Lecture 31 AcidBase Titartions Cont Kjeldahl Analysis Complexometric

Lecture 31 Acid-Base Titartions, Cont… Kjeldahl Analysis Complexometric Reactions 1

Kjeldahl Analysis An application of acid-base titrations that finds an important use in analytical chemistry is what is called Kjeldahl nitrogen analysis. This analysis is used for the determination of nitrogen in proteins and other nitrogen containing compounds. Usually, the quantity of proteins can be estimated from the amount of nitrogen they contain. The Kjeldahl analysis involves the following steps: 2

1. Digestion of the nitrogen containing compound and converting the nitrogen to ammonium hydrogen sulfate. This process is accomplished by decomposing the nitrogen containing compound with sulfuric acid. 2. The solution in step 1 is made alkaline by addition of concentrated Na. OH which coverts ammonium to gaseous ammonia , and the solution is distilled to drive the ammonia out. 3. The ammonia produced in step 2 is collected in a specific volume of a standard acid solution (dilute) where neutralization occurs. 4. The solution in step 3 is back-titrated against a standard Na. OH solution to determine excess acid. 5. mmoles of ammonia are then calculated and related 3 to mmol nitrogen.

Example A 0. 200 g of a urea (FW = 60, (NH 2)2 CO) sample is analyzed by the Kjeldahl method. The ammonia is collected in a 50 m. L of 0. 05 M H 2 SO 4. The excess acid required 3. 4 m. L of 0. 05 M Na. OH. Find the percentage of the compound in the sample. 2 NH 3 + H 2 SO 4 = (NH 4)2 SO 4 ½ mmol ammonia = mmol H 2 SO 4 reacted (NH 2)2 CO = 2 NH 3 mmol urea = ½ mmol ammonia mmol H 2 SO 4 titrated = ½ mmol Na. OH 4

mmol urea = mmol H 2 SO 4 taken – ½ mmol Na. OH mmol urea = 0. 05 x 50 – 1/2 x 0. 05 x 3. 4 = 2. 415 mg urea = 2. 415 x 60 = 144. 9 % urea = (144. 9/200)x 100 = 72. 5% 5

Modified Kjeldahl Analysis In conventional Kjeldahl method we need two standard solutions, an acid for collecting evolved ammonia and a base for back-titrating the acid. In a modified procedure, only a standard acid is required. In this procedure, ammonia is collected in a solution of dilute boric acid, the concentration of which need not be known accurately. The result of the reaction is the borate which is equivalent to ammonia. NH 3 + H 3 BO 3 g NH 4+ + H 2 BO 3 Borate is a strong conjugate base which can be titrated with a standard HCl solution. 6

Example A 0. 300 g feed sample is analyzed for its protein content by the modified Kjeldahl method. If 25 m. L of 0. 10 M HCl is required for the titration what is the percent protein content of the sample (mg protein = 6. 25 * mg N). Solution mmol N = mmol HCl mmol N = 0. 10 x 25 = 2. 5 mmol N = 2. 5 mg N = 2. 5 x 14 = 35 mg protein = 35 * 6. 25 = 218. 8 % protein = (218. 8/300) x 100 = 72. 9% 7

Complexometric Reactions and Titrations 8

Complexes are compounds formed from combination of metal ions with ligands (complexing agents). A metal ionis an electron deficient species while a ligand is an electron rich, and thus, electron donating species. A metal ion will thus accept electrons from a ligand where coordination bonds are formed. Electrons forming coordination bonds come solely from ligands. 9

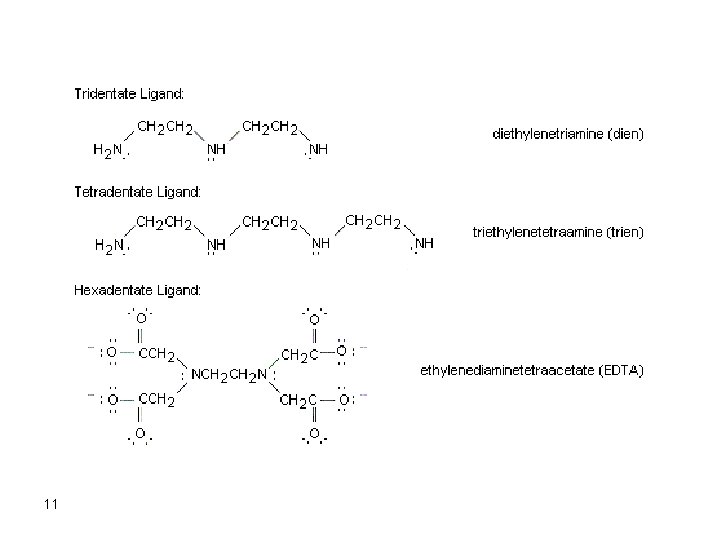
A ligand is called a monodentate if it donates a single pair of electrons (like : NH 3) while a bidentate ligand (like ethylenediamine, : NH 2 CH 2 H 2 N: ) donates two pairs of electrons. Ethylenediaminetetraacetic acid (EDTA) is a hexadentate ligand. The ligand can be as simple as ammonia which forms a complex with Cu 2+, for example, giving the complex Cu(NH 3)42+. When the ligand is a large organic molecule having two or more of the complexing groups, like EDTA, the ligand is called a chelating agent and the formed complex, in this case, is called a chelate. 10
11

