is dextrorotatory. Dietary carbohydrates are absorbed in the form of glucose.

ii. Mannose: is a subunit in glycoproteins and sialic acid. iii. Galactose: is a subunit of the milk sugar, lactose. It enters in structure of glycoproteins, glycolipids and mucopolysaccharides. Its pathological accumulation in blood and appearance in urine is called galactosemia.
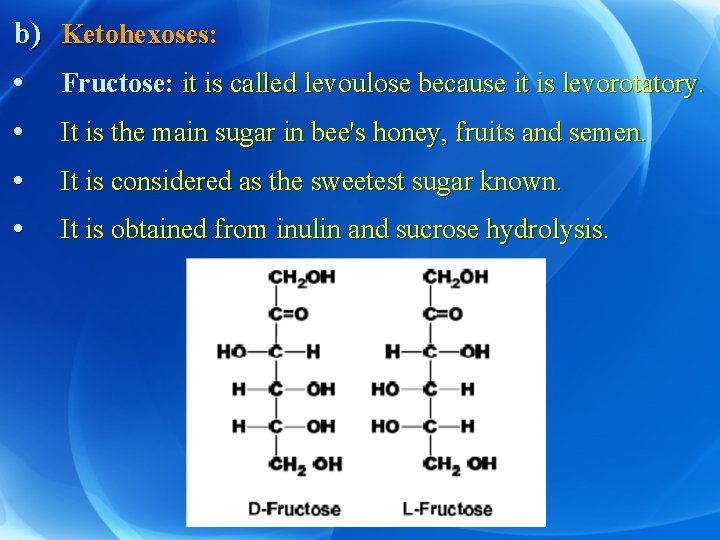
b) Ketohexoses: • Fructose: it is called levoulose because it is levorotatory. • It is the main sugar in bee's honey, fruits and semen. • It is considered as the sweetest sugar known. • It is obtained from inulin and sucrose hydrolysis.

Fructose is the semen sugar can be used as a fertelity index

v Asymmetric carbon atom and optical activity • Asymmetric carbon atom is attached to 4 different groups or atoms, e. g. , the central carbon in glyceraldehyde. • Any compound containing asymmetric carbon atom has the following two properties: 1) Isomerism which is created around the asymmetric carbon atom. 2) Optically activity.

Optical activity Ø Definition: It is the ability of a substance to rotate the plane of the plane polarized light. Ø While the plane polarized light vibrates in one plane and one direction, ordinary light vibrates in all directions. Ø Ordinary light can be changed to plane polarized light by passing it through a prism.
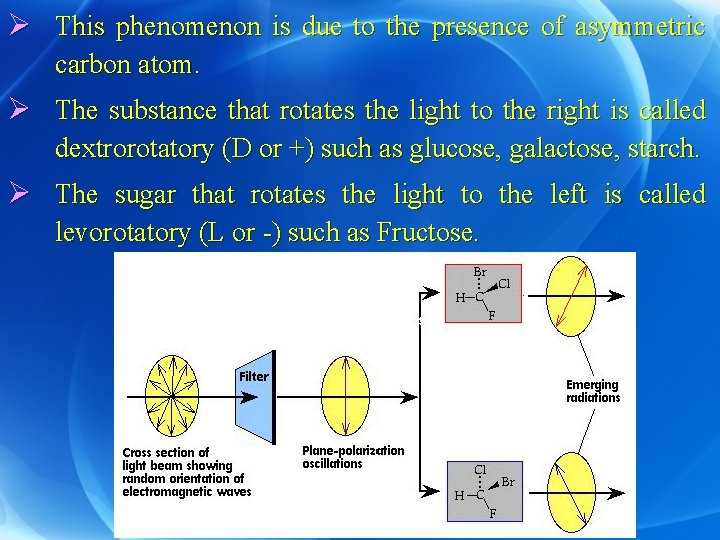
Ø This phenomenon is due to the presence of asymmetric carbon atom. Ø The substance that rotates the light to the right is called dextrorotatory (D or +) such as glucose, galactose, starch. Ø The sugar that rotates the light to the left is called levorotatory (L or -) such as Fructose.
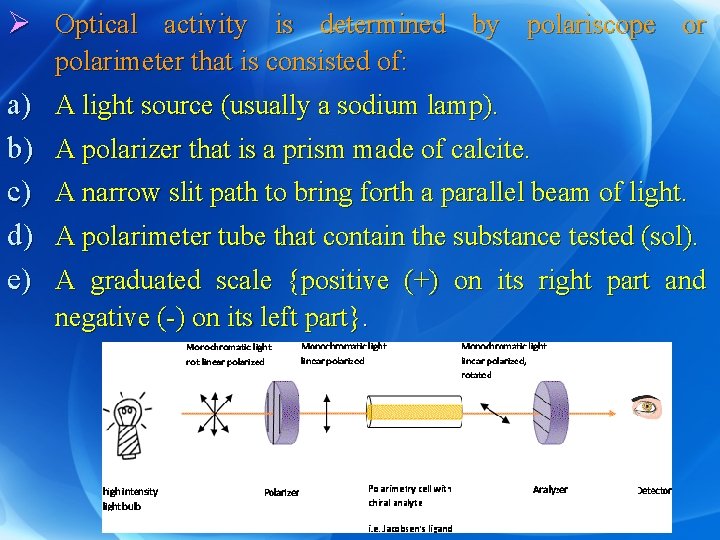
Ø Optical activity is determined by polariscope or polarimeter that is consisted of: a) b) c) d) e) A light source (usually a sodium lamp). A polarizer that is a prism made of calcite. A narrow slit path to bring forth a parallel beam of light. A polarimeter tube that contain the substance tested (sol). A graduated scale {positive (+) on its right part and negative (-) on its left part}.

