Lecture 6 Molecular orbitals of heteronuclear diatomic molecules

Lecture 6

Molecular orbitals of heteronuclear diatomic molecules

The general principle of molecular orbital theory Interactions of orbitals (or groups of orbitals) occur when the interacting orbitals overlap. the energy of the orbitals must be similar the interatomic distance must be short enough but not too short A bonding interaction takes place when: regions of the same sign overlap An antibonding interaction takes place when: regions of opposite sign overlap
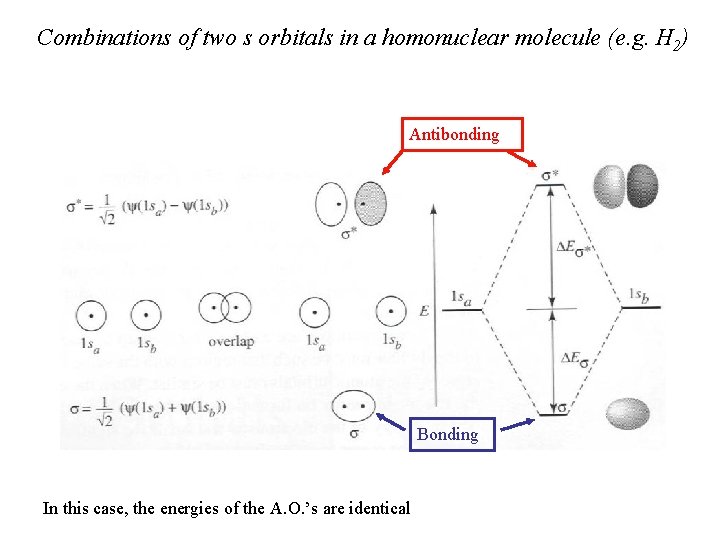
Combinations of two s orbitals in a homonuclear molecule (e. g. H 2) Antibonding Bonding In this case, the energies of the A. O. ’s are identical
![More generally: Y = N[ca. Y(1 sa) ± cb. Y (1 sb)] n A. More generally: Y = N[ca. Y(1 sa) ± cb. Y (1 sb)] n A.](http://slidetodoc.com/presentation_image/3093daf7ae55fe76e6c5eabe3a8ee601/image-5.jpg)
More generally: Y = N[ca. Y(1 sa) ± cb. Y (1 sb)] n A. O. ’s n M. O. ’s The same principle is applied to heteronuclear diatomic molecules But the atomic energy levels are lower for the heavier atom

Orbital potential energies (see also Table 5 -1 in p. 134 of textbook) Average energies for all electrons in the same level, e. g. , 3 p (use to estimate which orbitals may interact)
![The molecular orbitals of carbon monoxide Y = N[cc. Y(C) ± co. Y (O)] The molecular orbitals of carbon monoxide Y = N[cc. Y(C) ± co. Y (O)]](http://slidetodoc.com/presentation_image/3093daf7ae55fe76e6c5eabe3a8ee601/image-7.jpg)
The molecular orbitals of carbon monoxide Y = N[cc. Y(C) ± co. Y (O)] E(e. V) 2 s 2 p C -19. 43 -10. 66 O -32. 38 -15. 85 Each MO receives unequal contributions from C and O (cc ≠ co)

Group theory is used in building molecular orbitals
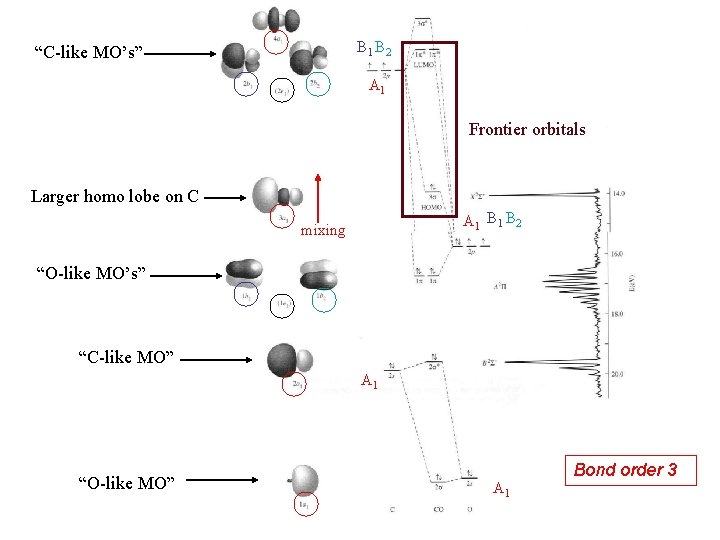
B 1 B 2 “C-like MO’s” A 1 Frontier orbitals Larger homo lobe on C A 1 B 2 mixing “O-like MO’s” “C-like MO” A 1 “O-like MO” A 1 Bond order 3

