Lecture 2 STUDY DESIGN IN CLINICAL RESEARCH SBM

Lecture 2 STUDY DESIGN IN CLINICAL RESEARCH SBM 4512 Clinical Research 1

Introduction Humans participate in research every day • Basic survey research (CENSES 2010) • Research designed to test the effectiveness of an intervention (drug, treatment etc) Research is a “systematic investigation” Clinical research differentiates between two types of physicians… 2

Which physician? ? ? Physician 1 Who systematically searches for facts (e. g. obtain a medical history, blood pressure and physical exam) to diagnose and treat a patient Physician 2: • Who systematically searches for facts among a sample of patients with the intention of drawing conclusions about the effectiveness of a treatment across a larger population. 3

Basic dimensions of a clinical research question To formulate the research question you need: • Understanding the biology and physiology of the problem • The epidemiology (i. e. determinants and distribution, prevalence, incidence and prognosis) • Frustrations in clinical management that lead to unsatisfactory results for patients. • To determine the value of treatment, that the intervention does more good than harm to those who receive it • Assess the efficacy and effectiveness 4

DESIGNS FOR CLINICAL STUDY/RESEARCH 1 5

Observational studies • Investigators observe the subjects and measure their outcomes. • The researchers do not actively manage the experiment. • Natural experiment • Example: Nurses’ Health Study 6
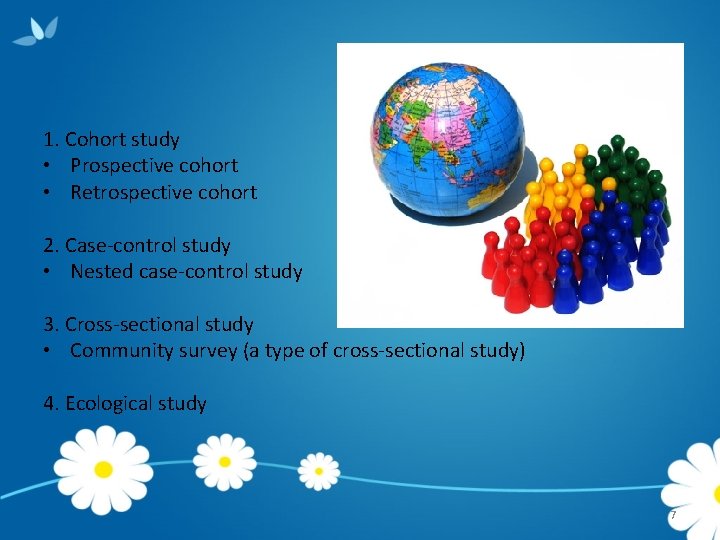
1. Cohort study • Prospective cohort • Retrospective cohort 2. Case-control study • Nested case-control study 3. Cross-sectional study • Community survey (a type of cross-sectional study) 4. Ecological study 7

COHORT STUDY • A cohort study or panel study is a form of longitudinal study (a type of observational study) used in medicine • It is an analysis of risk factors • Follows a group of people who do not have the disease • Life histories of segments of populations, and the individual people who constitute these segments • A cohort is a group of people who share a common characteristic or experience within a defined period (e. g. , are born, are exposed to a drug or a vaccine, etc. ). • Thus a group of people who were born on a day or in a particular period, say 1948, form a birth cohort. • The comparison group may be the general population from which the cohort is drawn, or it may be another cohort of persons thought to have had little or no exposure to the substance under investigation, but otherwise similar. • Data pair exposure/health is known for each individual. 8

