LECTURE 3 Basics of atmospheric composition and physics

LECTURE 3 Basics of atmospheric composition and physics AOSC 434 AIR POLLUTION Russell R. Dickerson


Ib. ATMOSPHERIC PHYSICS Seinfeld & Pandis: Chapter 1 Finlayson-Pitts: Chapter 2 1. Pressure: exponential decay 2. Composition 3. Temperature The motion of the atmosphere is caused by differential heating, that is some parts of the atmosphere receive more radiation than others and become unstable.
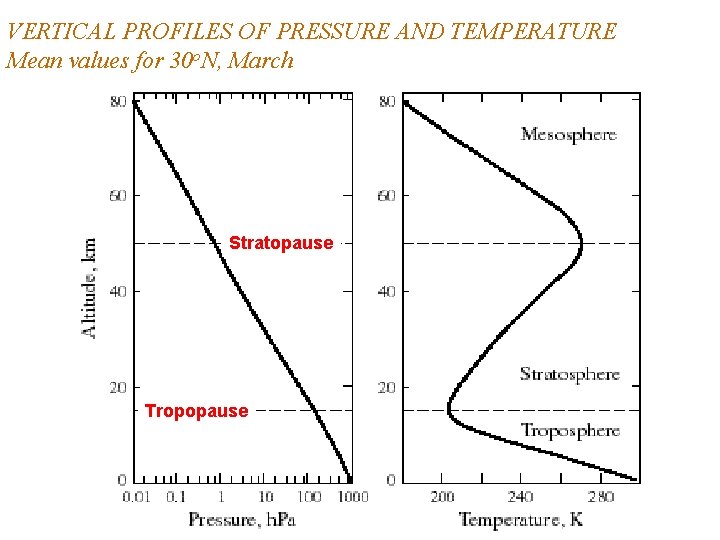
VERTICAL PROFILES OF PRESSURE AND TEMPERATURE Mean values for 30 o. N, March Stratopause Tropopause
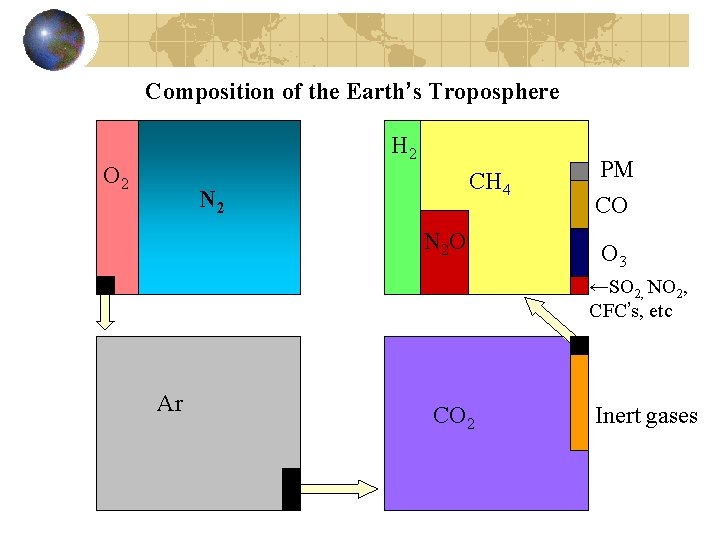
Composition of the Earth’s Troposphere H 2 O 2 CH 4 N 2 O PM CO O 3 ←SO 2, NO 2, CFC’s, etc Ar CO 2 Inert gases

Banded iron formation. Smithsonian in DC
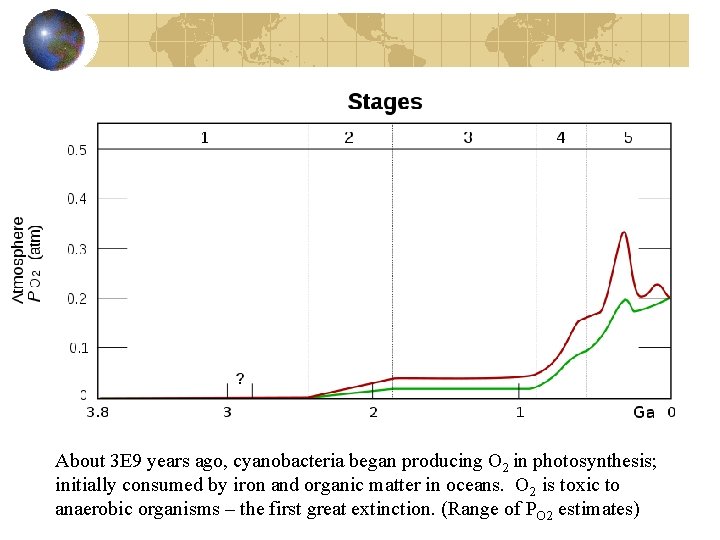
About 3 E 9 years ago, cyanobacteria began producing O 2 in photosynthesis; initially consumed by iron and organic matter in oceans. O 2 is toxic to anaerobic organisms – the first great extinction. (Range of PO 2 estimates)

Units of pressure: 1. 00 atm. = 14. 7 psi = 1, 013 mb = 760 mm Hg (torr) = 33. 9 ft H₂O = 29. 9” Hg = 101325 Pa A Pascal is a Nm⁻² (kg m s-2 m-2 ) thus h. Pa = mb. Units of Volume: liter, cc, ml, m³ Units of temp: K, °C Units of R: 0. 08206 L atm mole-1 K-1 8. 31 J mole-1 K-1 R’ = R/Mwt = 0. 287 J g-1 K-1 For a mole of dry air which has the mass 29 g. Problem for the student: calc Mwt. Wet (2% H₂O vapor by volume) air.

