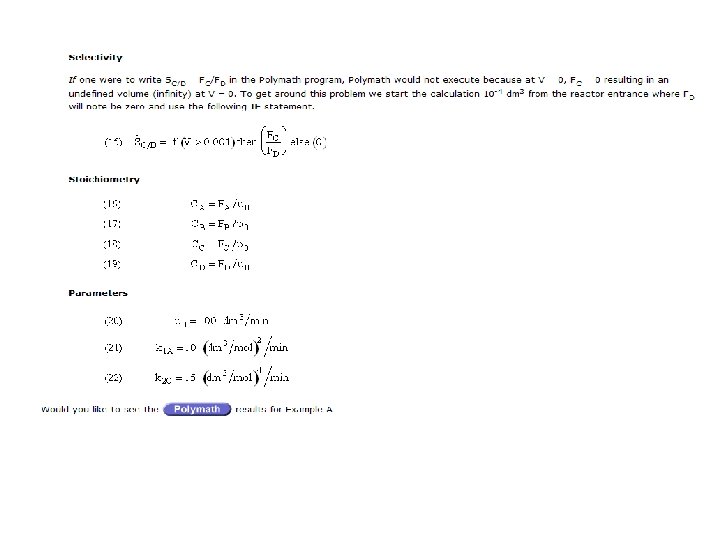
Lecture 22 Thursday 32708 Blowout Velocity CSTR Explosion

Lecture 22 Thursday 3/27/08 Blowout Velocity CSTR Explosion Batch Reactor Explosion
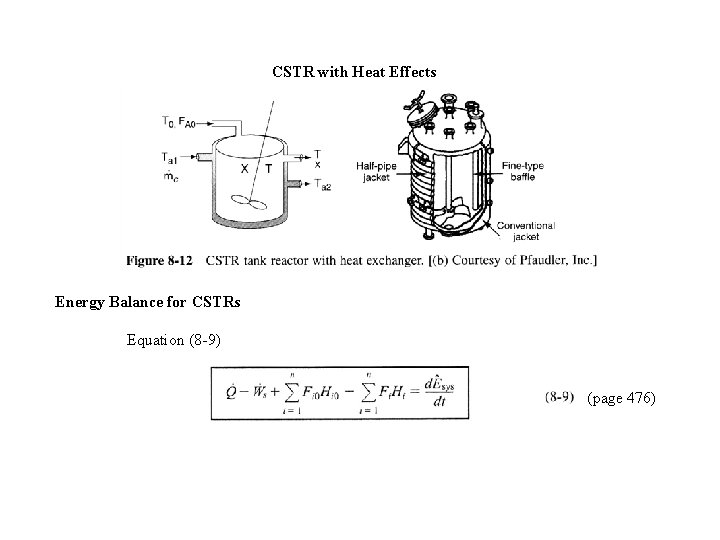
CSTR with Heat Effects Energy Balance for CSTRs Equation (8 -9) (page 476)

(page 593)

