LECTURE 5 ENERGY BALANCE Ch 61 ENERGY BALANCE

LECTURE 5 ENERGY BALANCE Ch 61

ENERGY BALANCE Concerned with energy changes and energy flow in a chemical process. Conservation of energy – first law of thermodynamics i. e. accumulation of energy in a system = energy input – energy output

Forms of energy Potential energy (mgh) Kinetic energy (1/2 mv 2) Thermal energy – heat (Q) supplied to or removed from a process Work energy – e. g. work done by a pump (W) to transport fluids Internal energy (U) of molecules m – mass (kg) g – gravitational constant, 9. 81 ms-2 v – velocity, ms-1
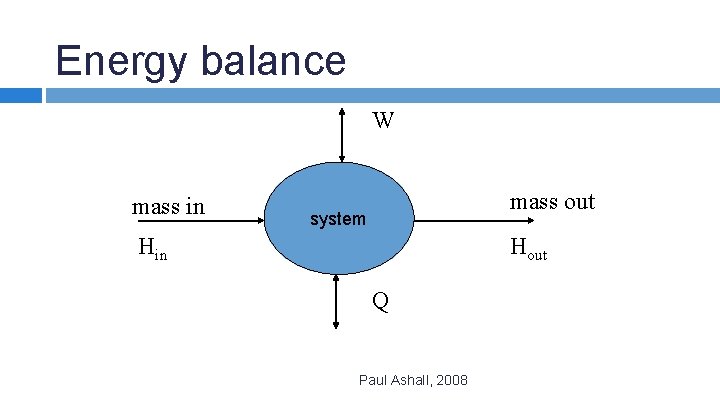
Energy balance W mass in mass out system Hin Hout Q Paul Ashall, 2008

IUPAC convention - heat transferred to a system is +ve and heat transferred from a system is –ve - work done on a system is+ve and work done by a system is -ve

STEADY STATE/NON-STEADY STATE Non steady state - accumulation/depletion of energy in system

Uses Heat required for a process Rate of heat removal from a process Heat transfer/design of heat exchangers Process design to determine energy requirements of a process Pump power requirements (mechanical energy balance) Pattern of energy usage in operation Process control Process design & development etc

1) No mass transfer (closed or batch) ∆E = Q + W 2) No accumulation of energy, no mass transfer (m 1 = m 2= m) ∆E = 0 Q = -W
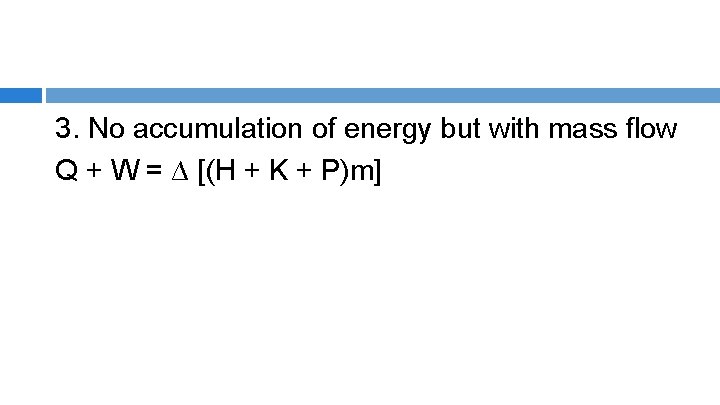
3. No accumulation of energy but with mass flow Q + W = ∆ [(H + K + P)m]

Air is being compressed from 100 k. Pa at 255 K (H = 489 k. J/kg) to 1000 k. Pa and 278 K (H = 509 k. J/kg) The exit velocity of the air from the compressor is 60 m/s. What is the power required (in k. W) for the compressor if the load is 100 kg/hr of air?

SAMPLE PROBLEM Water is pumped from the bottom of a well 4. 6 m deep at a rate of 760 L/hour into a vented storage tank to maintain a level of water in a tank 50 m above the ground. To prevent freezing in the winter a small heater puts 31, 600 k. J/hr into the water during its transfer from the well to the storage tank. Heat is lost from the whole system at a constant rate of 26, 400 k. J/hr. What is the temperature of the water as it enters the storage tank assuming that the well water is at 1. 6 o. C? A 2 -hp pump is used. About 55% of the rated horse power goes into the work of pumping and the rest is dissipated as heat to atmosphere.