1 A. Derive Hypsometric Equation Start with The Ideal Gas Law PV = n. RT or P = ρR’T or P = R’T/α Where R’ = R/Mwt = MOLE WT. AIR ρ = DENSITY AIR (g/l) α = SPECIFIC VOL AIR = 1/ρ We assume that the pressure at any given altitude is due to the weight of the atmosphere above that altitude. The weight is mass times acceleration. P = W = mg But m = Vρ For a unit area V = Z P = Zρg

For a second, higher layer the difference in pressure can be related to the difference in height. d. P = − g ρ d. Z But ρ = P/R’T d. P = − Pg/R’T * d. Z For an isothermal atmosphere g/R’T is a constant. By integrating both sides of the equation from the ground (Z = 0. 0) to altitude Z we obtain:

Where H₀ = R’T/g we can rewrite this as: *HYPSOMETRIC EQUATION* Note: Scale Height: H₀ ~ 8 km for T = 273 K For each 8 km of altitude the pressure is down by e⁻¹ or one “e-fold. ” Problems for the student: Derive an expression for pressure as a function of altitude using base two and base ten instead of base e. Calculate the scale height for the atmospheres of Venus and Mars. Ans. base 2 = H₀*ln(2) = 5. 5 km base 10 = H₀*ln(10) = 18 km

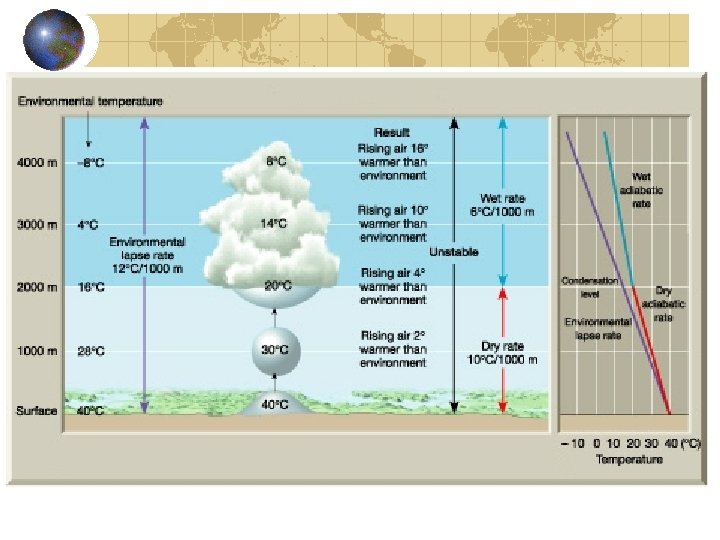
1 B. TEMPERATURE LAPSE RATE Going to the mountains in Shenandoah National Park the summer is a nice way to escape Washington’s heat. Why? Consider a parcel of air. If it rises it will expand cool. If we assume it exchanges no heat with the surroundings (a pretty good assumption, because air is a very poor conductor of heat) it will cool “adiabatically. ” Calculating the dry adiabatic lapse rate. First Law Thermodynamics*: d. U = đQ – đW Or đQ = d. U + đW = d. U + Pd. V *watch out for work done by the system or on the system.

WHERE U = Energy of system (also written E) Q = Heat across boundaries W = Work done by the system on the surroundings H = Internal heat or Enthalpy ASSUME: a) Adiabatic (d. Q = d. H = 0. 0) b) All work Pd. V work (remember α = 1/ρ) d. H = Cp d. T – α d. P Cpd. T = α d. P d. T = (α/Cp) d. P

Remember the Hydrostatic Equation OR Ideal Gas Law Result: This quantity, -g/Cp, is a constant in a dry atmosphere. It is called the dry adiabatic lapse rate and is given the symbol γ₀, defined as −d. T/d. Z. For a parcel of air moving adiabatically in the atmosphere:

Where Z₂ is higher than Z₁, but this presupposes that no heat is added to or lost from the parcel, and condensation, evaporation, and radiative heating can all produce a non-adiabatic system. The dry adiabatic lapse rate, g 0, is a general, thermodynamic property of the atmosphere and expresses the way a parcel of air will cool on rising or warm on falling, provided there is no exchange of heat with the surroundings and no water condensing or evaporating. The environmental lapse rate, G, is seldom equal to exactly the dry adiabatic lapse rate because radiative processes and phase changes constantly redistribute heat. The mean lapse rate is about 6. 5 K/km. Problem left to the students: Derive a new expression for the change in pressure with height for an atmosphere with a constant lapse rate,

