Lecture 9 Recombinant DNA Vector Design and Construction

Lecture 9 Recombinant DNA, Vector Design and Construction Neal Stewart and Dave Mann

Discussion questions 1. What basic elements should be included in the design and construction of an efficient ubiquitous and constitutive plant gene expression vector? 2. Discuss the advantages and disadvantages of recombination cloning technologies versus traditional restriction digestion and ligation technology. 3. Describe a novel strategy to generate a T-DNA vector that allows the expression of several genes from a single position in the genome. 4. Discuss the advantages and disadvantages of using plastid vectors for plant transformation and gene expression. 5. Describe ways in which transgene technology could be made more acceptable to the public.
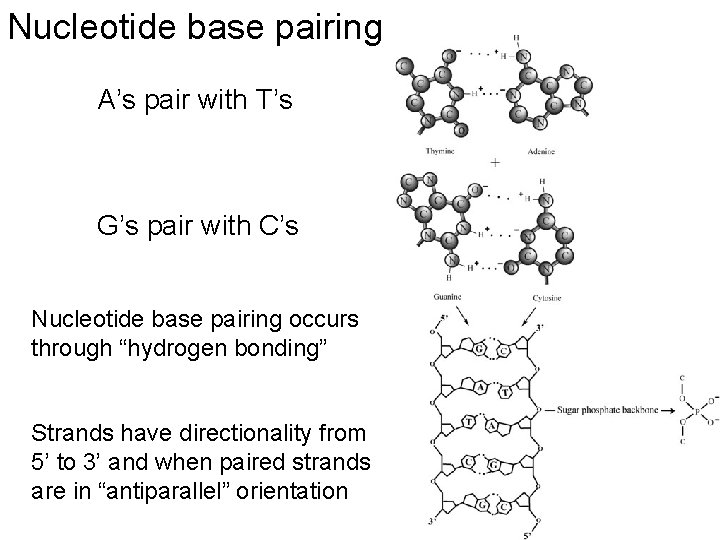
Nucleotide base pairing A’s pair with T’s G’s pair with C’s Nucleotide base pairing occurs through “hydrogen bonding” Strands have directionality from 5’ to 3’ and when paired strands are in “antiparallel” orientation
Figure 8. 1
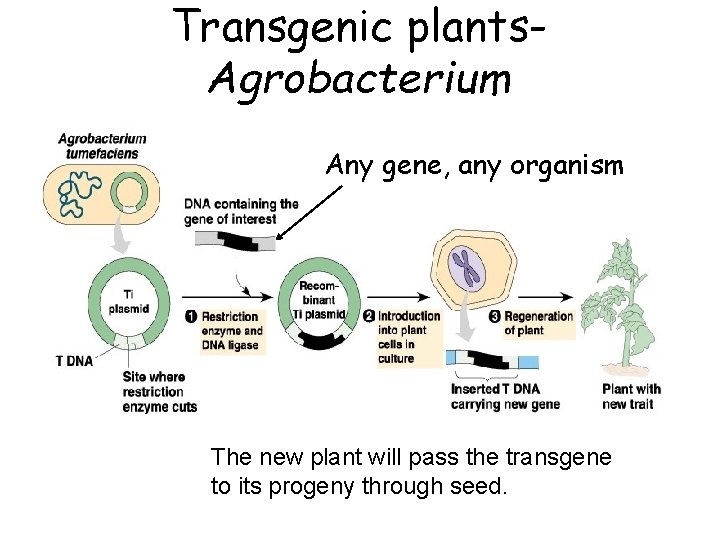
Transgenic plants. Agrobacterium Any gene, any organism The new plant will pass the transgene to its progeny through seed.

Biolistics Consumer reports, Sept. 1999

Steps to make transgenic plants— lotta transformation • Make transformation cloning plasmid vector • Transform bacteria (usually Escherichia coli) to maintain clone • Characterize plasmid (restriction digest and sequencing) • Transform Agrobacterium (if using Agrobacterium) and characterize • Transform plant

Recombinant DNA history 1966 The genetic code is deciphered when biochemical analysis reveals which codons determine which amino acids. 1970 Hamilton Smith, at Johns Hopkins Medical School, isolates the first restriction enzyme, an enzyme that cuts DNA at a very specific nucleotide sequence. Over the next few years, several more restriction enzymes will be isolated. 1972 Stanley Cohen and Herbert Boyer combine their efforts to create recombinant DNA. This technology will be the beginning of the biotechnology industry. 1976 Herbert Boyer cofounds Genentech, the first firm founded in the United States to apply recombinant DNA technology 1978 Somatostatin, which regulates human growth hormones, is the first human protein made using recombinant technology. 1983 Kary Mullis does PCR. 1985 Kary Mullis publishes method. Patents follow. 2000 Gateway cloning http: //www. accessexcellence. org/RC/AB/WYW/wkbooks/SFTS/sidebarmilestone. html

Key enzymes • Restriction endonuclease • DNA ligase • Taq DNA polymerase

Cloning platforms • Restriction enzymes/ligase • PCR-based methods • Gateway and other site specific recombination methods • Golden Gate and other “parts-based” modular construction methods

