Lecture 7 January 16 2016 Biotech 3 Lecture

Lecture 7 January 16, 2016 Biotech 3

Lecture Topics 1. Review Blue-White Colony Screen 2. Transformations 3. Competent cells 4. Standard Operating Procedure Review 5. - Primer Design Part II Start codon In frame Stop codon Designing tags 6. - PCR Mathematical concept and cycle number Polymerase rate and fidelity Primer design for point mutations PCR Parameters: DNA sequence, primers, type of polymerase, Tm, extension time, number of cycles
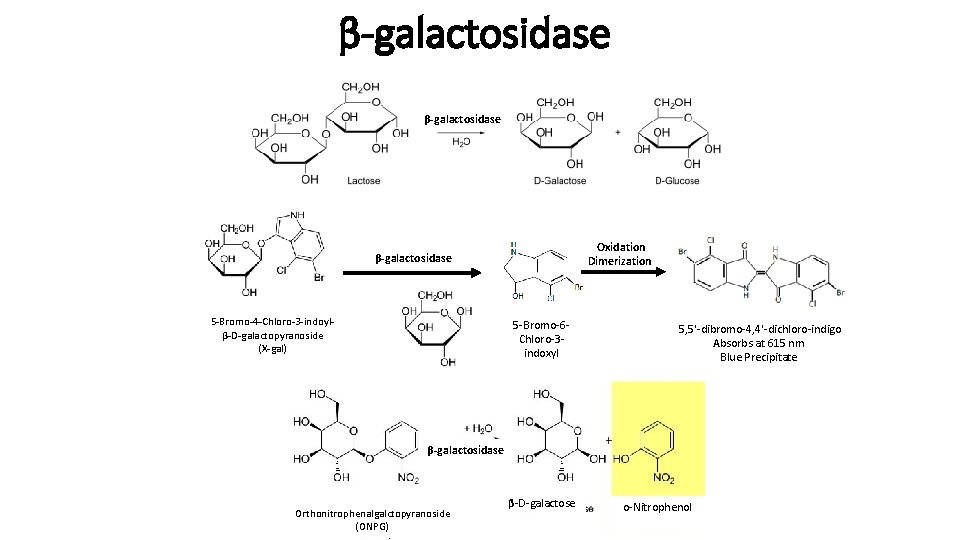
β-galactosidase Oxidation Dimerization β-galactosidase 5 -Bromo-4 -Chloro-3 -indoylβ-D-galactopyranoside (X-gal) 5 -Bromo-6 Chloro-3 indoxyl 5, 5'-dibromo-4, 4'-dichloro-indigo Absorbs at 615 nm Blue Precipitate β-galactosidase Orthonitrophenalgalctopyranoside (ONPG) β-D-galactose o-Nitrophenol
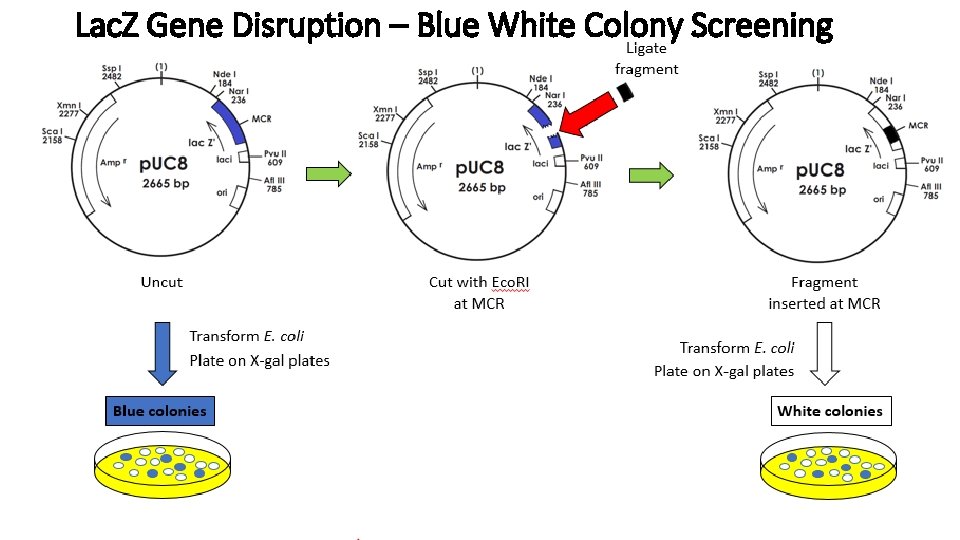
Lac. Z Gene Disruption – Blue White Colony Screening
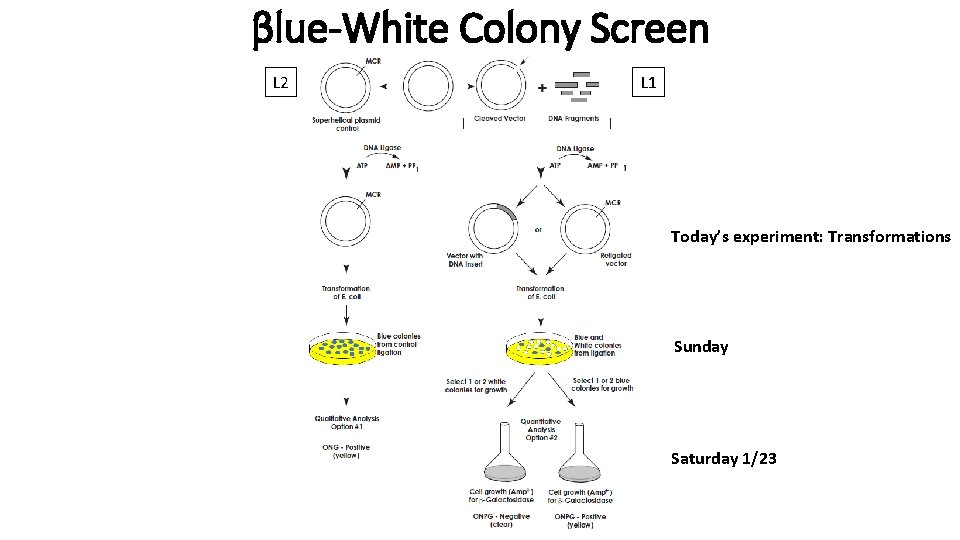
βlue-White Colony Screen L 2 L 1 Today’s experiment: Transformations Sunday Saturday 1/23
