Lecture 9 Ionising Radiation Generation Dr Sarah Bohndiek

Lecture 9: Ionising Radiation Generation Dr Sarah Bohndiek

Learning outcomes • After this lecture, you should be able to: • Describe how ionising radiation is generated for medical imaging • Explain the differences between an X-ray tube and a linear accelerator • Detail the process of generating radioisotopes and how they are incorporated into a radiopharmaceutical • Understand the difference between a positron emitter and a single photon emitter in terms of practical medical imaging

Ionising radiation can be used for imaging in three different configurations X-ray Single photon emission Positron emission Planar transmission X-ray CT Planar scintigraphy SPECT Positron Emission Tomography PET/CT

How to generate X-rays How to generate gamma rays using radiopharmaceuticals
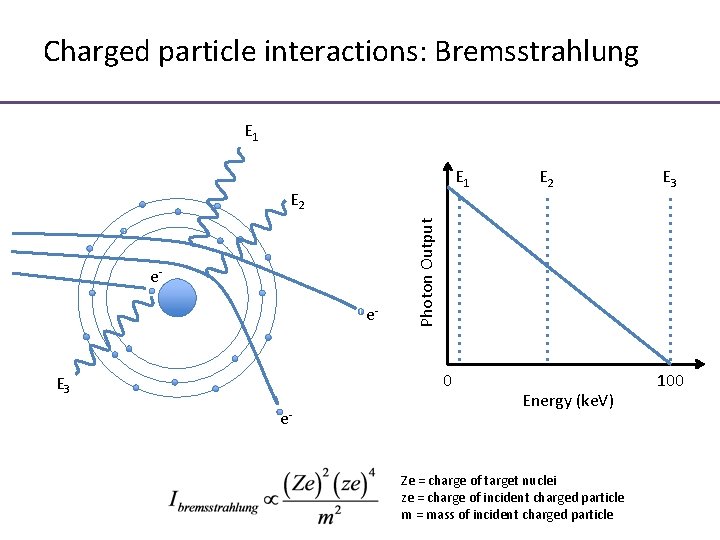
Charged particle interactions: Bremsstrahlung E 1 ee- 0 E 3 e- E 3 Photon Output E 2 Energy (ke. V) Ze = charge of target nuclei ze = charge of incident charged particle m = mass of incident charged particle 100

Charged particle interactions: Characteristic E 1 e- e. K L M Characteristic radiation occurs when incident electrons interact with the atomic electrons in the target material
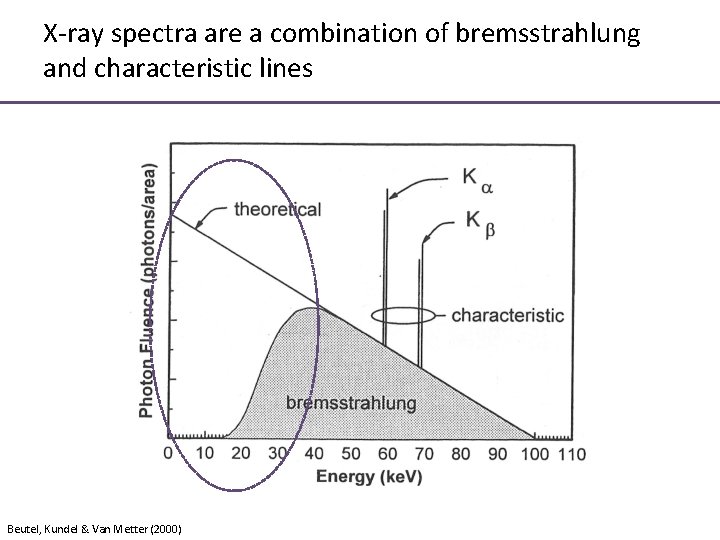
X-ray spectra are a combination of bremsstrahlung and characteristic lines Beutel, Kundel & Van Metter (2000)
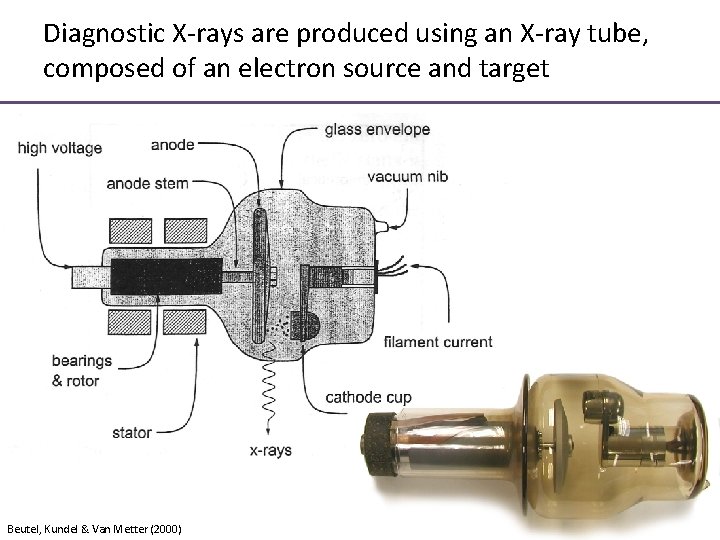
Diagnostic X-rays are produced using an X-ray tube, composed of an electron source and target Beutel, Kundel & Van Metter (2000)

