Lecture 2 OUTLINE Semiconductor Fundamentals contd Energy band

Lecture 2 OUTLINE • Semiconductor Fundamentals (cont’d) – Energy band model – Band gap energy – Density of states – Doping Reading: Pierret 2. 2 -2. 3, 3. 1. 5; Hu 1. 3 -1. 4, 1. 6, 2. 4
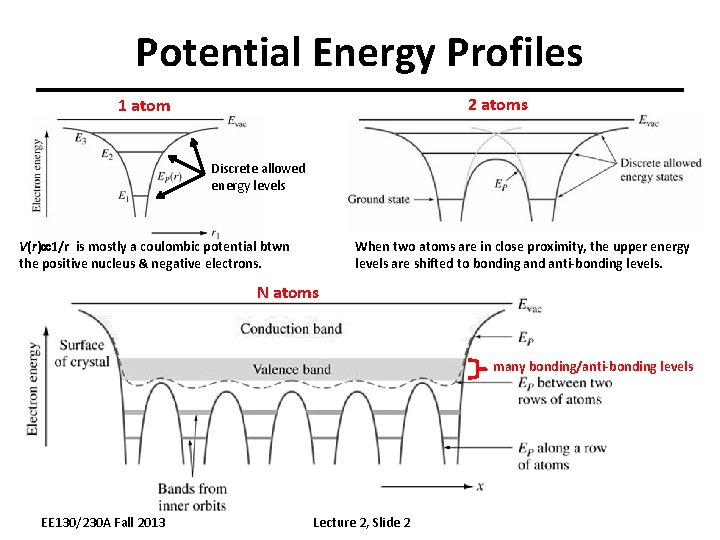
Potential Energy Profiles 2 atoms 1 atom Discrete allowed energy levels V(r) 1/r is mostly a coulombic potential btwn the positive nucleus & negative electrons. When two atoms are in close proximity, the upper energy levels are shifted to bonding and anti-bonding levels. N atoms many bonding/anti-bonding levels EE 130/230 A Fall 2013 Lecture 2, Slide 2
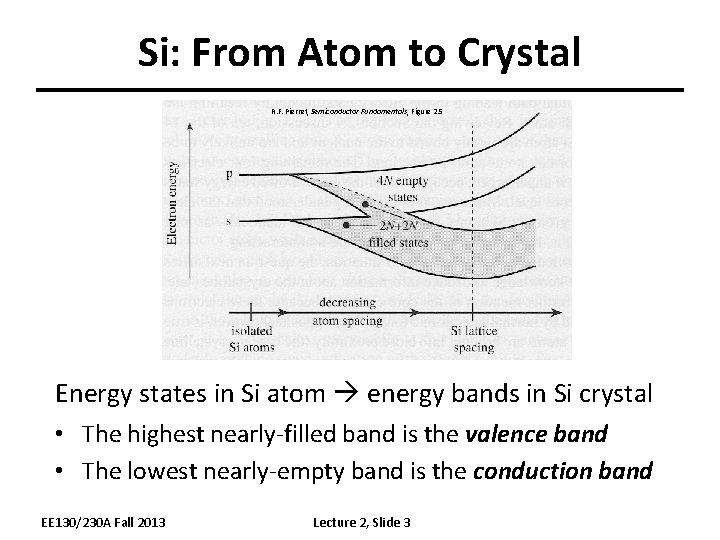
Si: From Atom to Crystal R. F. Pierret, Semiconductor Fundamentals, Figure 2. 5 Energy states in Si atom energy bands in Si crystal • The highest nearly-filled band is the valence band • The lowest nearly-empty band is the conduction band EE 130/230 A Fall 2013 Lecture 2, Slide 3
