Lecture 8 222 Cell Biology 1 Mitochondria Function

Lecture 8 222 Cell Biology 1

Mitochondria Function n n The most prominent roles of mitochondria are to produce ATP (i. e. , phosphorylation of ADP) through respiration And to regulate cellular metabolism. The central set of reactions involved in ATP production are collectively known as the citric acid cycle, or the Krebs Cycle. However, the mitochondrion has many other functions in addition to the production of ATP. 222 Cell Biology 2

Energy conversion n n A dominant role for the mitochondria is the production of ATP, as reflected by the large number of proteins in the inner membrane for this task. This is done by oxidizing the major products of glucose, pyruvate, and NADH, which are produced in the cytosol. This process of cellular respiration, also known as aerobic respiration, is dependent on the presence of oxygen. When oxygen is limited, the glycolytic products will be metabolized by anaerobic respiration, a process that is independent of the mitochondria. The production of ATP from glucose has an approximately 13 -fold higher yield during aerobic respiration compared to anaerobic respiration 222 Cell Biology 3

Cellular respiration n n n Cellular respiration, also known as 'oxidative metabolism', is one of the key ways a cell gains useful energy. It is the set of the metabolic reactions and processes that take place in organisms' cells To convert biochemical energy from nutrients into adenosine triphosphate (ATP), and then release waste products. The reactions involved in respiration are catabolic reactions that involve the oxidation of one molecule and the reduction of another. Nutrients commonly used by animal and plant cells in respiration include glucose, amino acids and fatty acids, and a common oxidizing agent (electron acceptor) is molecular oxygen (O 2 Organisms that use oxygen as a final electron acceptor in respiration are described as aerobic, while those that do not are referred to as anaerobic 222 Cell Biology 4
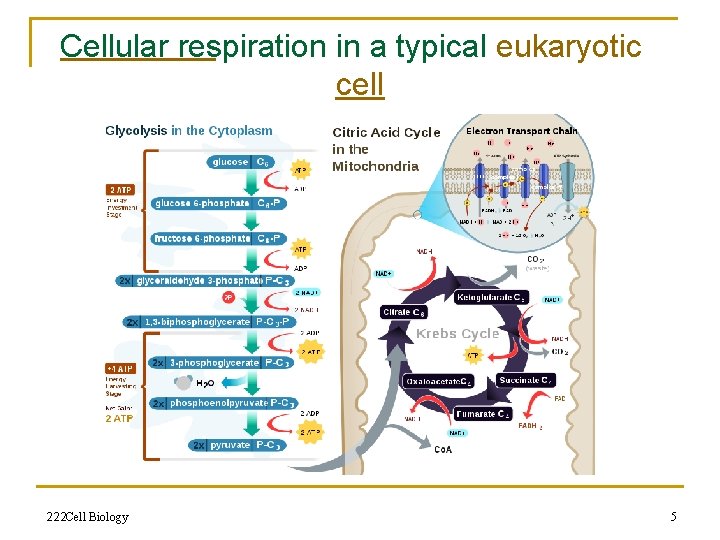
Cellular respiration in a typical eukaryotic cell 222 Cell Biology 5

Aerobic respiration n n Aerobic respiration requires oxygen in order to generate energy (ATP). It is the preferred method of pyruvate breakdown from glycolysis and requires that pyruvate enter the mitochondrion in order to be fully oxidized by the Krebs cycle. The product of this process is energy in the form of ATP (Adenosine Triphosphate), by substrate-level phosphorylation, NADH and FADH 2. Simplified reaction: C 6 H 12 O 6 (aq) + 6 O 2 (g) → 6 CO 2 (g) + 6 H 2 O 222 Cell Biology 6

Anaerobic respiration n In biology, anaerobic respiration is a way for an organism to produce usable energy without the involvement of oxygen; it is respiration without oxygen. Respiration is a redox reaction that processes energy in a form usable by an organism, chiefly the process of producing ATP. It employs an electron transport chain, with inorganic molecules other than oxygen used as a final electron acceptor. 222 Cell Biology 7

Pyruvate: the citric acid cycle n n Each pyruvate molecule produced by glycolysis is actively transported across the inner mitochondrial membrane, and into the matrix where it is oxidized and combined with coenzyme A to form CO 2, acetyl-Co. A, and NADH The acetyl-Co. A is the primary substrate to enter the citric acid cycle, also known as the tricarboxylic acid (TCA) cycle or Krebs cycle. The enzymes of the citric acid cycle are located in the mitochondrial matrix, with the exception of succinate dehydrogenase, which is bound to the inner mitochondrial membrane as part of Complex II. The citric acid cycle oxidizes the acetyl-Co. A to carbon dioxide, and, in the process, produces reduced cofactors (three molecules of NADH and one molecule of FADH 2) that are a source of electrons for the electron transport chain, and a molecule of GTP (that is readily converted to an ATP 222 Cell Biology 8