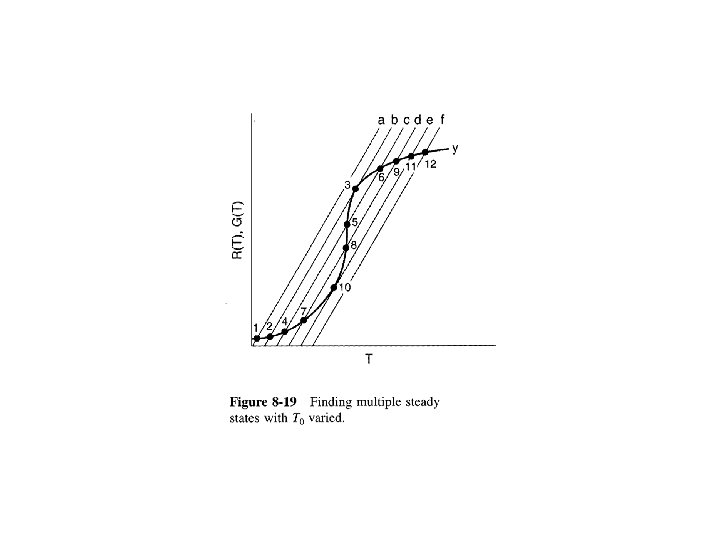
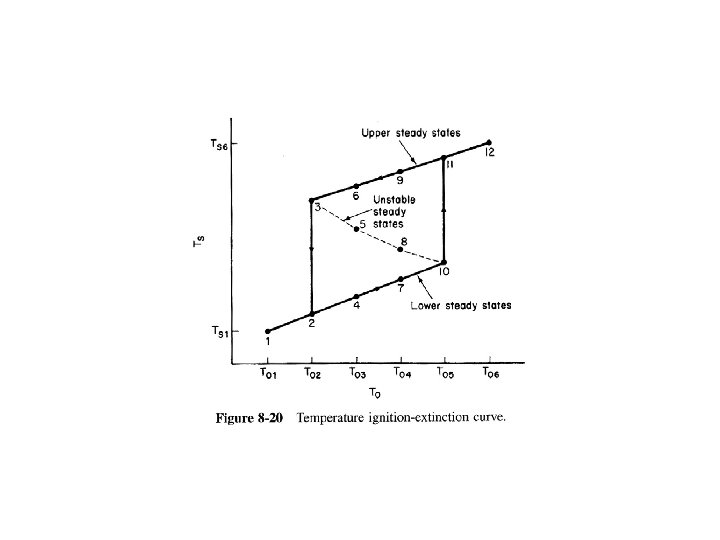
At Steady State Solving for X Solving for T

