Lecture 24 Valence bond theory Valence bond theory

Lecture 24 Valence bond theory

Valence bond theory l l l There are two major approximate theories of chemical bonds: valence bond (VB) theory and molecular orbital (MO) theory. While it is computationally less widely used than MO, VB has a special appeal to organic chemists studying reaction mechanisms and remains useful and important. The concepts of spn hybridization and lone pairs are introduced.

Orbital approximation l l In polyelectron atoms, we used the orbital approximation – an approximate separation of variables – where we filled hydrogenic orbitals with electrons to construct atomic wave functions. For polyatomic molecules, can we also use orbital approximation? Can we use hydrogenic atomic orbitals to construct molecular wave functions?
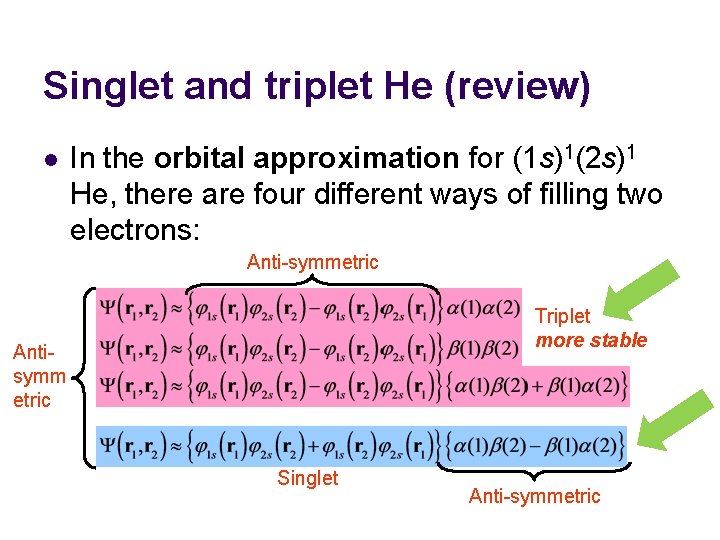
