Lecture 6 Quiz 6 A cube of ice

Lecture 6

Quiz 6 • A cube of ice is taken from the freezer at -8. 5 °C and placed in a 95 -g aluminum calorimeter filled with 210 g of water at room temperature of 20. 0 °C. The final water temperature is 17. 0 °C after the ice has melted. What was the mass of the ice cube? Phase Temp Specific Transitions eratu Latent (H 2 O) re Heat (°C) (J/kg) Fusion 0 333, 000 Phase Tempera Specific (S ↔ L) ture Heat (H 2 O) (°C) (J/kg-K) Vaporization 100 2, 260, 000
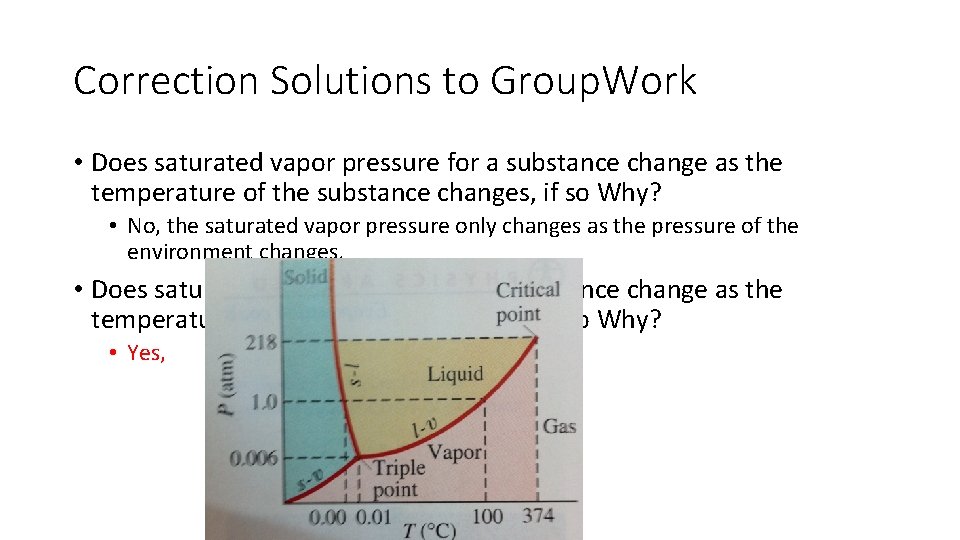
Correction Solutions to Group. Work • Does saturated vapor pressure for a substance change as the temperature of the substance changes, if so Why? • No, the saturated vapor pressure only changes as the pressure of the environment changes. • Does saturated vapor pressure for a substance change as the temperature of the substance changes, if so Why? • Yes,

Solutions to Groupwork 1. What are 10 subcooled liquids you come into contact with in the natural environment • Drinking water, tea, liquid dish soap, shampoo, rain water, gasoline, kerosene, etc. 2. What is a compressed liquid? And how is it different than a subcooled liquid? • They are the same thing 3. What are 3 examples of superheated vapors commonly used in Uganda? 1. 2. 3. 4. Refrigerant in a fridge is superheated during part of the cycle Oxygen in air Nitrogen in air CO 2 in air

4. Compare the specific latent heat of fusion and vaporization. Which latent heat is more important to our world, why? • Heat of vaporization is more important because the phase change between liquid and gas involved a large change of energy. Also, both liquids and gases flow, so they can easily be used in a cycle of flowing matter.

5. If ice melts in a glass of water, does the height of the water rise? If the ice contained a small piece of steel, when it melted, would the water level reduce? Why or why not? • No, if ice melts in a glass the water does not rise. Although ice takes up more space when it is froze the buoyance of the ice cube keeps the extra volume above the water line. • IF a piece of metal is imbedded? I’m not sure. Lets test. I’m thinking the answer is no, the level would be constant, because the ice-cube would just sit a bit lower in the water.