9
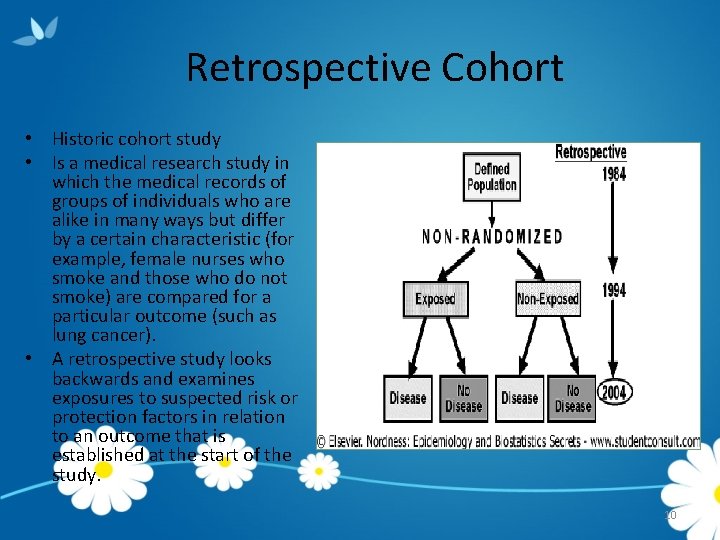
Retrospective Cohort • Historic cohort study • Is a medical research study in which the medical records of groups of individuals who are alike in many ways but differ by a certain characteristic (for example, female nurses who smoke and those who do not smoke) are compared for a particular outcome (such as lung cancer). • A retrospective study looks backwards and examines exposures to suspected risk or protection factors in relation to an outcome that is established at the start of the study. 10
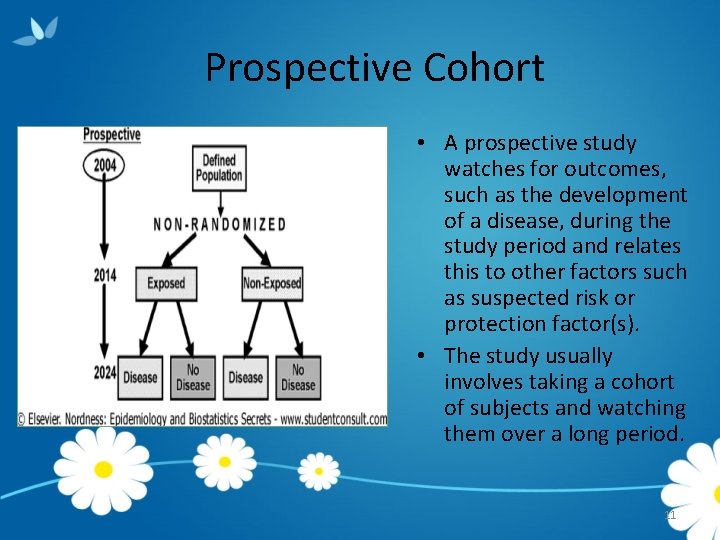
Prospective Cohort • A prospective study watches for outcomes, such as the development of a disease, during the study period and relates this to other factors such as suspected risk or protection factor(s). • The study usually involves taking a cohort of subjects and watching them over a long period. 11

Case-Control Study • Is a type of epidemiological, clinical study design. • It is typically used for retrospective studies. • In a case-control study, people with a disease (often, a specific diagnosis, perhaps lung cancer) are matched with people who do not have the disease (the 'controls'). • Further data are then collected on those individuals and the groups are compared to find out if other characteristics (perhaps a history of smoking) are also different between the two groups. • One of the most significant triumphs of the case-control study was the demonstration of the link between tobacco smoking and lung cancer, by Sir Richard Doll and others after him. Doll was able to show a statistically significant association between the two in a large case control study 12
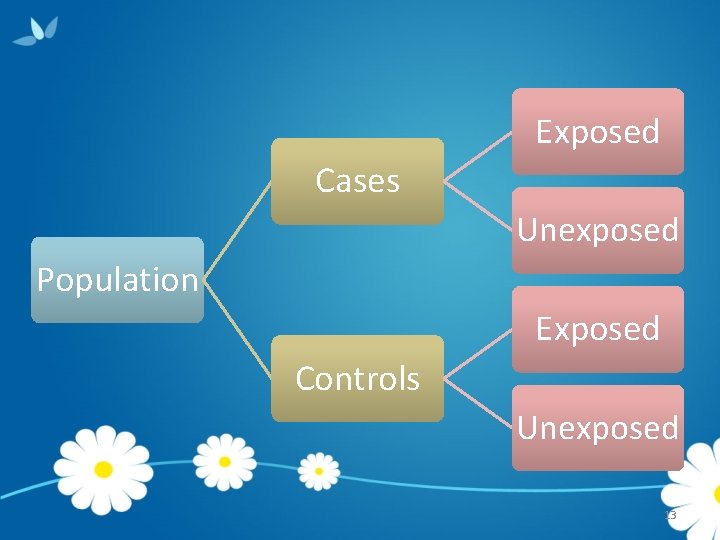
Exposed Cases Unexposed Population Exposed Controls Unexposed 13