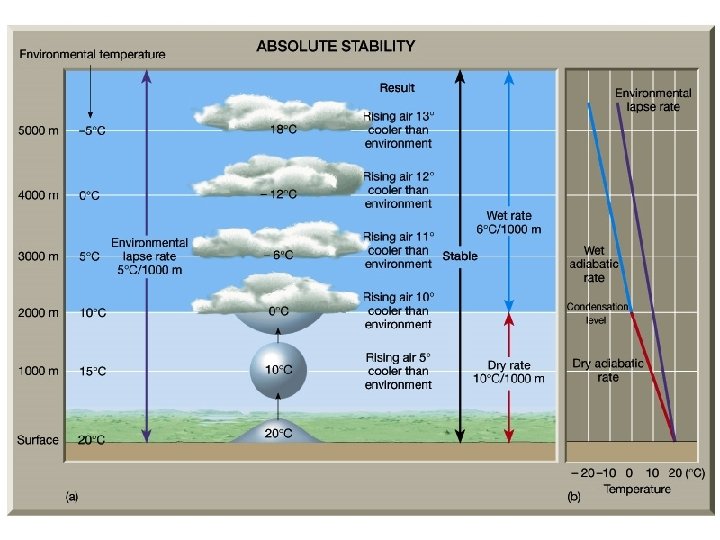
2. STABILITY AND THERMODYNAMIC DIAGRAMS Gray lines – thermodynamic property Black lines – measurements or soundings Three days Ga < γ₀ stable Gb = γ₀ neutrally stable Gc > γ₀ unstable On day a a parcel will cool more quickly than surroundings – air will be cooler and return to original altitude. On day b a parcel will always have same temperature as surroundings – no force of buoyancy. On day c a parcel will cool more slowly than surroundings – air will be warmer and rise.
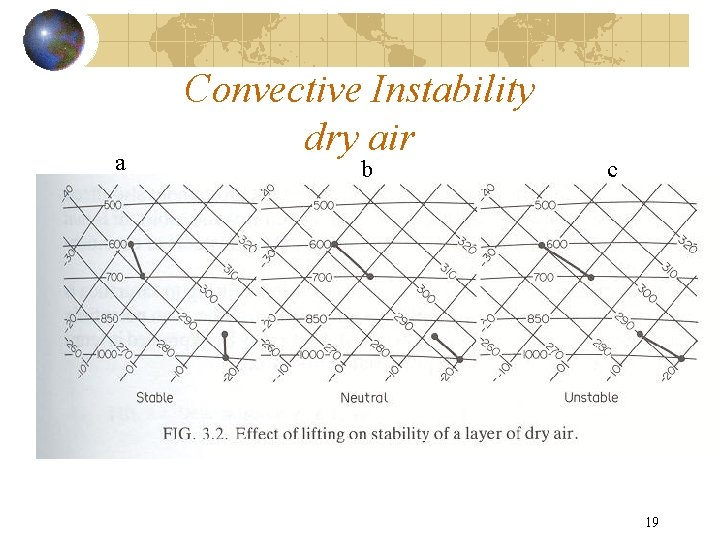
a Convective Instability dry air b c 19
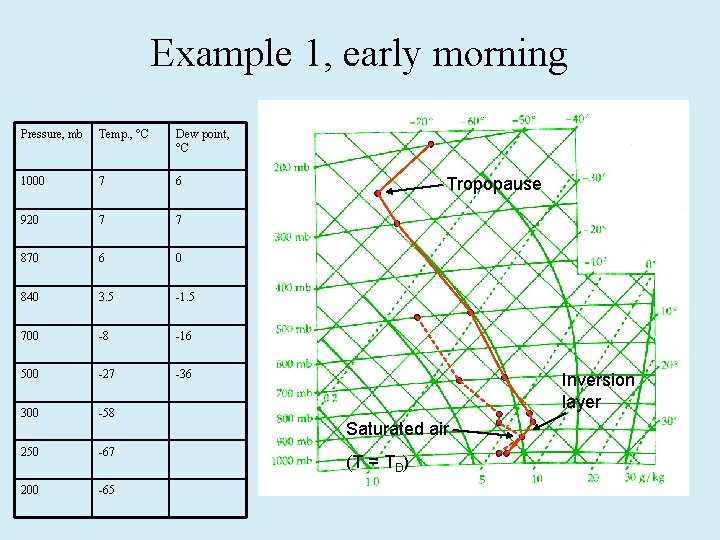
Example 1, early morning Pressure, mb Temp. , °C Dew point, °C 1000 7 6 920 7 7 870 6 0 840 3. 5 -1. 5 700 -8 -16 500 -27 -36 300 -58 250 -67 200 -65 Tropopause Inversion layer Saturated air (T = TD)
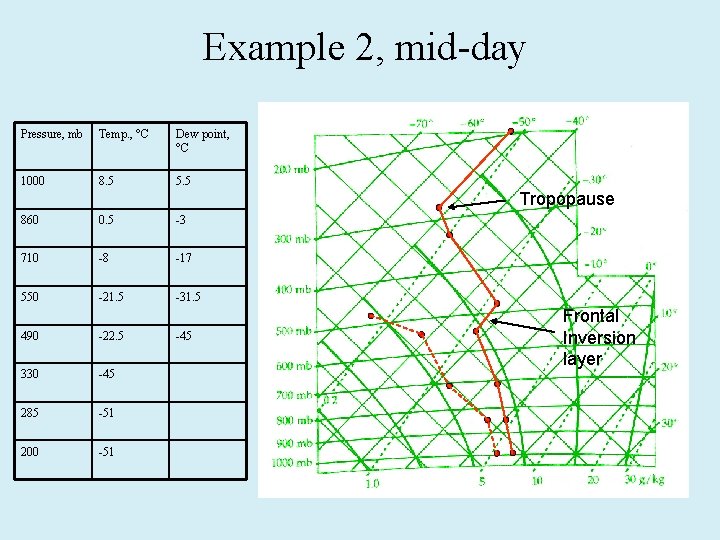
Example 2, mid-day Pressure, mb Temp. , °C Dew point, °C 1000 8. 5 5. 5 Tropopause 860 0. 5 -3 710 -8 -17 550 -21. 5 -31. 5 490 -22. 5 330 -45 285 -51 200 -51 -45 Frontal Inversion layer

Skew-T handout Very important for air pollution and mixing of emissions with free troposphere. Formation of thermal inversions. In the stratosphere the temperature increases with altitude, thus stable or stratified. DFN: Potential temperature, θ : The temperature that a parcel of air would have if it were brought to the 1000 h. Pa level (near the surface) in a dry adiabatic process. You can approximate it quickly, or a proper derivation from dq = Cpd. T – adp = 0 yields: θz = T (1000 h. Pa/Pz) R’/Cp = T (1000 h. Pa/Pz)0. 286
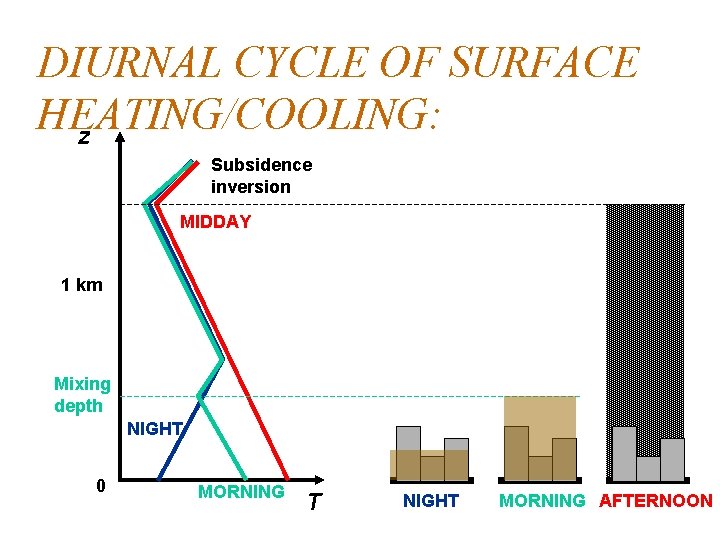
DIURNAL CYCLE OF SURFACE HEATING/COOLING: z Subsidence inversion MIDDAY 1 km Mixing depth NIGHT 0 MORNING T NIGHT MORNING AFTERNOON