A related example: HF s (A 1) H -13. 61 (1 s) F -40. 17 (2 s) No s-s int. (DE > 13 e. V) 2 p(A 1, B 2) -18. 65 Non-bonding (no symmetry match) Non-bonding (no E match)
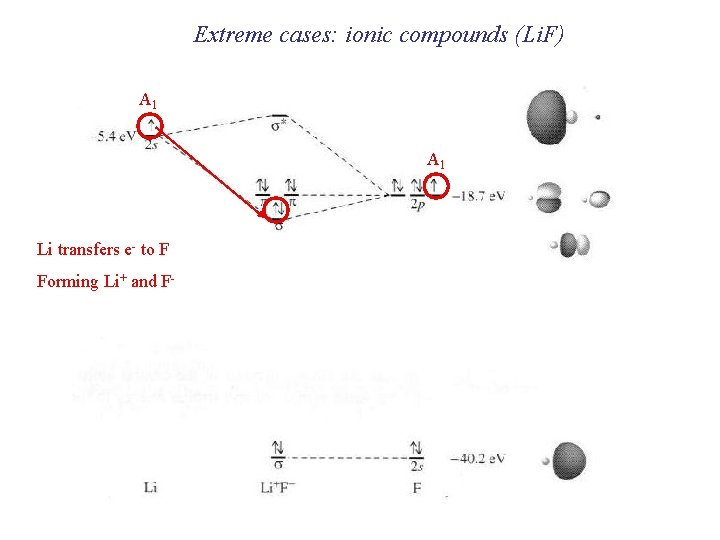
Extreme cases: ionic compounds (Li. F) A 1 Li transfers e- to F Forming Li+ and F-

Molecular orbitals for larger molecules 1. Determine point group of molecule (if linear, use D 2 h and C 2 v instead of D∞h or C∞v) 2. Assign x, y, z coordinates (z axis is higher rotation axis; if non-linear y axis in outer atoms point to central atom) 3. Find the characters of the representation for the combination of 2 s orbitals on the outer atoms, then for px, py, pz. (as for vibrations, orbitals that change position = 0, orbitals that do not change =1; and orbitals that remain in the same position but change sign = -1) 4. Find the reducible representations (they correspond to the symmetry of group orbitals, also called Symmetry Adapted Linear Combinations SALC’s of the orbitals). 5. Find AO’s in central atom with the same symmetry 6. Combine AO’s from central atom with those group orbitals of same symmetry and similar E

F-H-FD∞h, use D 2 h 1 st consider combinations of 2 s and 2 p orbitals from F atoms 8 GROUP ORBITALS DEFINED

Group orbitals can now be treated as atomic orbitals and combined with appropriate AO’s from H 1 s(H) is Ag so it matches two group orbitals 1 and 3 Both interactions are symmetry allowed, how about energies?
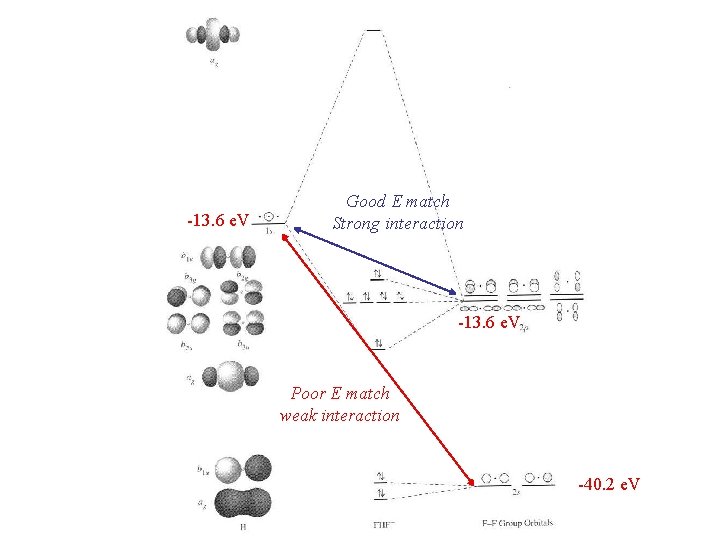
-13. 6 e. V Good E match Strong interaction -13. 6 e. V Poor E match weak interaction -40. 2 e. V