Restriction enzyme-ligation
Figure 8. 3
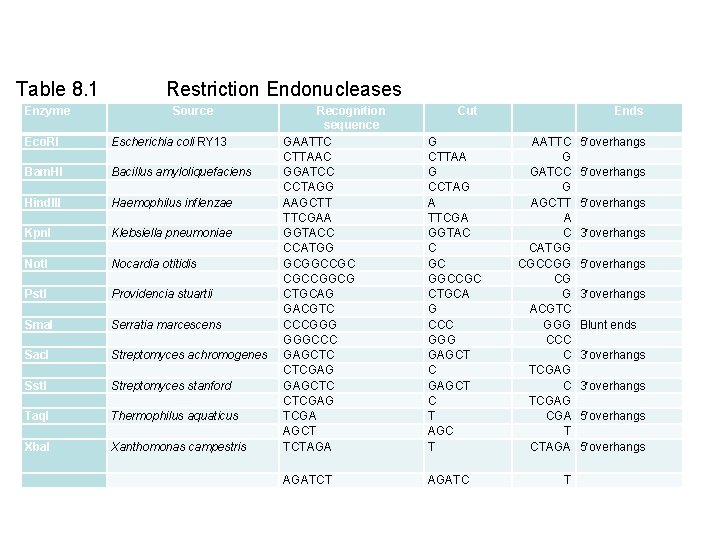
Table 8. 1 Restriction Endonucleases Enzyme Source Eco. RI Bam. HI Hind. III Kpn. I Not. I Pst. I Sma. I Sac. I Sst. I Taq. I Xba. I Escherichia coli RY 13 Bacillus amyloliquefaciens Haemophilus inflenzae Klebsiella pneumoniae Nocardia otitidis Providencia stuartii Serratia marcescens Streptomyces achromogenes Streptomyces stanford Thermophilus aquaticus Xanthomonas campestris Recognition sequence GAATTC CTTAAC GGATCC CCTAGG AAGCTT TTCGAA GGTACC CCATGG GCGGCCGC CGCCGGCG CTGCAG GACGTC CCCGGG GGGCCC GAGCTC CTCGAG TCGA AGCT TCTAGA AGATCT Cut G CTTAA G CCTAG A TTCGA GGTAC C GC GGCCGC CTGCA G CCC GGG GAGCT C T AGC T AGATC Ends AATTC G GATCC G AGCTT A C CATGG CGCCGG CG G ACGTC GGG CCC C TCGAG CGA T CTAGA 5 overhangs 3 overhangs Blunt ends 3 overhangs 5 overhangs T
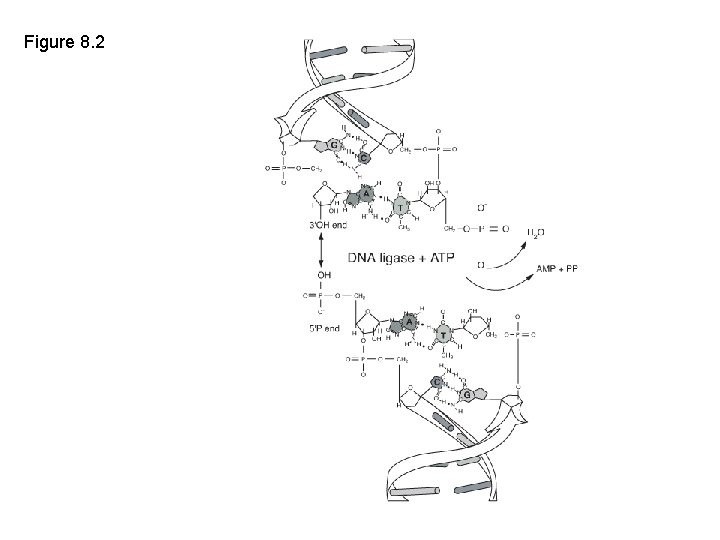
Figure 8. 2
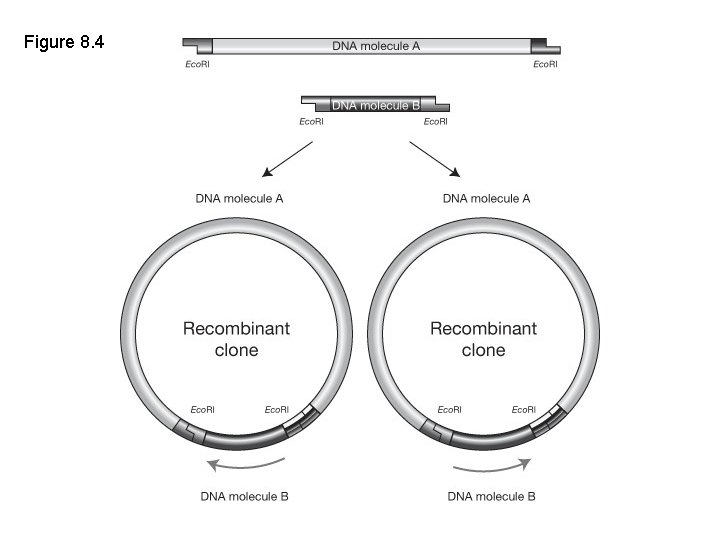
Figure 8. 4
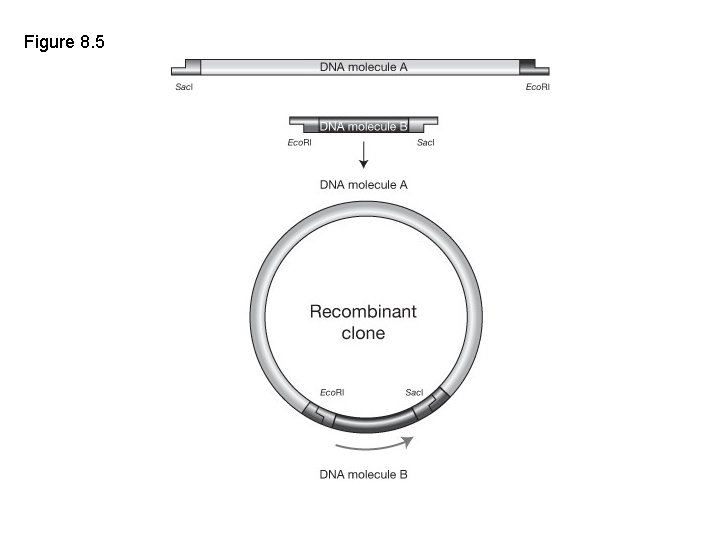
Figure 8. 5