Nested Case-control Study • In a nested case-control study, cases of a disease that occur in a defined cohort are identified and, for each, a specified number of matched controls is selected from among those in the cohort who have not developed the disease by the time of disease occurrence in the case. • Potentially offers impressive reductions in costs and efforts of data collection and analysis compared with the full cohort approach, with relatively minor loss in statistical efficiency (see restricted randomization). T • The nested case-control design is particularly advantageous for studies of biologic precursors of disease. • Compared with cohort studies can reduce cost and save time. 14
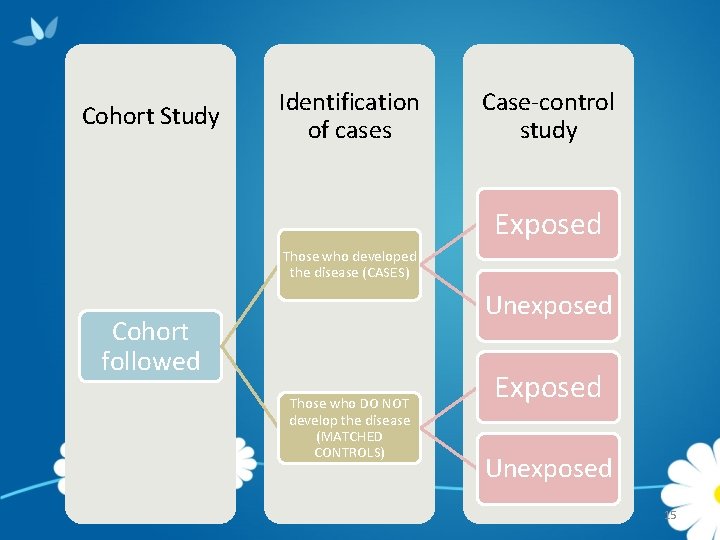
Cohort Study Identification of cases Case-control study Exposed Those who developed the disease (CASES) Unexposed Cohort followed Those who DO NOT develop the disease (MATCHED CONTROLS) Exposed Unexposed 15

Cross Sectional study • Involve observation of all of a population, or a representative subset, at a defined time. • Aim to provide data on the entire population under study • Used to describe some feature of the population, such as prevalence of an illness (acute or chronic conditions) or they may support inferences of cause and effect. • May also be described as censuses. 16

17

Ecological study • An epidemiological study in which the unit of analysis is a population rather than an individual. • E. g. looking at the association between smoking and lung cancer deaths in different countries. • Susceptible to the ecological fallacy. • An example of an ecological study is the analysis of the effects of disinfection byproducts on newborn babies, using 109 Massachusetts towns as units of analysis (Wright et al. 2004). • No information available about the individual members of the populations compared (e. g. comparing several states based on state-wide average air pollution and state-wide average prevalence of respiratory diseases • The results of ecological studies can provide the opportunity for later, more carefully designed studies (though more expensive and timeconsuming) to build on the initial observations 18

Ecological Fallacy: • Error in the interpretation of statistical data • Inferences about the nature of specific individuals are based solely upon aggregate statistics collected for the group to which those individuals belong. • This fallacy assumes that individual members of a group have the average characteristics of the group at large. • Stereotypes are one form of ecological fallacy, which assumes that groups are homogeneous. • For example, if a particular group of people are measured to have a lower average IQ than the general population, it is an error to assume that all members of that group have a lower IQ than the general population. 19

DESIGNS FOR CLINICAL STUDY/RESEARCH 2 20

Treatment studies Experimental (interventional) study • Investigators give the research subjects a particular medicine or other intervention. • Compare the treated subjects to subjects who receive no treatment or standard treatment. • Researchers measure how the subjects' health changes. • Example: testing the effectiveness of a drug

Randomized controlled trial aka THE CLINICAL TRIAL • Double-blind randomized trial • Single-blind randomized trial • Non-blind trial Nonrandomized trial (quasiexperiment)

CLINICAL TRIALS (how it all begins!)


How do we get from bench-to-bedside? ? ? • We begin the evaluation of therapeutic innovations with extensive testing in laboratory animals.