Lewis structure F-H-Fimplies 4 e around H ! MO analysis defines 3 c-2 e bond (2 e delocalized over 3 atoms) Bonding e Non-bonding e

CO 2 D∞h, use D 2 h (O O) group orbitals the same as for F F But C has more AO’s to be considered than H !
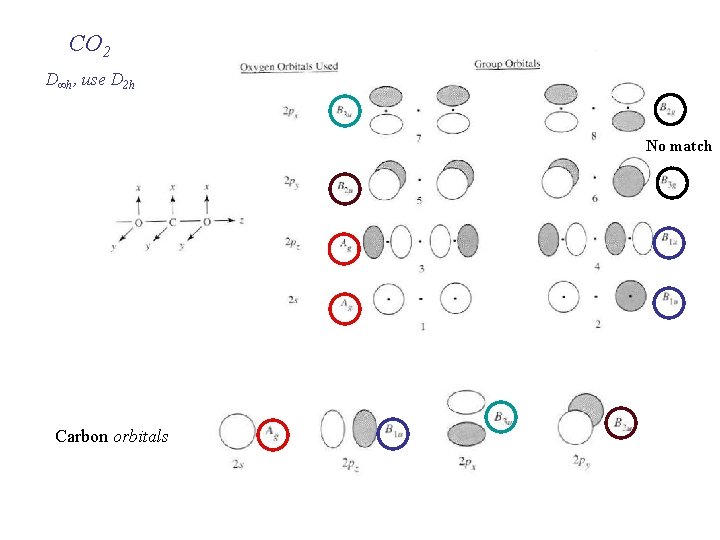
CO 2 D∞h, use D 2 h No match Carbon orbitals

Ag-Ag interactions All four are symmetry allowed B 1 u-B 1 u interactions

Primary B 1 u interaction Primary Ag interaction

Non-bonding p Bonding s Non-bonding s 4 bonds All occupied MO’s are 3 c-2 e
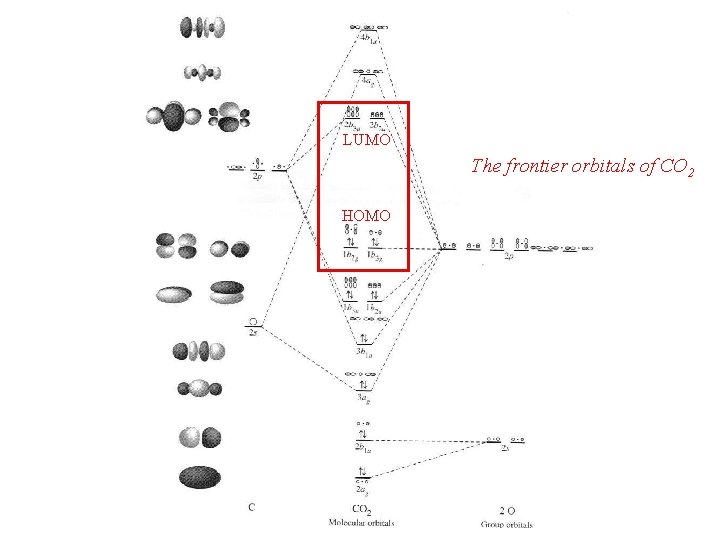
LUMO The frontier orbitals of CO 2 HOMO

Molecular orbitals for larger molecules: H 2 O 1. Determine point group of molecule: C 2 v 2. Assign x, y, z coordinates (z axis is higher rotation axis; if non-linear y axis in outer atoms point to central atom - not necessary for H since s orbitals are non-directional) 3. Find the characters of the representation for the combination of 2 s orbitals on the outer atoms, then for px, py, pz. (as for vibrations, orbitals that change position = 0, orbitals that do not change =1; and orbitals that remain in the same position but change sign = -1) 4. Find the irreducible representations (they correspond to the symmetry of group orbitals, also called Symmetry Adapted Linear Combinations SALC’s of the orbitals). 5. Find AO’s in central atom with the same symmetry 6. Combine AO’s from central atom with those group orbitals of same symmetry and similar E