Transformation vector requirements • • Origin of replication Bacterial selectable marker Gene constructs of interest T-DNA borders and other Agrobacterium genes if using Agrobacterium • Compatible with helper plasmid if using Agrobacterium
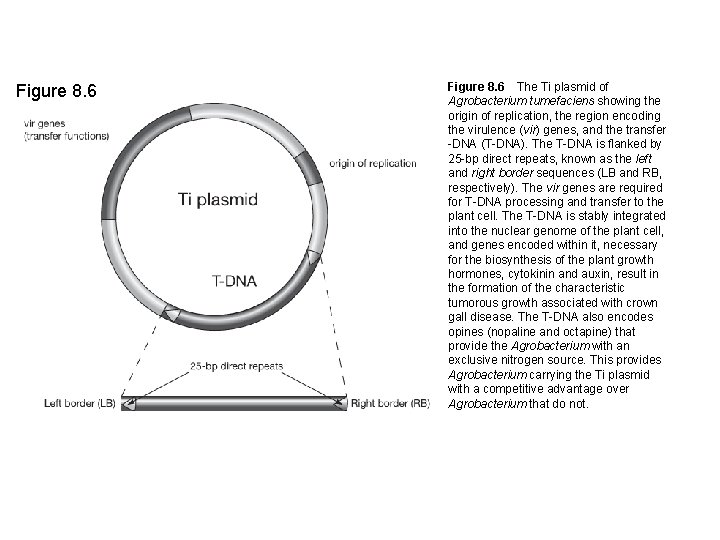
Figure 8. 6 The Ti plasmid of Agrobacterium tumefaciens showing the origin of replication, the region encoding the virulence (vir) genes, and the transfer -DNA (T-DNA). The T-DNA is flanked by 25 -bp direct repeats, known as the left and right border sequences (LB and RB, respectively). The vir genes are required for T-DNA processing and transfer to the plant cell. The T-DNA is stably integrated into the nuclear genome of the plant cell, and genes encoded within it, necessary for the biosynthesis of the plant growth hormones, cytokinin and auxin, result in the formation of the characteristic tumorous growth associated with crown gall disease. The T-DNA also encodes opines (nopaline and octapine) that provide the Agrobacterium with an exclusive nitrogen source. This provides Agrobacterium carrying the Ti plasmid with a competitive advantage over Agrobacterium that do not.
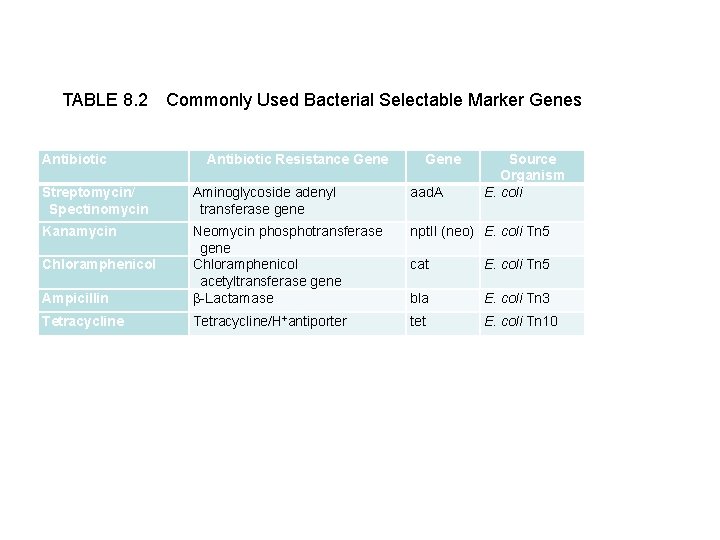
TABLE 8. 2 Commonly Used Bacterial Selectable Marker Genes Antibiotic Resistance Gene Source Organism E. coli Streptomycin/ Spectinomycin Aminoglycoside adenyl transferase gene aad. A Kanamycin npt. II (neo) E. coli Tn 5 Ampicillin Neomycin phosphotransferase gene Chloramphenicol acetyltransferase gene -Lactamase Tetracycline/H+antiporter Chloramphenicol cat E. coli Tn 5 bla E. coli Tn 3 tet E. coli Tn 10

Typical components of transformation vector: making a construct • Selectable marker cassette (with promoter and terminator) • Gene of interest cassette (with promoter and terminator) • Scorable marker cassette (with promoter and terminator) What happens if the promoter is missing? Is there ever a time when a promoterless construct is desirable?
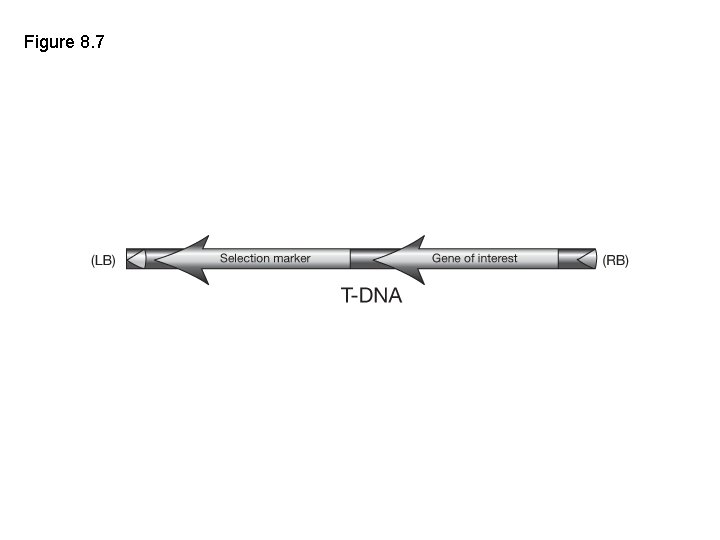
Figure 8. 7
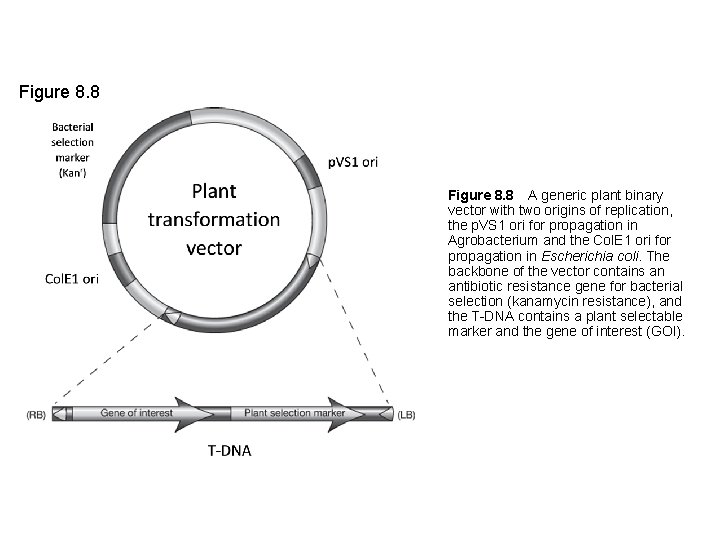
Figure 8. 8 A generic plant binary vector with two origins of replication, the p. VS 1 ori for propagation in Agrobacterium and the Col. E 1 ori for propagation in Escherichia coli. The backbone of the vector contains an antibiotic resistance gene for bacterial selection (kanamycin resistance), and the T-DNA contains a plant selectable marker and the gene of interest (GOI).
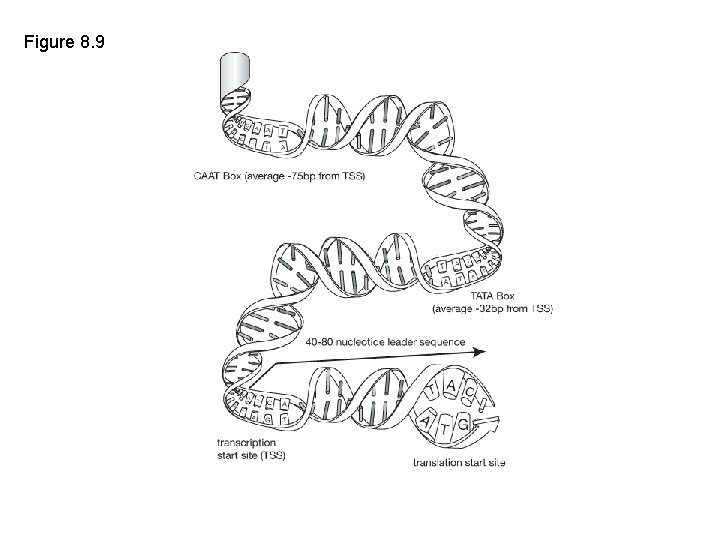
Figure 8. 9

