Lecture 5 Chemical Reaction Engineering CRE is the

















- Slides: 24

Lecture 5 Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place.

Lecture 5 – Thursday 1/24/2013 �Block 1: Mole Balances �Block 2: Rate Laws �Block 3: Stoichiometry �Stoichiometric Table: Flow �Definitions of Concentration: Flow �Gas Phase Volumetric Flow Rate �Calculate the Equilibrium Conversion Xe 2

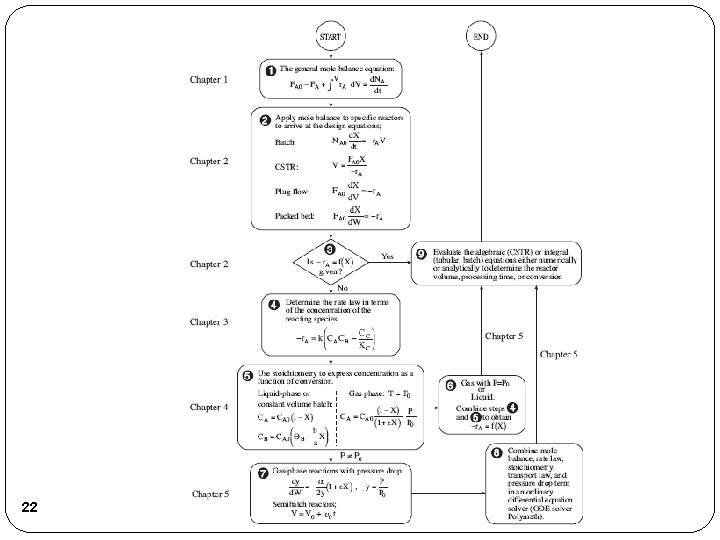
Review Lecture 2 Reactor Mole Balances Summary in terms of conversion, X Reactor Differential Algebraic Integral X Batch t CSTR PFR X PBR 3 W

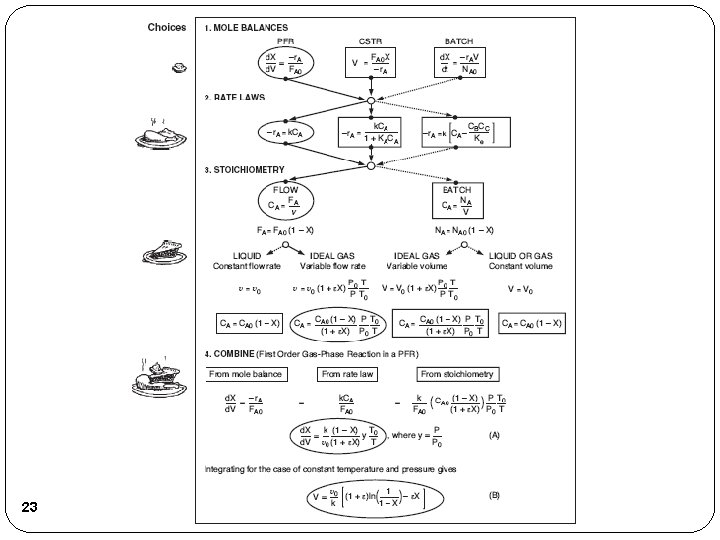
Review Lecture 3 Algorithm How to find Step 1: Rate Law Step 2: Stoichiometry Step 3: Combine to get 4

Review Lecture 3 Reaction Engineering Mole Balance Rate Laws Stoichiometry These topics build upon one another 5