Ø The angle of rotation obtained by optically active substance depends on: 1) Type and concentration of the substance. 2) The type of solvent used. 3) Type of light used. 4) Temperature. 5) Length of polarimeter tube in decimeters.

Ø Specific rotation: • It is the observed angle of deviation of the plane polarized light in degrees from the straight path. • It is measured when an aqueous solution of the substance is introduced in the path of the plane polarized light under the following conditions: • The light source used is sodium light, the temperature is 20 o. C, the concentration is 1 gm/ml and the polarimeter tube is one decimeter in length.

• Specific rotation is calculated by the following equation: • a = angle observed in degrees. • C = Concentration in gm/ml. • L = length of polarimeter tube in decimeter. Ø Each optically active substance has a characteristic specific rotation such as α-glucose (+112), β-glucose (+19) and fructose (- 92. 5).

Ø Importance of optical activity: 1) Determination whether a substance is optically active or not. 2) Identification of a substance by determining its specific rotation. 3) Determination of the concentration of a substance. 4) Differentiate between glucosuria and lactosuria. • This is important in late pregnancy to differentiate between diabetes mellitus (glucose) and the normal appearance of lactose produced by the mammary glands in urine.

Mutarotation • It is a temporary change in the specific rotation of the sugar when it is freshly prepared. • Mutarotation is due to the presence of a free anomeric carbon (C 1 in aldoses, C 2 in ketoses). • With time, it tends to stabilize at the specific rotation of the compound. • Therefore, it is recommended not to measure specific rotation of the solution directly after its preparation.

Ø For example: • α-glucose when freshly prepared has a specific rotation of +112, then the specific rotation decreases gradually till it stabilizes at +52. 5 (point of equilibrium). • β-glucose when freshly prepared has a specific rotation of +19, then the specific rotation gradually increases till it is stabilizes at +52. 5 (point of equilibrium).
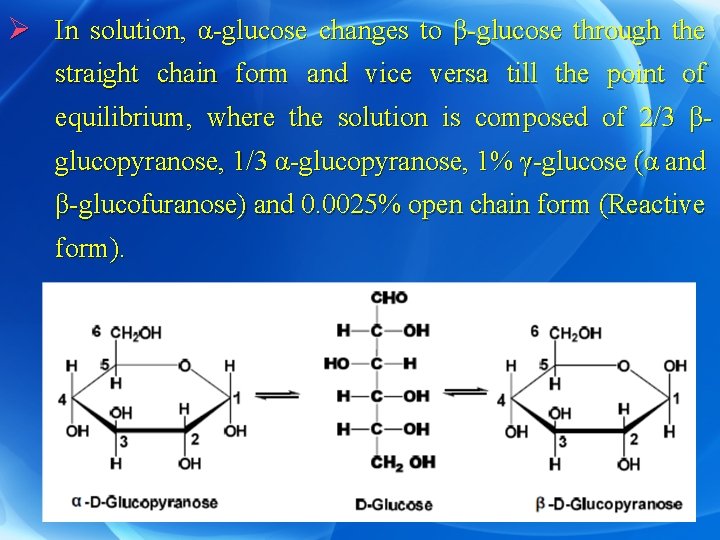
Ø In solution, α-glucose changes to β-glucose through the straight chain form and vice versa till the point of equilibrium, where the solution is composed of 2/3 βglucopyranose, 1/3 α-glucopyranose, 1% γ-glucose (α and β-glucofuranose) and 0. 0025% open chain form (Reactive form).