PCR videos! How PCR works: http: //youtube. com/watch? v=_Yg. Xc. J 4 n-k. Q PCR song! http: //youtube. com/watch? v=x 5 y. Pkx. CLads&feature=related

What is cloning?
http: //upload. wikimedia. org/wikipedia/commons/thumb/6/66/Scissors. sv g/540 px-Scissors. svg. png
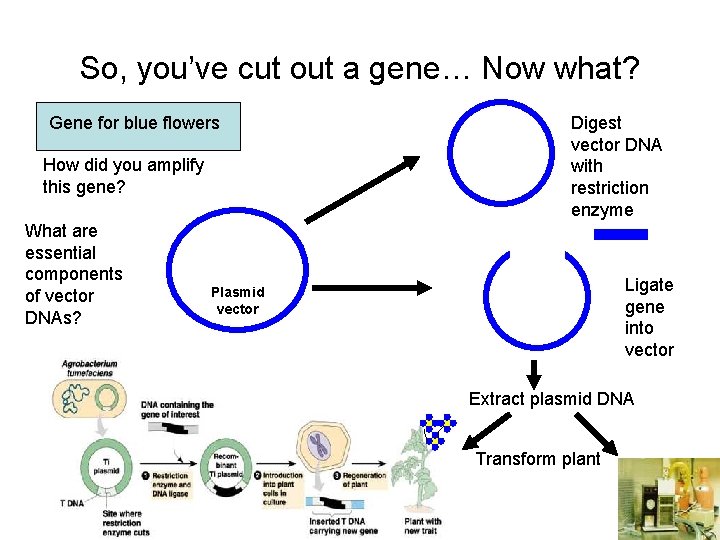
So, you’ve cut out a gene… Now what? Gene for blue flowers How did you amplify this gene? What are essential components of vector DNAs? Digest vector DNA with restriction enzyme Ligate gene into vector Plasmid vector Extract plasmid DNA Transform plant
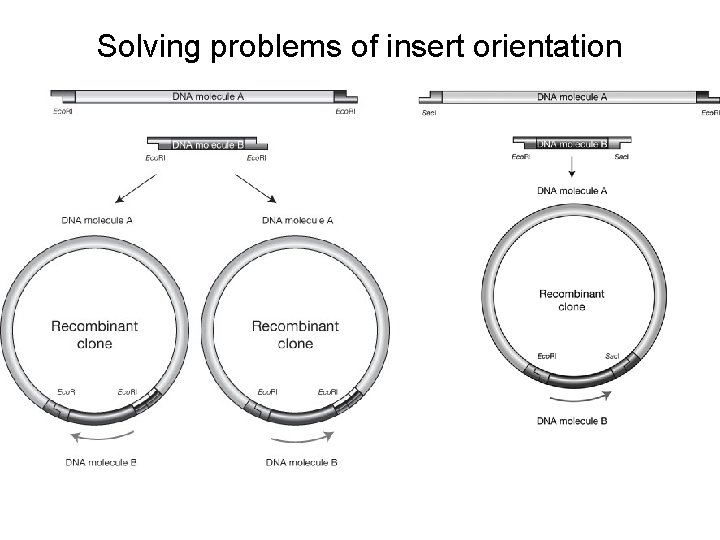
Solving problems of insert orientation

Problems with conventional cloning • Inconvenient restriction sites • Vector construction is laborious • Time-consuming reactions
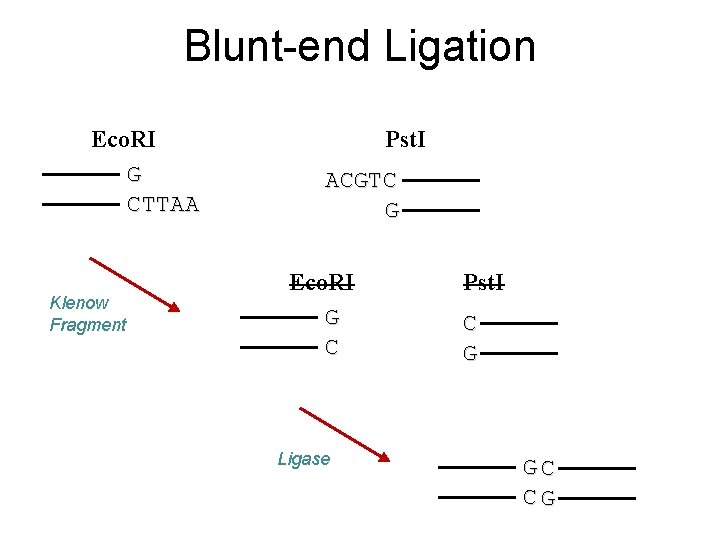
Blunt-end Ligation Eco. RI G CTTAA Klenow Fragment Pst. I ACGTC G Eco. RI G C Ligase Pst. I C G GC CG