Factors affecting the resulting X-ray spectrum • Tube current – Determines the number of electrons striking the anode, which determines the quantity of X-rays emitted • Exposure time – Similarly impacts quantity of X-rays emitted • Applied voltage – Energy of the X-rays produced increases with voltage – Efficiency of conversion of electron energy into X-rays also increases with applied voltage – Changing applied voltage therefore affects both the quantity of X-rays emitted and the shape of the spectrum • Anode material – Affects the efficiency of Bremsstrahlung production and position of characteristic lines, both of which increase with atomic number • Filtration
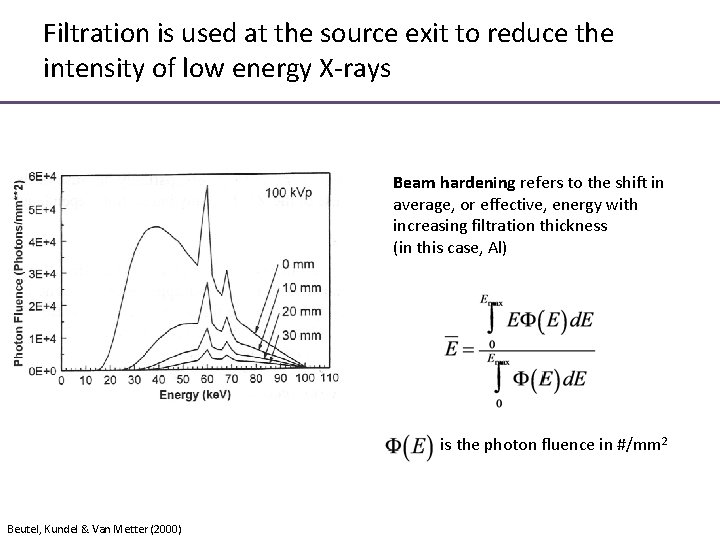
Filtration is used at the source exit to reduce the intensity of low energy X-rays Beam hardening refers to the shift in average, or effective, energy with increasing filtration thickness (in this case, Al) is the photon fluence in #/mm 2 Beutel, Kundel & Van Metter (2000)
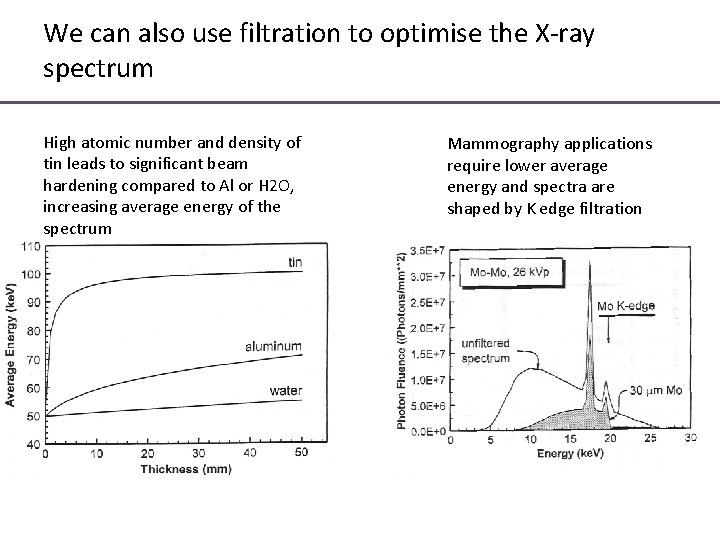
We can also use filtration to optimise the X-ray spectrum High atomic number and density of tin leads to significant beam hardening compared to Al or H 2 O, increasing average energy of the spectrum Mammography applications require lower average energy and spectra are shaped by K edge filtration

Therapeutic X-rays must reach energies several orders of magnitude higher than an X-ray tube can produce https: //www. youtube. com/watch? v=j. Sgn. Wfb. Ex 1 A
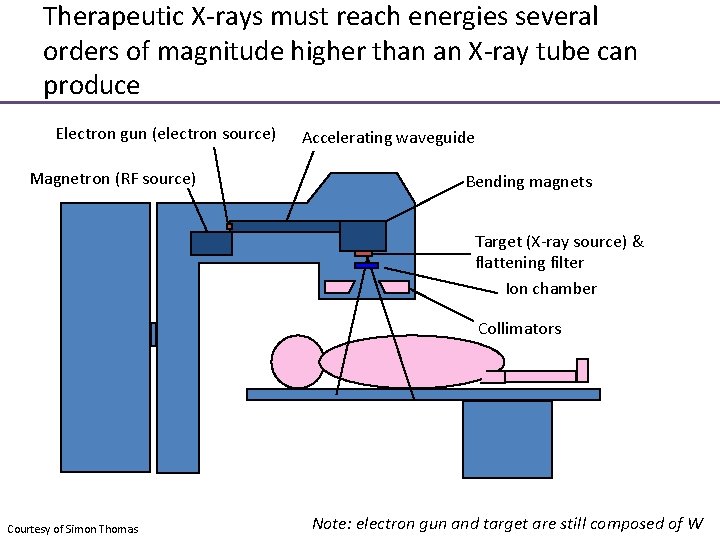
Therapeutic X-rays must reach energies several orders of magnitude higher than an X-ray tube can produce Electron gun (electron source) Magnetron (RF source) Accelerating waveguide Bending magnets Target (X-ray source) & flattening filter Ion chamber Collimators Courtesy of Simon Thomas Note: electron gun and target are still composed of W