Ø Cyclic structure of monosaccharide • The simple chain formula (Fisher's projection formula) fails to explain some reactions of sugars e. g. , glucose which has aldhydic group does not give all reactions of aldhyde. • This indicates that the –CHO group is masked or combined in some way.

• In solution, the sugar which has an aldhyde or keto group undergoes the following: • Condensation of a molecule of H 2 O with the aldehyde or keto group of the sugar to form aldenol or ketonol group. • The OH group from the aldenol group condenses with the OH on C 4 (Furanose) or C 5 (Pyranose) of the aldosugar to forms a ring or hemi acetal structure with the liberation of H 2 O again.


• Keto-sugar condenses only with C 5 (Furanose) or with C 6 (Pyranose).

Ø When the remaining OH on the aldehyde or the keto carbon atom in the cyclic form is located on the right side, the sugar form is called α-sugar and if it is located on the left side the sugar is called β-sugar. Ø Aldoses are more stable in pyranose form, whereas, Ketoses are more stable in furanose form.

Ø The OH group formed on the aldehyde or keto group due to cyclization of the sugar molecule makes C 1 in aldoses or C 2 in ketoses an asymmetric carbon atom. Ø Therefore, the two α and β forms of the sugar are isomers and called anomers because that carbon atom is called the anomeric carbon atom (i. e. , C 1 in aldoses and C 2 in ketoses).

Ø Haworth's projection formula: • Because Fisher’s formula could not explain some of the chemical and physical characteristics of sugars, Haworth put his projection formula. • C and O atoms of the ring are drawn in the plane of the page. • H and OH or other side groups are written on perpendicular plane. • All groups located on the left side of Fisher’s are written upwards. • All groups located on the right side of Fisher’s are written downwards.

• After C 4 participating in the ring, this role is oppositely applied, i. e. , right groups are written upwards and left groups are written downwards. • The radical of the molecule (the extra-cyclic part) is written upwards in D sugar and inside the ring (or down wards) in L-sugar. • In general, the OH on the α-anomer points down (ants down) while on the β-anomer it points up (butterflies up).