BASIC RESEARCH (from the bench) 1. Researchers analyze the physiological and chemical properties in vitro (laboratory testing or bench research) 2. Research the toxic effects in laboratory animals (animal testing or animal modeling)

Pre phase (The Basic research) Animal studies 1. Quantify the time course of the candidates drug distribution in various tissues and organs (pharmacokinetics) 2. Determine its effects on the structure and function of cells, tissues, organs and organ systems (pharmacodynamics) 3. Find out the substances into which it is metabolized, their effects, and their mechanisms of excretion 4. Test the dose response mechanics (LD 50, ED 50, ) Toxicology If the drug’s LD 50, is comfortably above the dose required for the desired pharmacodynamic effect and its other attributes are favorable, looks affordable and/or profitable…
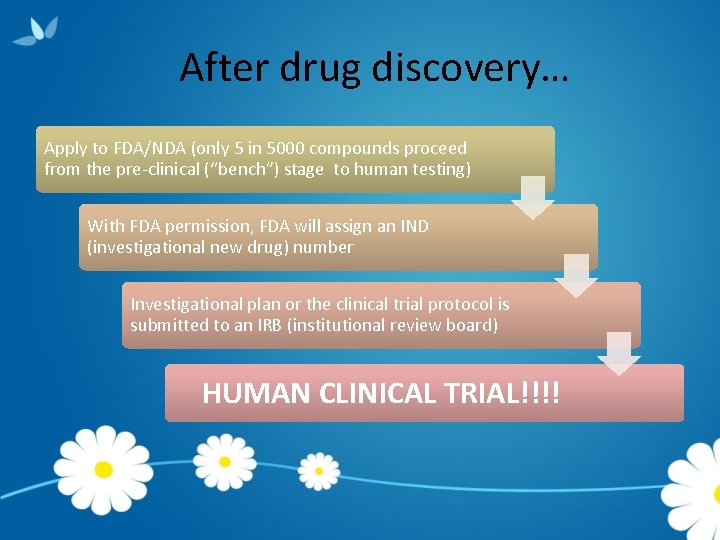
After drug discovery… Apply to FDA/NDA (only 5 in 5000 compounds proceed from the pre-clinical (“bench”) stage to human testing) With FDA permission, FDA will assign an IND (investigational new drug) number Investigational plan or the clinical trial protocol is submitted to an IRB (institutional review board) HUMAN CLINICAL TRIAL!!!!
HUMAN CLINICAL TRIAL!!

Human Clinical Trials • Drugs intended to treat people must be studied in people • Most humans prefer to learn about a drug from studies on someone else • Divided in to 4 distinct phases • Phase I, Phase III, Phase IV

Phase I • Assess the acute safety profile of a drug • Dose ranging – examine the maximum tolerated dose by volunteers for single and for multiple doses. • Clarify the pharmacokinetics (ADME), metabolic fate and freedom from toxicity as animals. • Give increasing amounts until the pharmacodynamic effects found in lab stage is replicated in humans • Conducted in normal healthy volunteers OR • Phase I trials are sometimes conducted in severely ill patients if adverse effects are expected.

Phase II Trials • If Phase I studies do not reveal any major concerns (e. g. unacceptable toxicities)…→ Phase II • Evaluate efficacy ( and safety) in selected populations of patients with the disease or condition to be treated, diagnosed, or prevented. • Larger sample size than Phase I trials Objectives: 1. To set and confirm the dose necessary for the desired pharmacodynamic effect 2. Estimate the proportion of patients who do (responders) and do not (non responders) display the desired effect (ED 50? ) 3. To confirm the results of the previous pharmacokinetic and metabolic studies 4. To continue to look for toxicity

Phase III trials • Full scale Phase III RCT with sufficient sample size and power to establish or refute the compounds predicted benefit. • Compare currently available or standard care treatments with the investigational drug. • These clinical trials are conducted in patient populations for which the medicine is eventually intended. • Patient volunteers chosen at random to receive the investigational treatment/drug or the other available comparator • best indicator of the benefit-versus-risk ratio • If drug demonstrates to be both safe and effective, marketing approval may be granted.