ENTHALPHY BALANCE p. e. , k. e. , W terms = 0 Q = H 2 – H 1 or Q = ΔH , where H 2 is the total enthalpy of output streams and H 1 is the total enthalpy of input streams, Q is the difference in total enthalpy i. e. the enthalpy (heat) transferred to or from the system

continued Q –ve (H 1>H 2), heat removed from system Q +ve (H 2>H 1), heat supplied to system.

Example – steam boiler Two input streams: stream 1 - 120 kg/min. water, 30 deg cent. , H = 125. 7 k. J/kg; stream 2 – 175 kg/min, 65 deg cent, H= 272 k. J/kg One output stream: 295 kg/min. saturated steam(17 atm. , 204 deg cent. ), H = 2793. 4 k. J/kg

continued Ignore k. e. and p. e. terms relative to enthalpy changes for processes involving phase changes, chemical reactions, large temperature changes etc Q = ΔH (enthalpy balance) Basis for calculation 1 min. Steady state Q = Hout – Hin Q = [295 x 2793. 4] – [(120 x 125. 7) + (175 x 272)] Q = + 7. 67 x 105 k. J/min

STEAM TABLES Enthalpy values (H k. J/kg) at various P, T

Enthalpy changes Change of T at constant P Change of P at constant T Change of phase Solution Mixing Chemical reaction crystallisation

INVOLVING CHEMICAL REACTIONS Heat of Reaction: ∆Hrxn= ∑∆Hfo(products) - ∑∆Hfo(reactants)

SAMPLE PROBLEM: Calculate the ∆Hrxn for the following reaction: 4 NH 3(g) + 5 O 2(g) → 4 NO(g) + 6 H 2 O(g) Given the following ∆Hfo/mole at 25 o. C. NH 3(g): -46. 191 k. J/mol NO(g): +90. 374 k. J/mol H 2 O(g): -214. 826 k. J/mol

∆H = -904. 696 k. J/mol

ENERGY BALANCE THAT ACCOUNT FOR CHEMICAL REACTIONS: Most common: 1) What is the temperature of the incoming or exit streams 2) How much material must be introduced into the entering stream to provide for a specific amount of heat transfer.
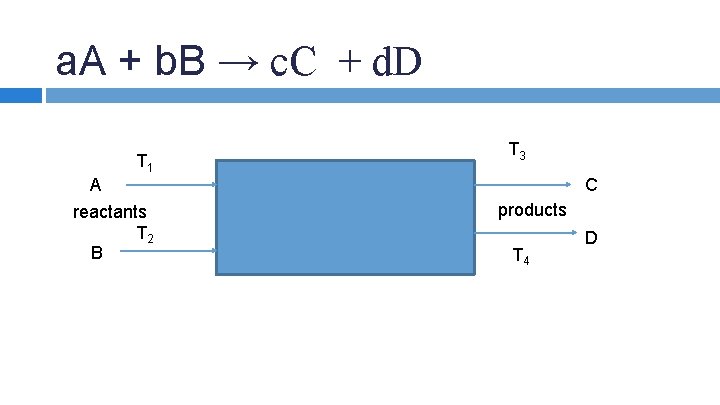
a. A + b. B → c. C + d. D T 1 A reactants T 2 B T 3 C products T 4 D