The core of the linear accelerator is the waveguide www. cnn. com

Summary 1: X-ray production • X-ray spectra at diagnostic energies are produced from X-ray tubes – Combination of Bremsstrahlung and characteristic rays – Tungsten is a common filament and target material – Filtration can be used to alter the spectral profile and mean energy – Achieve X-rays of energies ~ 10 -200 ke. V • Therapeutic X-rays are produced by linear accelerators (linacs) – Same filament and target materials – Accelerate electrons to far higher energies to achieve Xrays of energies ~ 1 -100 Me. V
How to generate X-rays How to generate gamma rays using radiopharmaceuticals
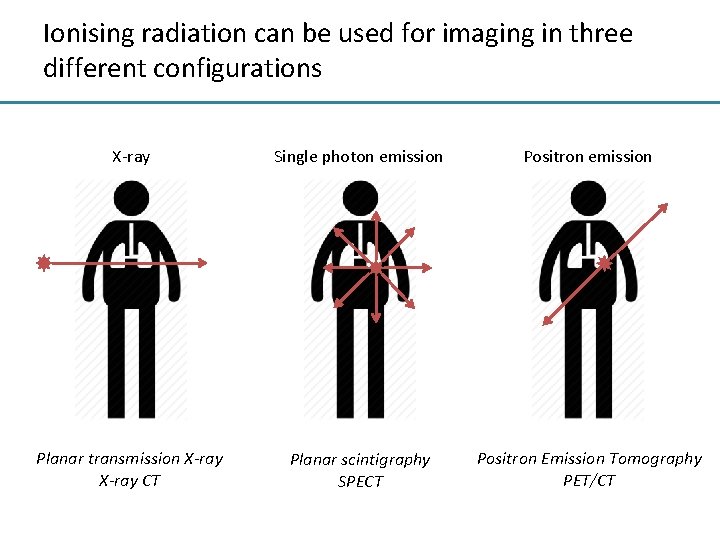
Ionising radiation can be used for imaging in three different configurations X-ray Single photon emission Positron emission Planar transmission X-ray CT Planar scintigraphy SPECT Positron Emission Tomography PET/CT
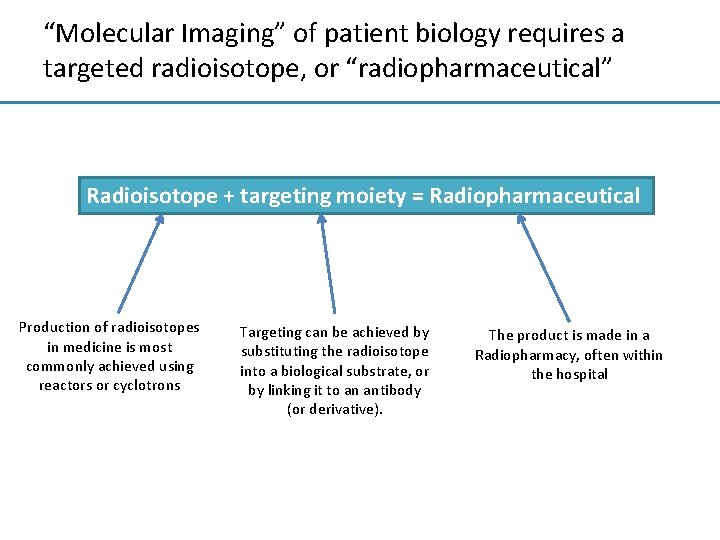
“Molecular Imaging” of patient biology requires a targeted radioisotope, or “radiopharmaceutical” Radioisotope + targeting moiety = Radiopharmaceutical Production of radioisotopes in medicine is most commonly achieved using reactors or cyclotrons Targeting can be achieved by substituting the radioisotope into a biological substrate, or by linking it to an antibody (or derivative). The product is made in a Radiopharmacy, often within the hospital
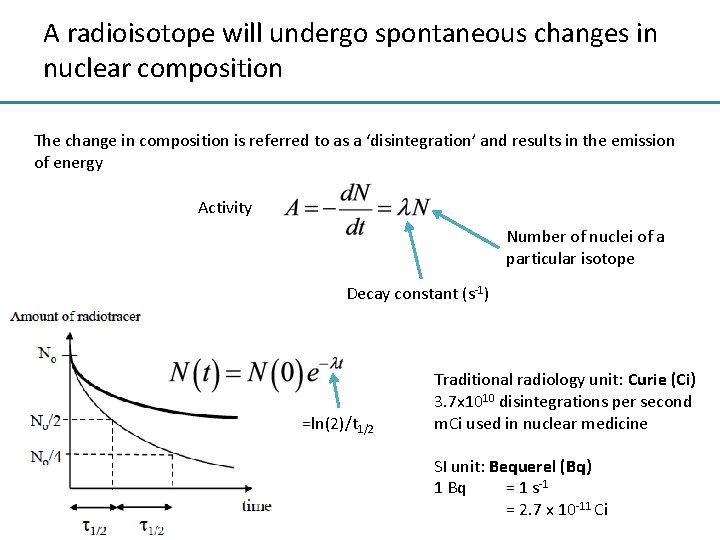
A radioisotope will undergo spontaneous changes in nuclear composition The change in composition is referred to as a ‘disintegration’ and results in the emission of energy Activity Number of nuclei of a particular isotope Decay constant (s-1) =ln(2)/t 1/2 Traditional radiology unit: Curie (Ci) 3. 7 x 1010 disintegrations per second m. Ci used in nuclear medicine SI unit: Bequerel (Bq) 1 Bq = 1 s-1 = 2. 7 x 10 -11 Ci