Singlet and triplet He (review) l In the orbital approximation for (1 s)1(2 s)1 He, there are four different ways of filling two electrons: Anti-symmetric Triplet more stable Antisymm etric Singlet Anti-symmetric
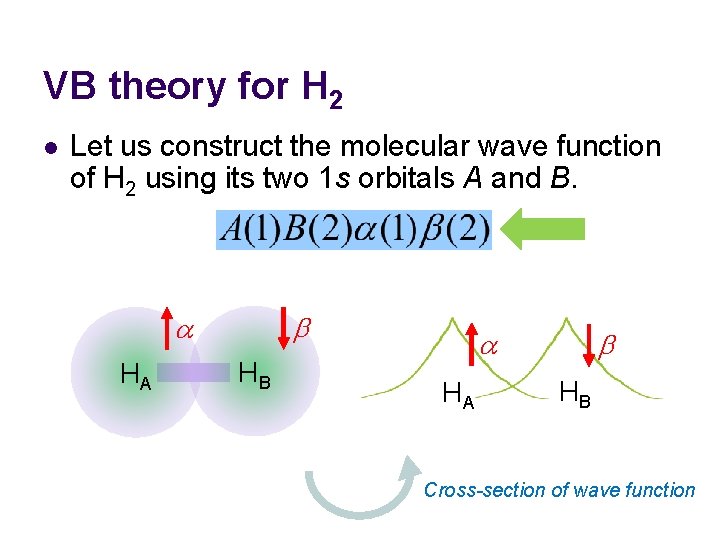
VB theory for H 2 l Let us construct the molecular wave function of H 2 using its two 1 s orbitals A and B. b a HA HB a HA b HB Cross-section of wave function
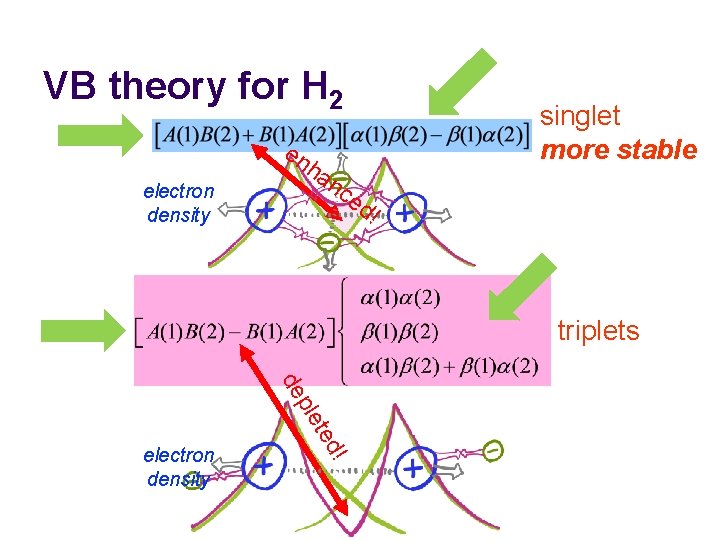
VB theory for H 2 en electron density ha singlet more stable nc ed ! triplets ! ted ple de electron density
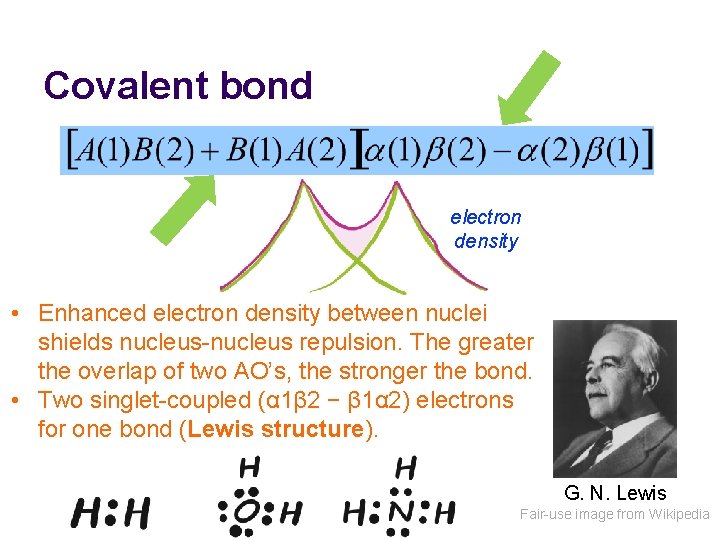
Covalent bond electron density • Enhanced electron density between nuclei shields nucleus-nucleus repulsion. The greater the overlap of two AO’s, the stronger the bond. • Two singlet-coupled (α 1β 2 − β 1α 2) electrons for one bond (Lewis structure). G. N. Lewis Fair-use image from Wikipedia