12

The tendency of complex formation is controlled by the formation constant of the reaction between the metal ion (Lewis acid) and the ligand (Lewis base). As the formation constant increases, the stability of the complex increases. Let us look at the complexation reaction of Ag+ with NH 3: Ag+ + NH 3 D Ag(NH 3)+ + NH 3 D Ag(NH 3)2+ 13 kf 1 = [Ag(NH 3)+]/[Ag+][NH 3] kf 2 = [Ag(NH 3)2+]/[Ag(NH 3)+][NH 3]
![Kf 1 x kf 2 = [Ag(NH 3)2+]/[Ag+][NH 3]2 Now look at the overall Kf 1 x kf 2 = [Ag(NH 3)2+]/[Ag+][NH 3]2 Now look at the overall](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-14.jpg)
Kf 1 x kf 2 = [Ag(NH 3)2+]/[Ag+][NH 3]2 Now look at the overall reaction: Ag+ + 2 NH 3 D Ag(NH 3)2+ kf = [Ag(NH 3)2+]/[Ag+][NH 3]2 It is clear from inspection of the values of the kf that: Kf = kf 1 x kf 2 For a multistep complexation reaction we will always have the formation constant of the overall reaction equals the product of all step 14 wise formation constants

The formation constant is also called the stability constant and if the equilibrium is written as a dissociation the equilibrium constant in this case is called the instability constant. Ag(NH 3)2+ D Ag+ + 2 NH 3 kinst = [Ag+][NH 3]2/[Ag(NH 3)2+] Therefore, we have: Kinst = 1/kf 15

Stability of Metal-Ligand Complexes The stability of complexes is influenced by a number of factors related to the ligand metal ions. 1. Nature of the metal ion: Small ions with high charges lead to stronger complexes. 2. Nature of the ligand: The ligands forming chelates impart extra stability (chelon effect). For example the complex of nickel with a multidentate ligand is more stable than the one formed with ammonia. 3. Basicity of the ligand: Greater basicity of the ligand results in greater stability of the complex. 16

4. Size of chelate ring: The formation of five- or six-membered rings provides the maximum stability. 5. Number of metal chelate rings: The stability of the complex is directly related to the number of chelate rings formed between the ligand metal ion. Greater the number of such rings, greater is the stability. 7. Steric effects: These also play an important role in the stability of the complexes. 17

Lecture 32 Complexometric Reactions, Cont…. Calculations EDTA Equilibria 18

Example A divalent metal ion reacts with a ligand to form a 1: 1 complex. Find the concentration of the metal ion in a solution prepared by mixing equal volumes of 0. 20 M M 2+ and 0. 20 M ligand (L). kf = 1. 0 x 108. The formation constant is very large and essentially the metal ions will almost quantitatively react with the ligand. The concentration of metal ions and ligand will be half that given as mixing of equal volumes of the ligand metal ion will make their concentrations half the original concentrations since the volume was doubled. 19
![[M 2+] = 0. 10 M, [L] = 0. 10 M M 2+ + [M 2+] = 0. 10 M, [L] = 0. 10 M M 2+ +](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-20.jpg)
[M 2+] = 0. 10 M, [L] = 0. 10 M M 2+ + L D ML 2+ Kf = ( 0. 10 –x )/x 2 Assume 0. 10>>x since kf is very large 1. 0 x 108 = 0. 10/x 2, x = 3. 2 x 10 -5 Relative error = (3. 2 x 10 -5/0. 10) x 100 = 3. 2 x 10 -2 % The assumption is valid. [M 2+] = 3. 2 x 10 -5 M 20

Silver ion forms a stable 1: 1 complex with trien. Calculate the silver ion concentration at equilibrium when 25 m. L of 0. 010 M silver nitrate is added to 50 m. L of 0. 015 M trien. Kf = 5. 0 x 107 Ag+ + trien D Ag(trien)+ mmol Ag+ added = 25 x 0. 01 = 0. 25 mmol trien added = 50 x 0. 015 = 0. 75 mmol trien excess = 0. 75 – 0. 25 = 0. 50 [Trien] = 0. 5/75 M [Ag(trien)+] = 0. 25/75 M 21
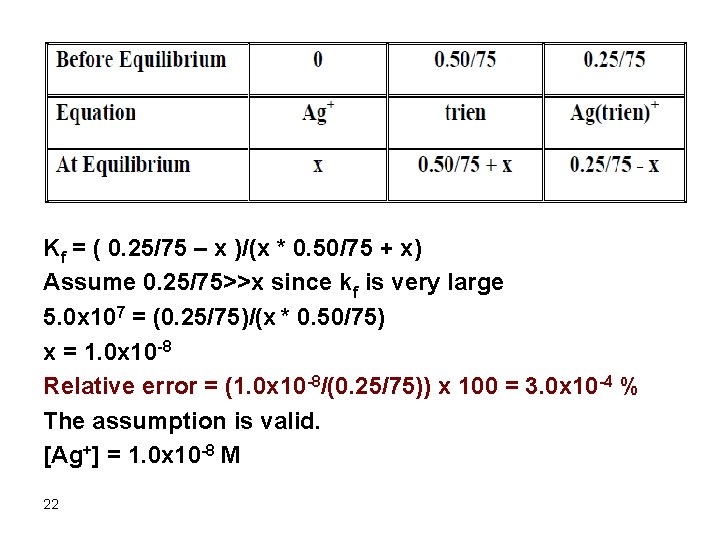
Kf = ( 0. 25/75 – x )/(x * 0. 50/75 + x) Assume 0. 25/75>>x since kf is very large 5. 0 x 107 = (0. 25/75)/(x * 0. 50/75) x = 1. 0 x 10 -8 Relative error = (1. 0 x 10 -8/(0. 25/75)) x 100 = 3. 0 x 10 -4 % The assumption is valid. [Ag+] = 1. 0 x 10 -8 M 22