Radioisotopes can be formed by bombarding a target nucleus with neutrons or charged particles • If a neutron is added without emission of a proton, the resulting radioisotope will have the same chemistry, differing only in mass • If the target nucleus is bombarded by a charged particle, the resulting nucleus will usually be a different element • Binding energy per nucleon is normally ~ 8 Me. V • Producing a specific radioisotope requires optimisation of: – The target nucleus – The bombarding particle – The energy of the bombarding particle
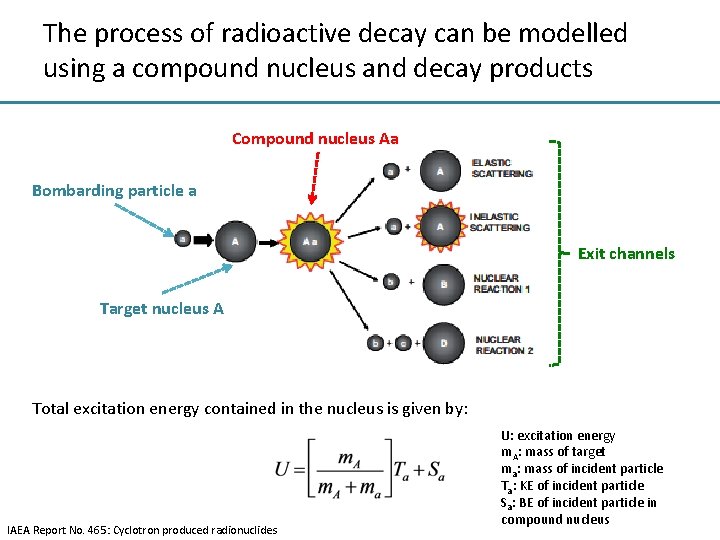
The process of radioactive decay can be modelled using a compound nucleus and decay products Compound nucleus Aa Bombarding particle a Exit channels Target nucleus A Total excitation energy contained in the nucleus is given by: IAEA Report No. 465: Cyclotron produced radionuclides U: excitation energy m. A: mass of target ma: mass of incident particle Ta: KE of incident particle Sa: BE of incident particle in compound nucleus
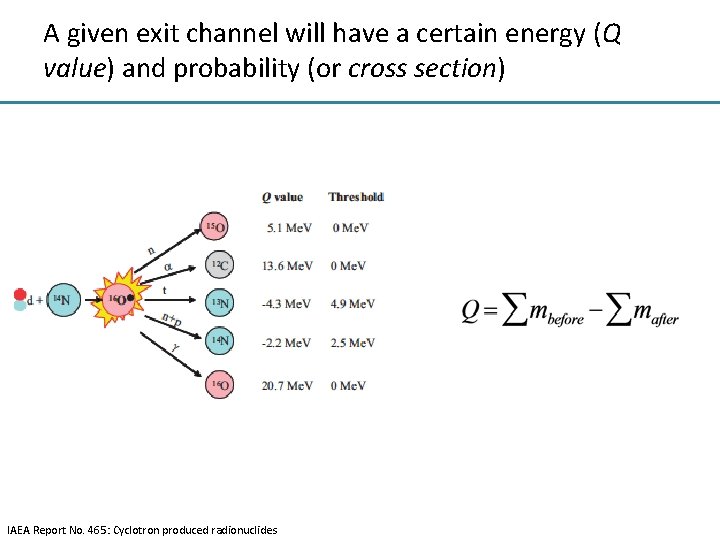
A given exit channel will have a certain energy (Q value) and probability (or cross section) IAEA Report No. 465: Cyclotron produced radionuclides
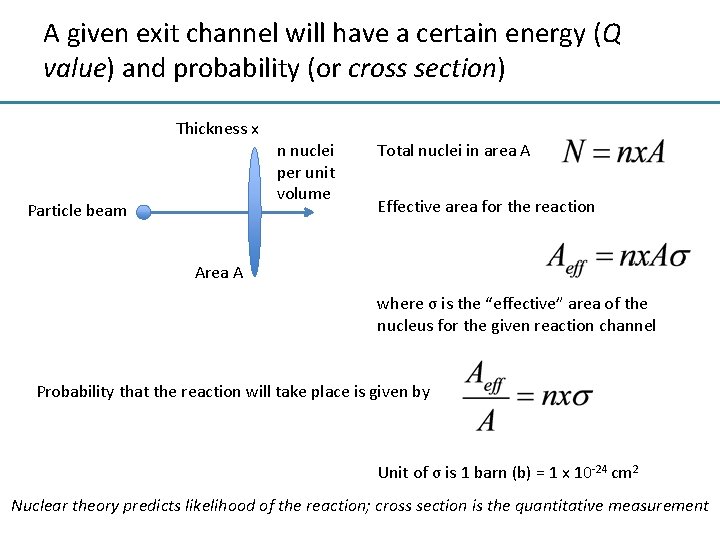
A given exit channel will have a certain energy (Q value) and probability (or cross section) Thickness x n nuclei per unit volume Particle beam Total nuclei in area A Effective area for the reaction Area A where σ is the “effective” area of the nucleus for the given reaction channel Probability that the reaction will take place is given by Unit of σ is 1 barn (b) = 1 x 10 -24 cm 2 Nuclear theory predicts likelihood of the reaction; cross section is the quantitative measurement

In nuclear reactors, radioisotopes are formed as byproducts of neutron bombardment Molybdenum-99 (Mo-99) 66 hour half life Highly enriched, weapons grade Uranium Ruth 2009 Rep Prog Phys 72 016701
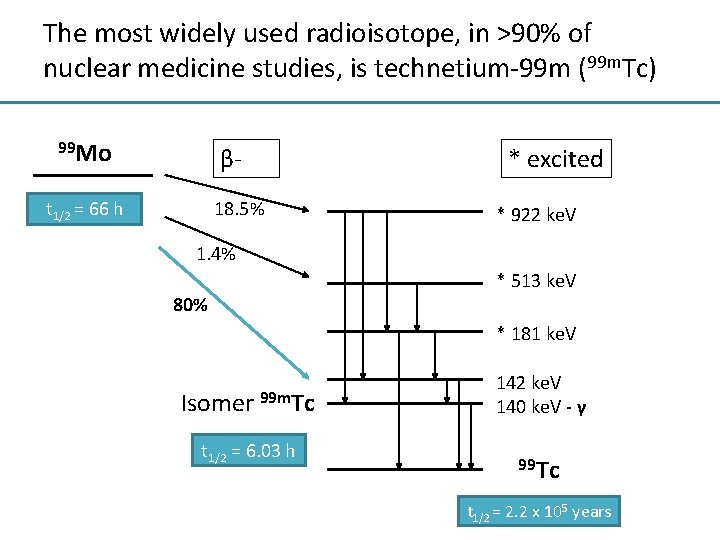
The most widely used radioisotope, in >90% of nuclear medicine studies, is technetium-99 m (99 m. Tc) 99 Mo β- t 1/2 = 66 h * excited 18. 5% * 922 ke. V 1. 4% * 513 ke. V 80% * 181 ke. V Isomer 99 m. Tc t 1/2 = 6. 03 h 142 ke. V 140 ke. V - γ 99 Tc t 1/2 = 2. 2 x 105 years
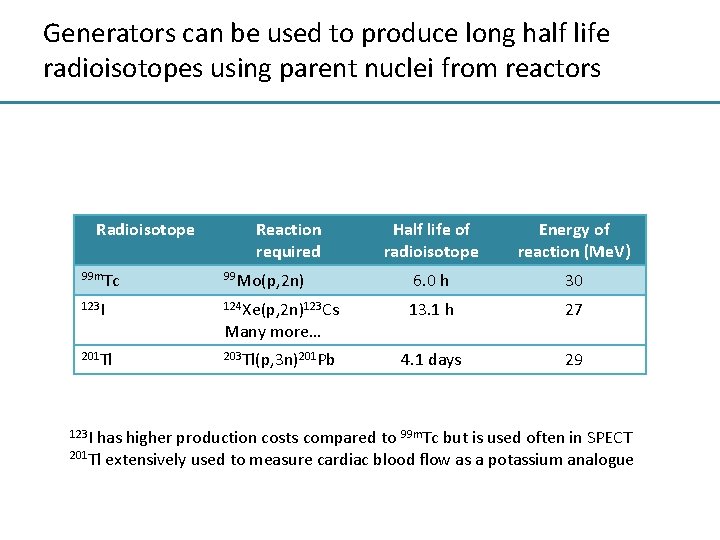
Generators can be used to produce long half life radioisotopes using parent nuclei from reactors Radioisotope Reaction required Half life of radioisotope Energy of reaction (Me. V) 6. 0 h 30 99 m. Tc 99 Mo(p, 2 n) 123 I 124 Xe(p, 2 n)123 Cs 13. 1 h 27 201 Tl 203 Tl(p, 3 n)201 Pb 4. 1 days 29 123 I Many more… has higher production costs compared to 99 m. Tc but is used often in SPECT 201 Tl extensively used to measure cardiac blood flow as a potassium analogue
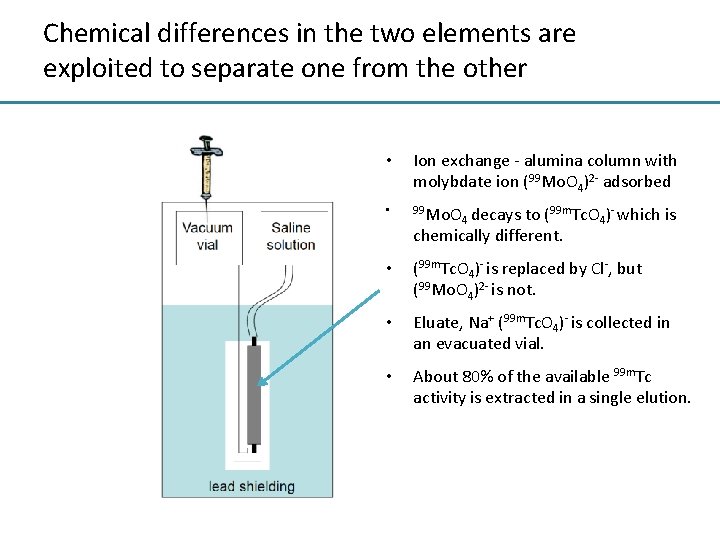
Chemical differences in the two elements are exploited to separate one from the other • Ion exchange - alumina column with molybdate ion (99 Mo. O 4)2 - adsorbed • 99 Mo. O • (99 m. Tc. O 4)- is replaced by Cl-, but (99 Mo. O 4)2 - is not. • Eluate, Na+ (99 m. Tc. O 4)- is collected in an evacuated vial. • About 80% of the available 99 m. Tc activity is extracted in a single elution. 4 decays to (99 m. Tc. O 4)- which is chemically different.

