Lecture 7 Web pollev comucibio Text To 37607

Lecture 7 Web: pollev. com/ucibio Text: To: 37607 Type in: 169964 <your question>

The importance of structure “Chaperones” & protein folding Misfolded proteins are very, very bad! E. g. Huntington’s disease E. g. Prions
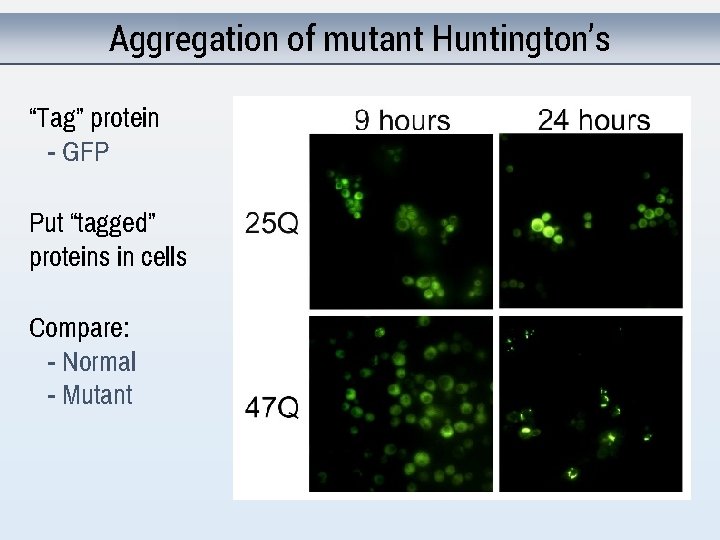
Aggregation of mutant Huntington’s “Tag” protein - GFP Put “tagged” proteins in cells Compare: - Normal - Mutant

Prions are WHACK proteins! Normal vs Disease = Same gene (no mutations!) Put Prion version into normal cells Prion Normal = Diseased! Prion protein changes 3 D conformation

Working with proteins Interested in studying Hexokinase How many proteins in cell? First step in studying Hexokinase? How do you know when you have purified Hexokinase?

Estimating purity First step – how do we know our protein is present? How do we know how pure our protein is?

Estimating purity Assume 100 proteins in cell. 1 g of each protein. Total protein? Ratio of our protein? Purify: Get rid of 50 unwanted proteins. Total protein? Ratio of our protein? Purify: Get rid of 40 remaining unwanted proteins. Total protein? Ratio of our protein? Purify: Get rid of 8 remaining unwanted proteins. Total protein? Ratio of our protein? Ratio of activity of protein: Total protein = Specific activity
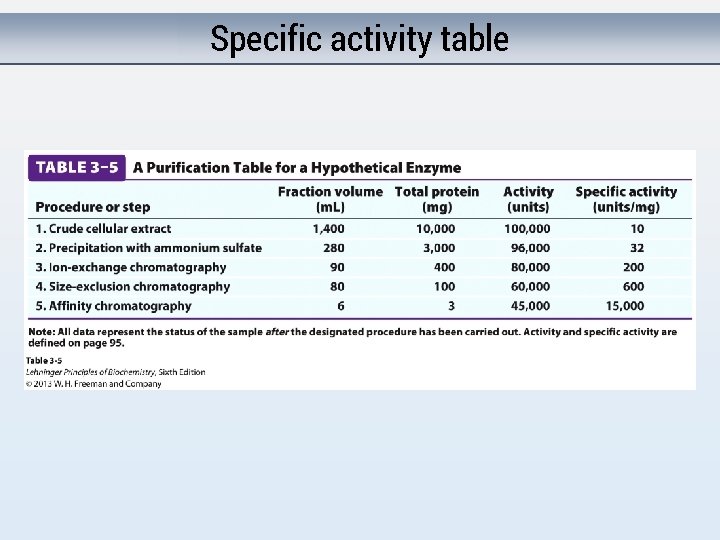
Specific activity table

Purifying proteins 1 st step = Getting protein! - Lyse - “Fractionate”
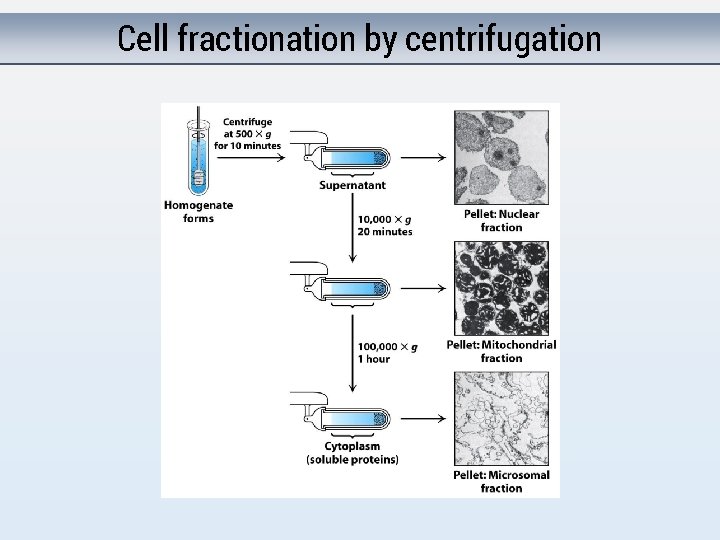
Cell fractionation by centrifugation