Calculating Heat of Vaporization – Experiment, Ramsey and Marshall Akrill and Osmond, “Work out Physics, A-Level” p. 163

Method •

Solution •

Discussion •

Method of mixtures determination of specific latent heat of vaporization • Is this a safe experiment to carry out? How could you make is safer? Sketch Initial Conditions Tw = 150 °C 1 kg water 0. 095 kg Al calorimeter 2 kg 1 kg hot oil Final Conditions Tmw = 125 °C 1 kg steam 2 kg of hot oil

Phase transitions are governed by the pressure, volume and temperature of the system • Changes 1. 2. 3. 4. 4. As Pressure is increased or decreases (P 1 ↔ P 2) As Temperature (heat) is added or removed (T 1 ↔ T 2) As Volume is expanded or shrunk (V 1 ↔ V 2) As Specific Volume is expanded or shrunk (v 1 ↔ v 2) Mass changes, which also changes the specific volume

P vs. V, vs. T diagrams/graphs • A state is defined by a pressure, temperature and volume. (except for phases 4 and 5) • A process (phase-change definition) is a change in P, V or T that results in a change in phase. • A process (thermodynamic definition) is a change in P, V, T or mass in a system that has an initial and final state • Graphs/Diagrams can be used to visualize the relationship between P, V and T as a process is taking place either for phase changes or for thermodynamic cycles.
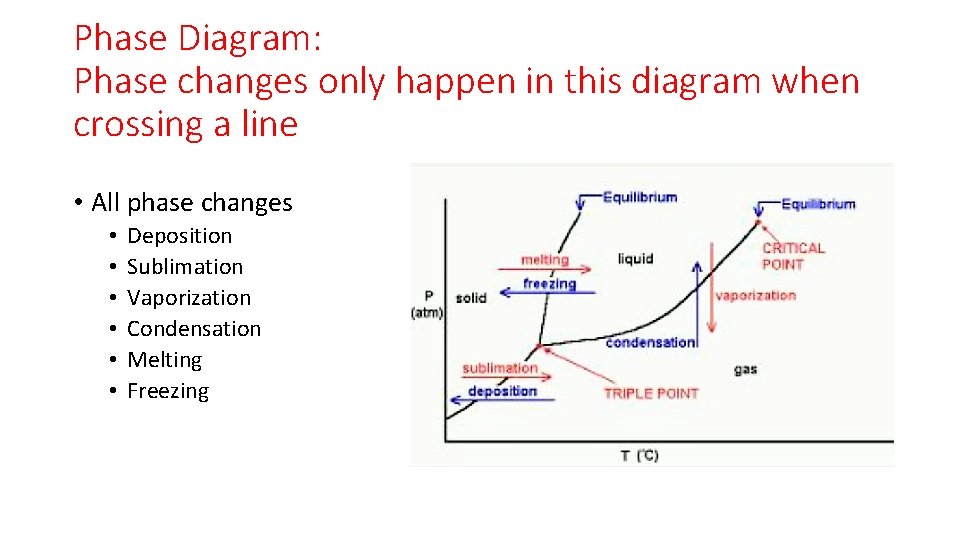
Phase Diagram: Phase changes only happen in this diagram when crossing a line • All phase changes • • • Deposition Sublimation Vaporization Condensation Melting Freezing
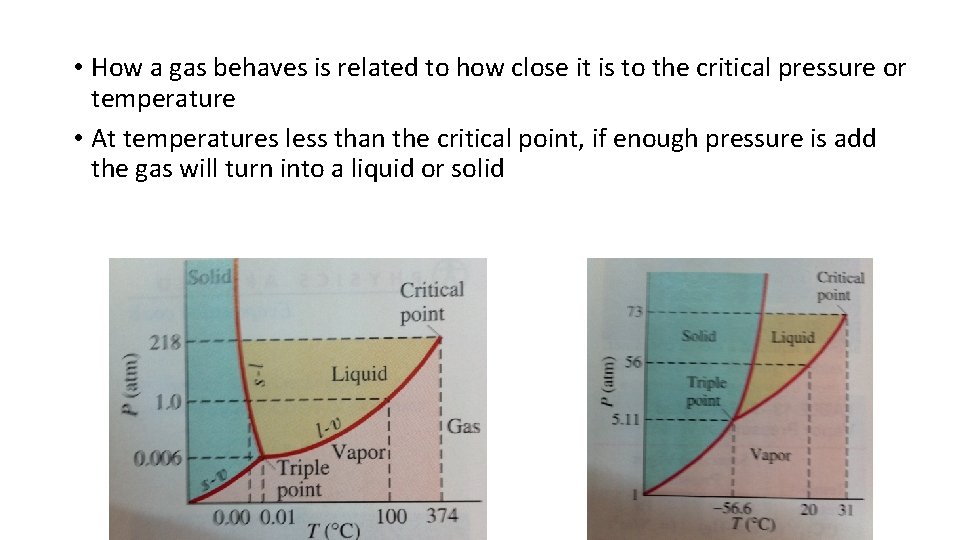
• How a gas behaves is related to how close it is to the critical pressure or temperature • At temperatures less than the critical point, if enough pressure is add the gas will turn into a liquid or solid
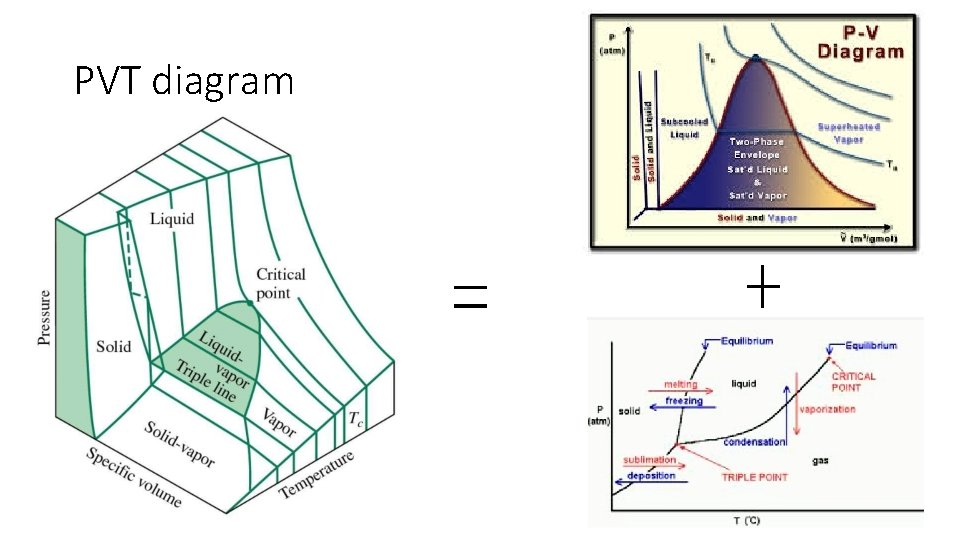
PVT diagram