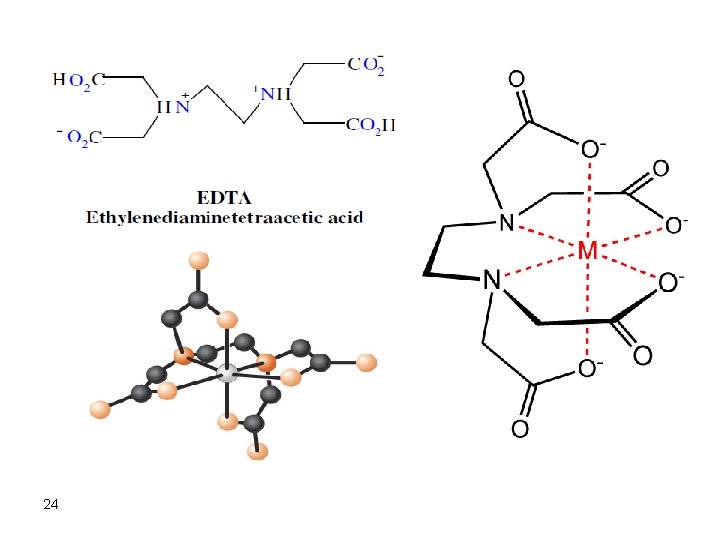
EDTA Titrations Ethylenediaminetetraacetic acid disodium salt (EDTA) is the most frequently used chelate in complexometric titrations. Usually, the disodium salt is used due to its good solubility. EDTA is used for titrations of divalent and polyvalent metal ions. The stoichiometry of EDTA reactions with metal ions is usually 1: 1. Therefore, calculations involved are simple and straightforward. Since EDTA is a polydentate ligand, it is a good chelating agent and its chelates with 23 metal ions have good stability.
24

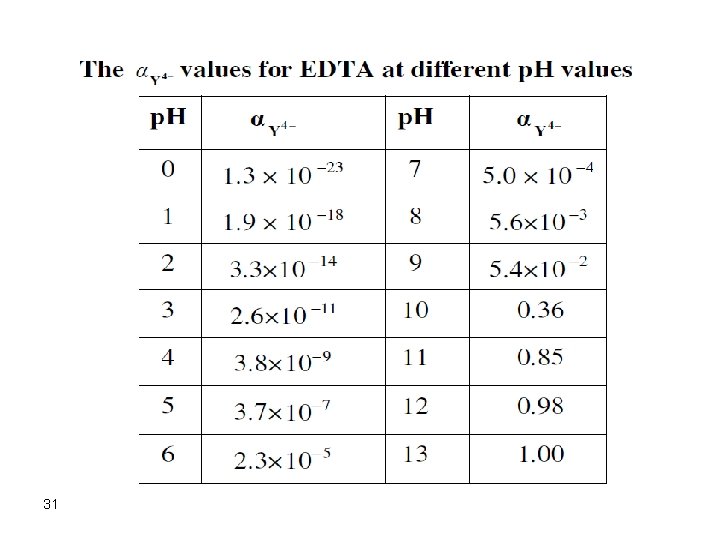
EDTA Equilibria EDTA can be regarded as H 4 Y where in solution we will have, in addition to H 4 Y, the following species: H 3 Y-, H 2 Y 2 -, HY 3 -, and Y 4 -. The amount of each species depends on the p. H of the solution where: a 4 = [Y 4 -]/CT where: CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2 -] + [HY 3 -] + [Y 4 -] a 4 = ka 1 ka 2 ka 3 ka 4/([H+]4 + ka 1 [H+]3 + ka 1 ka 2[H+]2 + ka 1 ka 2 ka 3[H+] + ka 1 ka 2 ka 3 ka 4) The species Y 4 - is the ligand species in EDTA titrations and thus should be looked at carefully. 25

26

The Formation Constant Reaction of EDTA with a metal ion to form a chelate is a simple reaction. For example, EDTA reacts with Ca 2+ ions to form a Ca-EDTA chelate forming the basis for estimation of water hardness. The reaction can be represented by the following equation: Ca 2+ + Y 4 - = Ca. Y 2 kf = 5. 0 x 1010 Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] The formation constant is very high and the reaction between Ca 2+ and Y 4 - can be considered quantitative. Therefore, if equivalent amounts of Ca 2+ and Y 4 - were mixed together, an equivalent amount of Ca. Y 2 - will be formed. 27

Formation Constants for EDTA Complexes Cation KMY Ag+ Mg 2+ Ca 2+ Sr 2+ Ba 2+ Mn 2+ Fe 2+ Co 2+ Ni 2+ 2. 1 x 107 4. 9 x 108 5. 0 x 1010 4. 3 x 108 5. 8 x 107 6. 2 x 1013 2. 1 x 1014 2. 0 x 1016 4. 2 x 1018 Cu 2+ Zn 2+ Cd 2+ Hg 2+ Pb 2+ Al 3+ Fe 3+ V 3+ Th 4+ 6. 3 x 1018 3. 2 x 1016 2. 9 x 1016 6. 3 x 1021 1. 1 x 1018 1. 3 x 1016 1. 3 x 1025 7. 9 x 1025 1. 6 x 1023 28