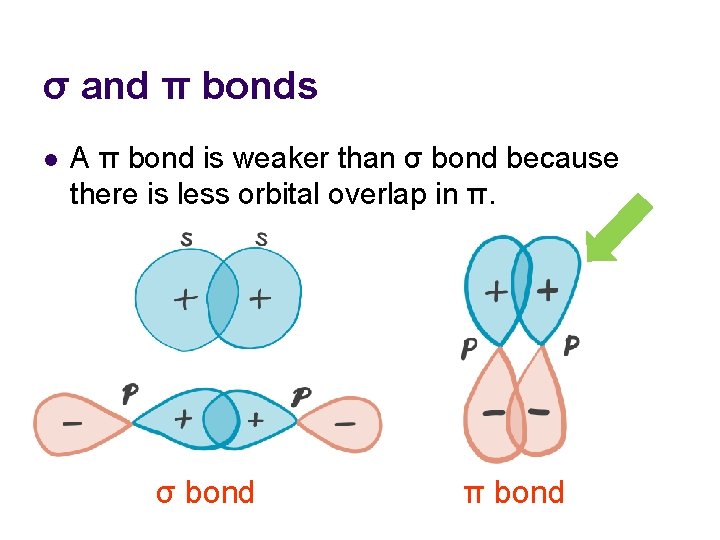
σ and π bonds l A π bond is weaker than σ bond because there is less orbital overlap in π. σ bond π bond
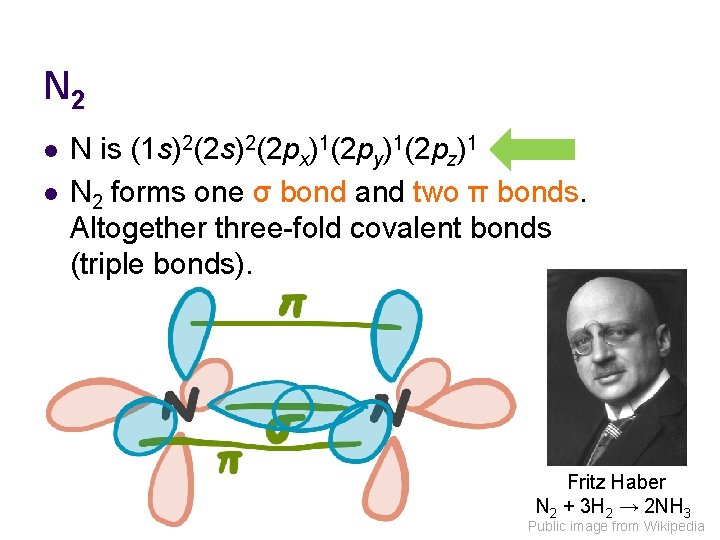
N 2 l l N is (1 s)2(2 px)1(2 py)1(2 pz)1 N 2 forms one σ bond and two π bonds. Altogether three-fold covalent bonds (triple bonds). Fritz Haber N 2 + 3 H 2 → 2 NH 3 Public image from Wikipedia
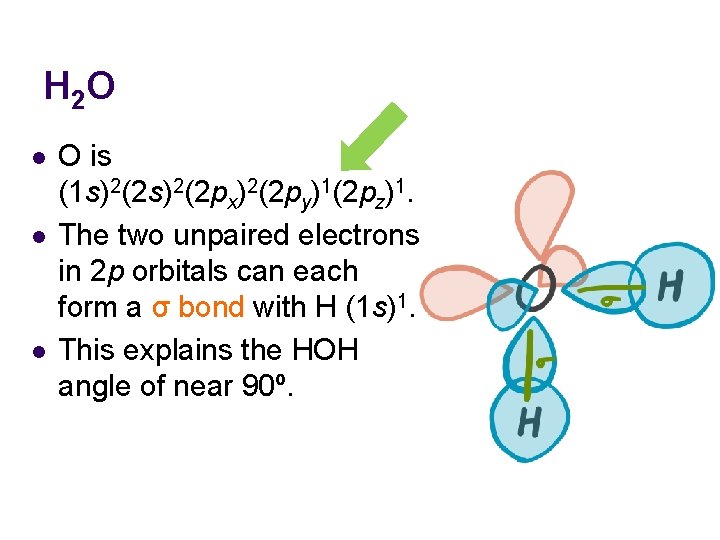
H 2 O l l l O is (1 s)2(2 px)2(2 py)1(2 pz)1. The two unpaired electrons in 2 p orbitals can each form a σ bond with H (1 s)1. This explains the HOH angle of near 90º.
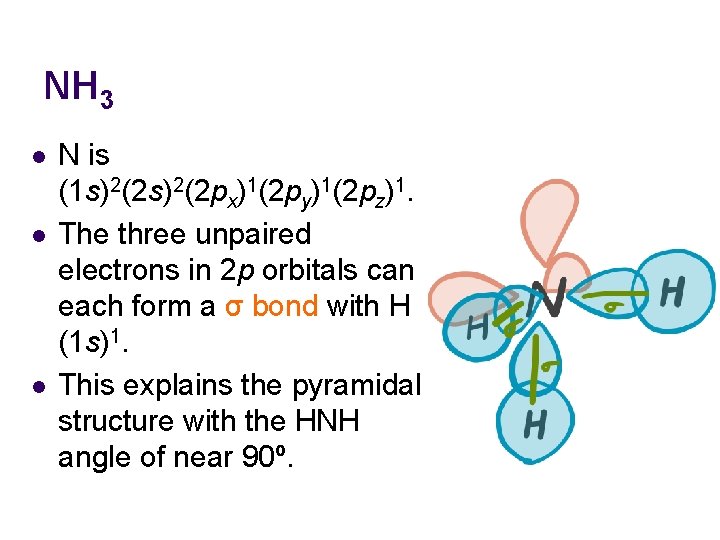
NH 3 l l l N is (1 s)2(2 px)1(2 py)1(2 pz)1. The three unpaired electrons in 2 p orbitals can each form a σ bond with H (1 s)1. This explains the pyramidal structure with the HNH angle of near 90º.