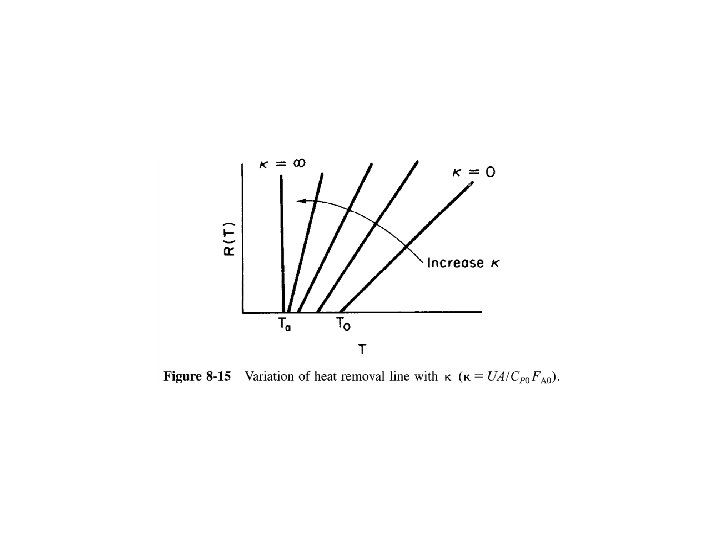

Mole Balance Rate Law Stoichiometry Combine

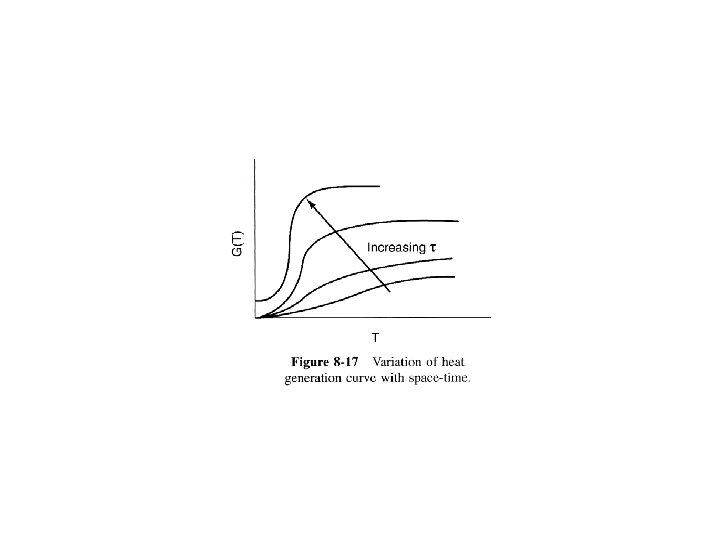
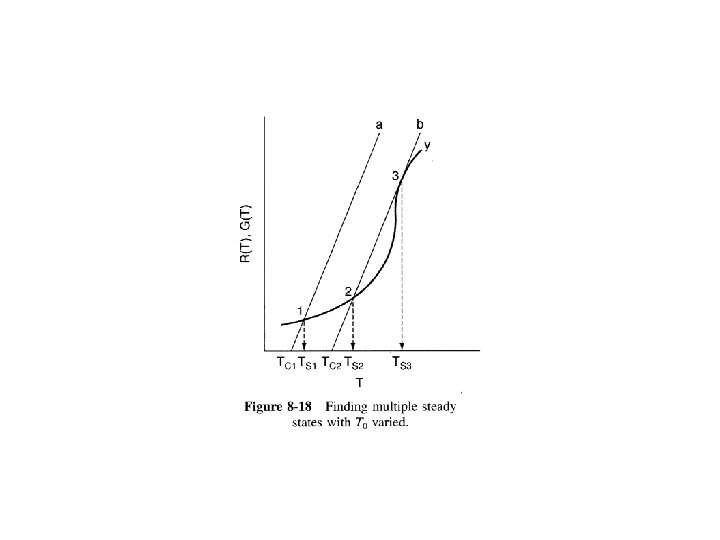
Heat Effects Isothermal Design Stoichiometry Rate Laws Mole Balance
Heat Effects Isothermal Design Stoichiometry Rate Laws Mole Balance








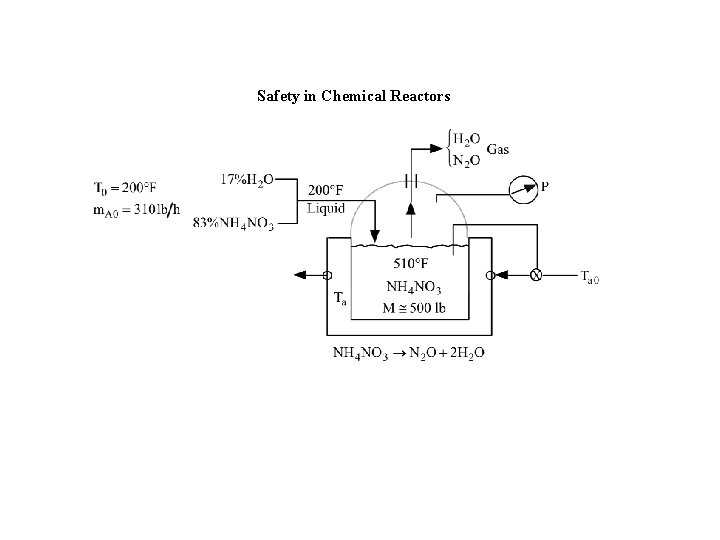
Safety in Chemical Reactors

Then rearranging Equation (9 -8)


Complete conversion FA = 0 Batch Reactors with Heat Effects Single Reactions Multiple Reactions Risk Rupture


Reactor Mole Balance Summary

Keeping MBAs Away From Chemical Reactors The process worked for 19 years before they showed up!! Why did they come? ? What did they want? ?