Bateman equations describe the growth of the daughter product from the parent in a generator Consider the decay series “parent” N 1 -> “daughter” N 2 -> “grand-daughter” N 3 The decay of the “parent” is As we can write Solving the differential equation yields:

Bateman equations describe the growth of the daughter product from the parent in a generator Equilibrium equations reflect the relative radioactivity (A) of daughter ‘d’ or parent ‘p’ Number of N 2 atoms decayed from N 1 and not yet to N 3 The number of N 2 atoms that remain from the initial N 2, 0 This is the Bateman Equation. It shows that the growth of the daughter product is a function of the decay of the parent and the disappearance of the daughter due to its own decay.
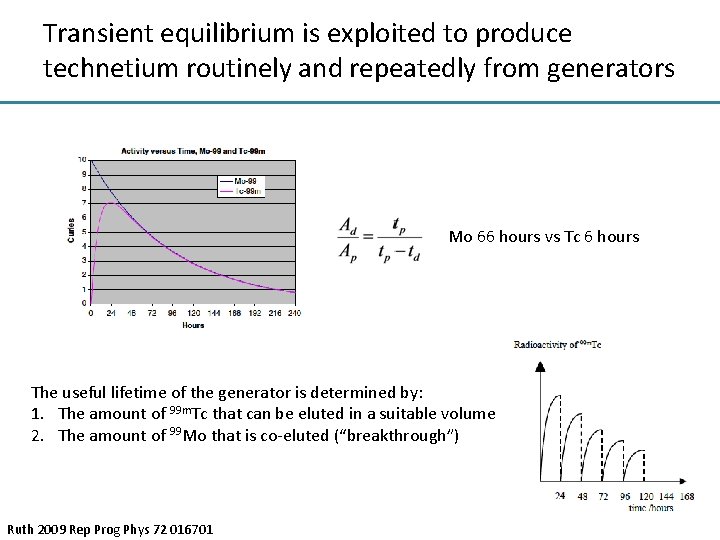
Transient equilibrium is exploited to produce technetium routinely and repeatedly from generators Mo 66 hours vs Tc 6 hours The useful lifetime of the generator is determined by: 1. The amount of 99 m. Tc that can be eluted in a suitable volume 2. The amount of 99 Mo that is co-eluted (“breakthrough”) Ruth 2009 Rep Prog Phys 72 016701
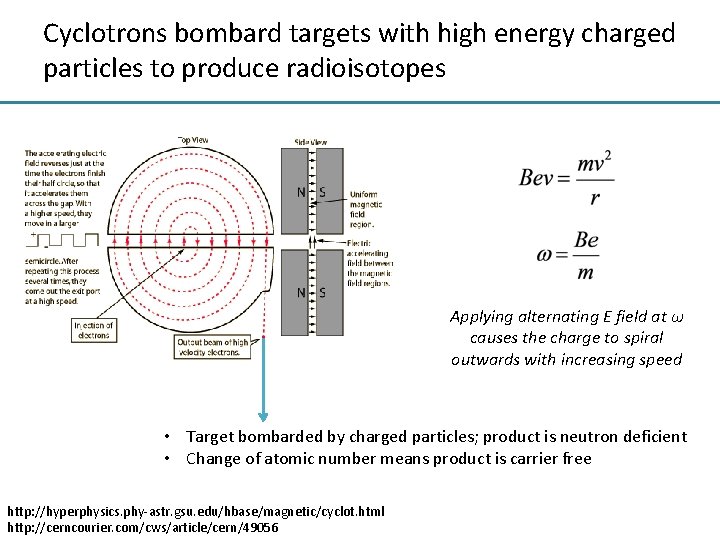
Cyclotrons bombard targets with high energy charged particles to produce radioisotopes Applying alternating E field at ω causes the charge to spiral outwards with increasing speed • Target bombarded by charged particles; product is neutron deficient • Change of atomic number means product is carrier free http: //hyperphysics. phy-astr. gsu. edu/hbase/magnetic/cyclot. html http: //cerncourier. com/cws/article/cern/49056
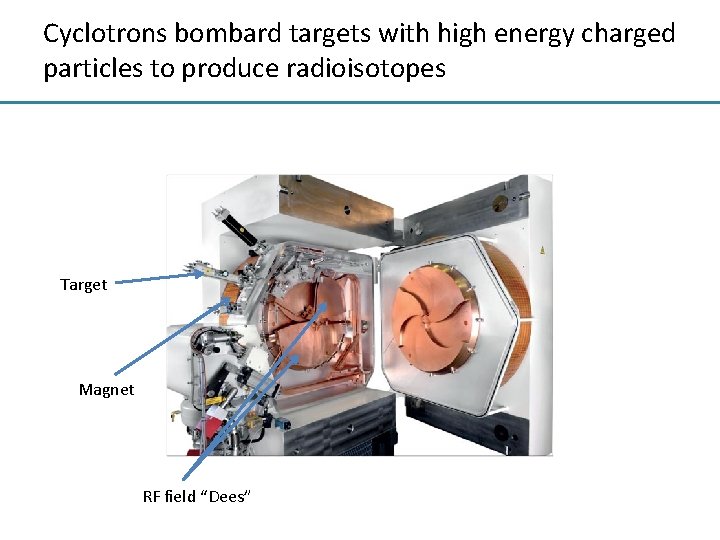
Cyclotrons bombard targets with high energy charged particles to produce radioisotopes Target Magnet RF field “Dees”
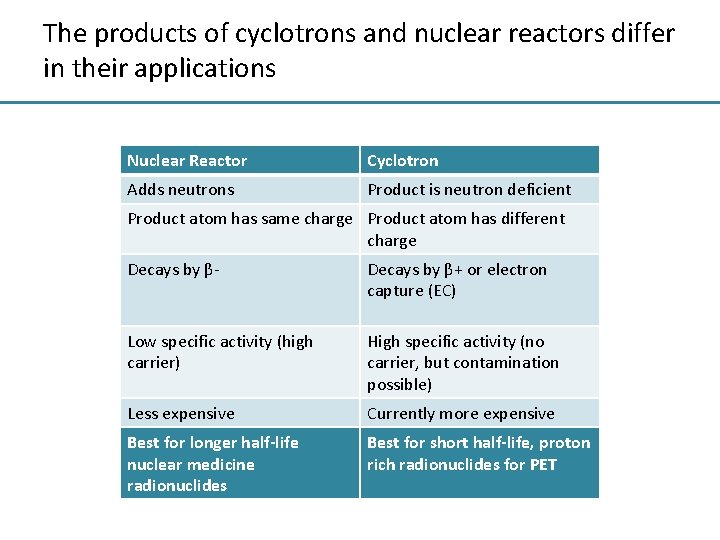