29 Minimum p. H for effective titrations of various metal ions with EDTA.

The question now is how to calculate the amount of Ca 2+ at equilibrium? Ca. Y 2 - D Ca 2+ + Y 4 However, [Ca 2+] # [Y 4 -] at this point since the amount of Y 4 - is p. H dependent and Y 4 - will disproportionate to form all the following species, depending on the p. H CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2 -] + [HY 3 -] + [Y 4 -] Where, CT is the sum of all species derived from Y 4 which is equal to [Ca 2+]. Therefore, the [Y 4 -] at equilibrium will be less than the [Ca 2+] and in fact it will only be a fraction of CT where: a 4 = [Y 4 -]/CT a 4 = ka 1 ka 2 ka 3 ka 4/([H+]4 + ka 1 [H+]3 + ka 1 ka 2[H+]2 + ka 1 ka 2 ka 3[H+] + ka 1 ka 2 ka 3 ka 4) 30
31

The Conditional Formation Constant We have seen that for the reaction Ca 2+ + Y 4 - D Ca. Y 2 - kf = 5. 0 x 1010 We can write the formation constant expression Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] However, we do not know the amount of Y 4 - at equilibrium but we can say that since a 4 = [Y 4 -]/CT, then we have: [Y 4 -] = a 4 CT Substitution in the formation constant expression we get: Kf = [Ca. Y 2 -]/[Ca 2+]a 4 CT or at a given p. H we can write Kf' = [Ca. Y 2 -]/[Ca 2+]CT Where Kf' is called the conditional formation constant. 32 It is conditional since it is now dependent on p. H.

Titration Curves In most cases, a titration is performed by addition of the titrant (EDTA) to the metal ion solution adjusted to appropriate p. H and in presence of a suitable indicator. The break in the titration curve is dependent on: 1. The value of the formation constant. 2. The concentrations of EDTA and metal ion. 3. The p. H of the solution As for acid-base titrations, the break in the titration curve increases as kf increases and as the concentration of reactants is increased. The p. H effect on the break of the titration curve is such that sharper breaks are obtained at higher p. H values. 33
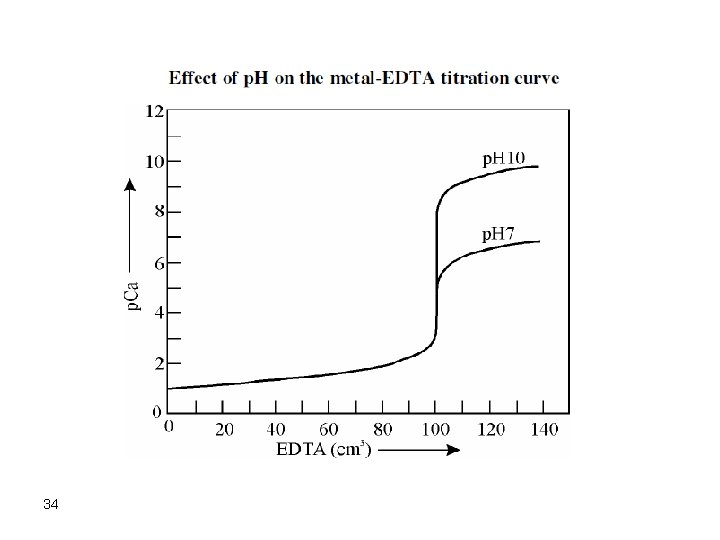
34
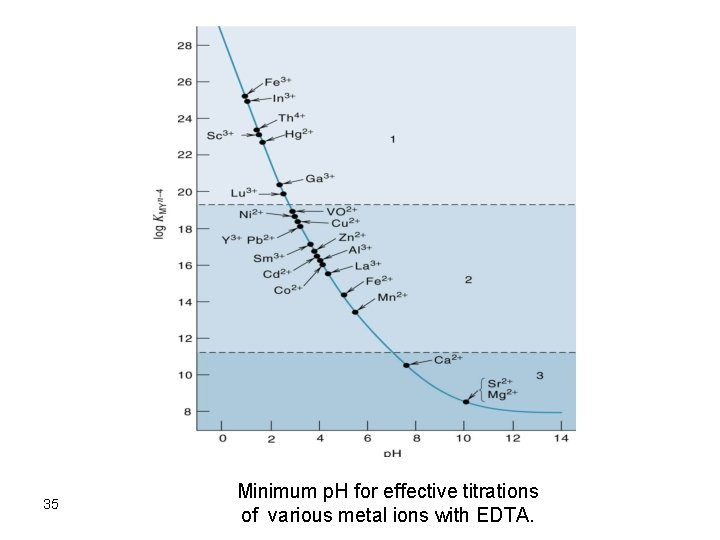
35 Minimum p. H for effective titrations of various metal ions with EDTA.