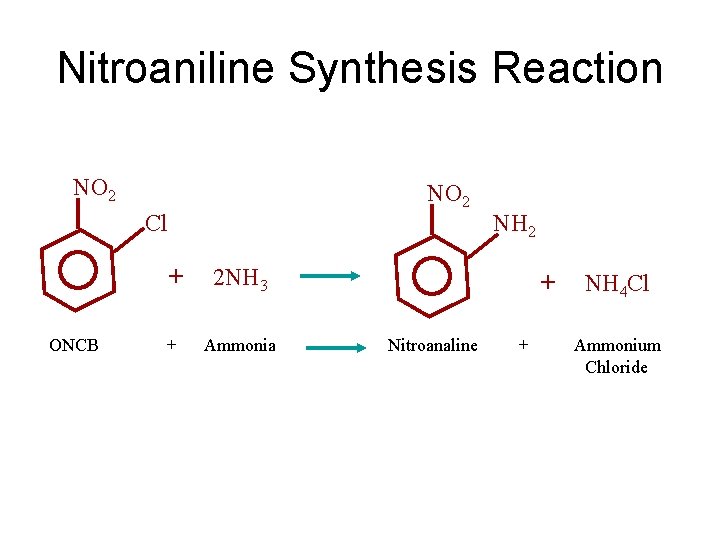
Nitroaniline Synthesis Reaction NO 2 Cl + 2 NH 3 NH 2 + NH 4 Cl ONCB + Ammonia Nitroanaline + Ammonium Chloride
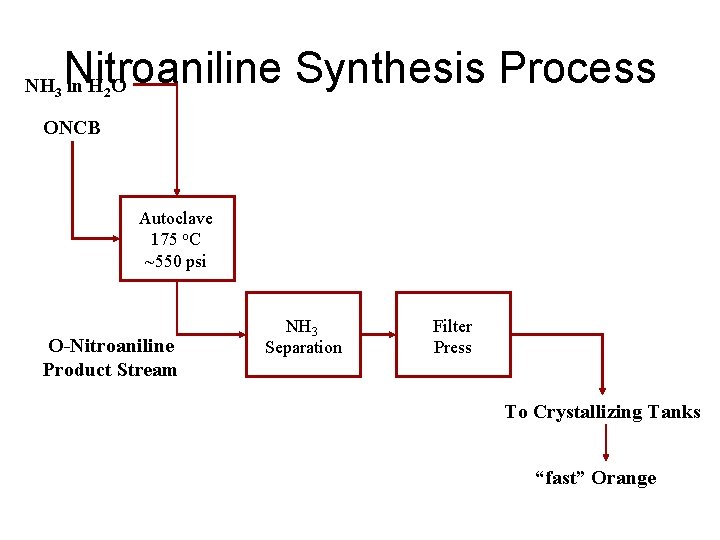
Nitroaniline Synthesis Process NH 3 in H 2 O ONCB Autoclave 175 o. C ~550 psi O-Nitroaniline Product Stream NH 3 Separation Filter Press To Crystallizing Tanks “fast” Orange
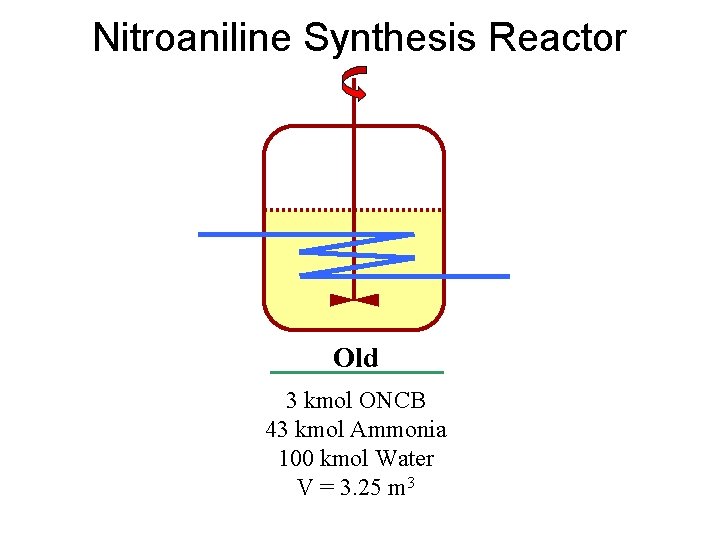