Promotion and hybridization l C (1 s)2(2 px)1(2 py)1 is known to form four equivalent bonds as in CH 4. valence 2 p 2 s 1 s Still not equivalent Promotion – we invest a small energy in C for a bigger energy gain (4 bonds instead of 2) in CH 4

sp 3 hybridization l From one s and three p orbitals, we form four equivalent bonds by linearly combing (hybridizing) them: z y x These are orthonormal
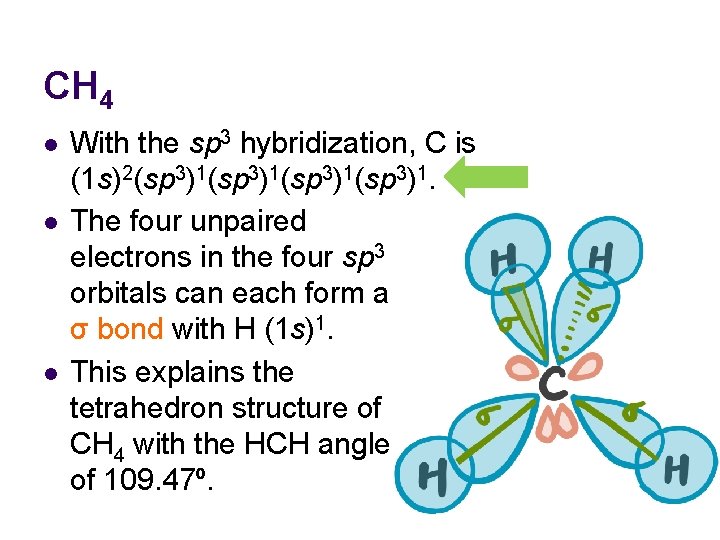
CH 4 l l l With the sp 3 hybridization, C is (1 s)2(sp 3)1(sp 3)1. The four unpaired electrons in the four sp 3 orbitals can each form a σ bond with H (1 s)1. This explains the tetrahedron structure of CH 4 with the HCH angle of 109. 47º.

sp 2 hybridization l From one s and two p orbitals, we form three equivalent bonds by linearly combing them: y x These are orthonormal

CH 2=CH 2 l l l With the sp 2 hybridization, C is (1 s)2(2 pz)1 (sp 2)1(sp 2)1. Three unpaired electrons in three sp 2 orbitals can each form a σ bond with H(1 s)1 or C(sp 2)1. C(2 pz)1 additionally forms a π bond. This explains the planar structure of ethylene with the HCH and CCH angles of near 120º. George O. Curme, Jr. Industrial production of ethylene Public image

sp hybridization l From one s and one p orbital, we form two equivalent bonds by linearly combing them: These are orthonormal
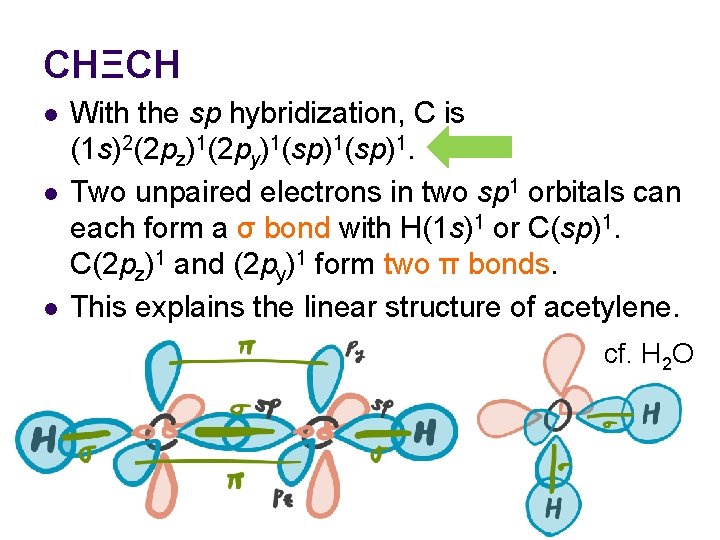
CHΞCH l l l With the sp hybridization, C is (1 s)2(2 pz)1(2 py)1(sp)1. Two unpaired electrons in two sp 1 orbitals can each form a σ bond with H(1 s)1 or C(sp)1. C(2 pz)1 and (2 py)1 form two π bonds. This explains the linear structure of acetylene. cf. H 2 O