SAMPLE PROBLEM: An iron pyrite ore containing 85. 0% Fe. S 2 and 15. 0% inert materials (G) is roasted with an amount of air equal to 200% excess air according to the reaction 4 Fe. S 2 + 11 O 2 → 2 Fe 2 O 3 + 8 SO 2 in order to produce SO 2. All the inert materials plus the Fe 2 O 3 end up in the solid waste product, whick analyzes at 4. 0% Fe. S 2. Determine the heat transfer per kg of ore to keep the product stream at 25 o. C if the entering streams are at 25 o. C.
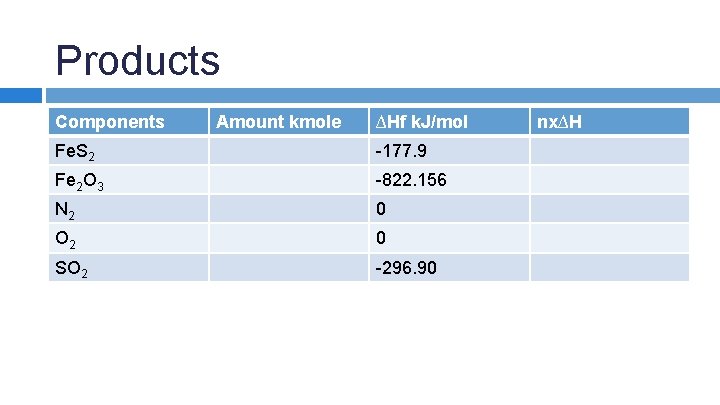
Products Components Amount kmole ∆Hf k. J/mol Fe. S 2 -177. 9 Fe 2 O 3 -822. 156 N 2 0 O 2 0 SO 2 -296. 90 nx∆H
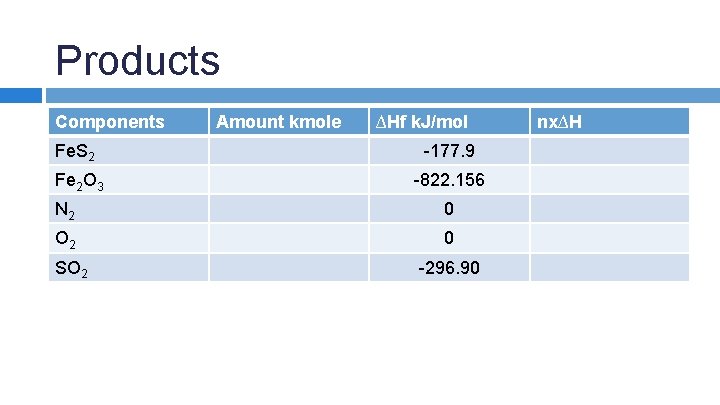
Products Components Amount kmole ∆Hf k. J/mol Fe. S 2 -177. 9 Fe 2 O 3 -822. 156 N 2 0 O 2 0 SO 2 -296. 90 nx∆H

Material Balance First!!!!
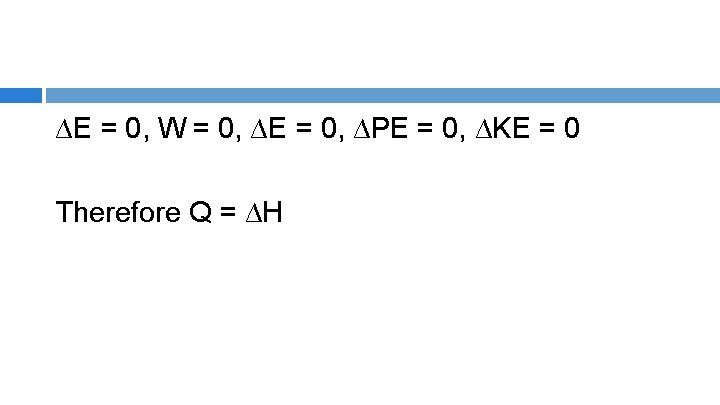
∆E = 0, W = 0, ∆E = 0, ∆PE = 0, ∆KE = 0 Therefore Q = ∆H
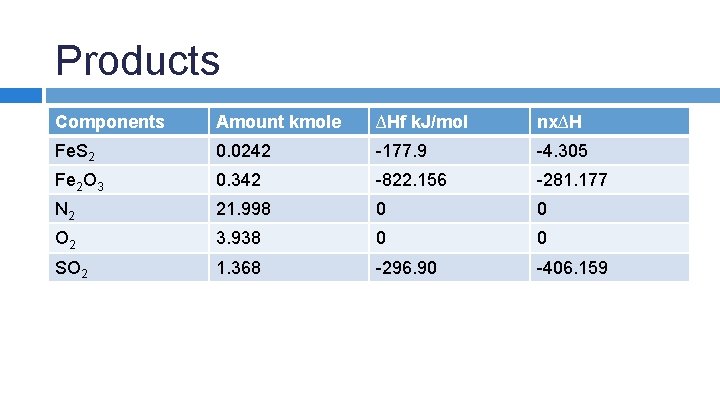
Products Components Amount kmole ∆Hf k. J/mol nx∆H Fe. S 2 0. 0242 -177. 9 -4. 305 Fe 2 O 3 0. 342 -822. 156 -281. 177 N 2 21. 998 0 0 O 2 3. 938 0 0 SO 2 1. 368 -296. 90 -406. 159
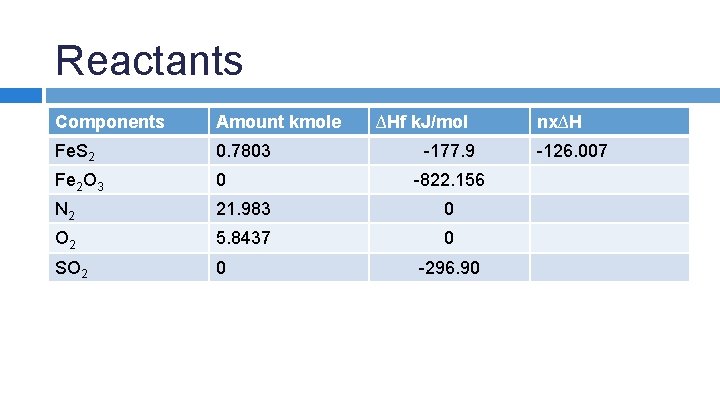
Reactants Components Amount kmole ∆Hf k. J/mol Fe. S 2 0. 7803 Fe 2 O 3 0 N 2 21. 983 0 O 2 5. 8437 0 SO 2 0 -177. 9 -822. 156 -296. 90 nx∆H -126. 007

