Lecture 8 Chemical Reaction Engineering CRE is the




























- Slides: 40

Lecture 8 Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place.

Today’s lecture �Block 1: Mole Balances on PFRs and PBR �Must Use the Differential Form �Block 2: Rate Laws �Block 3: Stoichiometry �Pressure Drop: 2 Liquid Phase Reactions: Pressure Drop does not affect the concentrations in liquid phase rx. Gas Phase Reactions: Epsilon not Equal to Zero d(P)/d(W)=. . Polymath will combine with d(X)/f(W)=. . for you Epsilon = 0 and Isothermal P=f(W) Combine then Separate Variables (X, W) and Integrate

Reactor Mole Balances in terms of conversion Reactor Differential Algebraic Integral X Batch t CSTR PFR X PBR 3 W

Concentration Flow System: Gas Phase Flow System: 4

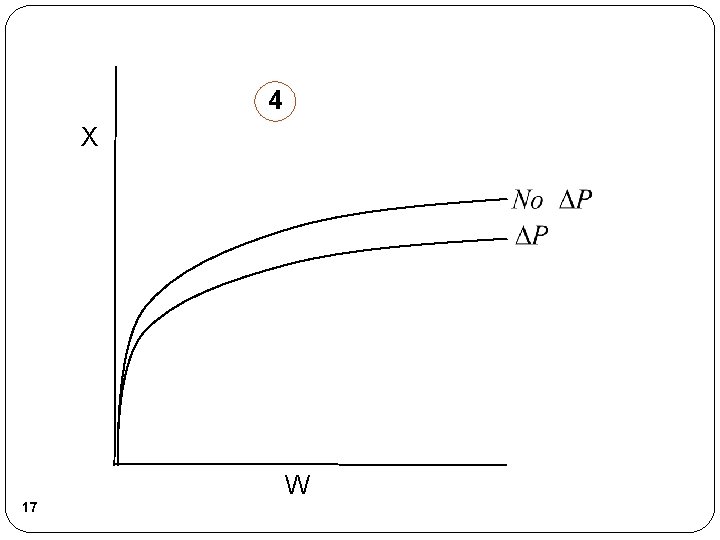
Pressure Drop in Packed Bed Reactors Note: Pressure drop does NOT affect liquid phase reactions Sample Question: Analyze the following second order gas phase reaction that occurs isothermally in a PBR: A B Mole Balance: Must use the differential form of the mole balance to separate variables: 5 Rate Law: Second order in A and

Pressure Drop in Packed Bed Reactors Stoichiometry: Isothermal, T=T 0 Combine: 6 Need to find (P/P 0) as a function of W (or V if you have a PFR)

Pressure Drop in Packed Bed Reactors Ergun Equation: Constant mass flow: 7

Pressure Drop in Packed Bed Reactors Variable Density Let 8

Pressure Drop in Packed Bed Reactors Catalyst Weight Where Let 9

Pressure Drop in Packed Bed Reactors We will use this form for single reactions: 10 Isothermal case

Pressure Drop in Packed Bed Reactors an or d The two expressions are coupled ordinary differential equations. We can only solve them simultaneously using an ODE solver such as Polymath. For the special case of isothermal operation and epsilon = 0, we can obtain an analytical solution. 11 Polymath will combine the mole balance, rate law

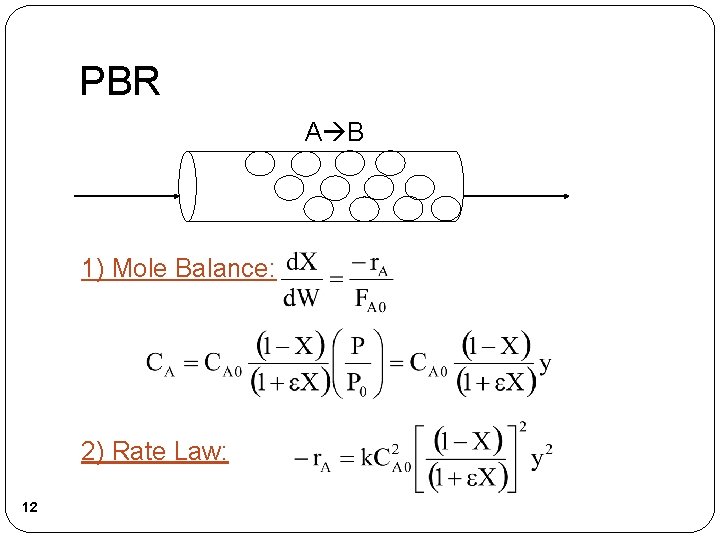
PBR A B 1) Mole Balance: 2) Rate Law: 12