The products of cyclotrons and nuclear reactors differ in their applications Nuclear Reactor Cyclotron Adds neutrons Product is neutron deficient Product atom has same charge Product atom has different charge Decays by β- Decays by β+ or electron capture (EC) Low specific activity (high carrier) High specific activity (no carrier, but contamination possible) Less expensive Currently more expensive Best for longer half-life nuclear medicine radionuclides Best for short half-life, proton rich radionuclides for PET
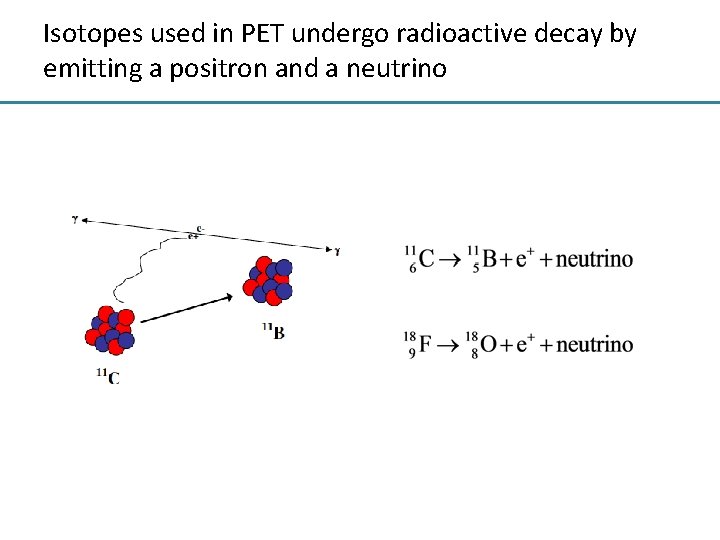
Isotopes used in PET undergo radioactive decay by emitting a positron and a neutrino
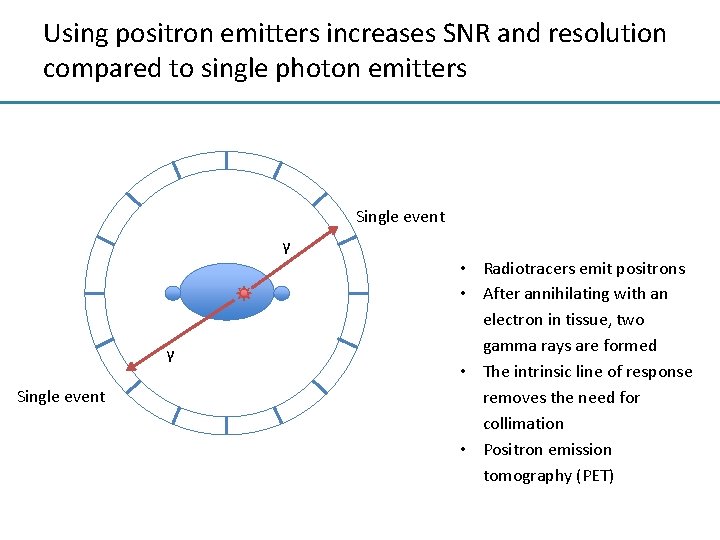
Using positron emitters increases SNR and resolution compared to single photon emitters Single event γ γ Single event • Radiotracers emit positrons • After annihilating with an electron in tissue, two gamma rays are formed • The intrinsic line of response removes the need for collimation • Positron emission tomography (PET)
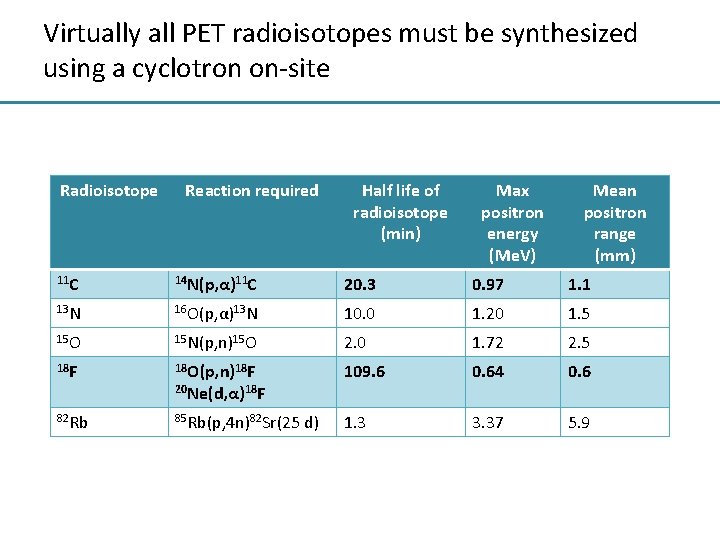
Virtually all PET radioisotopes must be synthesized using a cyclotron on-site Radioisotope Reaction required Half life of radioisotope (min) Max positron energy (Me. V) Mean positron range (mm) 11 C 14 N(p, α)11 C 20. 3 0. 97 1. 1 13 N 16 O(p, α)13 N 10. 0 1. 20 1. 5 15 O 15 N(p, n)15 O 2. 0 1. 72 2. 5 18 F 18 O(p, n)18 F 109. 6 0. 64 0. 6 82 Rb 85 Rb(p, 4 n)82 Sr(25 1. 3 3. 37 5. 9 20 Ne(d, α)18 F d)
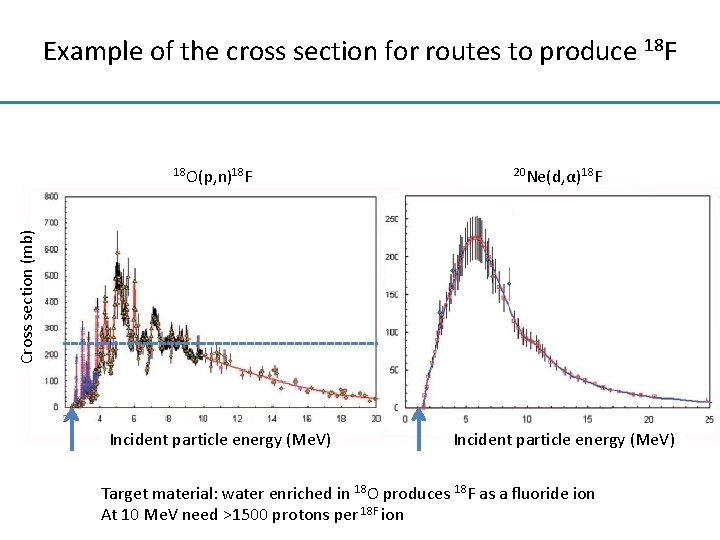
Example of the cross section for routes to produce 18 F 20 Ne(d, α)18 F Cross section (mb) 18 O(p, n)18 F Incident particle energy (Me. V) Target material: water enriched in 18 O produces 18 F as a fluoride ion At 10 Me. V need >1500 protons per 18 F ion
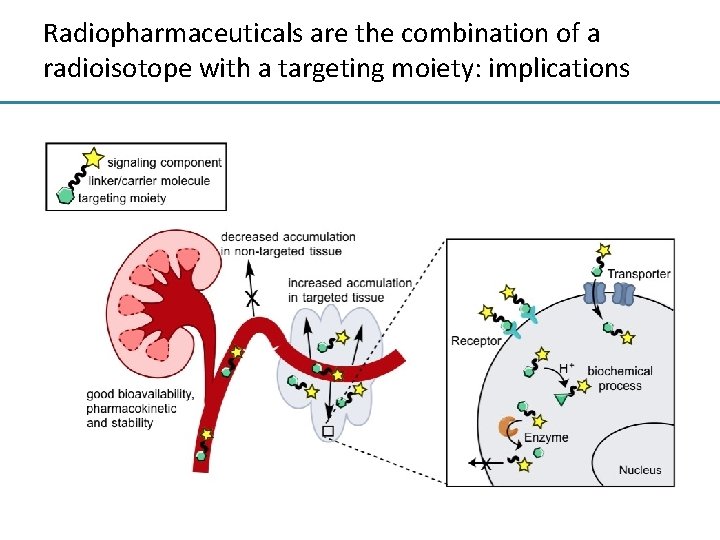
Radiopharmaceuticals are the combination of a radioisotope with a targeting moiety: implications