Nitroaniline Synthesis Reactor Old 3 kmol ONCB 43 kmol Ammonia 100 kmol Water V = 3. 25 m 3
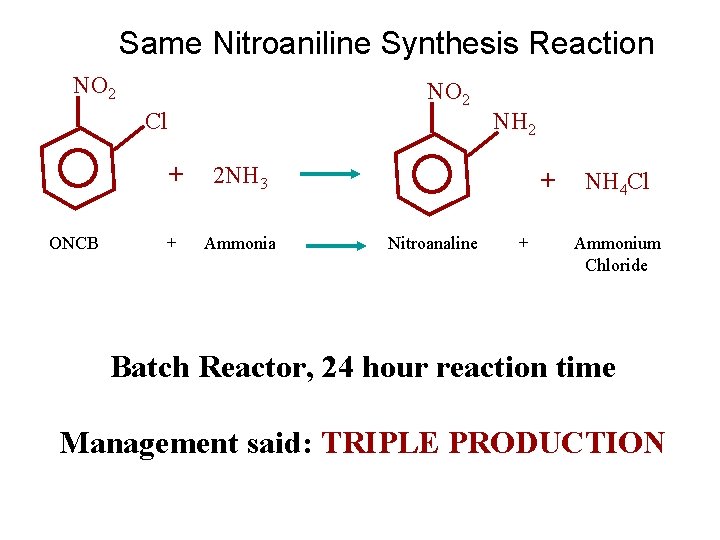
Same Nitroaniline Synthesis Reaction NO 2 Cl + 2 NH 3 NH 2 + NH 4 Cl ONCB + Ammonia Nitroanaline + Ammonium Chloride Batch Reactor, 24 hour reaction time Management said: TRIPLE PRODUCTION
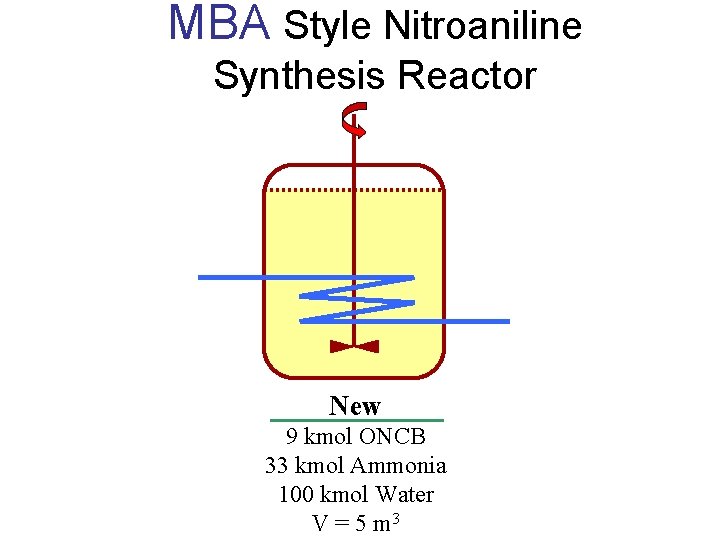
MBA Style Nitroaniline Synthesis Reactor New 9 kmol ONCB 33 kmol Ammonia 100 kmol Water V = 5 m 3