SAMPLE PROBLEM Q=

Latent heats (phase changes) Vapourisation (L to V) Melting (S to L) Sublimation (S to V)

Mechanical energy balance Consider mechanical energy terms only Application to flow of liquids ΔP + Δ v 2 + g Δh +F = W ρ 2 where W is work done on system by a pump and F is frictional energy loss in system (J/kg) ΔP = P 2 – P 1; Δ v 2 = v 22 –v 12; Δh = h 2 –h 1 Bernoulli equation (F=0, W=0)

Example - Bernoulli eqtn. Water flows between two points 1, 2. The volumetric flow rate is 20 litres/min. Point 2 is 50 m higher than point 1. The pipe internal diameters are 0. 5 cm at point 1 and 1 cm at point 2. The pressure at point 2 is 1 atm. . Calculate the pressure at point 2. Paul Ashall, 2008

continued ΔP/ρ + Δv 2/2 + gΔh +F = W ΔP = P 2 – P 1 (Pa) Δv 2 = v 22 – v 12 Δh = h 2 - h 1 (m) F= frictional energy loss (mechanical energy loss to system) (J/kg) W = work done on system by pump (J/kg) ρ = 1000 kg/m 3

continued Volumetric flow is 20/(1000. 60) m 3/s = 0. 000333 m 3/s v 1 = 0. 000333/(π(0. 0025)2) = 16. 97 m/s v 2 = 0. 000333/ (π(0. 005)2) = 4. 24 m/s (101325 - P 1)/1000 + [(4. 24)2 – (16. 97)2]/2 + 9. 81. 50 = 0 P 1 = 456825 Pa (4. 6 bar)

Sensible heat/enthalpy calculations ‘Sensible’ heat – heat/enthalpy that must be transferred to raise or lower the temperature of a substance or mixture of substances. Heat capacities/specific heats (solids, liquids, gases, vapours) Heat capacity/specific heat at constant P, Cp(T) = d. H/d. T or ΔH = integral Cp(T)d. T between limits T 2 and T 1 Use of mean heat capacities/specific heats over a temperature range Use of simple empirical equations to describe the variation of Cp with T

continued e. g. Cp = a + b. T + c. T 2 + d. T 3 , where a, b, c, d are coefficients ΔH = integral. Cpd. T between limits T 2, T 1 ΔH = [a. T + b. T 2 + c. T 3 + d. T 4] 2 3 4 Calculate values for T = T 2, T 1 and subtract Note: T may be in deg cent or K - check units for Cp!