Phase IV • Occurs once a drug has been marketed • Monitoring or Post marking Surveillance (i. e. , the period following marketing) • Conducted after a medicine is marketed to provide additional details about the medicine's efficacy or safety profile. • Adds valuable information to the profile of the new drug. PROVIDES DATA • Effect of different formulations, dosages, durations of treatment • Drug-disease and drug-drug interactions. • Drugs interaction with dietary components, the distribution and determinants of its use (pharmacoepidemiology) and the measurement of its cost-effectiveness (pharmacoeconomics) • New age groups, races, and other types of patients can be studied. • Detection and definition of previously unknown or inadequately quantified adverse reactions and related risk factors

Efficacy and effectiveness Efficacy To denote both the beneficial and harmful effects of an intervention when it is applied under ideal circumstances Effectiveness To denote both the beneficial and harmful effects of an intervention when it is applied under the usual circumstances that apply in health care. EFFICACY Does it work in the clinical trials? EFFECTIVENESS Does it work in clinical practice? EFFICIENCY Does it contribute to more efficient use of resources?

New drug development timeline • According to the FDA: new drug development takes on average 8. 5 years for marketing approval • Preclinical testing (initial synthesis → animal testing): 1 -3 years (average 18 month) • Clinical trials (Phases I to III): 2 -10 years (average 5 years) • FDA takes 2 months to 7 years to review the NDA (new drug application) before marketing approval

Clinical research…not an EXACT science What is the objective of the clinical trial? ? Louis Pasteur produced the IDEAL clinical trial: treated patients exposed to rabies with an experimental rabies vaccine → 100% survival →Pasteur's treatment was effective Drugs reduce the risk of death but do not entirely eliminate it • Relieve symptoms of the illness (e. g. pain, nausea, fatigue etc) • Alter clinical measurement of a physiological process (e. g. lowering b. p, cholesterol levels)

Clinical Research What you want? • • • High-quality science Rapid accrual Access to patients Adequate research network Unimpeachable data

Aim of a clinical trial • Decide on the target number of patients who will participate. • Goal: to determine the association between that exposure (intervention) and an outcome. Statistical significance • Power of the trial: the number of patients enrolled in a study has a large bearing on the ability of the study to reliably detect the size of the effect of the study intervention. • The larger the sample size or number of participants in the trial, the greater the statistical power. Expensive!

What is a clinical trial? Definition: • In health care, clinical trials are conducted to allow safety and efficacy data to be collected for new drugs or devices. • Clinical trials are used to test new drugs and therapies on human volunteers. • Such investigations are carried out using protocols that adhere to accepted standards of safety, patient care and data interpretation. However, history shows that patient welfare was not always such a high priority. • These trials can only take place once satisfactory information has been gathered on the quality of the product and its non-clinical safety, and Health Authority/Ethics Committee approval is granted in the country where the trial is taking place. • Clinical trials can vary in size from a single center in one country to multicenter trials in multiple countries.

Why? • Clinical trials or research is done when there is some reason to believe that the treatment being studied may be valuable to the patient. Some of the questions answered by doing such research: How helpful the treatment is? How does the experimental drug or new type of treatment work? If it works better than already existing treatment options? What are the side effects that occur as a result of the new treatment/ drug? Ø How serious the side effects are when compared to standard treatment options? Ø Do the benefits outweigh the side effects? Ø Which groups of people are most likely to benefit from the new drug / treatment? Ø Ø
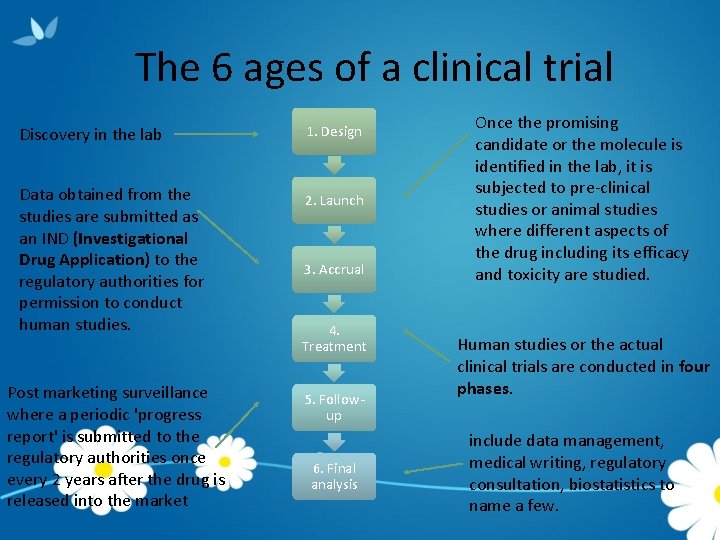
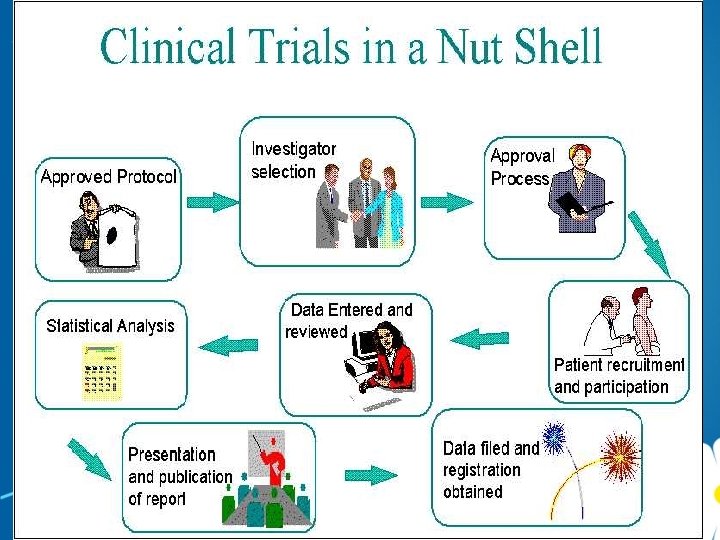
The 6 ages of a clinical trial Discovery in the lab 1. Design Data obtained from the studies are submitted as an IND (Investigational Drug Application) to the regulatory authorities for permission to conduct human studies. 2. Launch Post marketing surveillance where a periodic 'progress report' is submitted to the regulatory authorities once every 2 years after the drug is released into the market 3. Accrual 4. Treatment 5. Followup 6. Final analysis Once the promising candidate or the molecule is identified in the lab, it is subjected to pre-clinical studies or animal studies where different aspects of the drug including its efficacy and toxicity are studied. Human studies or the actual clinical trials are conducted in four phases. include data management, medical writing, regulatory consultation, biostatistics to name a few.