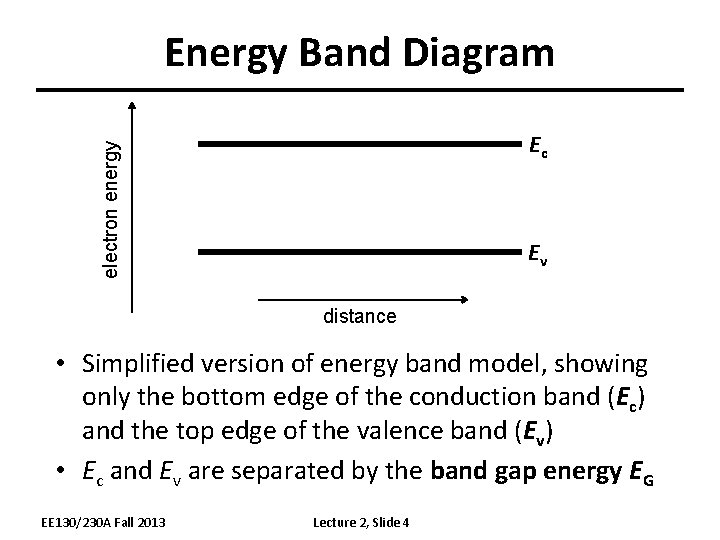
Energy Band Diagram electron energy Ec Ev distance • Simplified version of energy band model, showing only the bottom edge of the conduction band (Ec) and the top edge of the valence band (Ev) • Ec and Ev are separated by the band gap energy EG EE 130/230 A Fall 2013 Lecture 2, Slide 4
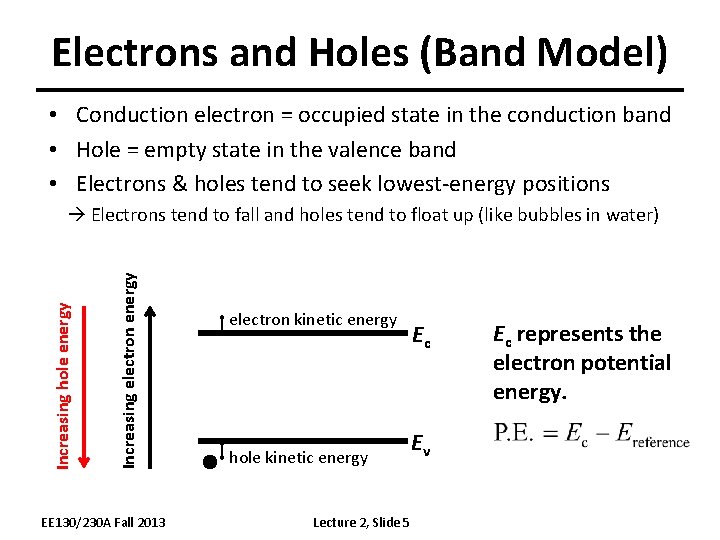
Electrons and Holes (Band Model) • Conduction electron = occupied state in the conduction band • Hole = empty state in the valence band • Electrons & holes tend to seek lowest-energy positions Increasing electron energy Increasing hole energy Electrons tend to fall and holes tend to float up (like bubbles in water) EE 130/230 A Fall 2013 electron kinetic energy hole kinetic energy Lecture 2, Slide 5 Ec Ev Ec represents the electron potential energy.