G For H 2 0 H group orbitals E two orbitals unchanged C 2 two orbitals interchanged sv two orbitals unchanged sv’ two orbitals interchanged G = A 1 + B 1

No match

antibonding px non-bonding pz py slightly bonding

Molecular orbitals for NH 3 Find reducible representation for 3 H’s G 3 0 1 Irreducible representations: G = A 1 + E


anti-bonding LUMO pz Slightly bonding HOMO bonding

Projection Operator Algorithm of creating an object forming a basis for an irreducible rep from an arbitrary function. Where the projection operator sums the results of using the symmetry operations multiplied by characters of the irreducible rep. j indicates the desired symmetry. lj is the dimension of the irreducible rep. h the order of the group. Starting with the 1 s. A create a function of A 1 sym ¼(E 1 s. A + C 21 s. A + sv’ 1 s. A) = ¼ (1 s. A + 1 s. B + 1 s. A)

Consider the bonding in NF 3 GA 3 0 -1 GB 3 0 1 GC 3 0 1 GD 3 0 1 3 GA = A 2 + E GB = GC = GD = A 1 + E 2 1 A B C D

Now construct SALC GA = A 2 + E PA 2(p 1) = 1/6 (p + p + (-1)(-p) + (-1)(-1 p ) + (-1)(-p ) 1 2 3 1 3 2 No AO on N is A 2

E: Apply projection operator to p 1 PA 2(p 1) = 2/6 (2 p 1 - p 2 - p 3) = E 1 But since it is two dimensional, E, there should be another SALC PA 2(p 2) = 2/6 (2 p 2 - p 3 - p 1) = E’ But E 1 and E’ should be orthogonal want sum of products of coefficients to be zero. E 2 = E’ + k E 1. = (-1 +k*2) p 1 + (2 + k(-1)) p 2 + (-1 + k(-1)) = 0 Have to choose k such that they are orthogonal. 0 = (2/6)2 (2(-1 + k*2) -1 (2 + k(-1)) -1 (-1 + k(-1)) k=½ E 2 = 2/6 (3/2 p 2 - 3/2 p 3)


Molecular shapes: When we discussed VSEPR theory The geometries of electron domains Can this be described in terms of MO’s?
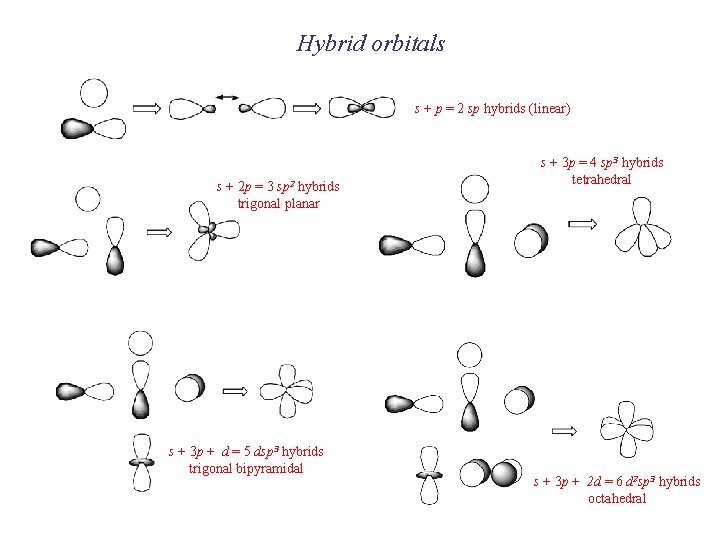
Hybrid orbitals s + p = 2 sp hybrids (linear) s + 2 p = 3 sp 2 hybrids trigonal planar s + 3 p + d = 5 dsp 3 hybrids trigonal bipyramidal s + 3 p = 4 sp 3 hybrids tetrahedral s + 3 p + 2 d = 6 d 2 sp 3 hybrids octahedral
- Slides: 36