Monsanto Accident


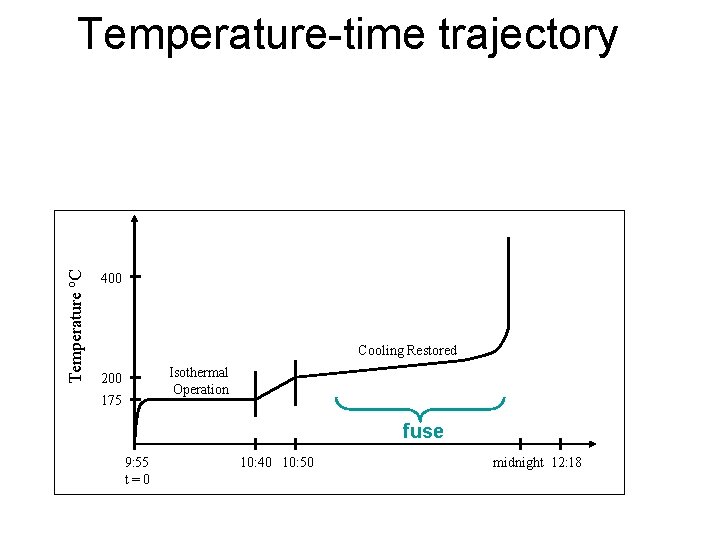
Temperature o. C Temperature-time trajectory 400 Cooling Restored Isothermal Operation 200 175 fuse 9: 55 t = 0 10: 40 10: 50 midnight 12: 18
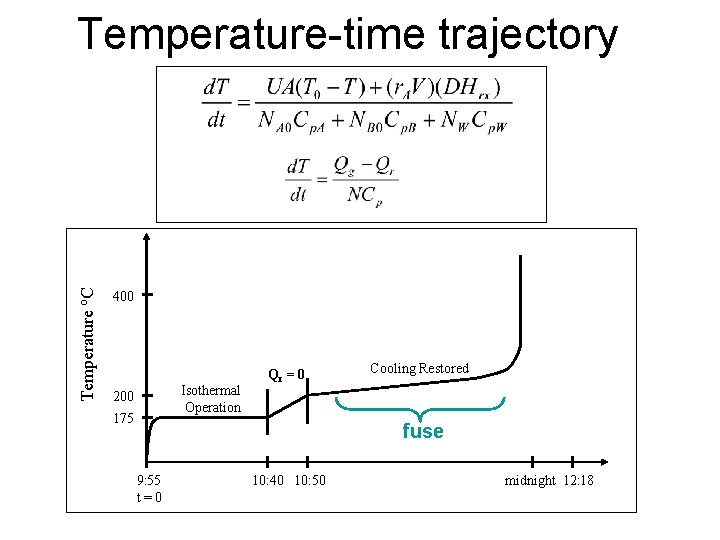
Temperature o. C Temperature-time trajectory 400 Isothermal Operation 200 175 Qr = 0 Cooling Restored fuse 9: 55 t = 0 10: 40 10: 50 midnight 12: 18

CSTR multiple reactions - =0

Example Mole balance) Rates) net rates, relative rates, rate laws…. . Stoichiometry) Energy Balance)
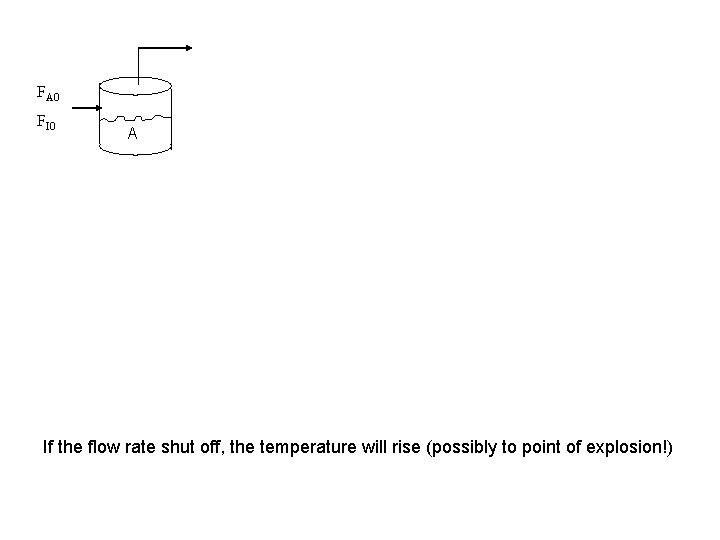
FA 0 FI 0 A If the flow rate shut off, the temperature will rise (possibly to point of explosion!)
- Slides: 47