Electron transport chain n An electron transport chain couples a chemical reaction between an electron donor (such as NADH) and an electron acceptor (such as O 2) to the transfer of H+ ions across a membrane, through a set of mediating biochemical reactions. These H+ ions are used to produce adenosine triphosphate (ATP), the main energy intermediate in living organisms. Electron transport chains are used for extracting energy from sunlight (photosynthesis) and from redox reactions such as the oxidation of sugars (respiration). 222 Cell Biology 9
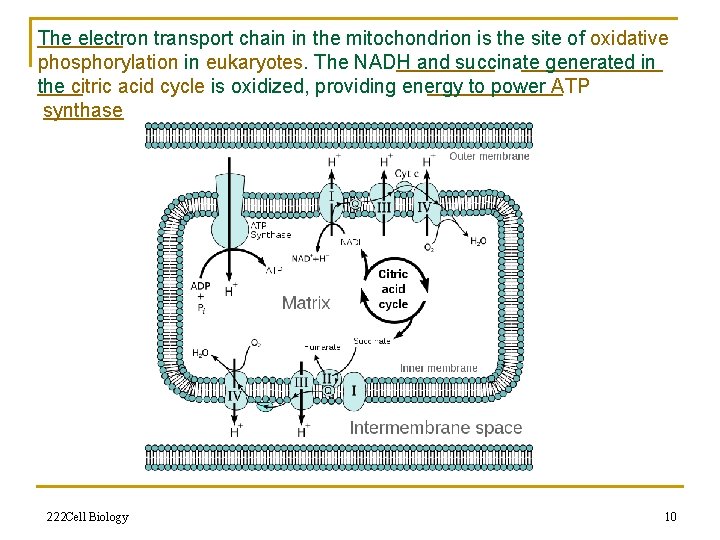
The electron transport chain in the mitochondrion is the site of oxidative phosphorylation in eukaryotes. The NADH and succinate generated in the citric acid cycle is oxidized, providing energy to power ATP synthase 222 Cell Biology 10

Mitochondrial DNA n n Mitochondrial DNA is localized to the matrix, which also contains a host of enzymes, as well as ribosomes for protein synthesis. Many of the critical metabolic steps of cellular respiration are catalyzed by enzymes that are able to diffuse through the mitochondrial matrix. The other proteins involved in respiration, including the enzyme that generates ATP, are embedded within the mitochondrial inner membrane. Infolding of the cristae dramatically increases the surface area available for hosting the enzymes responsible for cellular respiration. 222 Cell Biology 11

Heat production n n Under certain conditions, protons can re-enter the mitochondrial matrix without contributing to ATP synthesis. This process is known as proton leak or mitochondrial uncoupling and is due to the facilitated diffusion of protons into the matrix. The process results in the unharnessed potential energy of the proton electrochemical gradient being released as heat. The process is mediated by a proton channel called thermogenin, or UCP 1. 222 Cell Biology 12

Heat production n Thermogenin is primarily found in brown adipose tissue, or brown fat, and is responsible for non-shivering thermogenesis Brown adipose tissue is found in mammals, and is at its highest levels in early life and in hibernating animals. In humans, brown adipose tissue is present at birth and decreases with age. 222 Cell Biology 13

Storage of calcium ions n n n The concentrations of free calcium in the cell can regulate an array of reactions and is important for signal transduction in the cell. Mitochondria can store calcium, a contributing process for the cell's homeostasis of calcium. Their ability to rapidly take in calcium for later release makes them very good The endoplasmic reticulum (ER) is the most significant storage site of calcium There is a significant interplay between the mitochondrion and ER with regard to calcium 222 Cell Biology 14

Storage of calcium ions n n n The calcium is taken up into the matrix by a calcium uniporter on the inner mitochondrial membrane. It is primarily driven by the mitochondrial membrane potential. Release of this calcium back into the cell's interior can occur via a sodium-calcium exchange protein or via "calcium-induced-calciumrelease" pathways. This can initiate calcium spikes or calcium waves with large changes in the membrane potential. These can activate a series of second messenger system proteins that can coordinate processes such as neurotransmitter release in nerve cells and release of hormones in endocrine cells. 222 Cell Biology 15

Additional functions n Mitochondria play a central role in many other metabolic tasks, such as: Regulation of the membrane potential Apoptosis-programmed cell death Calcium signaling (including calcium-evoked apoptosis) Cellular proliferation regulation Regulation of cellular metabolism Certain heme synthesis reactions n Steroid synthesis. n n n 222 Cell Biology 16
- Slides: 16