Types of Research Organizations involved in Clinical Trials • Single institution/investigator • Study Group : Industry- or NIH-funded • Traditional Cooperative Group: (NCI Model) Government-funded • Contract Research Organization (CRO): F For-profit Often division of larger entity (contract labs, practice management etc)

Problems in clinical trials Exponential increase in number of new drugs Limited number of patients Limited number of clinical investigators Improvements in symptoms and clinical measurements are difficult to evaluate • Chronic conditions (arthritis, depression, MS, Parkinson’s, asthma) are subject to remission “on/off” • Some case measurements are subjective (interpretation by the investigator or patient) • •

• Diseases are highly variable due to multicausality

Classifying trials by their purpose Treatment trials: Test experimental treatments, new combinations of drugs, or new approaches to surgery or radiation therapy. Prevention trials: • Look for better ways to prevent disease in people who have never had the disease or to prevent a disease from returning. • These approaches may include medicines, vitamins, vaccines, minerals, or lifestyle changes. Diagnostic trials: Conducted to find better tests or procedures for diagnosing a particular disease or condition. Screening trials: Test the best way to detect certain diseases or health conditions. Quality of Life: Trials (or Supportive Care trials) explore ways to improve comfort and the quality of life for individuals with a chronic illness.


Randomized controlled/clinical trial (RCT) • Definition: Intervention studies or experiments Randomized trials (RCTs) Prospective, analytical, experimental study. To evaluate the effectiveness of interventions (e. g pharmacological treatments, interventions, diagnostic tests) • Method: Consists of one treatment group and one control group current standard treatment or placebo). Randomization or random allocation: Patients randomly assigned to each group. Outcomes of the groups are compared after sufficient follow-up time. • Properly executed, the RCT is the strongest evidence of the clinical efficacy of preventive and therapeutic procedures in the clinical setting.

Example: • Research topic and Aim: A randomized, controlled community trial was conducted to evaluate the effectiveness of vitamin A supplementation to prevent childhood mortality in Indonesia (Sommer et al. , 1986). • Method: In 229 villages, children aged 1 -5 years were given two doses of vitamin A while children in the 221 control villages were not given vitamin A until after the study. • Result: Mortality among children in the control villages was 50% higher that than in the villages given vitamin A • Conclusion/inference: Vitamin A is an effective supplement to prevent childhood mortality

NEXT LECTURE… • DESIGN AND CONDUCT OF A CLINICAL TRIAL (PART 1)
- Slides: 51