Pressure PVT diagram Spe cifi c V o e lum e ur t a r e T p m e
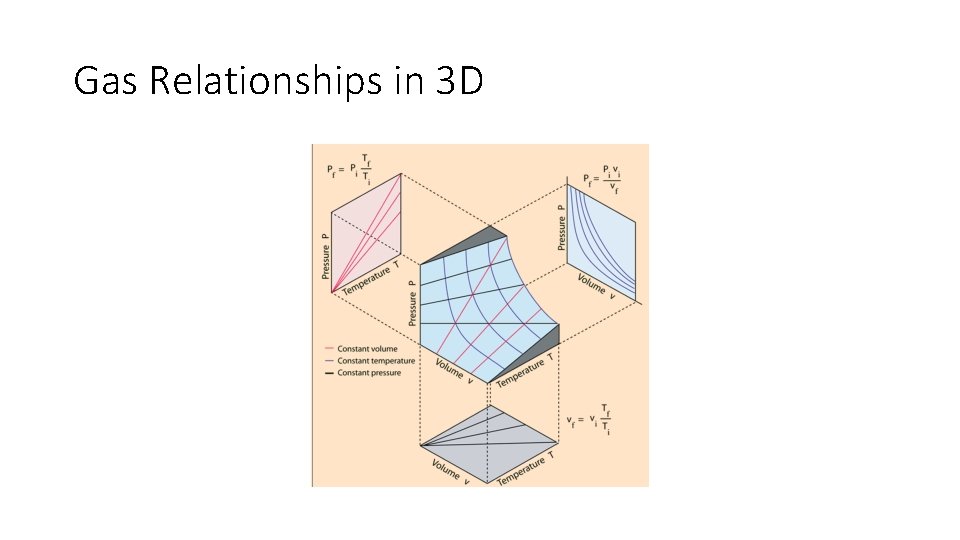
Gas Relationships in 3 D
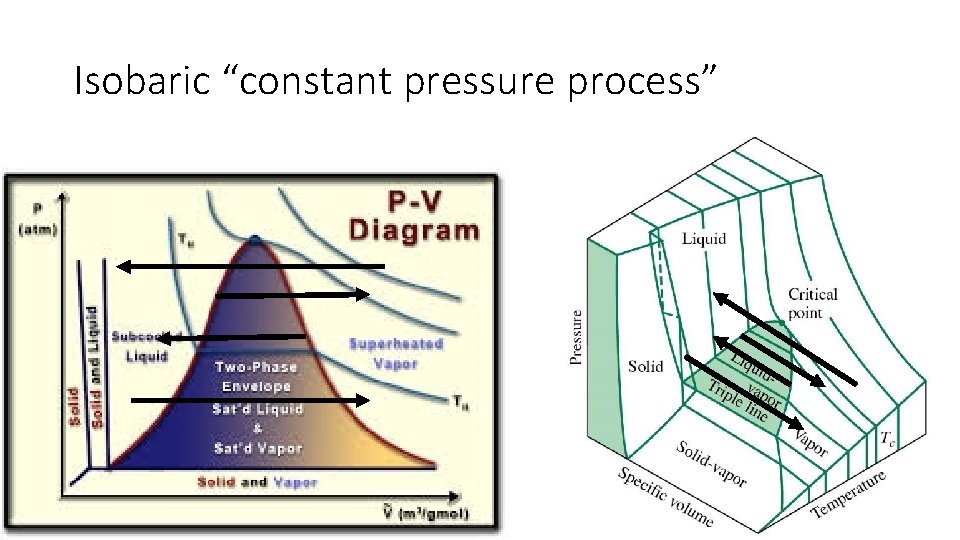
Isobaric “constant pressure process”
What are some isobaric examples from your life?