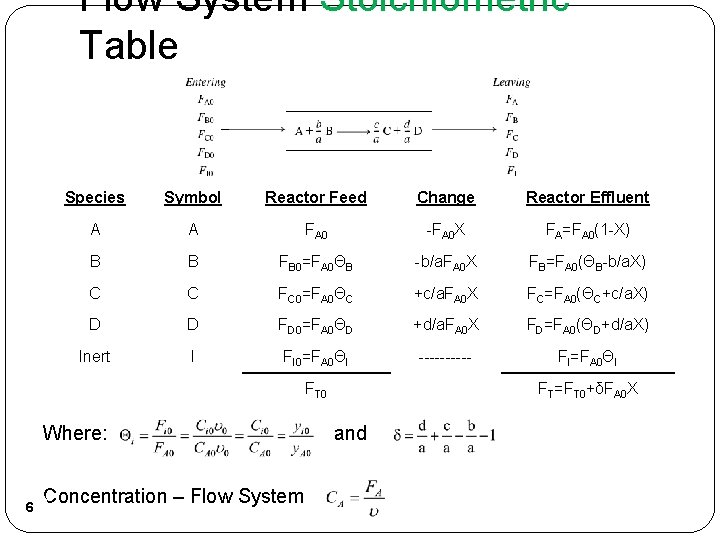
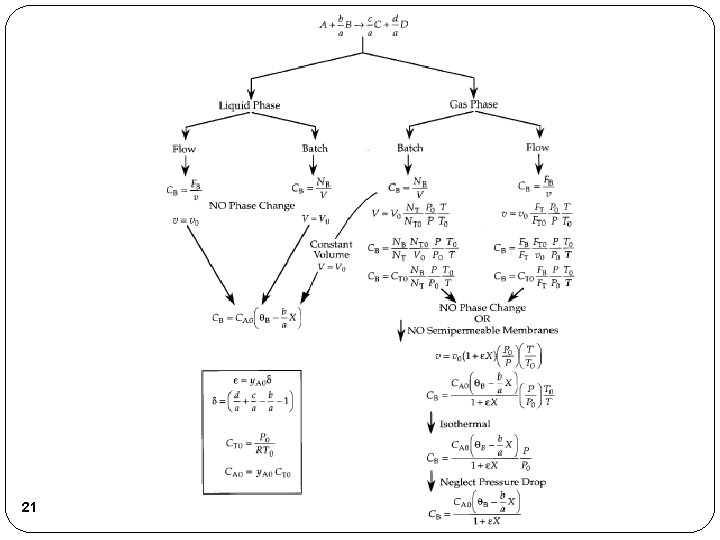
Flow System Stoichiometric Table Species Symbol Reactor Feed Change Reactor Effluent A A FA 0 -FA 0 X FA=FA 0(1 -X) B B FB 0=FA 0ΘB -b/a. FA 0 X FB=FA 0(ΘB-b/a. X) C C FC 0=FA 0ΘC +c/a. FA 0 X FC=FA 0(ΘC+c/a. X) D D FD 0=FA 0ΘD +d/a. FA 0 X FD=FA 0(ΘD+d/a. X) Inert I FI 0=FA 0ΘI ----- FI=FA 0ΘI FT 0 Where: 6 Concentration – Flow System FT=FT 0+δFA 0 X and

Stoichiometry Concentration Flow System: Liquid Phase Flow System: Liquid Systems Flow Liquid Phase etc. 7

Liquid Systems If the rate of reaction were then we would have This gives us 8

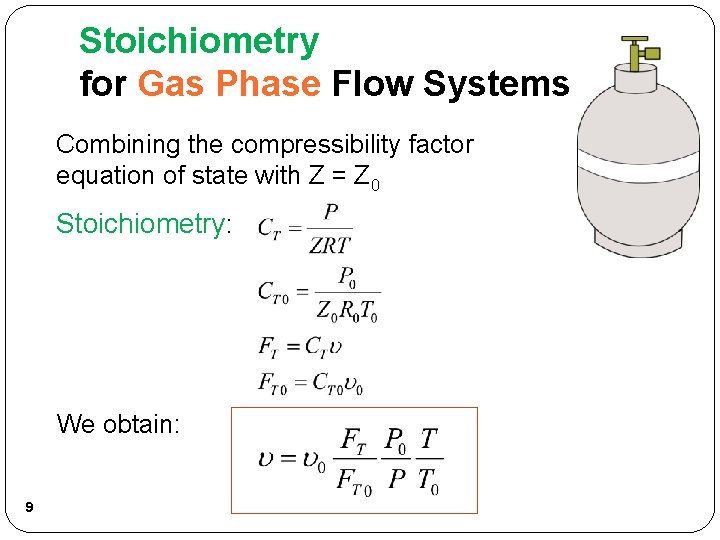
Stoichiometry for Gas Phase Flow Systems Combining the compressibility factor equation of state with Z = Z 0 Stoichiometry: We obtain: 9