Isomerism Ø Isomers are substances which have the same molecular formula but differ in distribution of their atoms in the space around carbon atoms. Ø There are 2 types of isomerism; I. Structural isomerism. II. Stereo-isomerism.
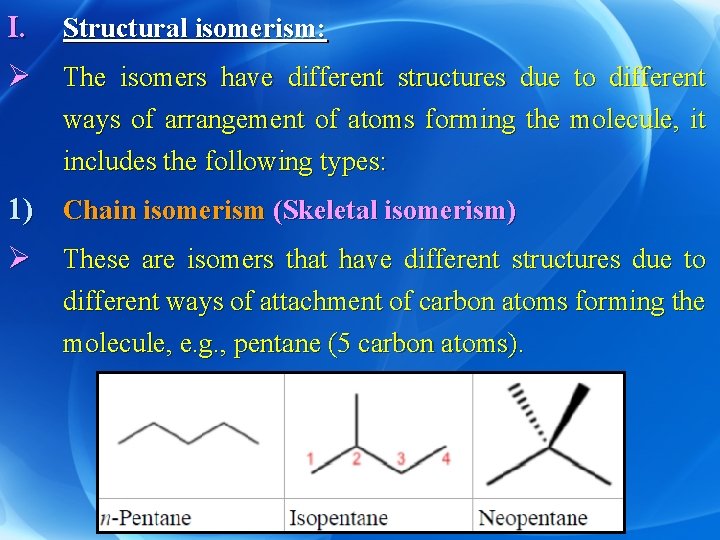
I. Structural isomerism: Ø The isomers have different structures due to different ways of arrangement of atoms forming the molecule, it includes the following types: 1) Chain isomerism (Skeletal isomerism) Ø These are isomers that have different structures due to different ways of attachment of carbon atoms forming the molecule, e. g. , pentane (5 carbon atoms).
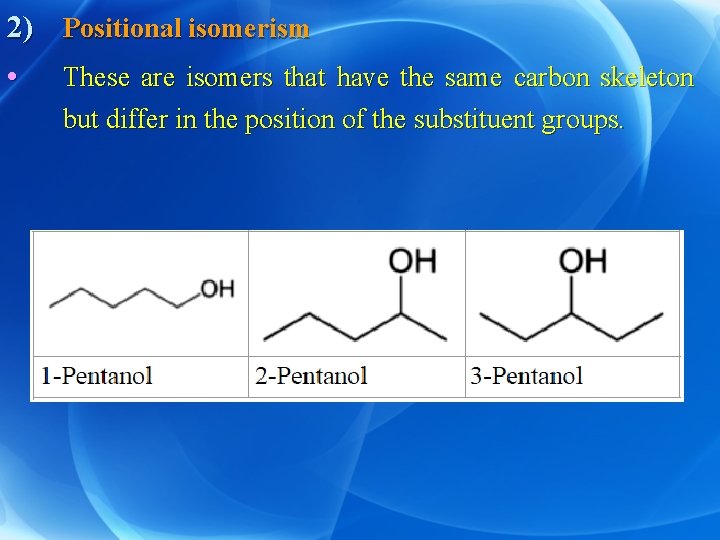
2) Positional isomerism • These are isomers that have the same carbon skeleton but differ in the position of the substituent groups.
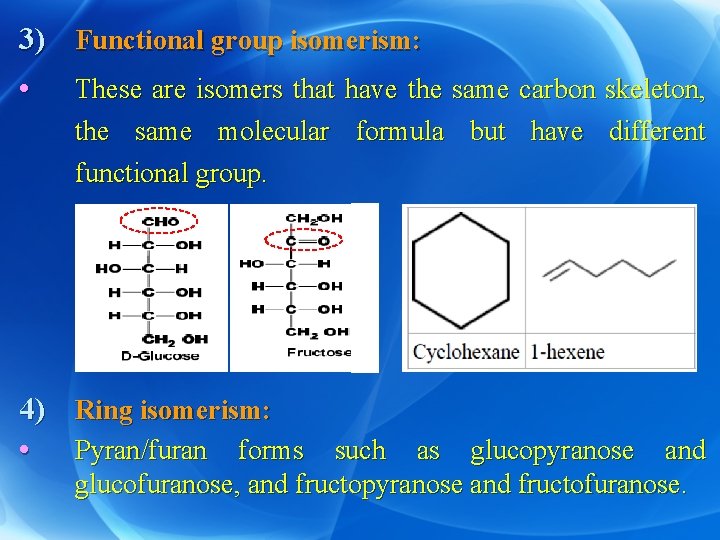
3) Functional group isomerism: • These are isomers that have the same carbon skeleton, the same molecular formula but have different functional group. 4) • Ring isomerism: Pyran/furan forms such as glucopyranose and glucofuranose, and fructopyranose and fructofuranose.

II. Stereo-isomerism: • They are molecules having the same structure but differ in position of their different groups and atoms in the space, i. e. , in spatial configuration. • The number of stereoisomers = 2 n, where n is the number of asymmetric carbon atoms. • There are four types of stereo-isomerism as follows:

For example Glocose has 4 assymetric carbon atomes So N= 16 isomers

1) D and L isomerism (enantiomers): • They differ in distribution of H and OH groups around all assymetric carbon so they are mirror image. • They are called optical antipodes, if one is D the second is L (have the same optical activity but to the other side). • A mixture containing equal amounts of the two isomers is called racemic mixture which is optically inactive because each isomer equalizes the effect of the other e. g.