Lecture 33 Complexometric Titrations, Cont… Complexometric Indicators 36

Indicators The indicator is usually a weaker chelate forming ligand. The indicator has a color when free in solution and has a clearly different color in the chelate. The following equilibrium describes the function of an indicator (H 3 In) in a Mg 2+ reaction with EDTA: Mg. In- (Color 1) + Y 4 - D Mg. Y 2 - + In 3 - (Color 2) 37
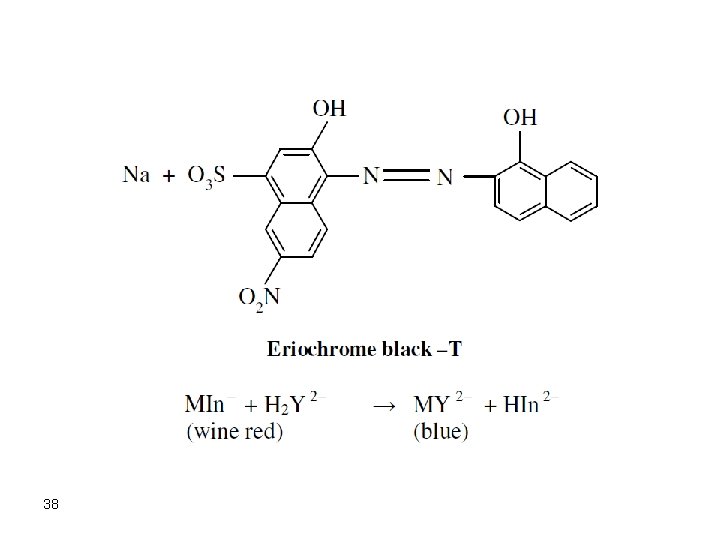
38
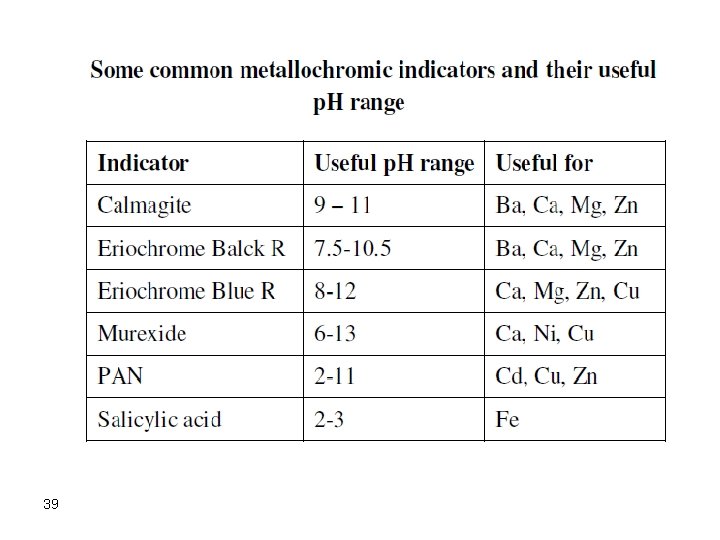
39

Problems Associated with Complexometric Indicators There could be some complications which may render some complexometric titrations useless or have great uncertainties. Some of these problems are discussed below: 1. Slow reaction rates In some EDTA titrations, the reaction is not fast enough to allow acceptable and successful determination of a metal ion. An example is the titration of Cr 3+ where direct titration is not possible. The best way to overcome this problem is to perform a back titration. However, we are faced with the problem of finding a suitable indicator that is weaker than the chelate but is not extremely weak to be displaced at the first drop of the titrant. 40
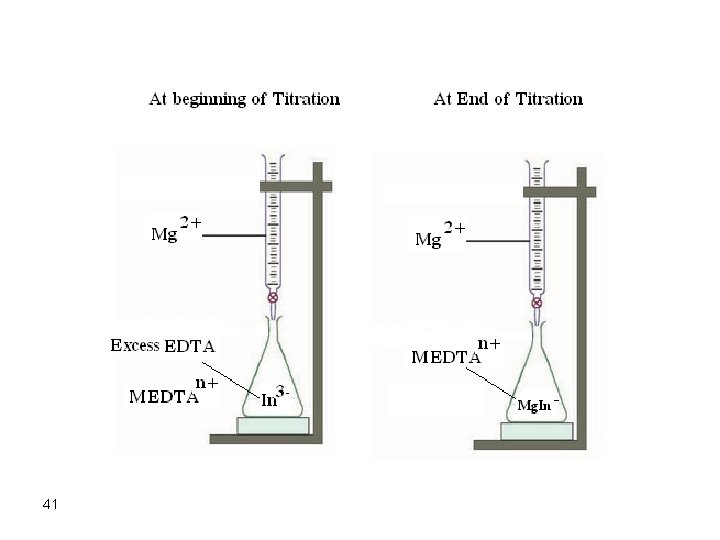
41

2. Lack of a suitable indicator This is the most severe problem in EDTA titrations and one should be critical about this issue and pay attention to the best method which may be used to overcome this problem. First let us take a note of the fact that Mg 2+-EDTA titration has excellent indicators that show very good change in color at the end point. Look at the following situations: 42

a. A little of a known standard Mg 2+ is added to the metal ion of interest. Now the indicator will form a clear cut color with magnesium ions. Titration of the metal ion follows and after it is over, added EDTA will react with Mg-In chelate to release the free indicator, thus changing color. This procedure requires performing the same titration on a blank containing the same amount of Mg 2+. 43
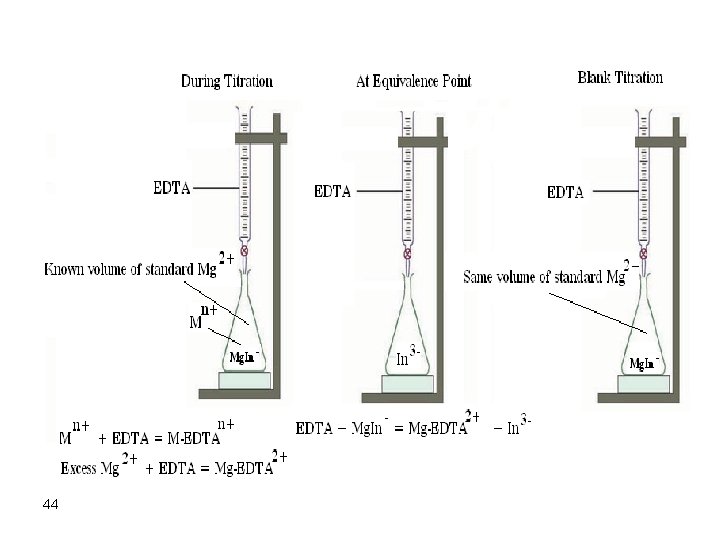
44

b. A blank experiment will not be necessary if we add a little of Mg-EDTA complex to the metal ion of interest. The metal ion will replace the Mg 2+ in the Mg-EDTA complex thus releasing Mg 2+ which immediately forms a good color with the indicator in solution. No need to do any corrections since the amount of EDTA in the added complex is exactly equal to the Mg 2+ in the complex. 45