Examples of isobaric process • Ice melting in a glass • Vaporization of sweat of a person • Tea (water) vaporization out of a cup • Most processes that happen in daily life
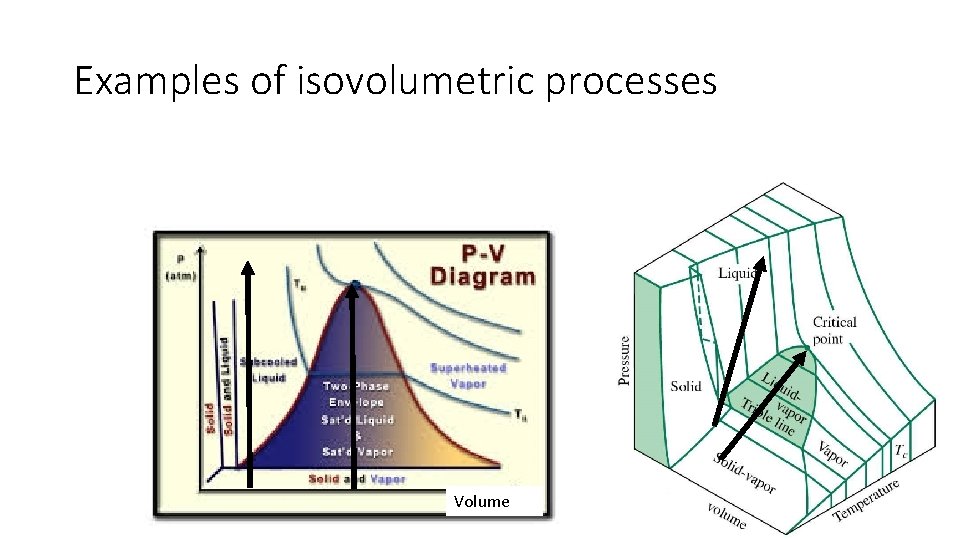
Examples of isovolumetric processes Volume

What are some isovolumetric examples from your life?