Electrostatic Potential, V and Electric Field, E • The potential energy of a particle with charge -q is related to the electrostatic potential V(x): 0. 7 e. V EE 130/230 A Fall 2013 • Variation of Ec with position is called “band bending. ” Lecture 2, Slide 6

Measuring the Band Gap Energy • EG can be determined from the minimum energy of photons that are absorbed by the semiconductor Ec photon hn > E G Ev Band gap energies of selected semiconductors Semiconductor Ge Si Ga. As Band gap energy (e. V) 0. 67 1. 12 1. 42 EE 130/230 A Fall 2013 Lecture 2, Slide 7

Density of States E Ec Ev density of states, g(E)d. E = number of states per cm 3 in the energy range between E and E+d. E Near the band edges: Electron and hole density-of-states effective masses for E Ec Si Ge Ga. As mn, DOS*/mo 1. 08 0. 56 0. 067 for E Ev EE 130/230 A Fall 2013 Lecture 2, Slide 8 mp, DOS*/mo 0. 81 0. 29 0. 47

Effective Mass, m* • When an electron is moving inside a solid material, the potential field will affect its movement. Schrödinger equation: E : total energy Y : wave function ħ : reduced Planck constant • For low kinetic energy where p is the crystal momentum i. e. a conduction electron behaves as a particle but with an effective mass m* EE 130/230 A Fall 2013 Lecture 2, Slide 9
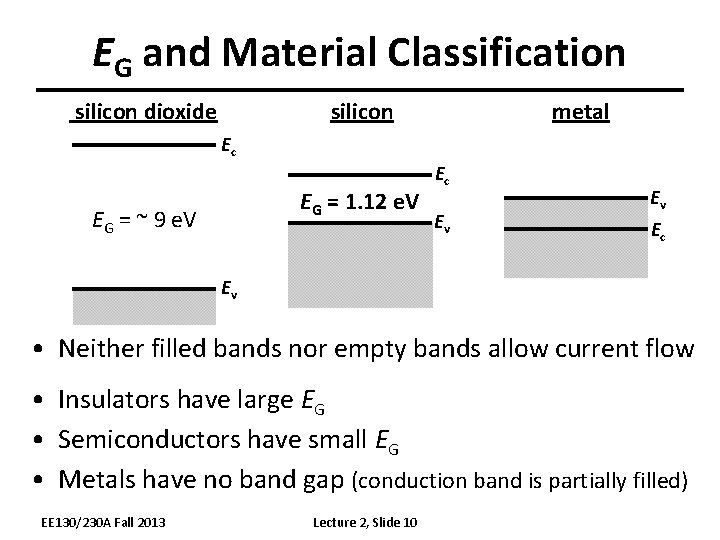
EG and Material Classification silicon dioxide metal Ec EG = 1. 12 e. V EG = ~ 9 e. V Ec Ev Ev Ec Ev • Neither filled bands nor empty bands allow current flow • Insulators have large EG • Semiconductors have small EG • Metals have no band gap (conduction band is partially filled) EE 130/230 A Fall 2013 Lecture 2, Slide 10
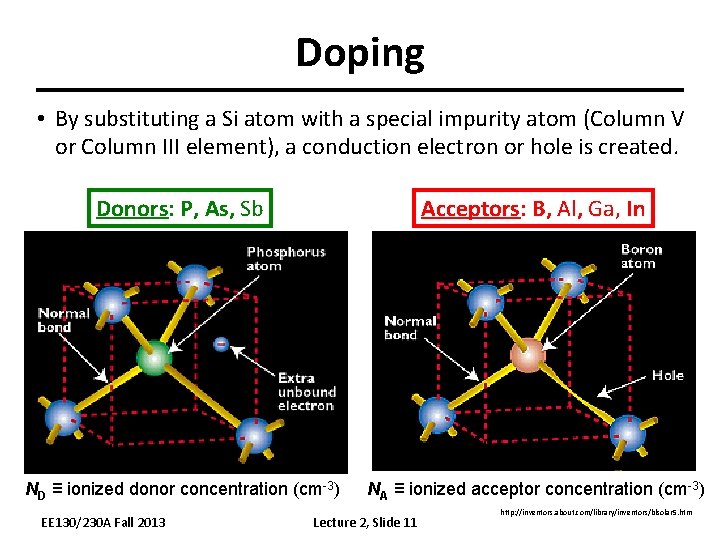
Doping • By substituting a Si atom with a special impurity atom (Column V or Column III element), a conduction electron or hole is created. Donors: P, As, Sb Acceptors: B, Al, Ga, In ND ≡ ionized donor concentration (cm-3) NA ≡ ionized acceptor concentration (cm-3) EE 130/230 A Fall 2013 Lecture 2, Slide 11 http: //inventors. about. com/library/inventors/blsolar 5. htm

Doping Silicon with a Donor Example: Add arsenic (As) atom to the Si crystal Si Si As Si Si The loosely bound 5 th valence electron of the As atom “breaks free” and becomes a mobile electron for current conduction. EE 130/230 A Fall 2013 Lecture 2, Slide 12