If the parcel reaches saturation, the condensation of water adds heat, and the rising air cools at a new, slower rate, Gs. For details, see Wallace and Hobbs. Since Gs< Gd, we must also consider conditions between the two stability criteria for dry and saturated. G = parcel lapse (thermodynamically determined) rate. g = environment (observed) lapse rate. 24

Plume looping, Baltimore ~2 pm.



Plume Lofting, Beijing in Winter ~7 am.

High levels of air pollution in Beijing contain a large quantity of PM 2. 5. © i. Stock. Photo. com/beijingstory. Marshall J PNAS 2013; 110: 8756 -8756 © 2013 by National Academy of Sciences

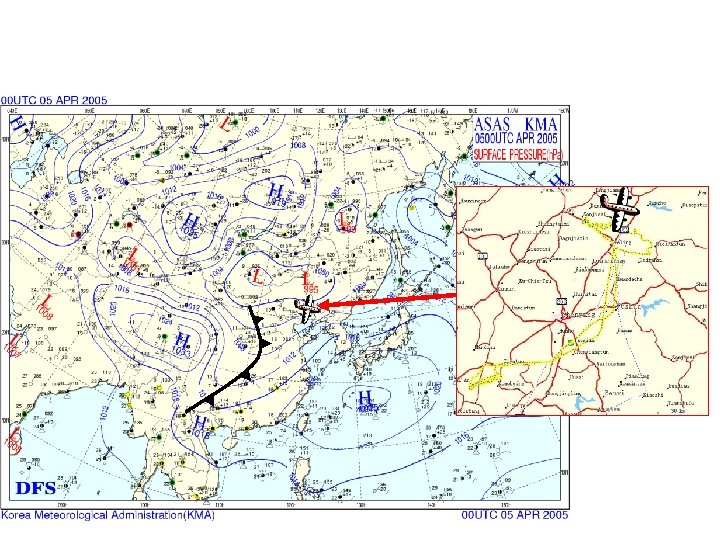
Chinese Y 12 (Twin Otter) for the airborne campaign over NE China April 2005
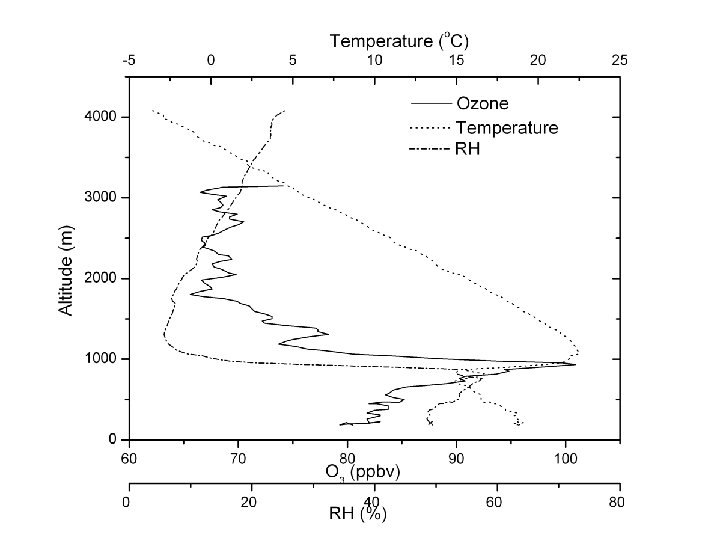
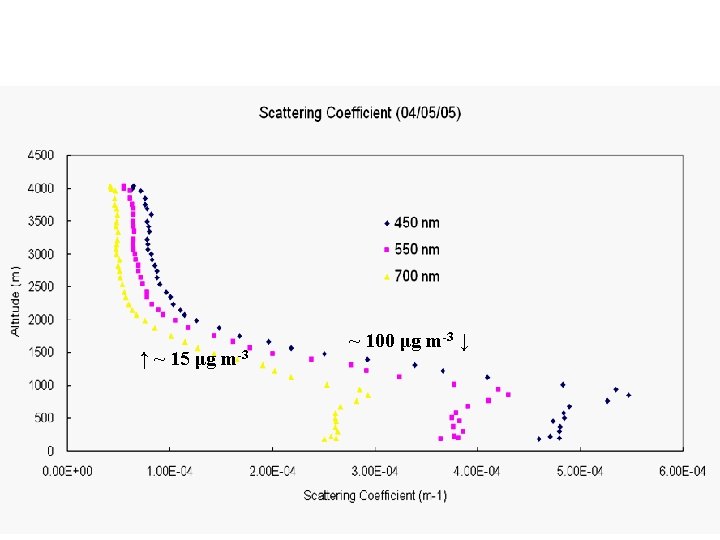
↑ ~ 15 μg m-3 ~ 100 μg m-3 ↓
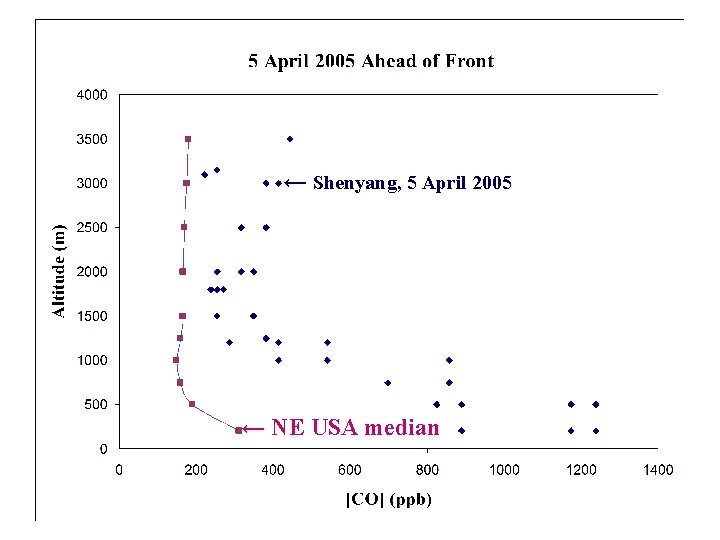
← Shenyang, 5 April 2005 ← NE USA median
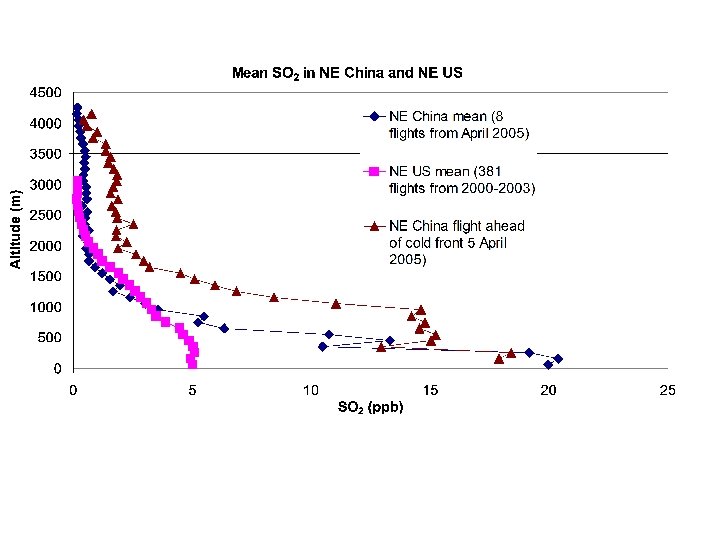