2) Epimers: • They are optical isomers which differ in distribution of H and OH groups around a single asymmetric carbon atom other than the anomeric (carbon of aldhyde or ketone). • Ribose is an epimer to each of arabinose and xylose.
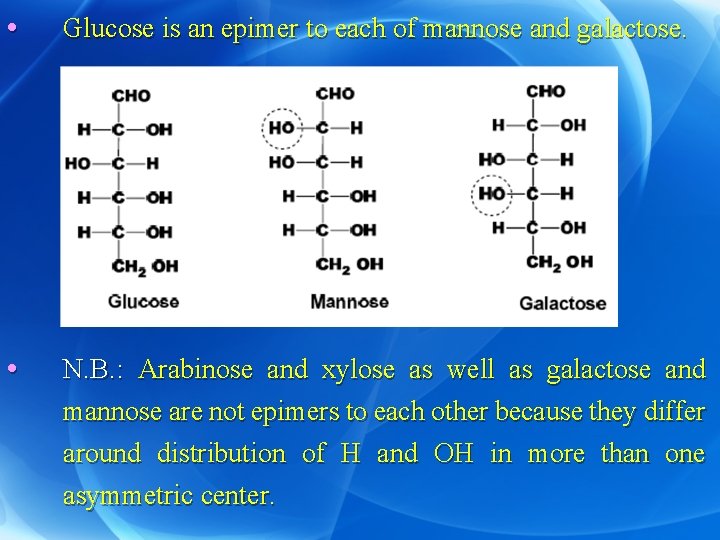
• Glucose is an epimer to each of mannose and galactose. • N. B. : Arabinose and xylose as well as galactose and mannose are not epimers to each other because they differ around distribution of H and OH in more than one asymmetric center.
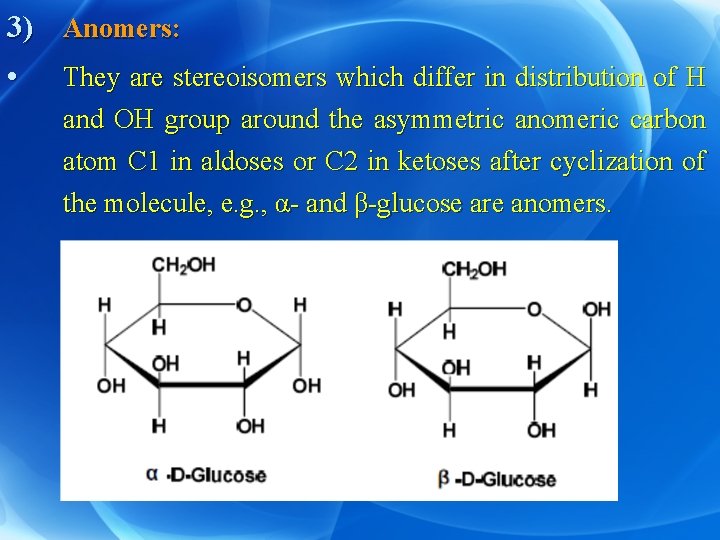
3) Anomers: • They are stereoisomers which differ in distribution of H and OH group around the asymmetric anomeric carbon atom C 1 in aldoses or C 2 in ketoses after cyclization of the molecule, e. g. , α- and β-glucose are anomers.
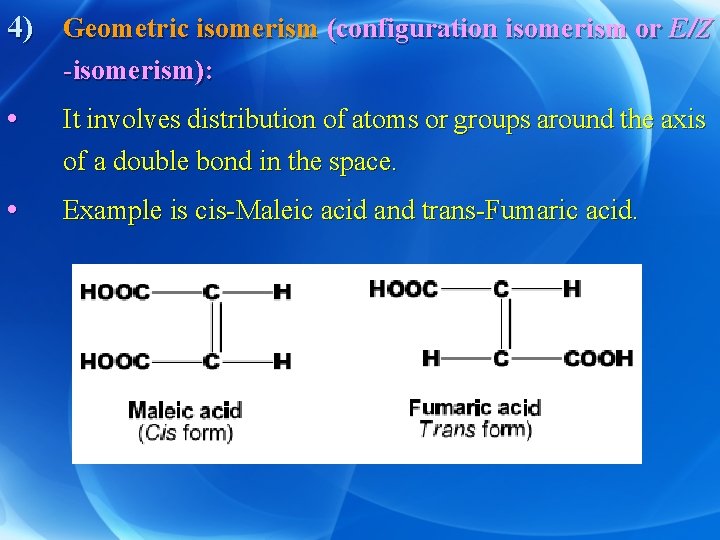
4) Geometric isomerism (configuration isomerism or E/Z -isomerism): • It involves distribution of atoms or groups around the axis of a double bond in the space. • Example is cis-Maleic acid and trans-Fumaric acid.

Chemical reactions of monosaccharides Next lecture ? ?

- Slides: 53