Examples of Isothermal processes • All phase changes • • • Deposition Sublimation Vaporization Condensation Melting Freezing
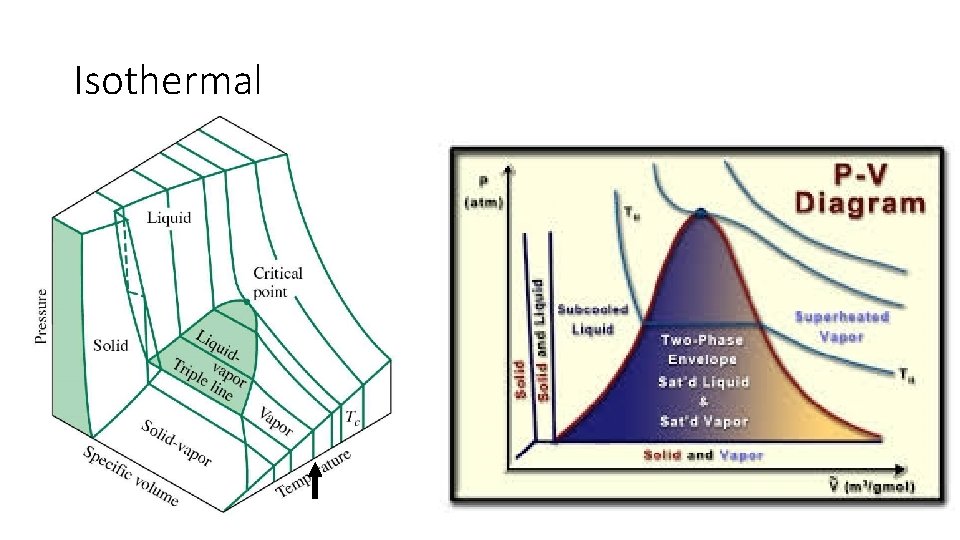
Isothermal