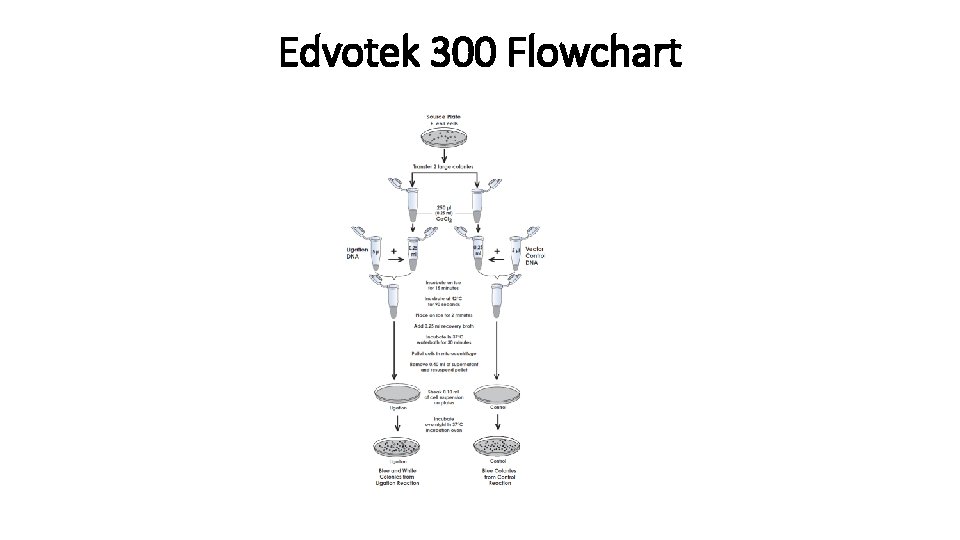
Edvotek 300 Flowchart
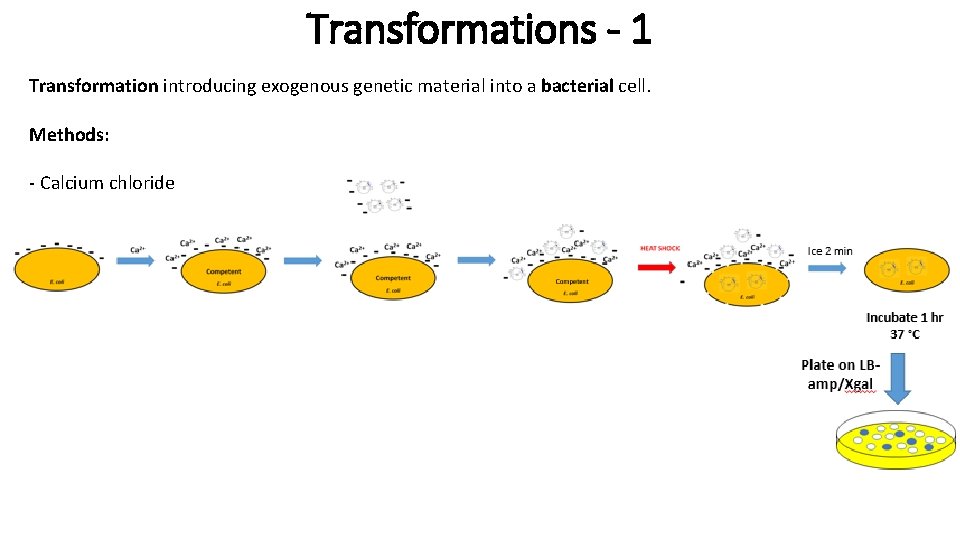
Transformations - 1 Transformation introducing exogenous genetic material into a bacterial cell. Methods: - Calcium chloride
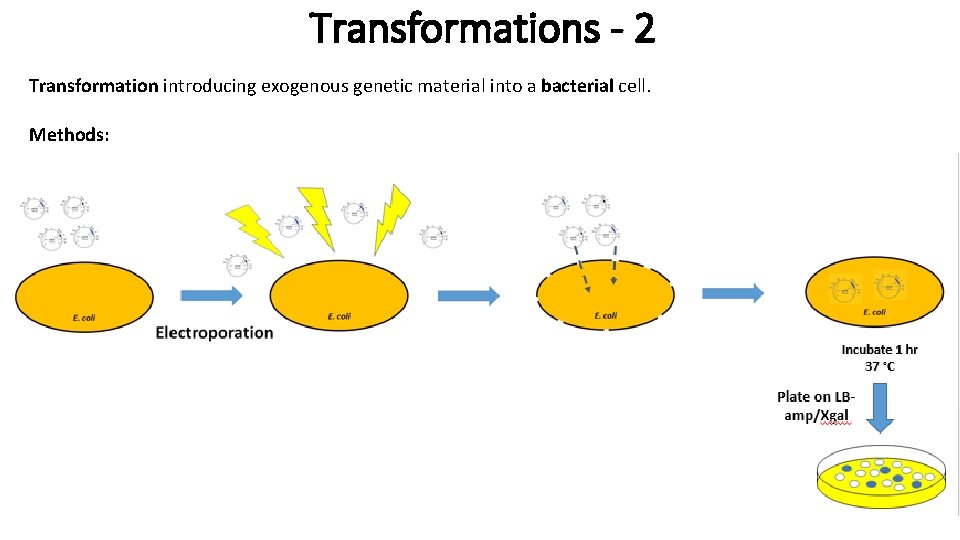
Transformations - 2 Transformation introducing exogenous genetic material into a bacterial cell. Methods: - Electroporation
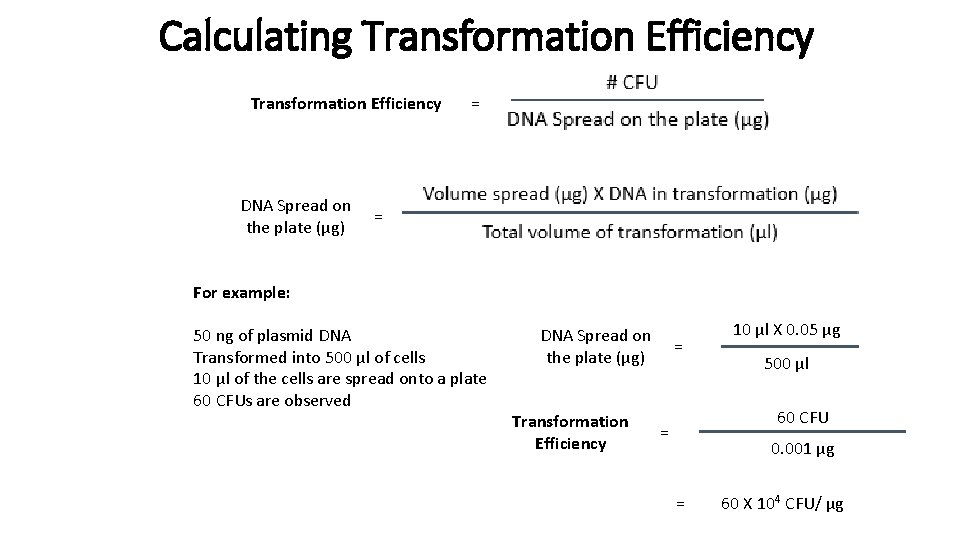