Preliminary Results from RF 6 Y 12 in Hebei, China 05/21/2016 PRELIMINARY DATA DO NOT CITE! Hao He, Xinrong Ren, Zhibin Wang, Fei Wang, Zhanqing Li Russ Dickerson

Fair weather Cu ~20 km southeast of Xingtai 5: 23 GMT (13: 23 LST) 5/21/2016. Aircraft alt 2500 m; heavy loadings of aerosols observed throughout PBL. Photo credit Hao He.

Picture of the Xingtai Supersite
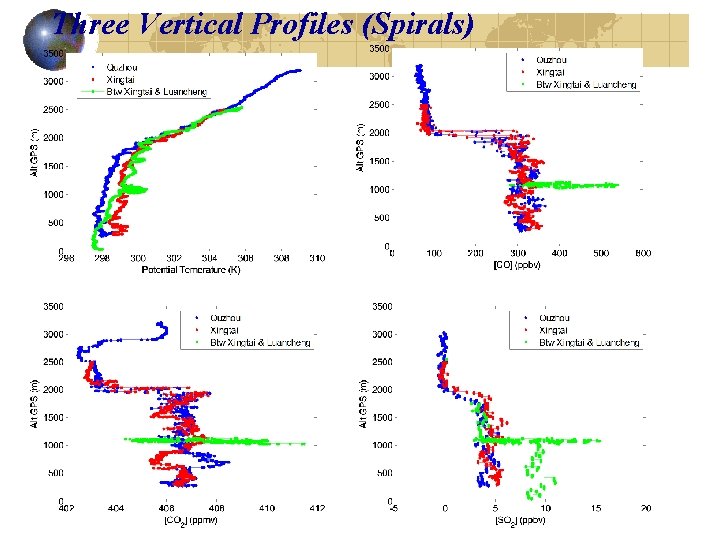
Three Vertical Profiles (Spirals)
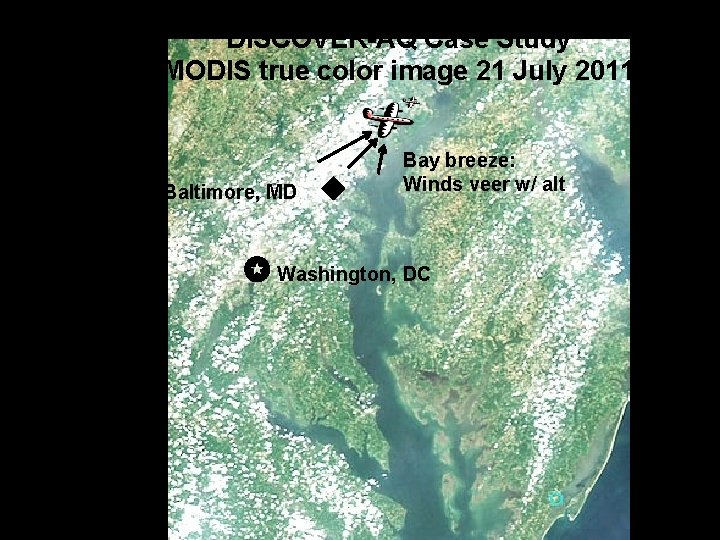