Example Calculate the enthalpy required to heat a stream of nitrogen gas flowing at 100 mole/min. , through a gas heater from 20 to 100 deg. cent. (use mean Cp value 29. 1 J mol-1 K-1 or Cp = 29 + 0. 22 x 10 -2 T + 0. 572 x 10 -5 T 2 – 2. 87 x 10 -9 T 3, where T is in deg cent)

Heat capacity/specific heat data Felder & Rousseau pp 372/373 and Table B 10 Perry’s Chemical Engineers Handbook The properties of gases and liquids, R. Reid et al, 4 th edition, Mc. Graw Hill, 1987 Estimating thermochemical properties of liquids part 7 heat capacity, P. Gold & G. Ogle, Chem. Eng. , 1969, p 130 Coulson & Richardson Chem. Eng. , Vol. 6, 3 rd edition, ch. 8, pp 321 -324 ‘Phys. Props’

Example – change of phase A feed stream to a distillation unit contains an equimolar mixture of benzene and toluene at 10 deg cent. The vapour stream from the top of the column contains 68. 4 mol % benzene at 50 deg cent. and the liquid stream from the bottom of the column contains 40 mol% benzene at 50 deg cent. [Need Cp (benzene, liquid), Cp (toluene, liquid), Cp (benzene, vapour), Cp (toluene, vapour), latent heat of vapourisation benzene, latent heat of vapourisation toluene. ]

Energy balances on systems involving chemical reaction Standard heat of formation (ΔHof) – heat of reaction when product is formed from its elements in their standard states at 298 K, 1 atm. (k. J/mol) a. A + b. B c. C + d. D -a -b +c +d (stoichiometric coefficients, νi) ΔHof. A, ΔHof. B, ΔHof. C, ΔHof. D (heats of formation) ΔHo. R = c ΔHof. C + d ΔHof. D - a ΔHof. A - bΔHof. B Paul Ashall, 2008

Heat (enthalpy) of reaction ΔHo. R –ve (exothermic reaction) ΔHo. R +ve (endothermic reaction) Paul Ashall, 2008

ENTHALPHY BALANCE REACTOR Qp = Hproducts – Hreactants + Qr Qp – heat transferred to or from process Qr – reaction heat (ζ ΔHo. R), where ζ is extent of reaction and is equal to [moles component, i, out – moles component i, in]/ νi Paul Ashall, 2008
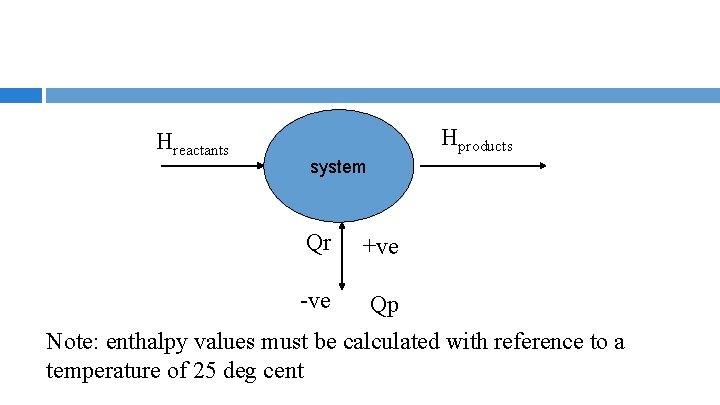
Hreactants Hproducts system Qr +ve -ve Qp Note: enthalpy values must be calculated with reference to a temperature of 25 deg cent

ENERGY BALANCE TECHNIQUES Complete mass balance/molar balance Calculate all enthalpy changes between process conditions and standard/reference conditions for all components at start (input) and finish (output). Consider any additional enthalpy changes Solve enthalpy balance equation Paul Ashall, 2008

ENERGY BALANCE TECHNIQUES Adiabatic temperature: Qp = 0 Paul Ashall, 2008

EXAMPLES Reactor Crystalliser Drier Distillation

References The Properties of Gases and Liquids, R. Reid Elementary Principles of Chemical Processes, R. M. Felder and R. W. Rousseau Paul Ashall, 2008

SAMPLE PROBLEM: Limestone (Ca. CO 3) is converted into Ca. O in a continuous vertical kiln. Heat is supplied by combustion of natural gas (CH 4) in direct contact with limestone using 50% excess air. Determine the kg of Ca. CO 3 that can be processed per kg of natural gas. Assume that the following heat capacities apply. Cp of Ca. CO 3 = 234 J/mole –o. C Cp of Ca. O = 111 j/mole - o. C

- Slides: 50