PBR 13

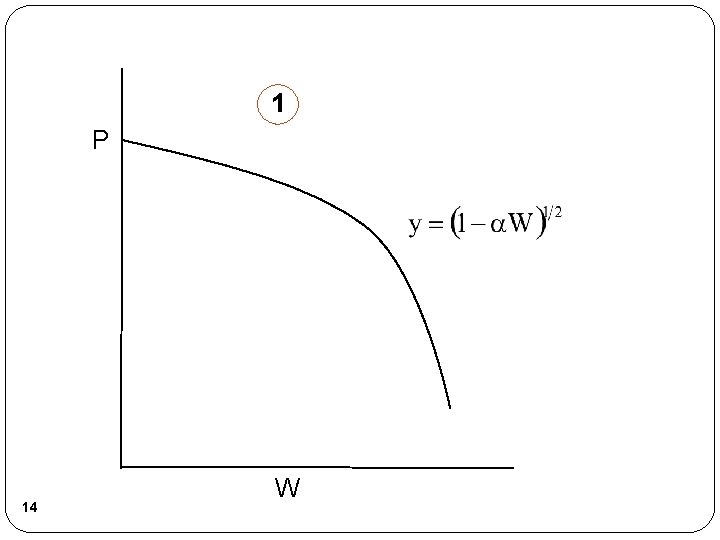
1 P 14 W

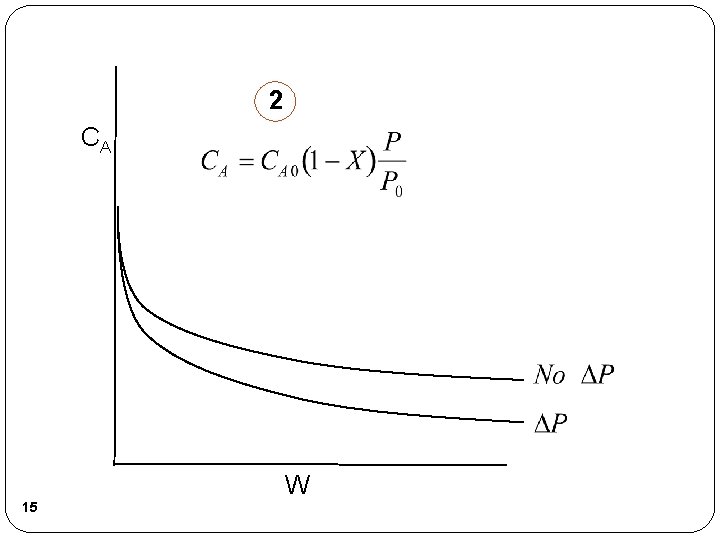
2 CA W 15

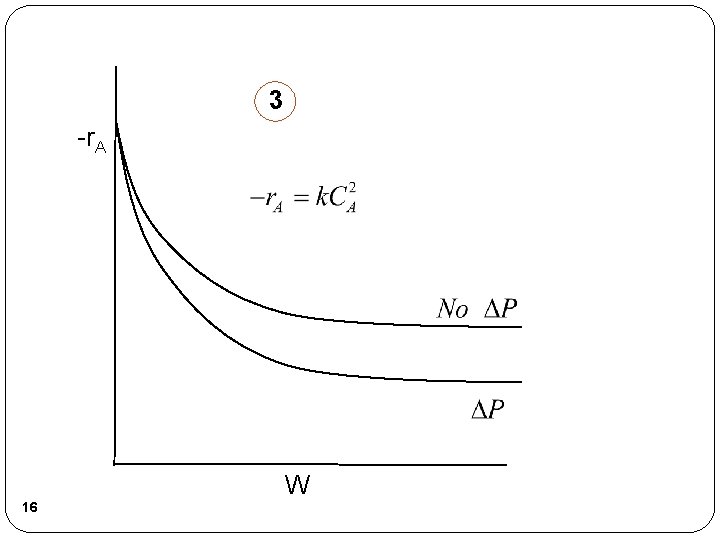
3 -r. A W 16

4 X W 17

18

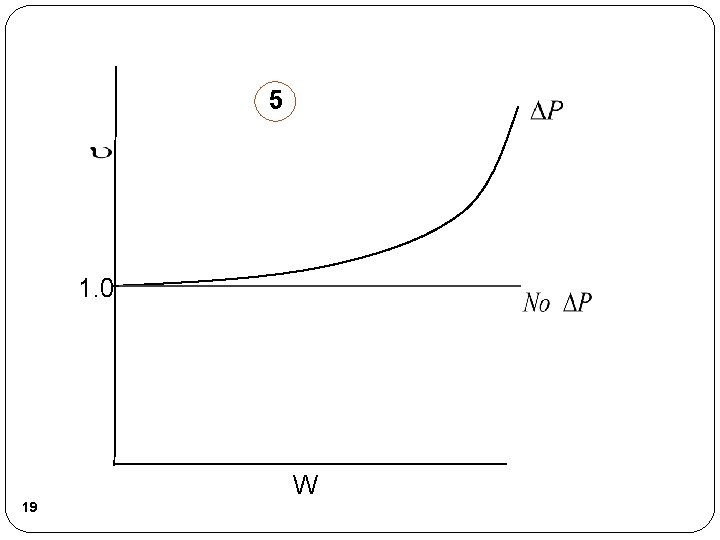
5 1. 0 W 19

Example 1: Gas Phase Reaction in PBR for δ = 0 Gas Phase Reaction in PBR with δ = 0 (Polymath Solution) A + B 2 C Repeat the previous one with equil molar feed of A and B and k. A = 1. 5 dm 9/mol 2/kg/min α = 0. 0099 kg-1 Find X at 100 kg 20

Example 1: Gas Phase Reaction in PBR for δ = 0 A + B 2 C Case 1: Case 2: 21