Doping Silicon with an Acceptor Example: Add boron (B) atom to the Si crystal Si Si B Si Si The B atom accepts an electron from a neighboring Si atom, resulting in a missing bonding electron, or “hole”. The hole is free to roam around the Si lattice, carrying current as a positive charge. EE 130/230 A Fall 2013 Lecture 2, Slide 13
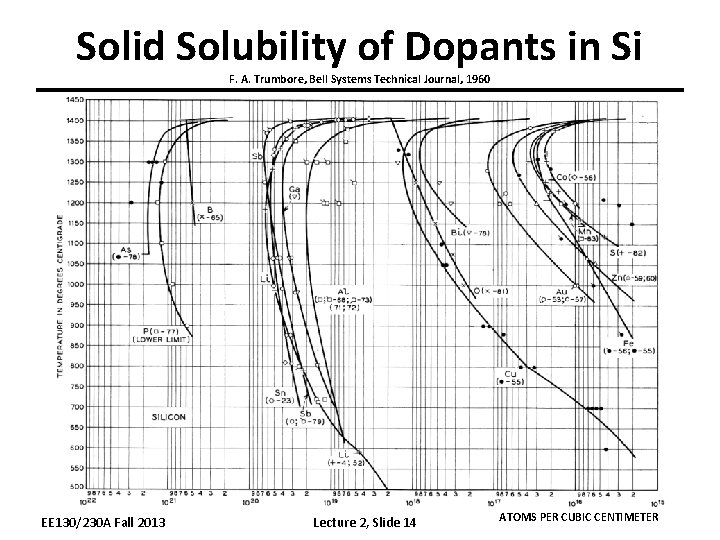
Solid Solubility of Dopants in Si F. A. Trumbore, Bell Systems Technical Journal, 1960 EE 130/230 A Fall 2013 Lecture 2, Slide 14 ATOMS PER CUBIC CENTIMETER
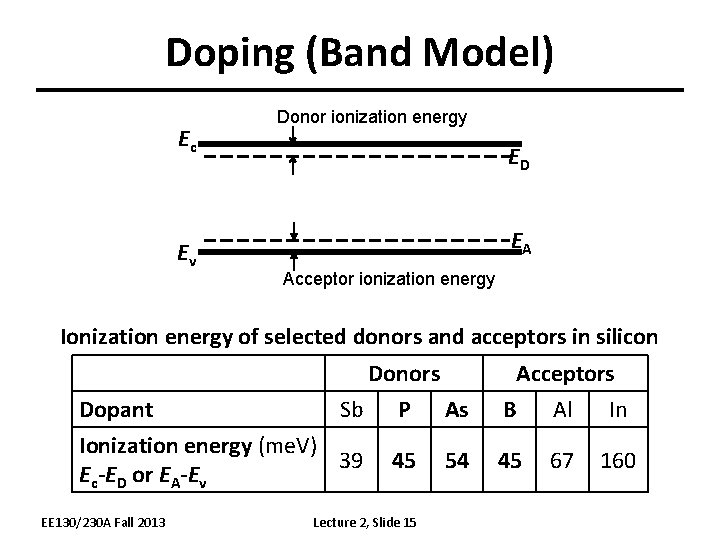
Doping (Band Model) Ec Ev Donor ionization energy ED EA Acceptor ionization energy Ionization energy of selected donors and acceptors in silicon Donors Sb P As Dopant Ionization energy (me. V) 39 Ec-ED or EA-Ev EE 130/230 A Fall 2013 45 Lecture 2, Slide 15 54 Acceptors B Al In 45 67 160
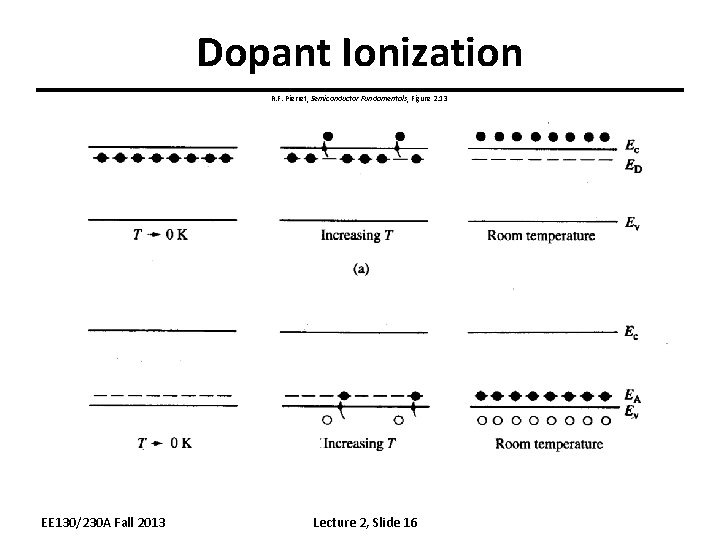
Dopant Ionization R. F. Pierret, Semiconductor Fundamentals, Figure 2. 13 EE 130/230 A Fall 2013 Lecture 2, Slide 16