Site-specific Recombination: Gateway™ Cloning
Bacteriophage λ
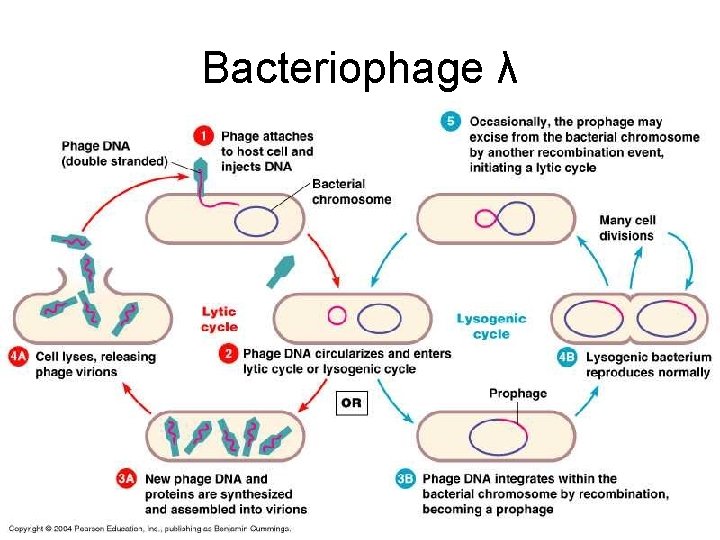
Bacteriophage λ
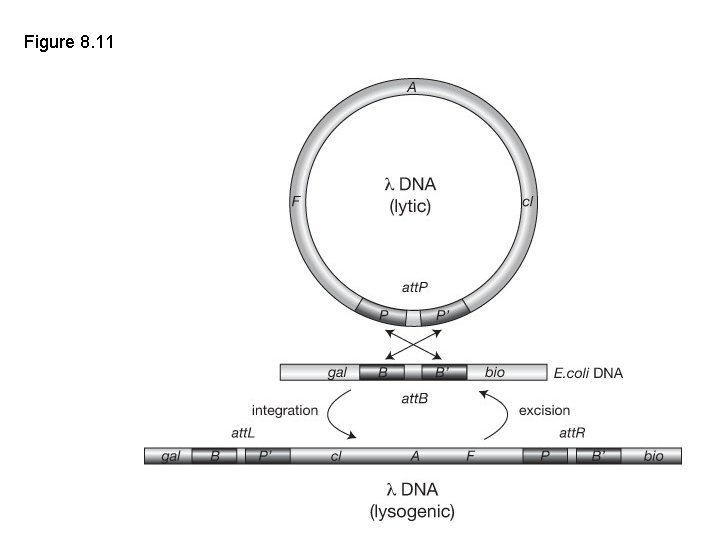
Figure 8. 11

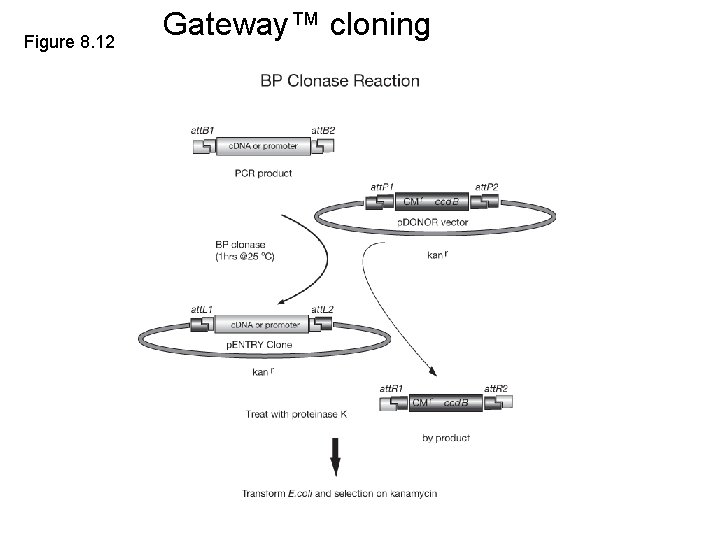
Figure 8. 12 Gateway™ cloning
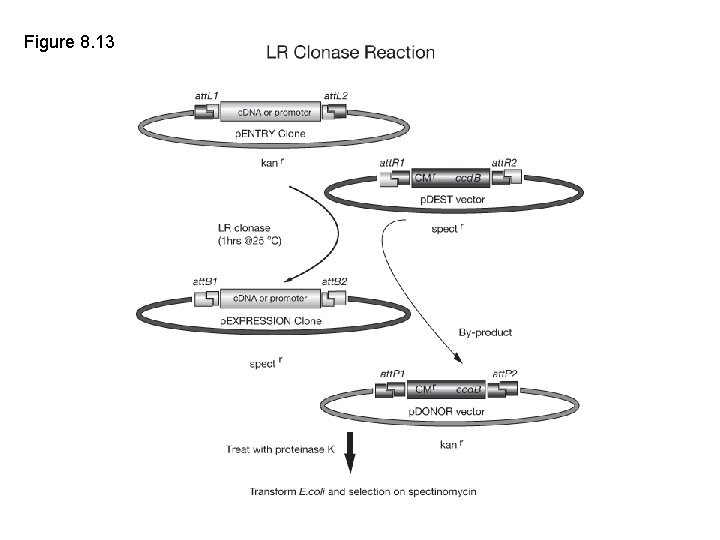
Figure 8. 13
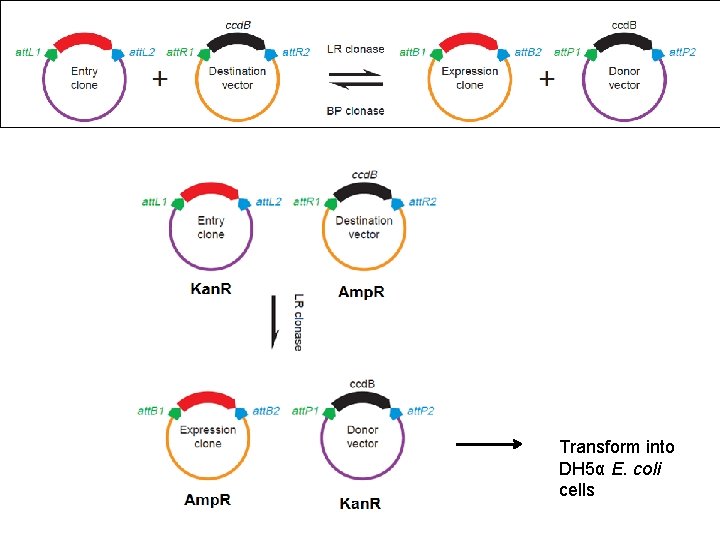
Transform into DH 5α E. coli cells
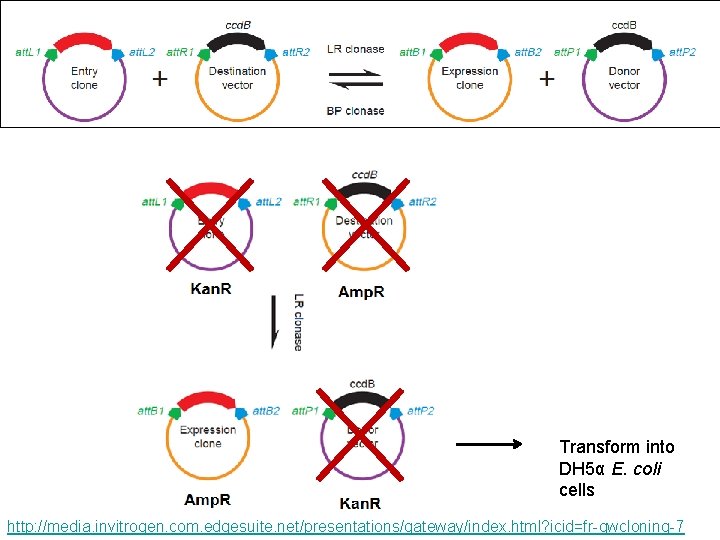
Transform into DH 5α E. coli cells http: //media. invitrogen. com. edgesuite. net/presentations/gateway/index. html? icid=fr-gwcloning-7