Calculating Transformation Efficiency DNA Spread on the plate (μg) = = For example: 50 ng of plasmid DNA Transformed into 500 μl of cells 10 μl of the cells are spread onto a plate 60 CFUs are observed DNA Spread on the plate (μg) Transformation Efficiency = 10 μl X 0. 05 μg 500 μl 60 CFU = 0. 001 μg = 60 X 104 CFU/ μg
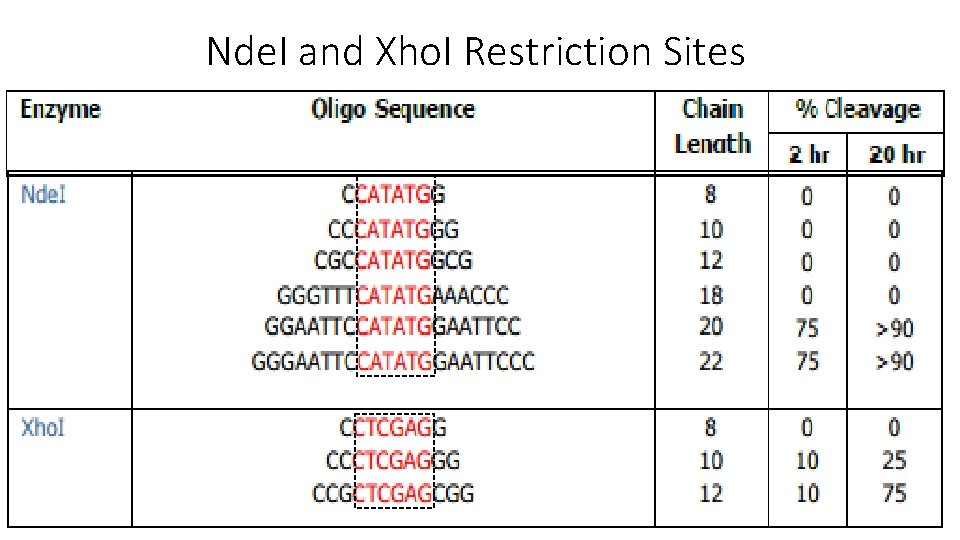
Nde. I and Xho. I Restriction Sites
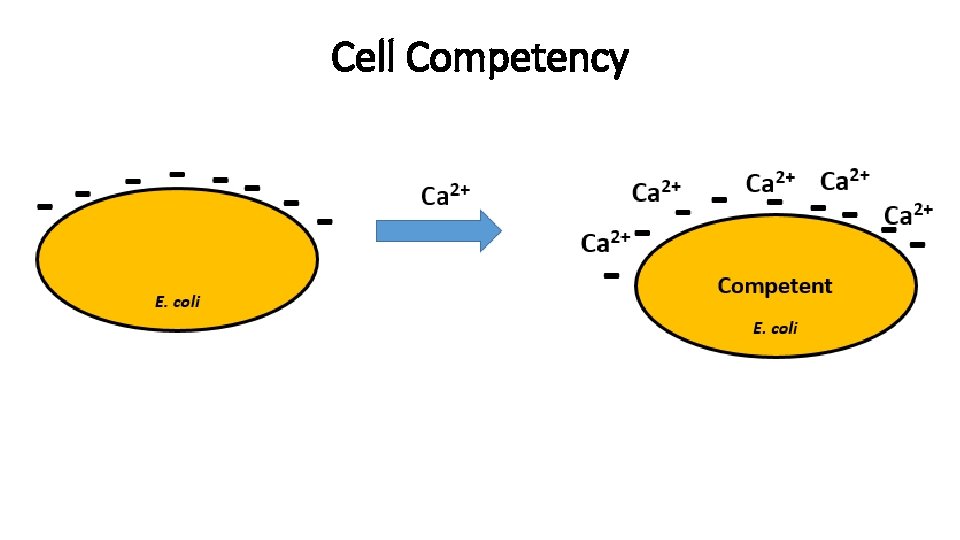
Cell Competency
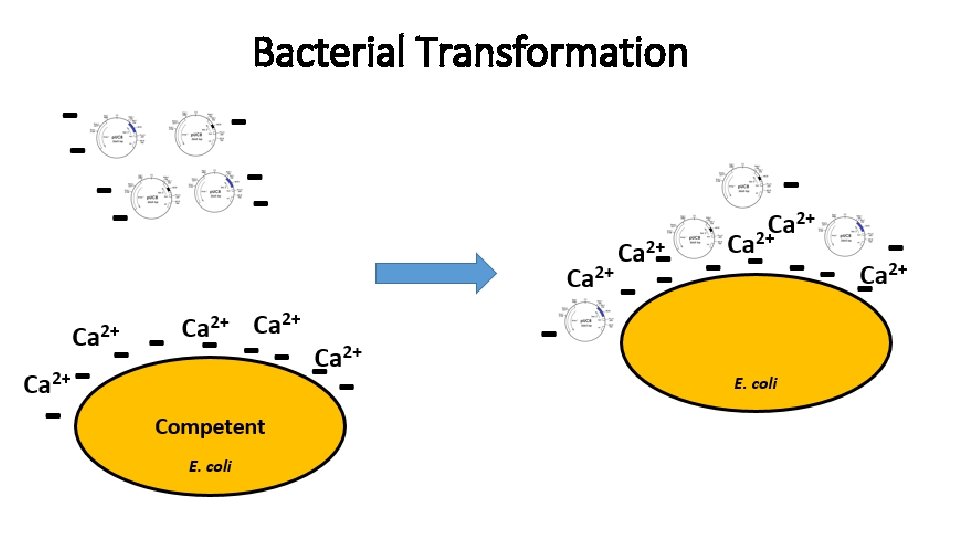
Bacterial Transformation
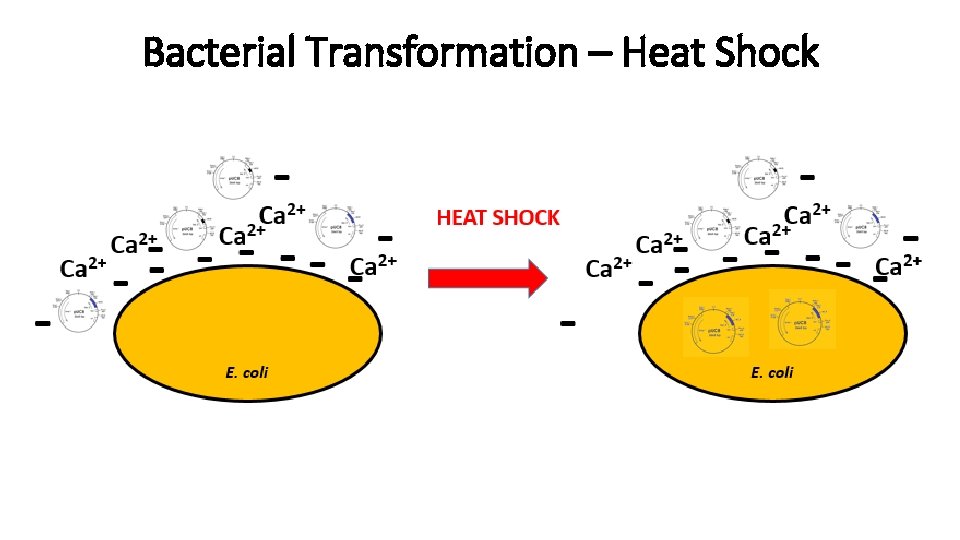