Charge-Carrier Concentrations Charge neutrality condition: ND + p = NA + n At thermal equilibrium, np = ni 2 (“Law of Mass Action”) Note: Carrier concentrations depend on net dopant concentration! EE 130/230 A Fall 2013 Lecture 2, Slide 17

n-type Material (n > p) ND > NA (more specifically, ND – NA >> ni): EE 130/230 A Fall 2013 Lecture 2, Slide 18

p-type Material (p > n) NA > ND (more specifically, NA – ND >> ni): EE 130/230 A Fall 2013 Lecture 2, Slide 19
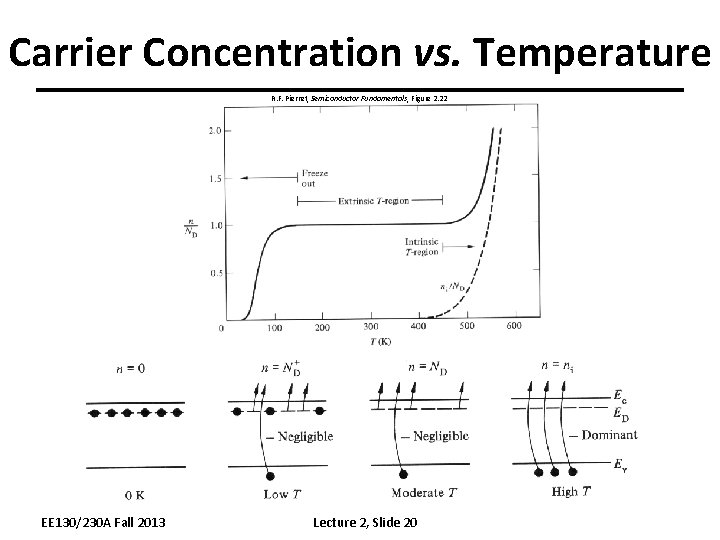
Carrier Concentration vs. Temperature R. F. Pierret, Semiconductor Fundamentals, Figure 2. 22 EE 130/230 A Fall 2013 Lecture 2, Slide 20

Terminology donor: impurity atom that increases n acceptor: impurity atom that increases p n-type material: contains more electrons than holes p-type material: contains more holes than electrons majority carrier: the most abundant carrier minority carrier: the least abundant carrier intrinsic semiconductor: n = p = ni extrinsic semiconductor: doped semiconductor such that majority carrier concentration = net dopant concentration EE 130/230 A Fall 2013 Lecture 2, Slide 21

Summary • Allowed electron energy levels in an atom give rise to bands of allowed electron energy levels in a crystal. – The valence band is the highest nearly-filled band. – The conduction band is the lowest nearly-empty band. • The band gap energy is the energy required to free an electron from a covalent bond. – EG for Si at 300 K = 1. 12 e. V – Insulators have large EG; semiconductors have small EG EE 130/230 A Fall 2013 Lecture 2, Slide 22

Summary (cont’d) • Ec represents the electron potential energy Variation in Ec(x) variation in electric potential V Electric field • E - Ec represents the electron kinetic energy EE 130/230 A Fall 2013 Lecture 2, Slide 23

Summary (cont’d) • Dopants in silicon: – Reside on lattice sites (substituting for Si) – Have relatively low ionization energies (<50 me. V) ionized at room temperature – Group-V elements contribute conduction electrons, and are called donors – Group-III elements contribute holes, and are called acceptors Dopant concentrations typically range from 1015 cm-3 to 1020 cm-3 EE 130/230 A Fall 2013 Lecture 2, Slide 24
- Slides: 24