46

c. If it is not easy to get a Mg-EDTA complex, just add a little Mg 2+ to the EDTA titrant. Standardize the EDTA and start titration. At the very first point of EDTA added, some Mg 2+ is released forming a chelate with the indicator and thus giving a clear color. It is wise to consult the literature for suitable indicators of a specific titration. There a lot of data and information on titrations of all metals you may think of. Therefore, use this wealth of information to conduct successful EDTA titrations. 47
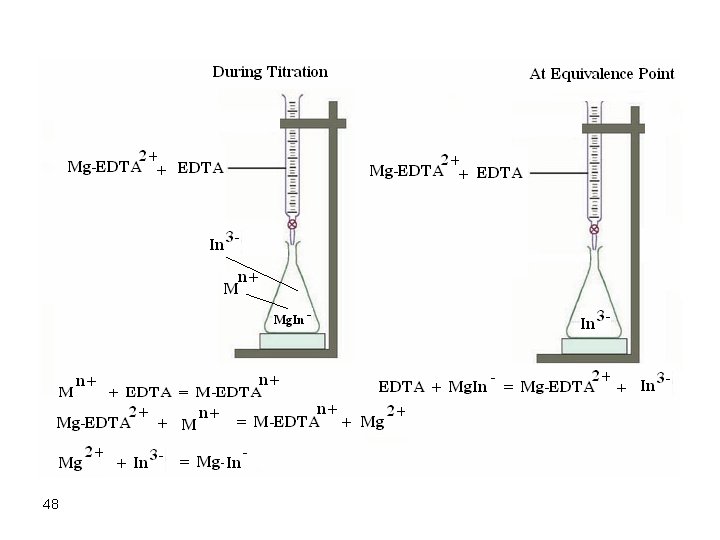
48

Example Find the concentrations of all species in solution at equilibrium resulting from mixing 50 m. L of 0. 200 M Ca 2+ with 50 m. L of 0. 100 M EDTA adjusted to p. H 10. a 4 at p. H 10 is 0. 35. kf = 5. 0 x 1010 Solution Ca 2+ + Y 4 - g Ca. Y 2 mmol Ca 2+ added = 0. 200 x 50 = 10. 0 mmol EDTA added = 0. 100 x 50 = 5. 00 mmol Ca 2+ excess = 10. 0 – 5. 00 = 5. 00 [Ca 2+]excess = 5. 00/100 = 0. 050 M mmol Ca. Y 2 - formed = 5. 00 [Ca. Y 2 -] = 5. 00/100 = 0. 050 Ca. Y 2 - D Ca 2+ + Y 449
![CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2 CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-50.jpg)
CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2 -] + [HY 3 -] + [Y 4 -] Kf = [Ca. Y 2 -]/[Ca 2+]a 4 CT [Ca 2+] = CT Using the same type of calculation we are used to perform, one can write the following: 50
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 = Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 =](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-51.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 = (0. 05 – x)/((0. 050 + x)* a 4 x) assume that 0. 05>>x x = 5. 6 x 10 -11 Relative error will be very small value The assumption is valid [Ca 2+] = 0. 050 + x = 0. 050 M [Ca. Y 2 -] = 0. 050 – x = 0. 050 M [Y 4 -] = 0. 35 * 5. 6 x 10 -11 = 1. 9 x 10 -11 M 51

Example Calculate the p. Ca of a solution at p. H 10 after addition of 100 m. L of 0. 10 M Ca 2+ to 100 m. L of 0. 10 M EDTA. a 4 at p. H 10 is 0. 35. kf = 5. 0 x 1010 Solution Ca 2+ + Y 4 - = Ca. Y 2 mmol Ca 2+ = 0. 10 x 100 = 10 mmol EDTA = 0. 10 x 100 = 10 mmol Ca. Y 2 - = 10 [Ca. Y 2 -] = 10/200 = 0. 05 M Therefore, Ca 2+ will be produced from partial 52 dissociation of the complex

Ca 2+ + Y 4 - D Ca. Y 2 CT = [H 4 Y] + [H 3 Y-] + [H 2 Y 2 -] + [HY 3 -] + [Y 4 -] Kf = [Ca. Y 2 -]/[Ca 2+]a 4 CT [Ca 2+] = CT 5. 0 x 1010 = 0. 05/([Ca 2+]2 x 0. 35) [Ca 2+] = 1. 7 x 10 -6 M p. Ca = 5. 77 Using the same type of calculation we are used to perform, one can write the following: 53
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 = Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 =](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-54.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5. 0 x 1010 = (0. 05 – x)/(x* a 4 x) assume that 0. 05>>x x = 1. 7 x 10 -6 Relative error = (1. 7 x 10 -6/0. 05) x 100 = 3. 4 x 10 -3% [Ca 2+] = 1. 7 x 10 -6 M p. Ca = 5. 77 54