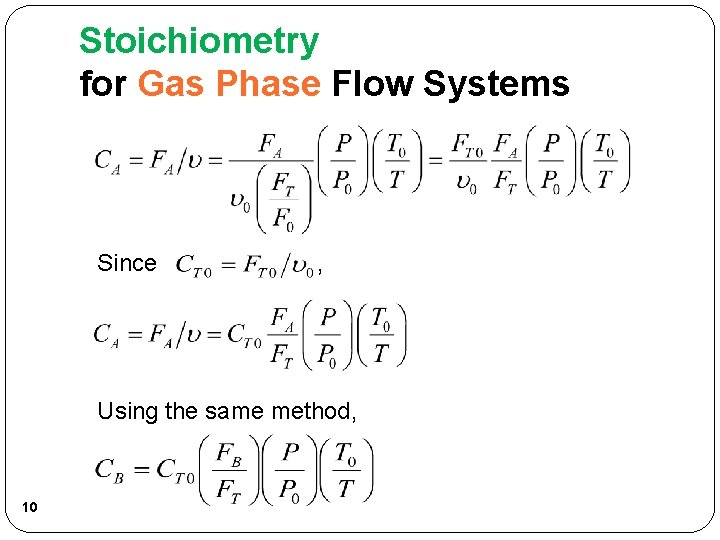
Stoichiometry for Gas Phase Flow Systems Since , Using the same method, 10

Stoichiometry for Gas Phase Flow Systems The total molar flow rate is: Substituting FT gives: Where 11

For Gas Phase Flow Systems Concentration Flow System: Gas Phase Flow System: 12

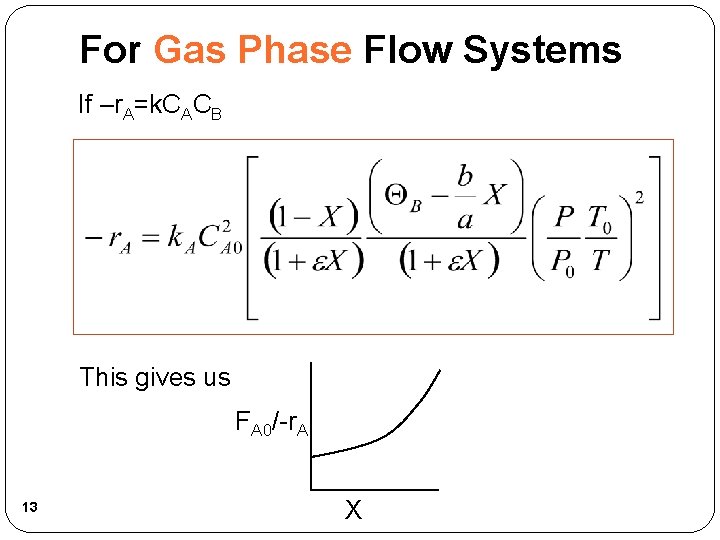
For Gas Phase Flow Systems If –r. A=k. CACB This gives us FA 0/-r. A 13 X

For Gas Phase Flow Systems 14

Example: Calculating the equilibrium conversion (Xef) for gas phase reaction in a flow reactor Consider the following elementary reaction where KC=20 dm 3/mol and CA 0=0. 2 mol/dm 3. Calculate Equilibrium Conversion or both a batch reactor (Xeb) and a flow reactor (Xef). 15

Gas Flow Example (Xef) Solution: Rate Law: 16

Gas Flow Example (Xef) Species Fed Change Remaining A FA 0 -FA 0 X FA=FA 0(1 -X) B 0 +FA 0 X/2 FB=FA 0 X/2 FT 0=FA 0 17 FT=FA 0 -FA 0 X/2

Gas Flow Example (Xef) A FA 0 -FA 0 X FA=FA 0(1 -X) B 0 FA 0 X/2 FB=FA 0 X/2 Stoichiometry: 18 Gas isothermal Gas isobaric T=T 0 P=P 0

Gas Flow Example (Xef) Pure A y. A 0=1, CA 0=y. A 0 P 0/RT 0, CA 0=P 0/RT 0 At equilibrium: -r. A=0 19

Gas Flow Example (Xef) Flow: 20 Recall Batch:

21

22

23

End of Lecture 5 24