Bacterial Transformation – Heat Shock
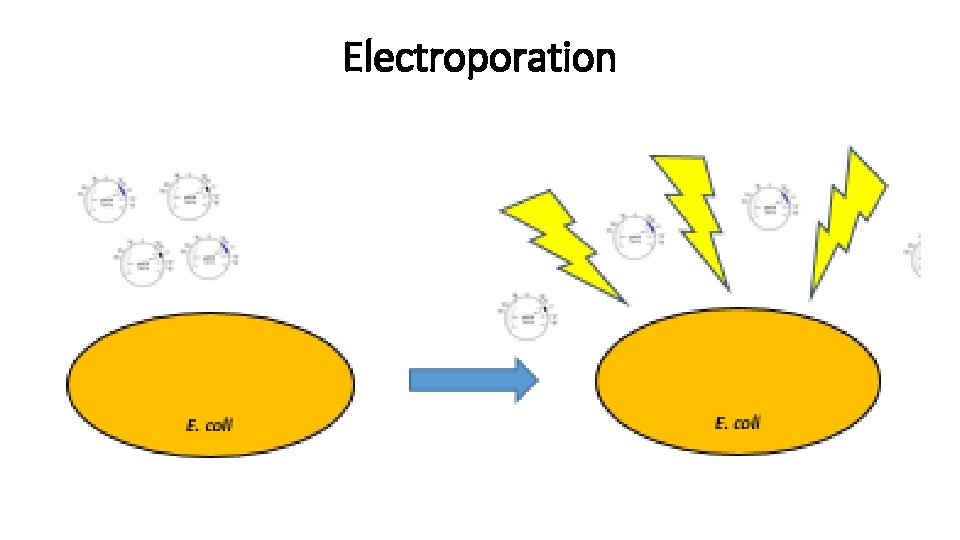
Electroporation

Standard Operating Procedure 1. Title Page - Title of the procedure - SOP identification number - Date of issue or revision - Name of the agency/division/branch to where the SOP applies - Signatures of those who prepared and approved of the SOP 2. Table of Contents 3. Quality Assurance/Quality Control - Performance evaluation samples 4. Reference - Cite all significant references. 5. Scope and applicability - Purpose of the process, limits, standards, etc. 6. Methodology and procedures - List all steps with necessary details including what equipment is needed. - Cover sequential procedures 7. Terminology - Identify acronyms and abbreviations that are not commonly known 8. Health and Safety 9. Equipment and supplies - Complete list of what is required to perform the procedure 10. Cautions and interferences - Cover what could go wrong and what may interfere with the final product
- Slides: 15