Purifying proteins What properties of proteins can you exploit?
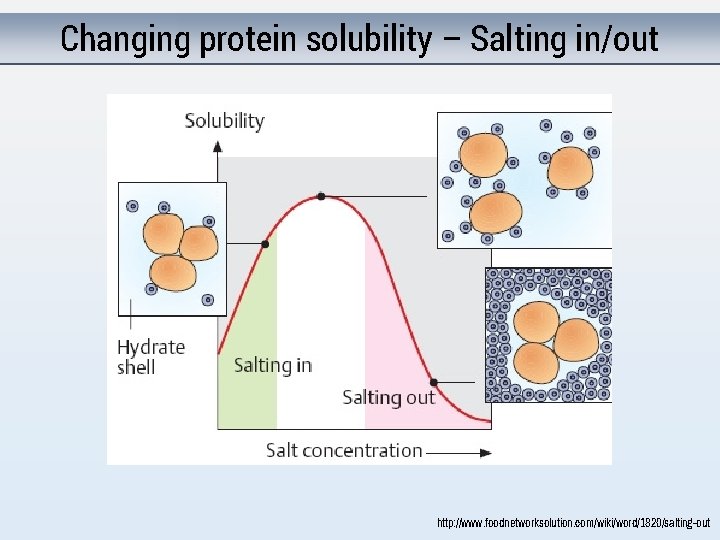
Changing protein solubility – Salting in/out http: //www. foodnetworksolution. com/wiki/word/1820/salting-out
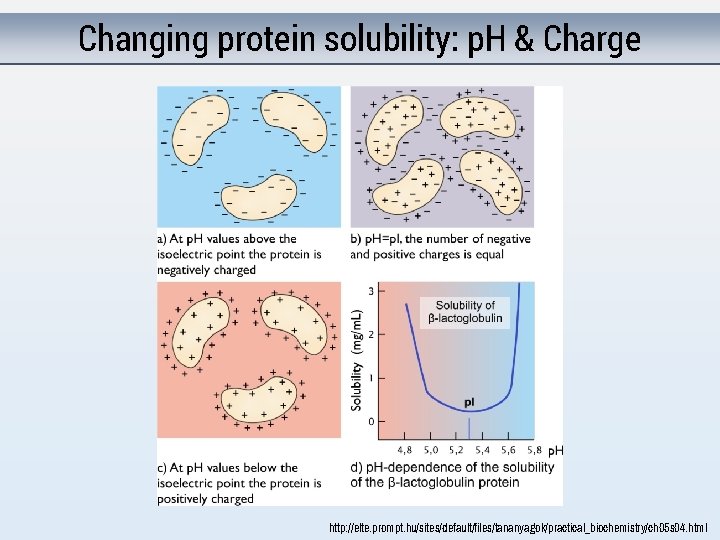
Changing protein solubility: p. H & Charge http: //elte. prompt. hu/sites/default/files/tananyagok/practical_biochemistry/ch 05 s 04. html
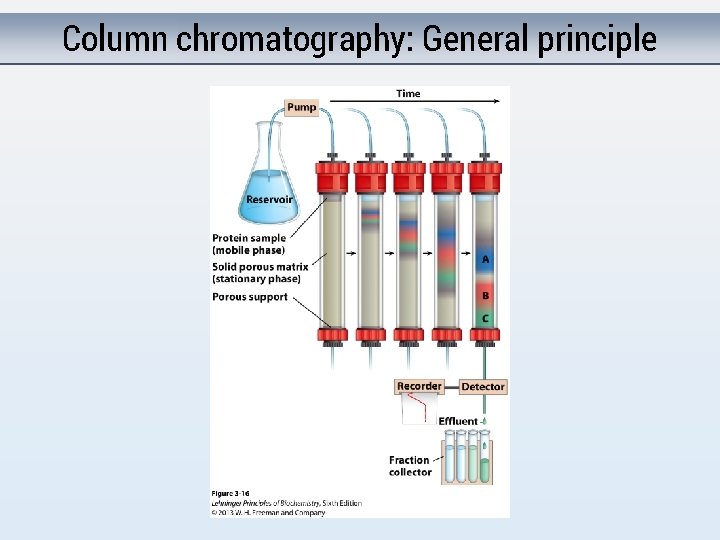
Column chromatography: General principle
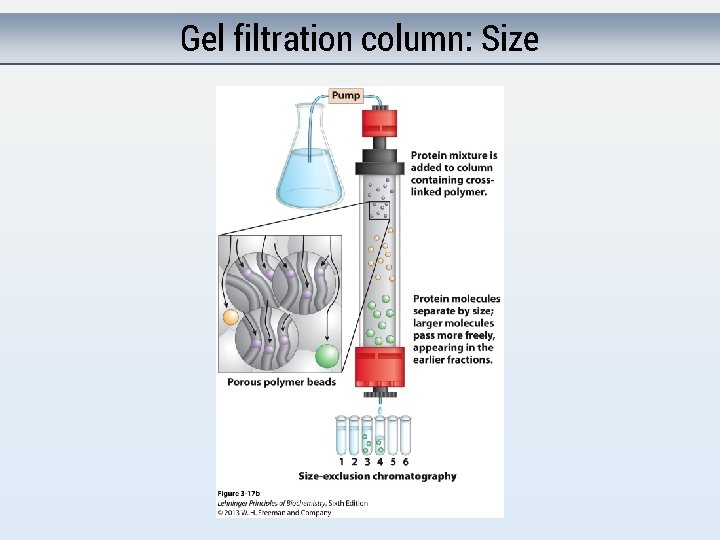
Gel filtration column: Size