Lecture 34 Complexometric Reactions, Cont… EDTA Titrations 55

Calculate the titer of a 0. 100 M EDTA solution in terms of mg Ca. CO 3 (FW = 100. 0) per m. L EDTA The EDTA concentration is 0. 100 mmol/m. L, therefore, the point here is to calculate the mg Ca. CO 3 reacting with 0. 100 mmol EDTA. We know that EDTA reacts with metal ions in a 1: 1 ratio. Therefore 0. 100 mmol EDTA will react with 0. 100 mmol Ca. CO 3 = mmol EDTA mg Ca. CO 3/100 = 0. 1 * 1 mg Ca. CO 3 = 10. 0 56

Example An EDTA solution is standardized against high purity Ca. CO 3 by dissolving 0. 3982 g of Ca. CO 3 in HCl and adjusting the p. H to 10. The solution is then titrated with EDTA requiring 38. 26 m. L. Find the molarity of EDTA. Solution EDTA reacts with metal ions in a 1: 1 ratio. Therefore, mmol Ca. CO 3 = mmol EDTA mg/FW = Molarity x Vm. L 398. 2/100. 0 = M x 38. 26, MEDTA = 0. 1041 57

Example Find the concentration of Ca 2+ in a 20 m. L of 0. 20 M solution at p. H 10 after addition of 100 m. L of 0. 10 M EDTA. a 4 at p. H 10 is 0. 35. kf = 5 x 1010 Solution Initial mmol Ca 2+ = 0. 20 x 20 = 4. 0 mmol EDTA added = 0. 10 x 100 = 10 mmol EDTA excess = 10 – 4. 0 = 6. 0 CT = 6. 0/120 = 0. 050 M mmol Ca. Y 2 - = 4. 0 [Ca. Y 2 -] = 4. 0/120 = 0. 033 M Ca 2+ + Y 4 - D Ca. Y 2 kf = 5. 0 x 1010 58
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-59.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. 033 – x)/(x* a 4(0. 050 + x) ) assume that 0. 033>>x x = 3. 9 x 10 -11 The assumption is valid by inspection of the values and no need to calculate the relative error. [Ca 2+] = 3. 9 x 10 -11 M p. Ca = 10. 41 59

Example Find p. Ca in a 100 m. L solution of 0. 10 M Ca 2+ at p. H 10 after addition of 0, 25, 50, 100, 150, and 200 m. L of 0. 10 M EDTA. a 4 at p. H 10 is 0. 35. kf = 5 x 1010 Solution Again, we should remember that EDTA reactions with metal ions are 1: 1 reactions. Therefore, we have: Ca 2+ + Y 4 - D Ca. Y 2 kf = 5. 0 x 1010 1. After addition of 0 m. L EDTA [Ca 2+] = 0. 10 M p. Ca = 1. 00 60

2. After addition of 25 m. L EDTA Initial mmol Ca 2+ = 0. 10 x 100 = 10 mmol EDTA added = 0. 10 x 25 = 2. 5 mmol Ca 2+ left = 10 – 2. 5 = 7. 5 [Ca 2+]left = 7. 5/125 = 0. 06 M In fact, this calcium concentration is the major source of calcium in solution since the amount of calcium coming from dissociation of the chelate is very small, especially in presence of Ca 2+ left in solution. However, let us calculate the amount of calcium released from the chelate: mmol Ca. Y 2 - formed = 2. 5 [Ca. Y 2 -] = 2. 5/125 = 0. 02 M 61
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-62.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. 02 – x)/((0. 06 + x) * a 4 x) assume that 0. 02>>x x = 1. 9 x 10 -11 The assumption is valid even without verification. [Ca 2+] = 0. 06 + 1. 9 x 10 -11 = 0. 06 M p. Ca = 1. 22 62

3. After addition of 50 m. L EDTA mmol EDTA added = 0. 10 x 50 = 5. 0 mmol Ca 2+ left = 10 – 5. 0 = 5. 0 [Ca 2+]left = 5. 0/150 = 0. 033 M We will see by similar calculation as in step above that the amount of Ca 2+ coming from dissociation of the chelate is exceedingly small as compared to amount left. However, for the sake of practice let us perform the calculation: mmol Ca. Y 2 - formed = 5. 0 [Ca. Y 2 -] = 5. 0/150 = 0. 033 M 63
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-64.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. 033 – x)/((0. 033 + x)* a 4 x) assume that 0. 033>>x x = 5. 7 x 10 -11 The assumption is valid even without verification. [Ca 2+] = 0. 033+ 5. 7 x 10 -11 = 0. 033 M p. Ca = 1. 48 64

4. After addition of 100 m. L EDTA mmol EDTA added = 0. 10 x 100 = 10 mmol Ca 2+ left = 10 – 10 = 0 This is the equivalence point. The only source for Ca 2+ is the dissociation of the Chelate mmol Ca. Y 2 - formed = 10 [Ca. Y 2 -] = 10/200 = 0. 05 M Ca 2+ + Y 4 - D Ca. Y 2 - kf = 5. 0 x 1010 65
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 105 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 105 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-66.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 105 = (0. 05 – x)/(x* a 4 x) assume that 0. 05>>x, x = 1. 7 x 10 -6 Relative error = (1. 7 x 10 -6/0. 05) x 100 = 3. 4 x 10 -3% [Ca 2+] = 1. 7 x 10 -6 M, p. Ca = 5. 77 5. After addition of 150 m. L EDTA mmol EDTA added = 0. 10 x 150 = 15 mmol EDTA excess = 15 – 10 = 5. 0 CT = 5. 0/250 = 0. 02 M mmol Ca. Y 2 - = 10 [Ca. Y 2 -] = 10/250 = 0. 04 M Ca 2+ + Y 4 - D Ca. Y 2 - kf = 5. 0 x 1010 66
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-67.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. 04 – x)/(x* a 4(0. 02 + x) ) assume that 0. 02>>x x = 1. 1 x 10 -10 The assumption is valid [Ca 2+] = 1. 1 x 10 -10 M p. Ca = 9. 95 67