What are some isothermal examples from your life?
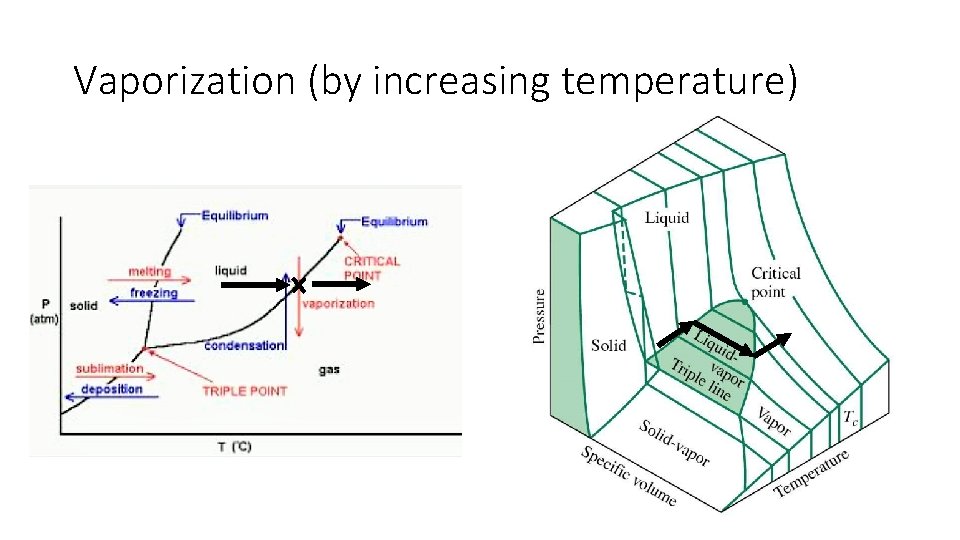
Vaporization (by increasing temperature)
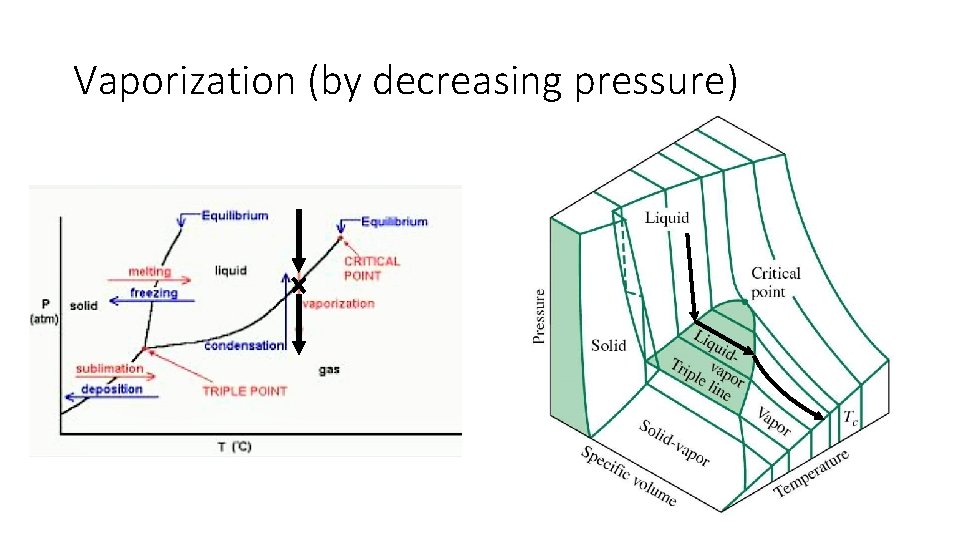
Vaporization (by decreasing pressure)
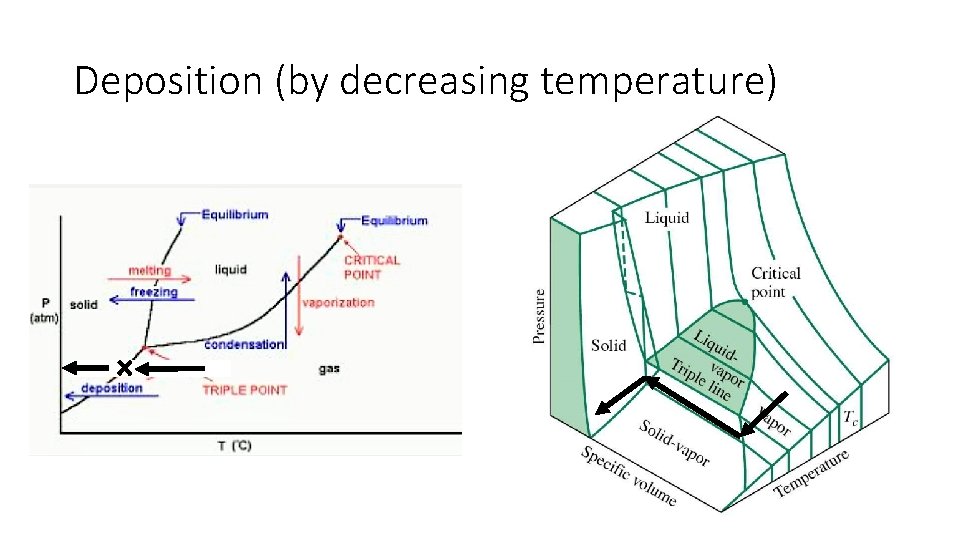
Deposition (by decreasing temperature)
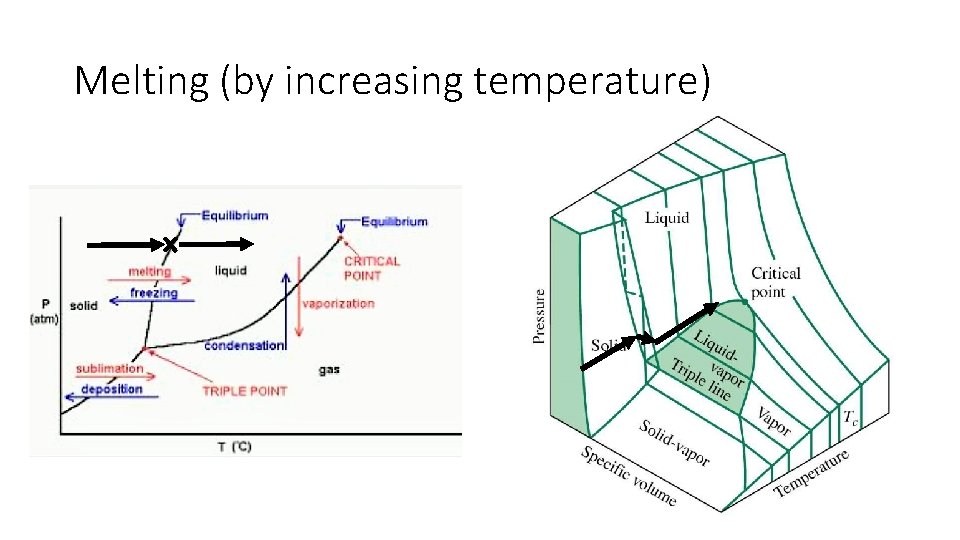
Melting (by increasing temperature)
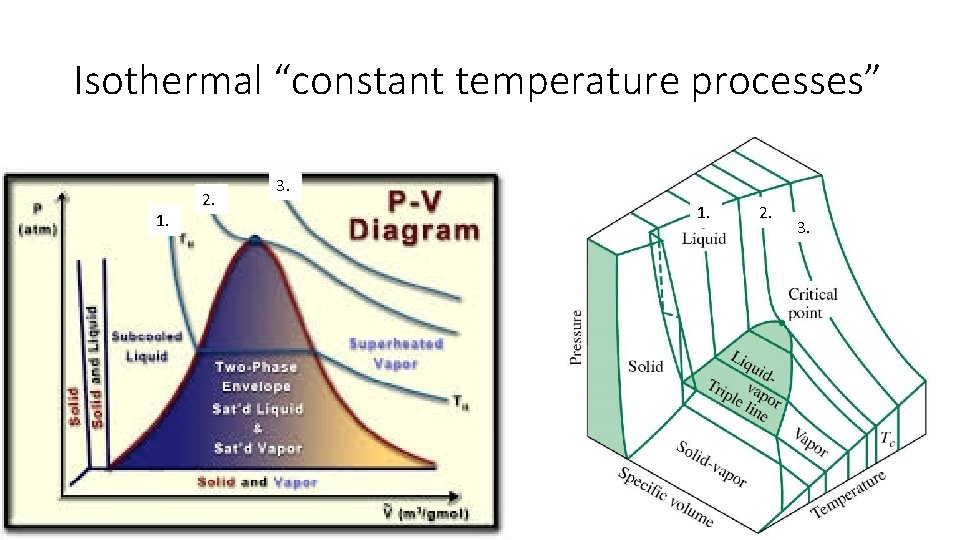
Isothermal “constant temperature processes” 1. 2. 3.
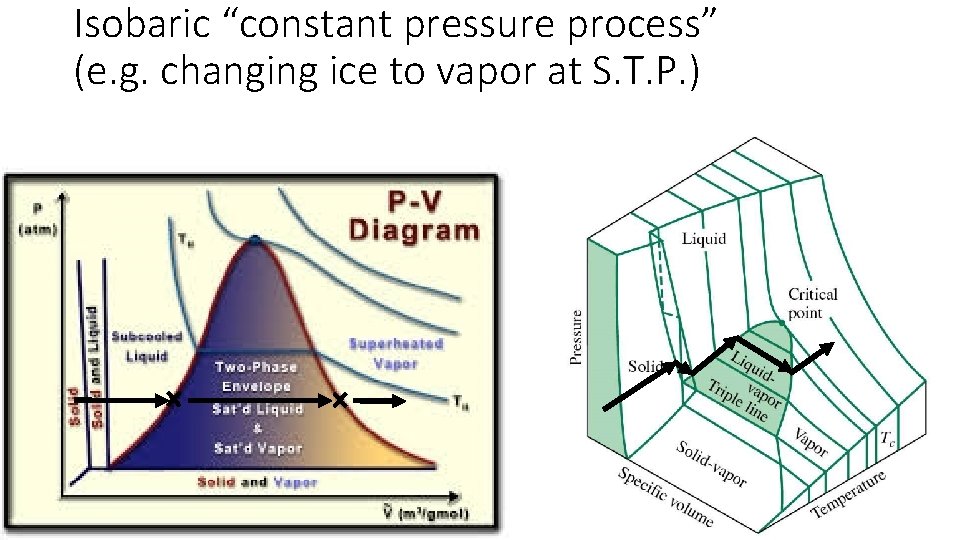
Isobaric “constant pressure process” (e. g. changing ice to vapor at S. T. P. )

Microscopic explanation of melting and freezing • Energy input (heat) is required to remove the bond of particles in a solid at a given P, V&T or energy released when a liquid changes to a solid • Energy (heat) required to remove the bond of particles in a liquid at a given P, V&T or energy released when a gas changes to a liquid

Groupwork: Determining Latent Heat by mixing • An Al calorimeter of 50 g has 20 grams of a solid melted in it. The initial temperature of the substance is Tim = -30 °C. Tiw of the calorimeter is 27 °C with 200 ml of water. The final temperature of the mixture is 20 °C. The SH of the substance at all temperatures is 500 J/kg. The melting point of the substance is -20 °C. What is the heat of fusion of the substance in J/kg? Sketch Initial Conditions Tw = 27 °C Tm = -30 °C 200 ml Water 0. 5 kg Al calorimeter Final Conditions Tmw = 20 °C 0. 2 kg of substance and water
- Slides: 34