DISCOVER-AQ Case Study MODIS true color image 21 July 2011 Baltimore, MD Bay breeze: Winds veer w/ alt Was ✪ Washington, DC hing ton, DC
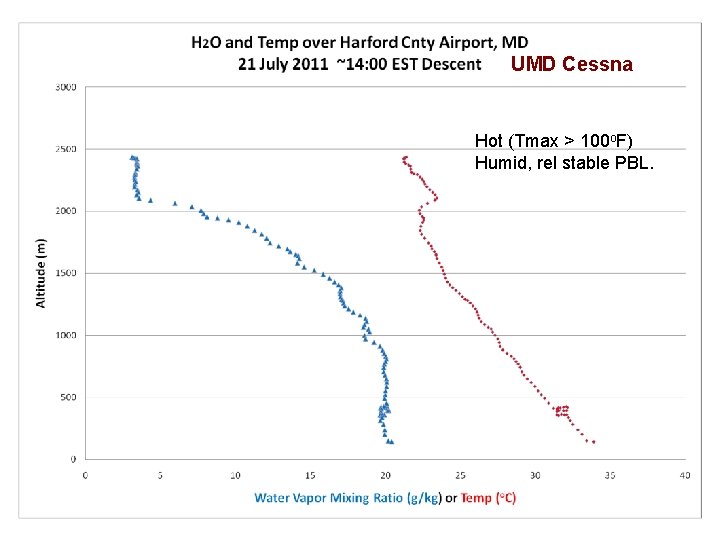
UMD Cessna Hot (Tmax > 100 o. F) Humid, rel stable PBL.
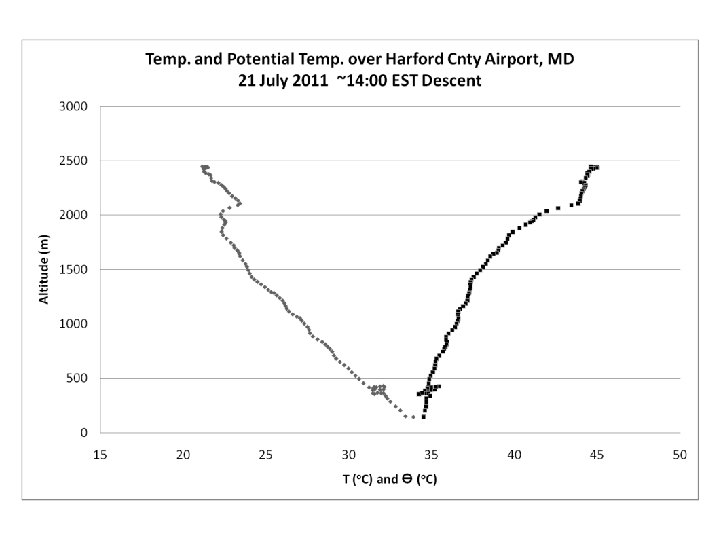
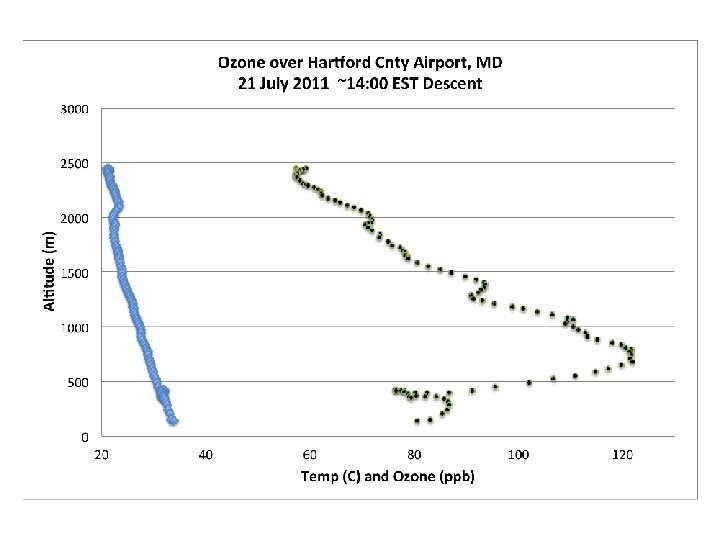
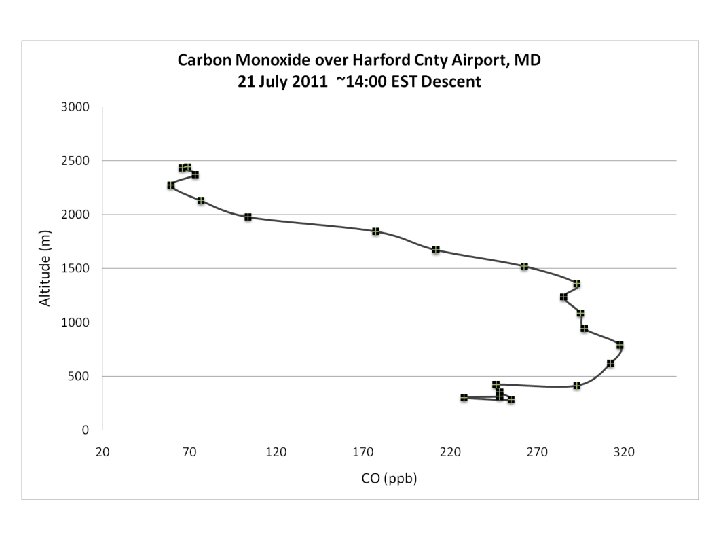
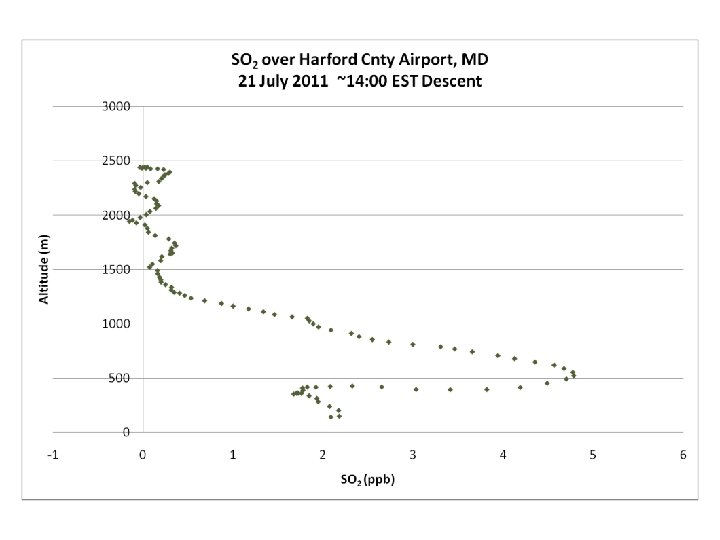
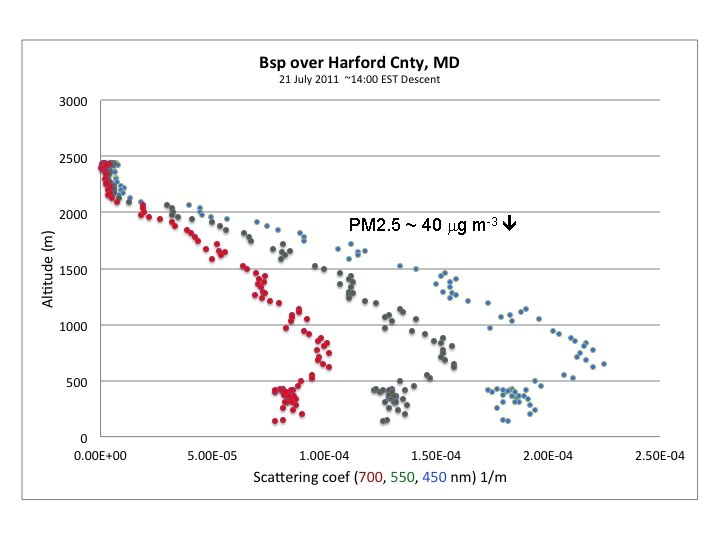
PM 2. 5 ~ 40 mg m-3
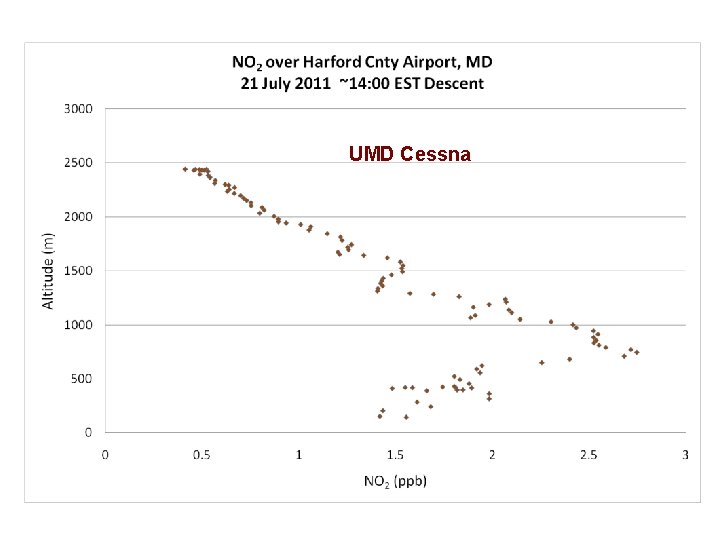
UMD Cessna

