LECTURE 4 1 BALANCING CHEMICAL EQUATIONS What is

LECTURE 4. 1 – BALANCING CHEMICAL EQUATIONS

What is the law of conservation of mass?

I. Law of Conservation of Mass • The law of conservation of mass states that matter cannot be created or destroyed, it can only change forms. • Therefore, we cannot destroy something. All we can do is change the form.

How are states indicated in chemical reactions?

TNT Explosion C 7 H 5 N 3 O 6 → N 2 + H 2 O + C

I. Chemical Reactions • Substances react to form new substances • Chemical equations are used to explain a reaction that occurs.

TNT Reaction C 7 H 5 N 3 O 6 → N 2 + H 2 O + C Element Before Reaction? C H N O ✔ ✔ After Reaction? ✔ ✔
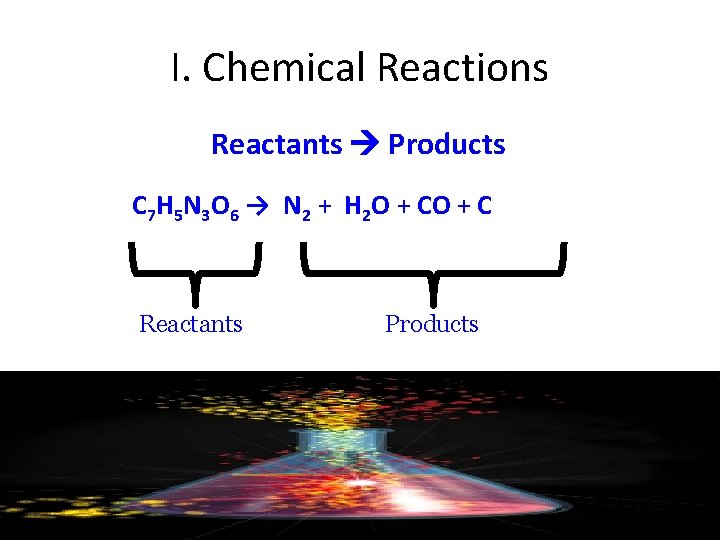
I. Chemical Reactions Reactants Products C 7 H 5 N 3 O 6 → N 2 + H 2 O + C Reactants Products

II States of Molecules • A molecule can be in a solid, liquid, gas, or aqueous state • Aqueous means dissolved in water • We abbreviate these as: – (s) = solid – (l) = liquid – (g) = gas – (aq) = aqueous

Table Talk • With your table, label the products, the reactants, and the state of each compound. NH 4 OH (aq) + Fe. Cl 3 (s) NH 4 Cl (s) + Fe(OH) 3 (aq)

Stop and Jot • For the following reaction, label the products, the reactants, and the state of each compound: H 2 O 2 (aq) + Na. Br (s) Na. OH (aq) + H 2 O (l) + O 2 (g)

How do you balance chemical reactions?

I. Balancing Chemical Reactions • Need same amount of products and reactants to satisfy law of conservation of mass • We balance chemical equations to achieve this

II. Tips for Balancing Chemical Reactions • We CANNOT change the subscripts. • We can change the number in front of the substance, known as the coefficient. Coefficient 2 Na. Cl This means we have 2 Na and 2 Cl
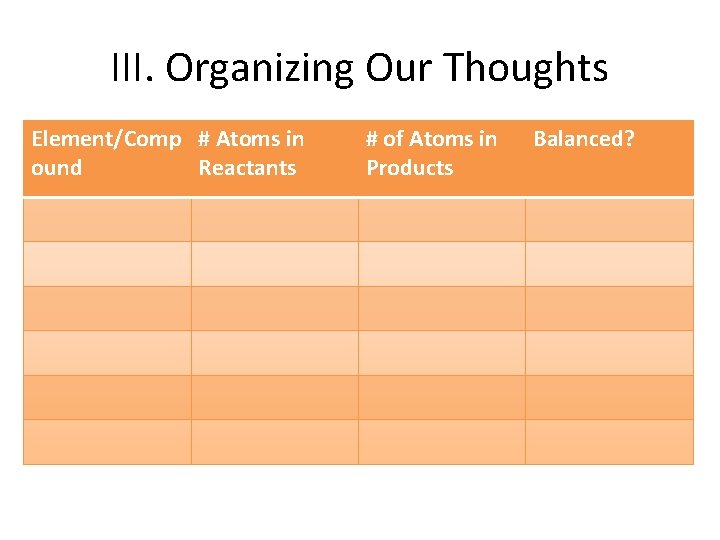
III. Organizing Our Thoughts Element/Comp # Atoms in ound Reactants # of Atoms in Products Balanced?

Class Example • Balance the following chemical reaction: ____H 2 O 2 ___ H 2 O + ____O 2

Class Example • Balance the following equation: __Ag. NO 3 + ___Na 2 Cr. O 4 __ Ag 2 Cr. O 4 +___ Na. NO 3

Table Talk • Balance the following equation: Mg + Fe. Cl 3 Mg. Cl 2 + Fe

Extra Examples • ___ C 3 H 6 + ____ H 2 O ___ C 3 H 8 + __ O 2 • ___ Mg + ___ Fe. Cl 3 ___ Mg. Cl 2 + ___ Fe • __ H 2 O + __ Fe 2 (SO 4) 3 __ Fe 2 O 3 + __ H 2 SO 4 + ___ H 2 O + ___ H 2
- Slides: 19