Why do we need so many types of vectors? What are some different applications in plants? • Functional analysis of open reading frame (ORFs) • Overexpression and knockdown (RNAi) of specific genes. • Multigenic traits for crop improvement • Analysis of the expression level/specificity/ inducibility of promoters
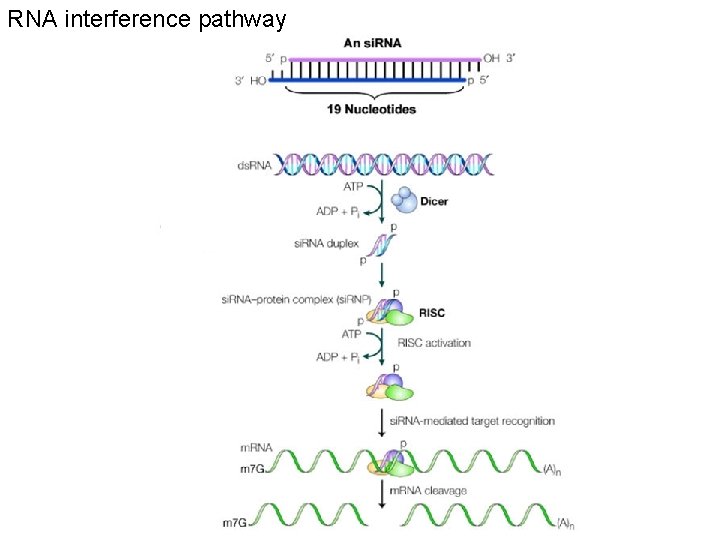
RNA interference pathway
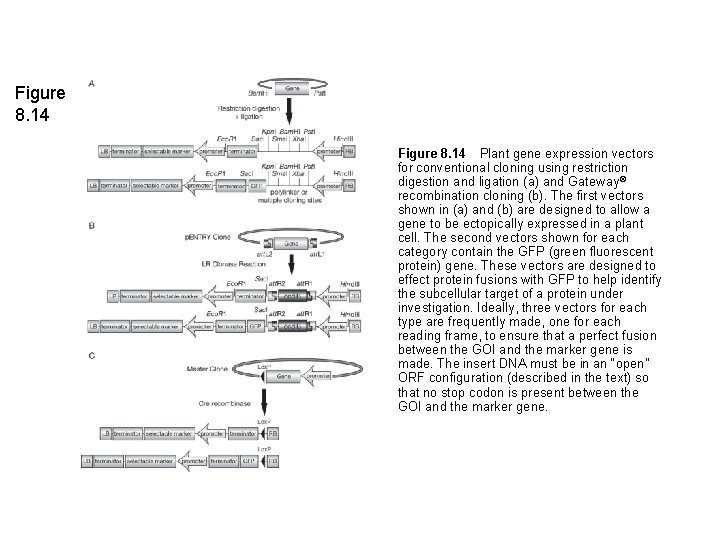
Figure 8. 14 Plant gene expression vectors for conventional cloning using restriction digestion and ligation (a) and Gateway ® recombination cloning (b). The first vectors shown in (a) and (b) are designed to allow a gene to be ectopically expressed in a plant cell. The second vectors shown for each category contain the GFP (green fluorescent protein) gene. These vectors are designed to effect protein fusions with GFP to help identify the subcellular target of a protein under investigation. Ideally, three vectors for each type are frequently made, one for each reading frame, to ensure that a perfect fusion between the GOI and the marker gene is made. The insert DNA must be in an “open” ORF configuration (described in the text) so that no stop codon is present between the GOI and the marker gene.
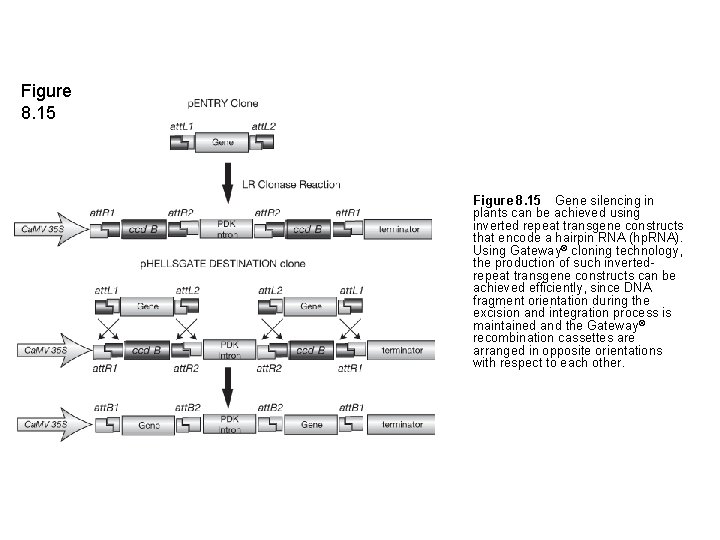
Figure 8. 15 Gene silencing in plants can be achieved using inverted repeat transgene constructs that encode a hairpin RNA (hp. RNA). Using Gateway® cloning technology, the production of such invertedrepeat transgene constructs can be achieved efficiently, since DNA fragment orientation during the excision and integration process is maintained and the Gateway ® recombination cassettes are arranged in opposite orientations with respect to each other.
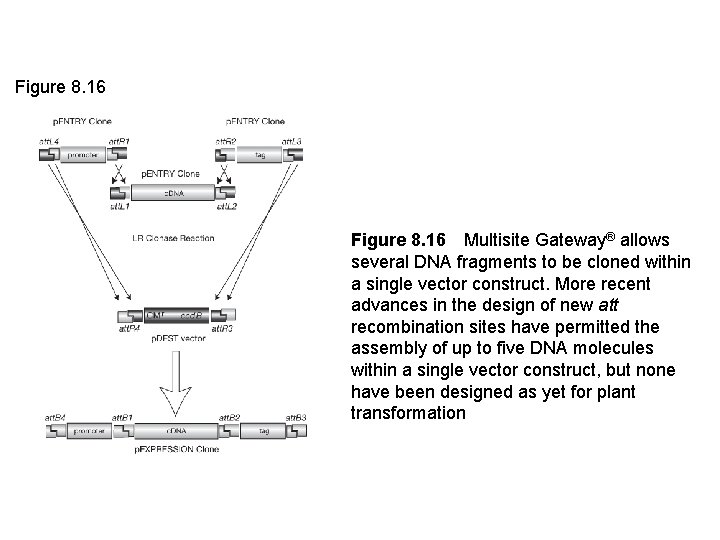
Figure 8. 16 Multisite Gateway® allows several DNA fragments to be cloned within a single vector construct. More recent advances in the design of new att recombination sites have permitted the assembly of up to five DNA molecules within a single vector construct, but none have been designed as yet for plant transformation