Atmospheric Circulation and Winds Copyright R. R. Dickerson 2011 48
ITCZ Copyright R. R. Dickerson 2011 49
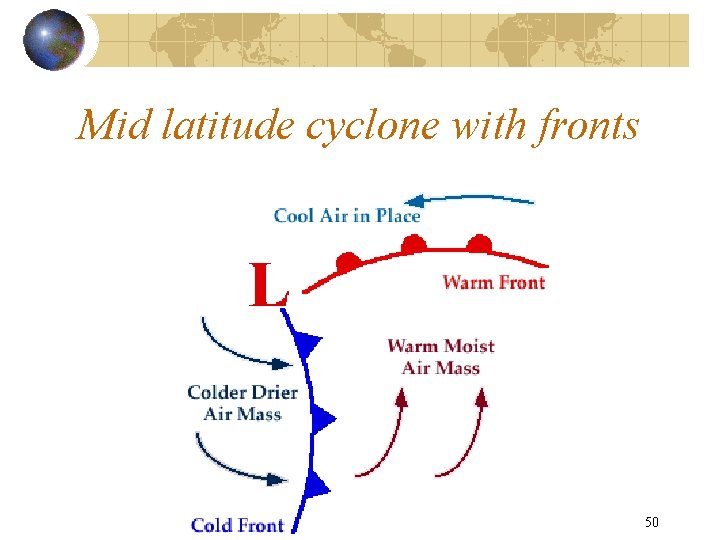
Mid latitude cyclone with fronts Copyright R. R. Dickerson 2011 50
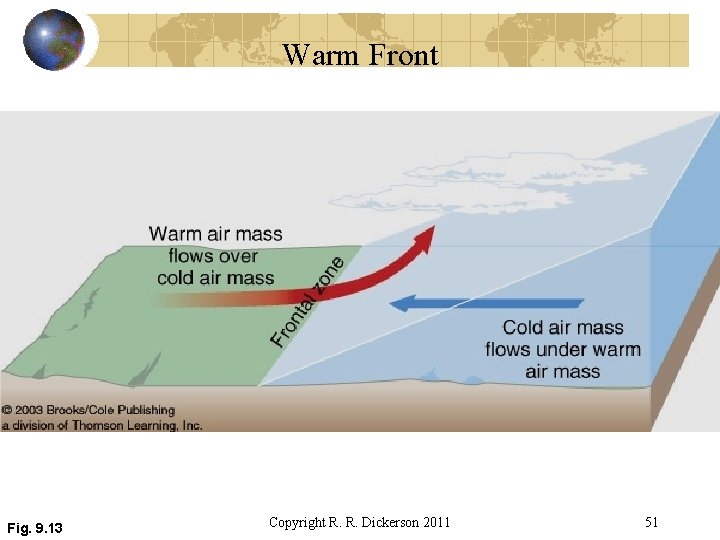
Warm Front Fig. 9. 13 Copyright R. R. Dickerson 2011 51
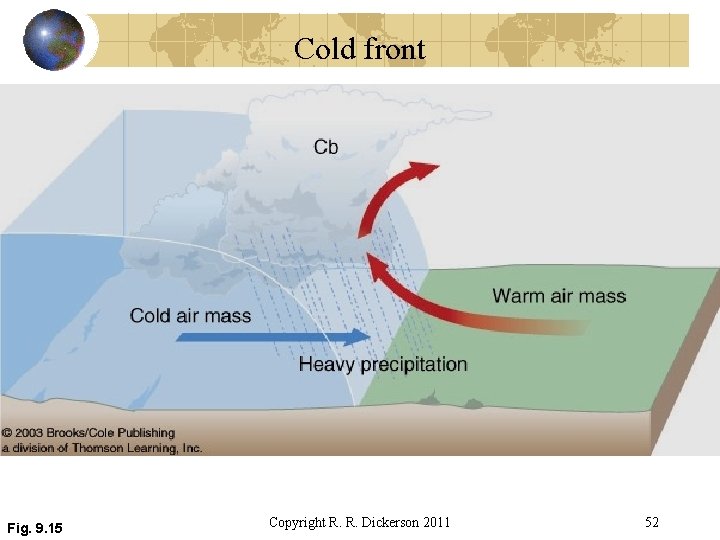
Cold front Fig. 9. 15 Copyright R. R. Dickerson 2011 52