Example 1: Gas Phase Reaction in PBR for δ = 0 1) Mole Balance: 2) Rate Law: 3) 4) 22

Example 1: Gas Phase Reaction in PBR for δ = 0 5) 23

Example 1: Gas Phase Reaction in PBR for δ = 0 24

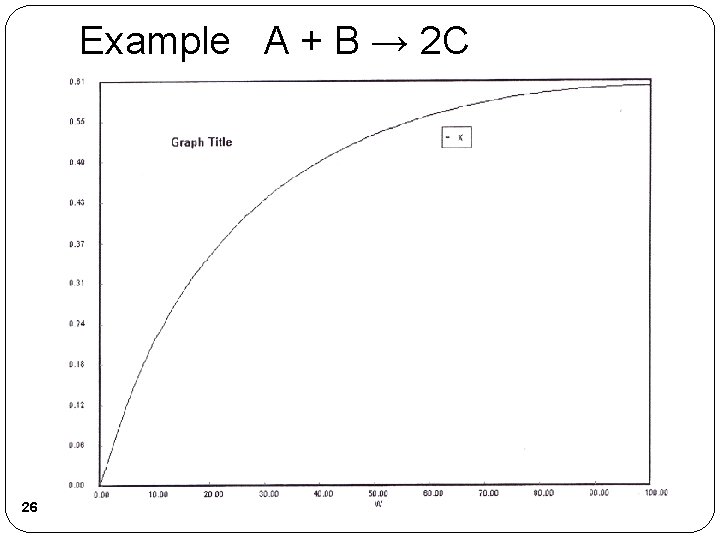
Example A + B → 2 C 25

Example A + B → 2 C 26

Example 2: Gas Phase Reaction in PBR for δ ≠ 0 Polymath Solution A + 2 B C is carried out in a packed bed reactor in which there is pressure drop. The fed is stoichiometric in A and B. Plot the conversion and pressure ratio y = P/P 0 as a function of catalyst weight upto 100 kg. Additional Information k. A = 6 dm 9/mol 2/kg/min α = 0. 02 kg-1 27