Multiple promoters from the MRS 2/MGT gene family fused to the GUS gene and expressed in Arabidopsis thaliana Gebert et al. , The Plant Cell, Vol. 21: 4018– 4030, December 2009
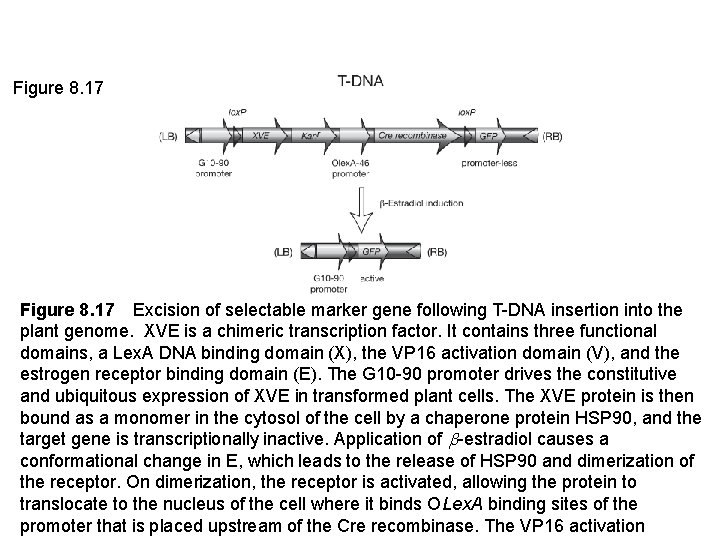
Figure 8. 17 Excision of selectable marker gene following T-DNA insertion into the plant genome. XVE is a chimeric transcription factor. It contains three functional domains, a Lex. A DNA binding domain (X), the VP 16 activation domain (V), and the estrogen receptor binding domain (E). The G 10 -90 promoter drives the constitutive and ubiquitous expression of XVE in transformed plant cells. The XVE protein is then bound as a monomer in the cytosol of the cell by a chaperone protein HSP 90, and the target gene is transcriptionally inactive. Application of -estradiol causes a conformational change in E, which leads to the release of HSP 90 and dimerization of the receptor. On dimerization, the receptor is activated, allowing the protein to translocate to the nucleus of the cell where it binds OLex. A binding sites of the promoter that is placed upstream of the Cre recombinase. The VP 16 activation
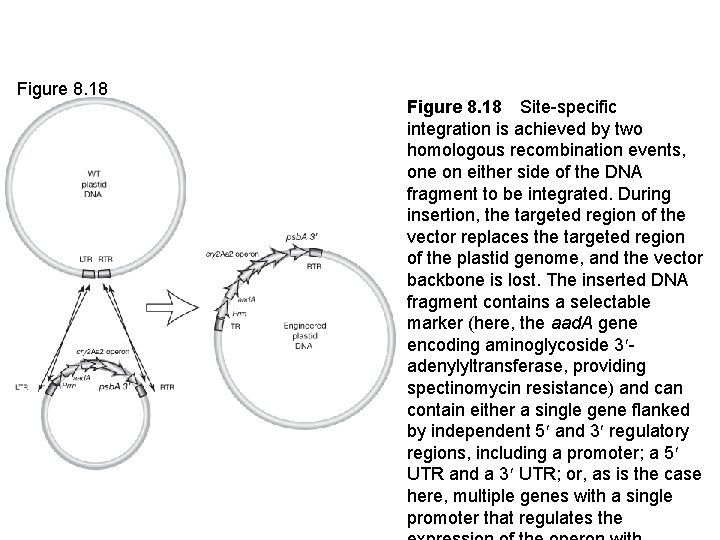
Figure 8. 18 Site-specific integration is achieved by two homologous recombination events, one on either side of the DNA fragment to be integrated. During insertion, the targeted region of the vector replaces the targeted region of the plastid genome, and the vector backbone is lost. The inserted DNA fragment contains a selectable marker (here, the aad. A gene encoding aminoglycoside 3 adenylyltransferase, providing spectinomycin resistance) and can contain either a single gene flanked by independent 5 and 3 regulatory regions, including a promoter; a 5 UTR and a 3 UTR; or, as is the case here, multiple genes with a single promoter that regulates the

Vectors derived from plant sequences • Public acceptance of GMOs linked to concerns of the origin of DNA employed • Ironically, wild-type plant cells already contain bacterially-derived genomes • T-DNA could be replaced with P-DNA • Replace viral promoters with plant promoters
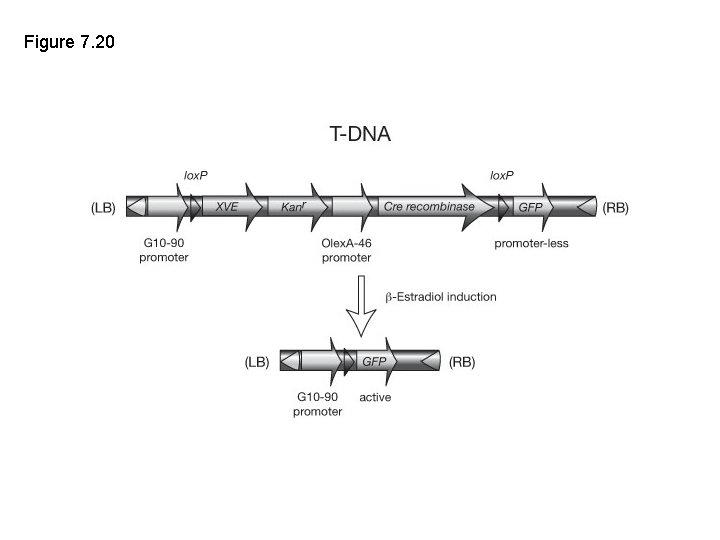
Figure 7. 20
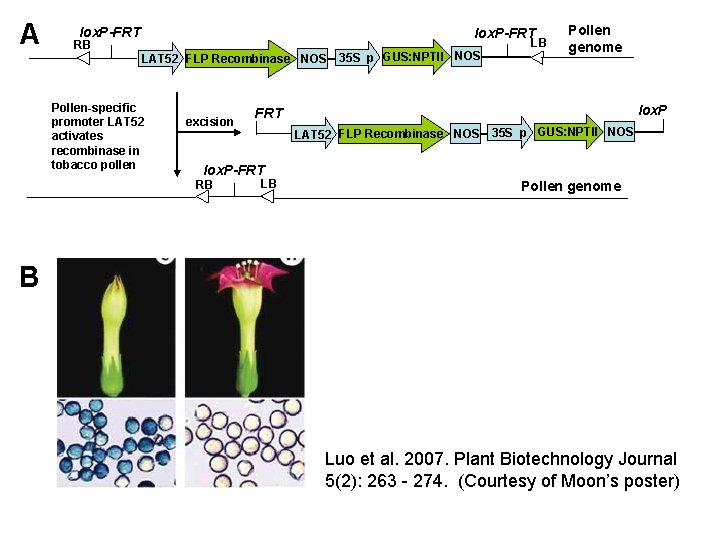
A lox. P-FRT RB LAT 52 FLP Recombinase NOS Pollen-specific promoter LAT 52 activates recombinase in tobacco pollen excision 35 S p GUS: NPTII NOS LB Pollen genome lox. P FRT LAT 52 FLP Recombinase NOS 35 S p GUS: NPTII NOS lox. P-FRT RB LB Pollen genome B Luo et al. 2007. Plant Biotechnology Journal 5(2): 263 - 274. (Courtesy of Moon’s poster)