Strict criteria define an ideal radiopharmaceutical • Optimal half life – Short enough to produce significant radioactivity without requiring a large injected dose – Long enough to survive post injection delay for background (blood, organ) clearance • Decays into a non-radioactive (or long half life) daughter product • Energy of gamma rays from single photon emitters 100 – 300 ke. V – <100 ke. V photons attenuated – >300 ke. V harder to collimate and detect • Decays via emission of a mono-energetic gamma ray without alpha or beta particles • Energy of beta particles from positron emitters < 1 Me. V – Minimise tissue damage

Practical considerations are also important • Chemistry for radiolabeling should be versatile and not alter the biological behaviour of the target • The resulting radiopharmaceutical should have: – High uptake and residence time in the organ of interest – Low non-specific uptake – No adverse reactions / toxicity to the patient • The specific activity should be high • Very short half life materials create problems of availability • Low cost desirable for widespread use

Clean production of radiopharmaceuticals requires a radiopharmacy: Example for 99 m. Tc 1. Injections prepared each morning 2. Radiopharmacy performed in a “clean room” cabinet 1. Positive air pressure 2. Interlocked room 3. Sterile environment 3. Generator delivered once per week 4. Extracted 99 m. Tc added to pharmaceutical vial and mixed 5. Radioactive dose assessed in MBq 6. Syringe prepared and placed in lead shield and carrier box http: //www. nuclearmedicine. ie/Radiopharmacy. html