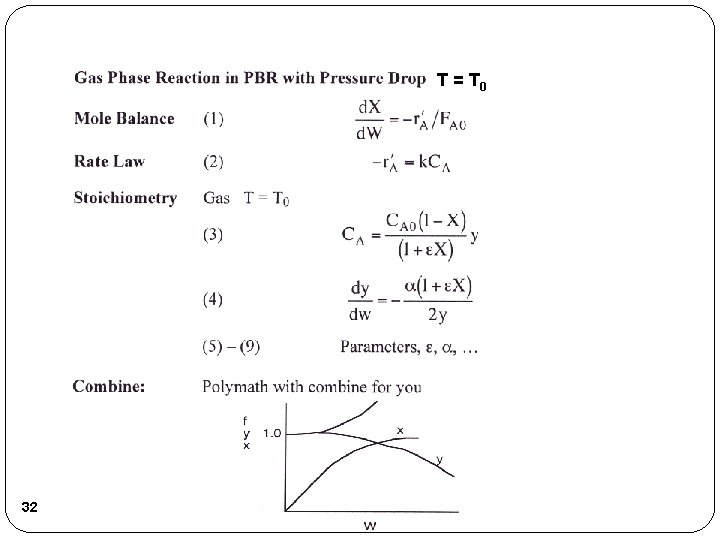
Example 2: Gas Phase Reaction in PBR for δ ≠ 0 A + 2 B C 1) Mole Balance: 2) Rate Law: 3) Stoichiometry: Gas, Isothermal 28

Example 2: Gas Phase Reaction in PBR for δ ≠ 0 4) 5) 6) 7) Initial values: W=0, X=0, y=1 W=100 Combine with Polymath. 29 If δ≠ 0, polymath must be used to solve.

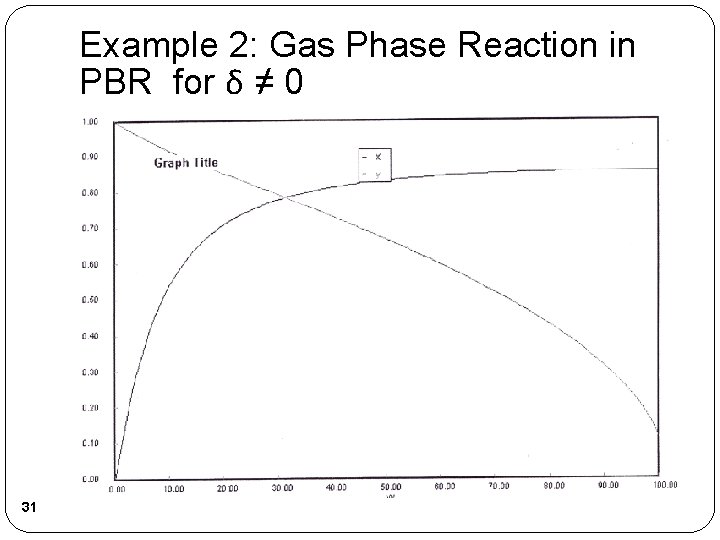
Example 2: Gas Phase Reaction in PBR for δ ≠ 0 30

Example 2: Gas Phase Reaction in PBR for δ ≠ 0 31

T = T 0 32

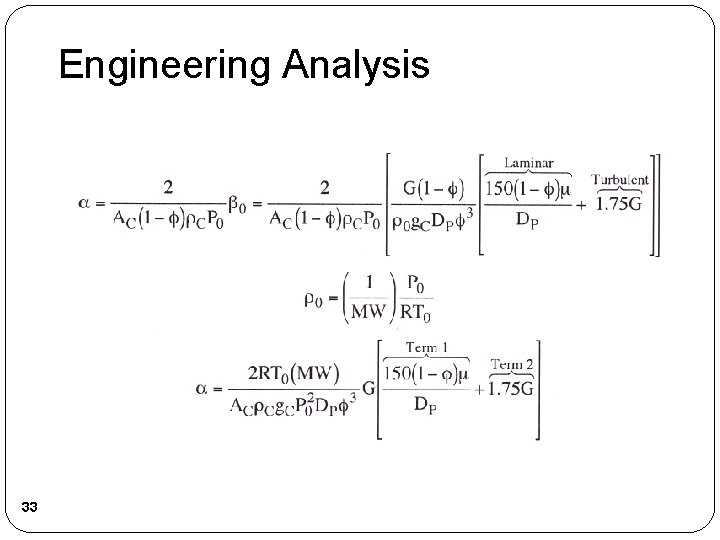
Engineering Analysis 33

Engineering Analysis 34

Engineering Analysis 35

Pressure Change – Molar Flow Rate Use for heat effects, multiple rxns 36 Isothermal: T = T 0

Heat Effects Isothermal Design Stoichiometry Rate Laws Mole Balance 37

Gas Phase Reaction with Pressure Drop Concentration Flow System: Gas Phase Flow System: 38

Example 2: Gas Phase Reaction in PBR for δ ≠ 0 A + 2 B C 1) Mole Balance: 2) Rate Law: 3) Stoichiometry: Gas, Isothermal 39

End of Lecture 8 40