6. After addition of 200 m. L EDTA mmol EDTA added = 0. 10 x 200 = 20 mmol EDTA excess = 20 – 10 = 10 CT = 10/300 = 0. 033 M mmol Ca. Y 2 - = 10 [Ca. Y 2 -] = 10/300 = 0. 033 M Ca 2+ + Y 4 - D Ca. Y 2 - kf = 5. 0 x 1010 68
![Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0.](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-69.jpg)
Kf = [Ca. Y 2 -]/[Ca 2+][Y 4 -] 5 x 1010 = (0. 033 – x)/(x* a 4(0. 033 + x) ) assume that 0. 033>>x x = 5. 7 x 10 -11 The assumption is undoubtedly valid [Ca 2+] = 5. 7 x 10 -11 M p. Ca = 10. 24 69

Fractions of Dissociating Species in Polyligand Complexes When polyligand complexes are dissociated in solution, metal ions, ligand, and intermediates are obtained in equilibrium with the complex. For example, look at the following equilibria Ag+ + NH 3 D Ag(NH 3)+ + NH 3 D Ag(NH 3)2+ kf 1 = [Ag(NH 3)+]/[Ag+][NH 3] kf 2 = [Ag(NH 3)2+]/[Ag(NH 3)+][NH 3] We have Ag+, NH 3, Ag(NH 3)+, and Ag(NH 3)2+ all present in solution at equilibrium where CAg = [Ag+] + [Ag(NH 3)2+] 70
![The fraction of each Ag+ species can be defined as: b 0 = [Ag+]/ The fraction of each Ag+ species can be defined as: b 0 = [Ag+]/](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-71.jpg)
The fraction of each Ag+ species can be defined as: b 0 = [Ag+]/ CAg b 1 = [Ag(NH 3)+]/ CAg b 2 = [Ag(NH 3)2+]/ CAg As seen for fractions of a polyprotic acid dissociating species, one can look at the b values as b 0 for the fraction with zero ligand (free metal ion, Ag+), b 1 as the fraction of the species having one ligand (Ag(NH 3)+) while b 2 as the fraction containing two ligands (Ag(NH 3)2+). The sum of all fractions will necessarily add up to unity (b 0 + b 1 + b 2 = 1) 71

For the case of b 0, we make all terms as a function of Ag+ since b 0 is a function of Ag+. We use the equilibrium constants of each step: CAg = [Ag+] + [Ag(NH 3)2+] kf 1 = [Ag(NH 3)+]/[Ag+][NH 3] [Ag(NH 3)+] = kf 1 [Ag+][NH 3] Kf 1 x kf 2 = [Ag(NH 3)2+]/[Ag+][NH 3]2 [Ag(NH 3)2+] = Kf 1 x kf 2 [Ag+][NH 3]2 Substitution in the CAg relation gives: CAg = [Ag+] + kf 1 [Ag+][NH 3] + Kf 1 x kf 2 [Ag+][NH 3]2 CAg = [Ag+]( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) [Ag+]/ CAg = 1/( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) 72

The inverse of this equation gives: b 0 = 1/ ( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) If we use the same procedure for the derivation of relations for other fractions we will get the same denominator but the nominator will change according to the species of interest: b 0 = 1/ ( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) b 1 = kf 1 [NH 3] / ( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) b 2 = Kf 1 kf 2 [NH 3]2/ ( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) 73

Example Calculate the concentration of the different ion species of silver for 0. 010 M Ag+ in a 0. 10 M NH 3 solution. Kf 1 = 2. 5 x 103, kf 2 = 1. 0 x 104 Solution Ag+ + 2 NH 3 D Ag(NH 3)2+ kf = kf 1*kf 2 = 2. 5*107 [NH 3]left = 0. 08 M 74
![b 0 = 1/ ( 1 + kf 1 [NH 3] + Kf 1 b 0 = 1/ ( 1 + kf 1 [NH 3] + Kf 1](http://slidetodoc.com/presentation_image_h/8a89c97a2f0273fc1113702a23117b0a/image-75.jpg)
b 0 = 1/ ( 1 + kf 1 [NH 3] + Kf 1 kf 2 [NH 3]2) Substitution in the above equation yields: b 0 = 1/ ( 1 + 2. 5 x 103 * 0. 08 + 2. 5 x 103 * 1. 0 x 104 *( 0. 08)2) b 0 = 6. 2 x 10 -6 b 0 = [Ag+]/ CAg 6. 2 x 10 -6 = [Ag+]/0. 010 [Ag+] = 6. 2 x 10 -8 M In the same manner calculations give: b 1 = 1. 2 x 10 -3 b 1 = [Ag(NH 3)+]/ CAg 1. 2 x 10 -3 = [Ag(NH 3)+]/ 0. 010 [Ag(NH 3)+] = 1. 2 x 10 -5 M • 75

b 2 = 0. 999 or 1. 0 if we consider significant figures. b 2 = [Ag(NH 3)2+]/ CAg 1. 0 = [Ag(NH 3)2+]/ 0. 010 [Ag(NH 3)2+] = 0. 010 M Therefore, it is clear that most Ag+ will be in the complex form Ag(NH 3)2+ since the formation constant is large for the overall reaction: Kf = kf 1*kf 2 Kf = 2. 5 x 103 * 1. 0 x 104 = 2. 5 x 107 76
- Slides: 76