Ion exchange column: Charge
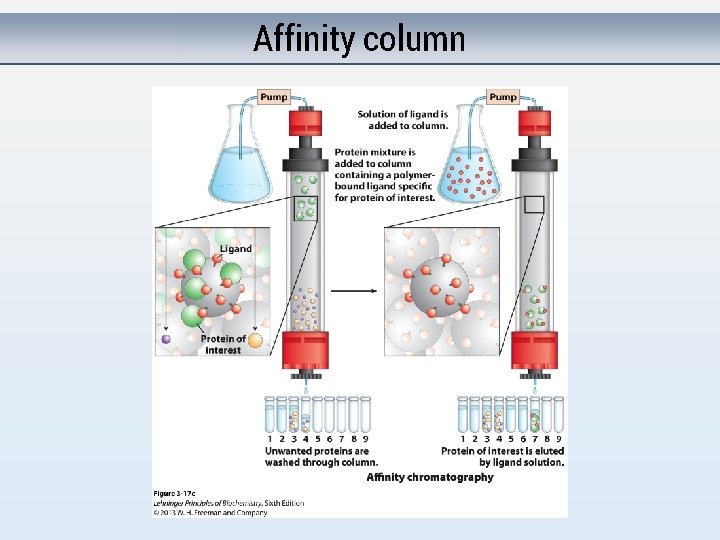
Affinity column
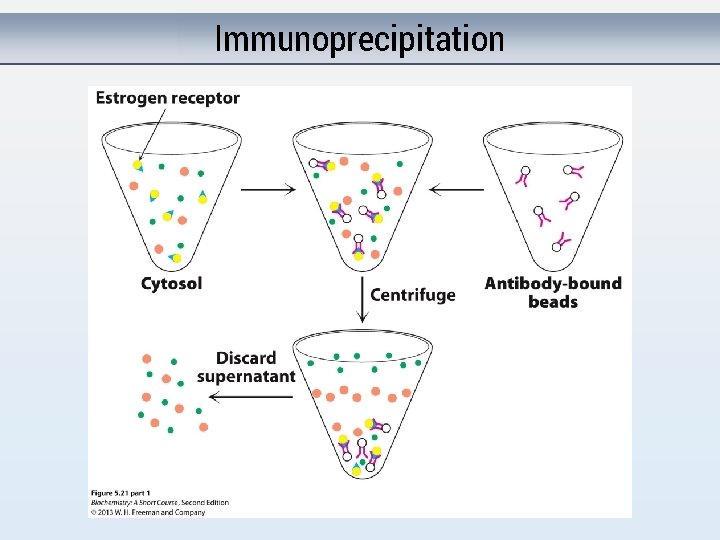
Immunoprecipitation
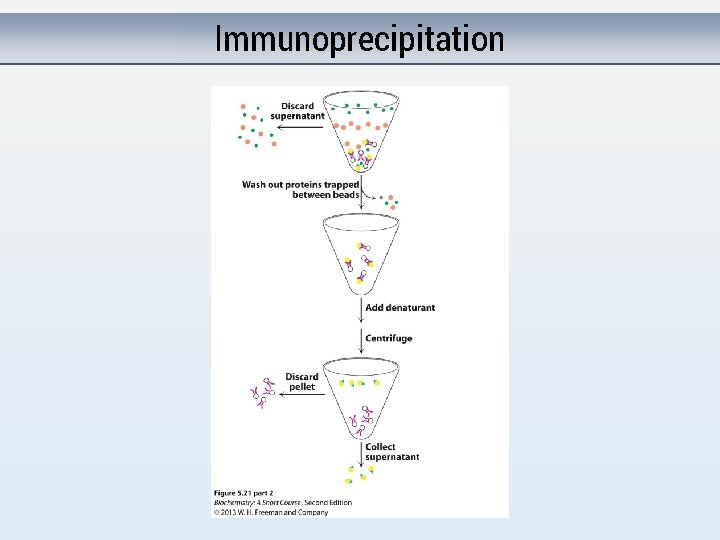
Immunoprecipitation
- Slides: 19