LECTURE 7 2 ARRHENIUS AND BRONSTED LOWRY ACIDSBASES
LECTURE 7. 2 – ARRHENIUS AND BRONSTED LOWRY ACIDS/BASES
What are Arrhenius and Bronsted – Lowry acid/bases?

I. Arrhenius Acids/Bases • An Arrhenius acid gives a H+ • Arrhenius acid has an H in its compound • An Arrhenius base gives an OH • An Arrhenius base has an OH in its compound

II. Bronsted-Lowry Acids/Bases • Bronsted-Lowry Acid – Donate (give) a H+ to solution • Bronsted-Lowry Base – Accept (take) a H+ from solution.

Class Example • Label the acid, base, and salt in the following reaction. Identify the acids and bases as either Arrhenius, Bronsted-Lowery, or both. HCl. O 3 + KOH H 2 O + KCl. O 3
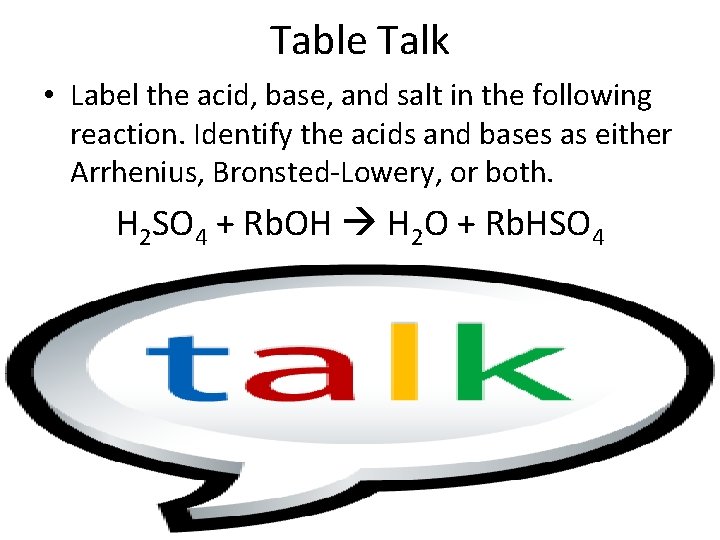
Table Talk • Label the acid, base, and salt in the following reaction. Identify the acids and bases as either Arrhenius, Bronsted-Lowery, or both. H 2 SO 4 + Rb. OH H 2 O + Rb. HSO 4

What are strong and weak acid and bases?

I. Strength of Acids/Bases • Acids/Bases can be classified as strong or weak based on how much it disassociates. • Disassociate – To break apart • Acids/Bases that completely dissociate are called strong acids/bases • Acids/Bases that only partially dissociate are called weak acids/bases.
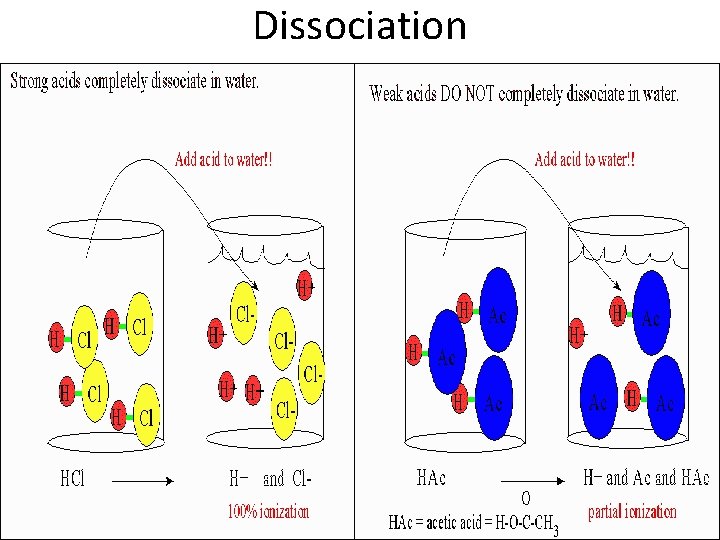
Dissociation

II. Strong Acids 1. HI 2. HBr 3. HCl 4. HNO 3 5. HCl. O 4 6. HCl. O 3 7. H 2 SO 4 You must memorize these!
III. Strong Bases 1. Ca(OH)2 2. Sr(OH)2 3. Ba(OH)2 4. Na. OH 5. KOH 6. Rb. OH 7. Cs. OH You must memorize these!

ALL ACIDS/BASES THAT ARE NOT ON THIS LIST ARE WEAK ACIDS/BASES
- Slides: 12