Copyright R. R. Dickerson 2011 Fig. 53 1 -17, p. 21
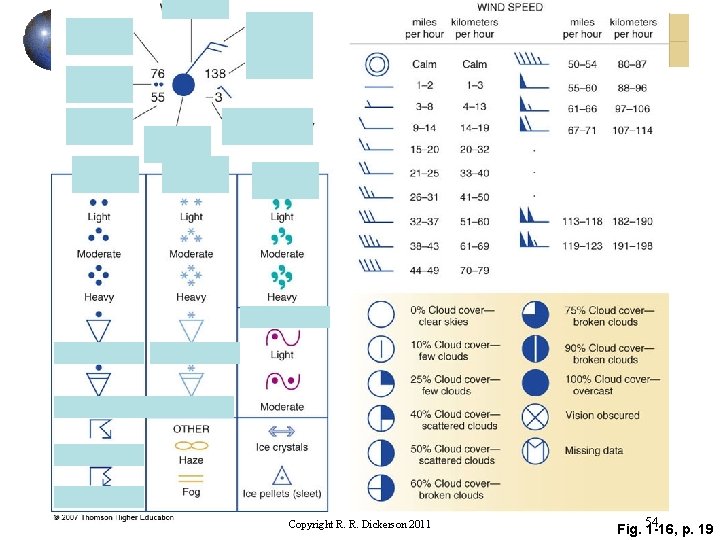
Copyright R. R. Dickerson 2011 Fig. 54 1 -16, p. 19
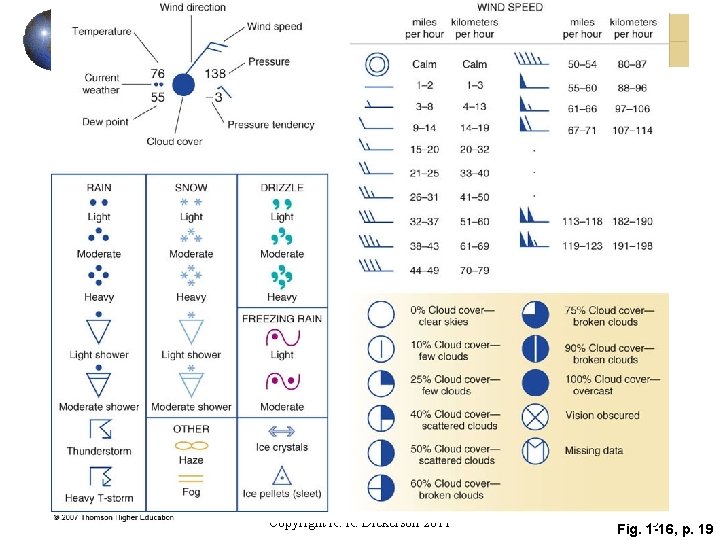
Copyright R. R. Dickerson 2011 Fig. 55 1 -16, p. 19
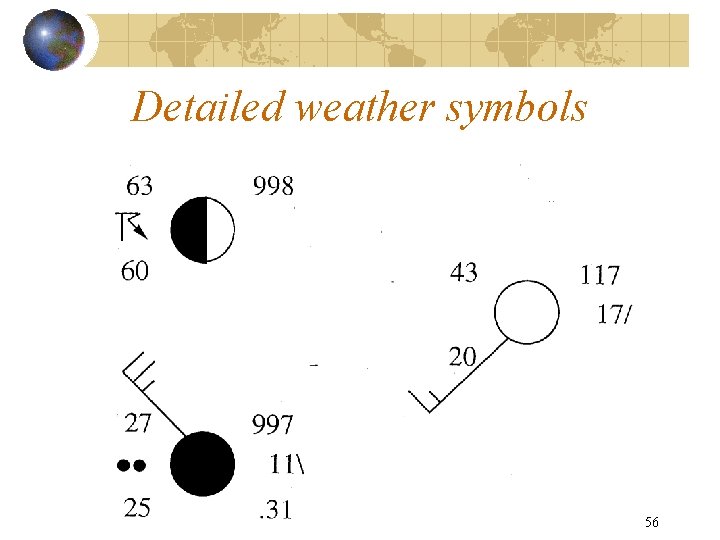
Detailed weather symbols Copyright R. R. Dickerson 2011 56

Inversion under a high pressure system over NYC Looking north January 2016

NYC Looking South
- Slides: 58