Example of a plant expression vector set • • p. ANIC Made for switchgrass transformation: Bio. Energy Science Center (BESC) http: //plantsciences. utk. edu/stewart. htm
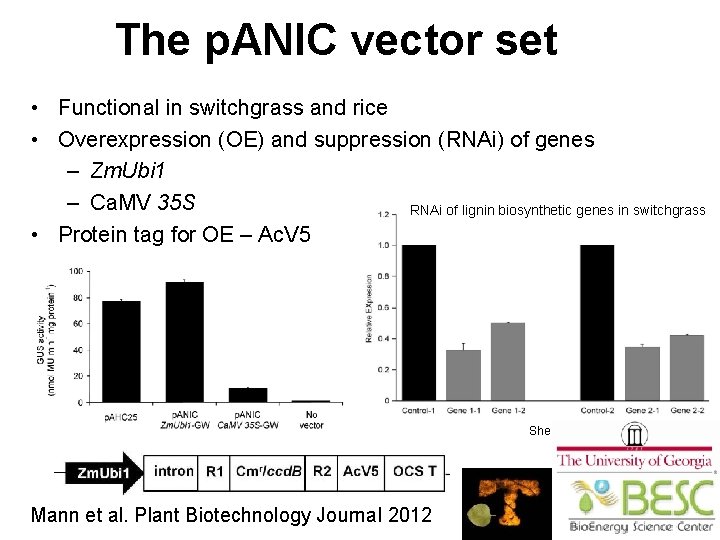
The p. ANIC vector set • Functional in switchgrass and rice • Overexpression (OE) and suppression (RNAi) of genes – Zm. Ubi 1 – Ca. MV 35 S RNAi of lignin biosynthetic genes in switchgrass • Protein tag for OE – Ac. V 5 Shen & Dixon, Noble Foundation Mann et al. Plant Biotechnology Journal 2012 53
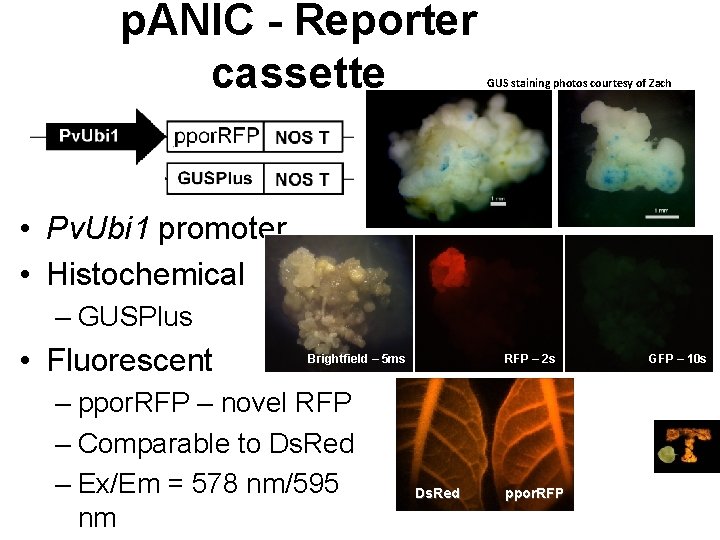
p. ANIC - Reporter cassette GUS staining photos courtesy of Zach King • Pv. Ubi 1 promoter • Histochemical – GUSPlus • Fluorescent Brightfield – 5 ms – ppor. RFP – novel RFP – Comparable to Ds. Red – Ex/Em = 578 nm/595 nm RFP – 2 s Ds. Red ppor. RFP GFP – 10 s
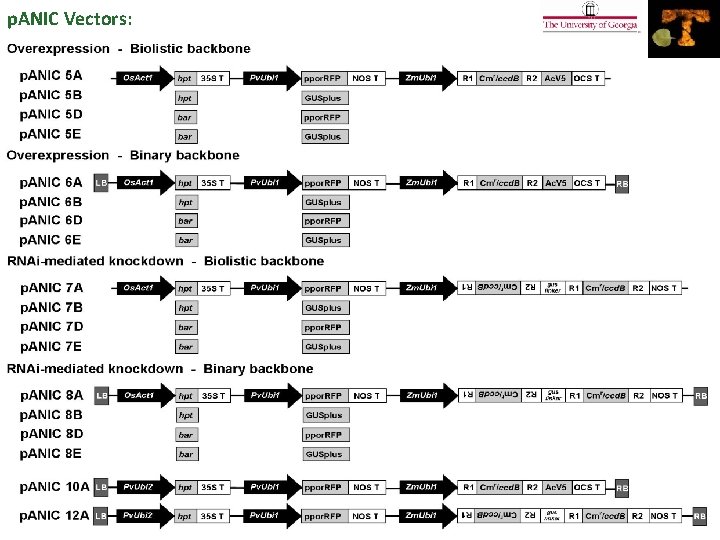
p. ANIC Vectors:

Modular “parts” based cloning Golden Gate is one method https: //www. neb. com/applications/cloning-andsynthetic-biology/dna-assembly-andcloning/golden-gate-assembly
- Slides: 56