Nuclear medicine radiopharmaceuticals are used in a range of clinical applications Radiopharmaceutical Clinical application 99 m. Tc Thyroid function, cardiac ejection fraction Pertechnetate 99 m. Tc Dimercaptosuccinic acid (DMSA) Kidney function 99 m. Tc MDP / HDP Bone function 99 m. Tc MAG 3 / DTPA Bladder filling 123 I Ioflupane Parkinson’s Disease
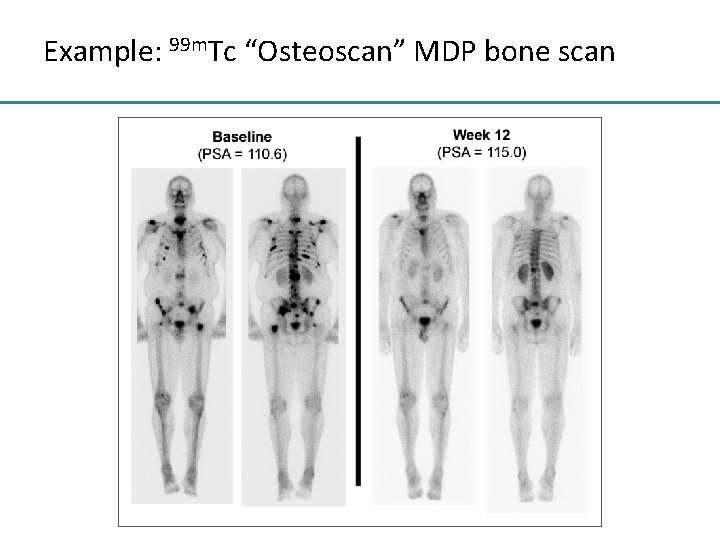
Example: 99 m. Tc “Osteoscan” MDP bone scan
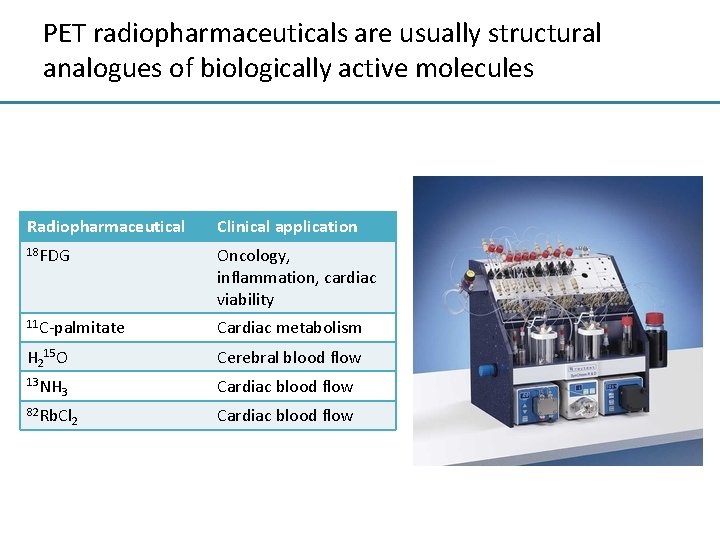
PET radiopharmaceuticals are usually structural analogues of biologically active molecules Radiopharmaceutical Clinical application 18 FDG Oncology, inflammation, cardiac viability 11 C-palmitate Cardiac metabolism H 215 O Cerebral blood flow 13 NH Cardiac blood flow 3 82 Rb. Cl 2 Cardiac blood flow
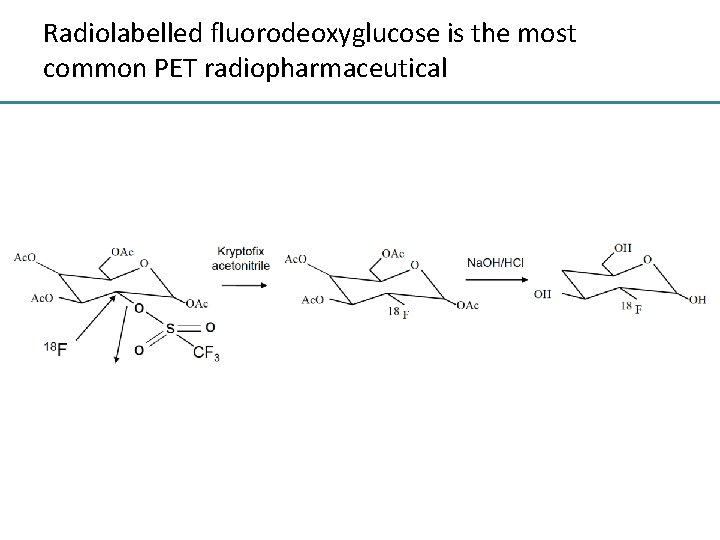
Radiolabelled fluorodeoxyglucose is the most common PET radiopharmaceutical
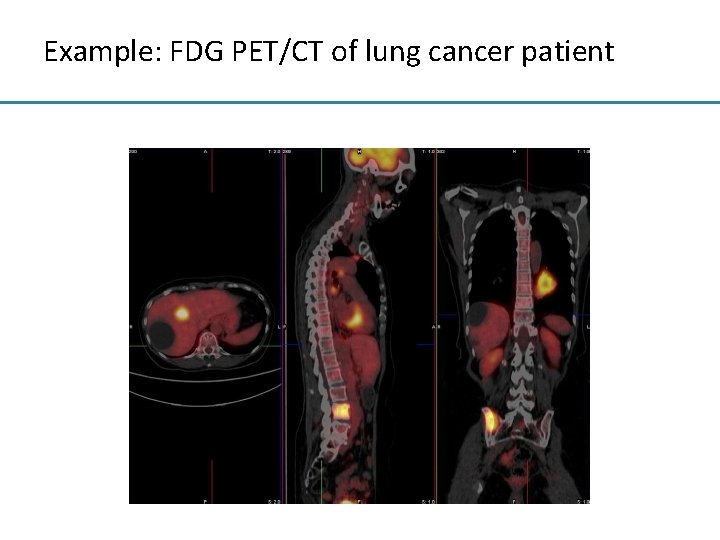
Example: FDG PET/CT of lung cancer patient

Summary 2: Radiopharmaceuticals • A radiopharmaceutical uses a radioactive isotope to label a substrate of biological interest – It must be prepared in a clean radiopharmacy • Radioisotopes can be produced in: – Nuclear reactors (long half life single photon emitters) – Cyclotrons (short half life positron emitters) • The Bateman equations describe the balance of decays of parent and daughter nuclei, leading to transient equilibrium • Ideal radioisotopes produce very long half life daughters without other emissions (e. g. alpha/beta)
How to generate X-rays How to generate gamma rays using radiopharmaceuticals
- Slides: 48