Lone pairs l l Revisit H 2 O. O is (1 s)2(2 px)2(2 py)1(2 pz)1. Two unpaired electrons each form a covalent bond: O(2 py)1 H(1 s)1 and O(2 pz)1 H(1 s)1 Two valence electrons that do not participate in chemical bond are called a lone pair: O(2 s)2 and O(2 px)2. Lone pairs are part of electron density not shielding nucleus-nucleus repulsion and are also not stabilized by nuclear charges. They are naked electron pairs that repel other lone pairs or bonding electron pairs.
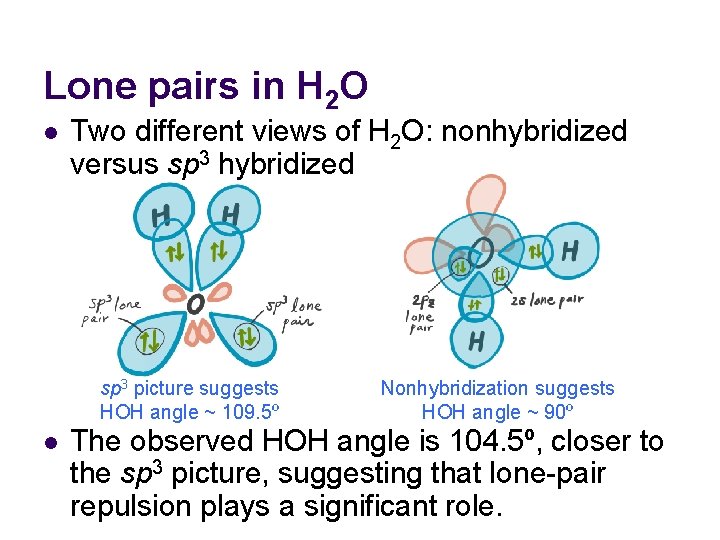
Lone pairs in H 2 O l Two different views of H 2 O: nonhybridized versus sp 3 hybridized sp 3 picture suggests HOH angle ~ 109. 5º l Nonhybridization suggests HOH angle ~ 90º The observed HOH angle is 104. 5º, closer to the sp 3 picture, suggesting that lone-pair repulsion plays a significant role.
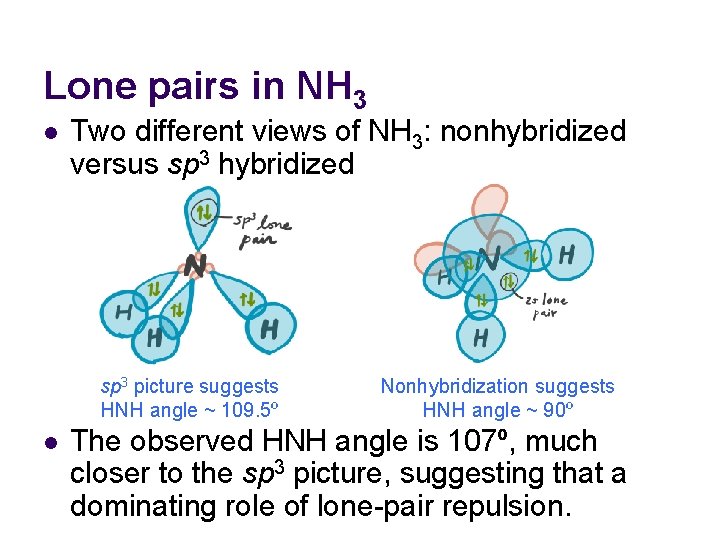
Lone pairs in NH 3 l Two different views of NH 3: nonhybridized versus sp 3 hybridized sp 3 picture suggests HNH angle ~ 109. 5º l Nonhybridization suggests HNH angle ~ 90º The observed HNH angle is 107º, much closer to the sp 3 picture, suggesting that a dominating role of lone-pair repulsion.

Lone pairs in H 2 X l The larger the central atom in the isovalence H 2 X series, the more widely spread valence p and s orbitals and the smaller the lone-pair repulsions. H 2 Te has no need to promote and hybridize (HTe. H angle of 89. 5º) to minimize the lone-pair repulsion, whereas H 2 O can lower its energy by promoting and hybridizing into sp 3 and separating the lone pairs more widely. H 2 X HXH angle H 2 O 104. 5 H 2 S 92. 2 H 2 Se 91. 0 H 2 Te 89. 5

Challenge homework #7 l l C is (1 s)2(2 px)1(2 py)1. Is methylene CH 2 bent (nonhybridized p, sp 2, sp 3) or linear (sp 1)? Find the answer in the following paper and report. “Methylene: A Paradigm for Computational Quantum Chemistry” by Henry F. Schaefer III, Science, volume 231, page 1100, 7 March 1986.

Summary l l VB theory is an orbital approximation for molecules. The orbitals used are hydrogenic atomic orbitals. VB theory explains the Lewis structure (two singlet-coupled electrons – α and β spins – per bond). This explains σ and π bonds, promotion and spn hybridization, lone pairs. Lone-pair repulsion is important in determining molecular structures.
- Slides: 24