Lecture 2 Introduction to Chromatography and Size Exclusion


























- Slides: 56

Lecture 2: Introduction to Chromatography and Size. Exclusion Chromatography Yaj

What is chromatography? It is a physical method of separation in which the components to be separated are distributed between two phases, one of which is stationary (stationary phase, SP) while the other moves (mobile phase, MP or‘eluant’) in a definite direction Yaj

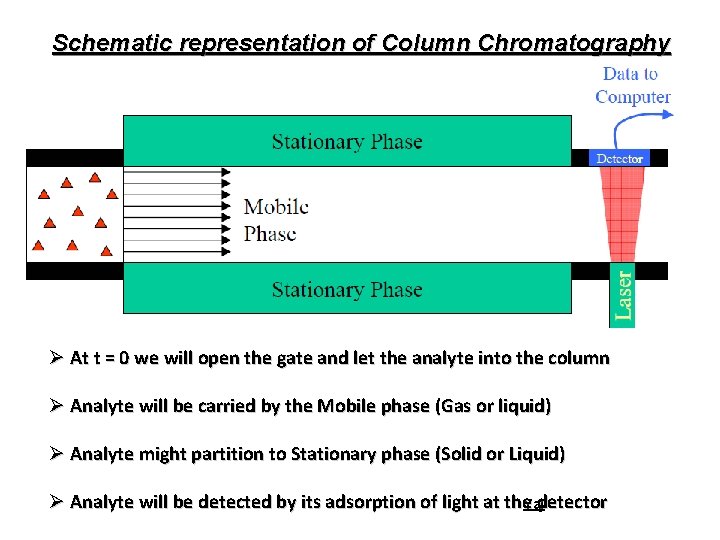
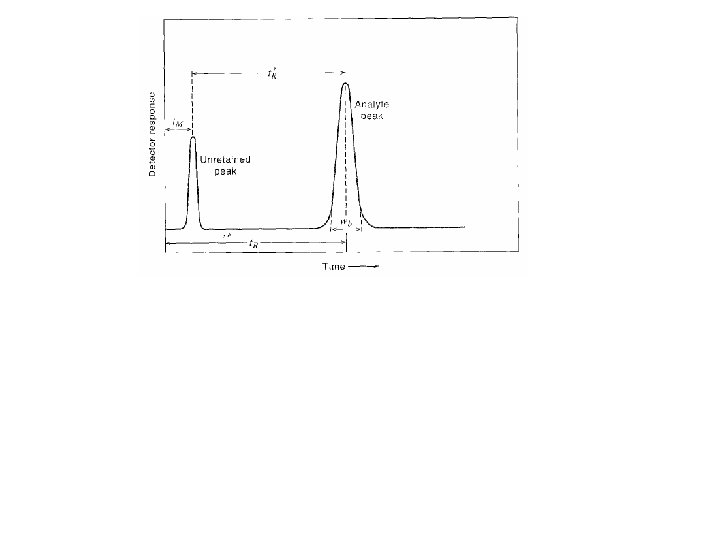
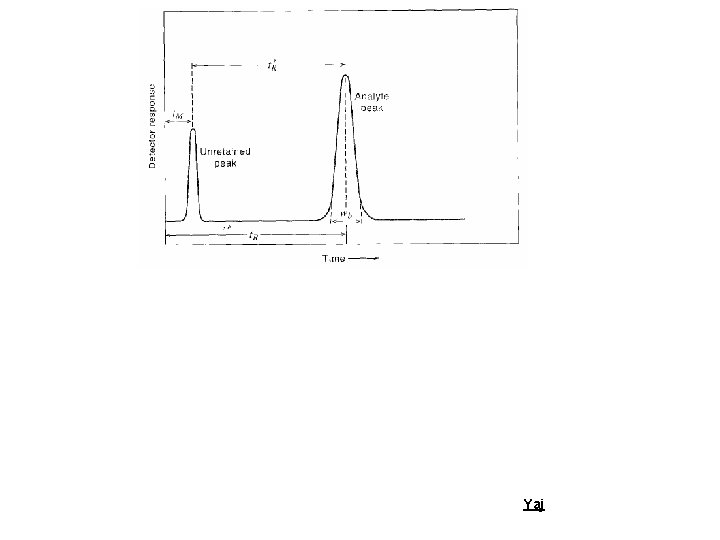
Schematic representation of Column Chromatography Ø At t = 0 we will open the gate and let the analyte into the column Ø Analyte will be carried by the Mobile phase (Gas or liquid) Ø Analyte might partition to Stationary phase (Solid or Liquid) Ø Analyte will be detected by its adsorption of light at the. Yajdetector

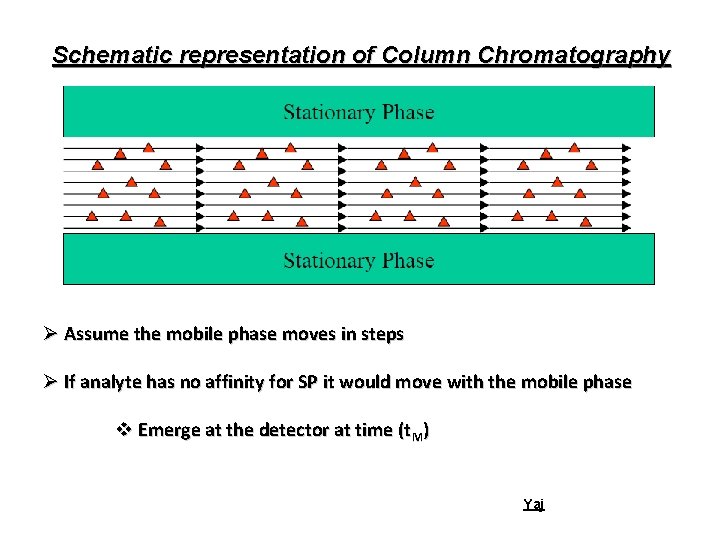
Schematic representation of Column Chromatography Ø Assume the mobile phase moves in steps Ø If analyte has no affinity for SP it would move with the mobile phase v Emerge at the detector at time (t. M) Yaj

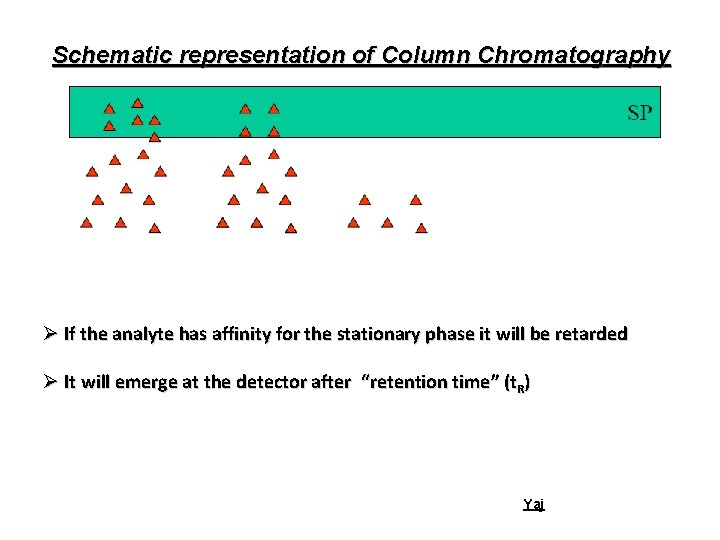
Schematic representation of Column Chromatography Ø If the analyte has affinity for the stationary phase it will be retarded Ø It will emerge at the detector after “retention time” (t. R) Yaj

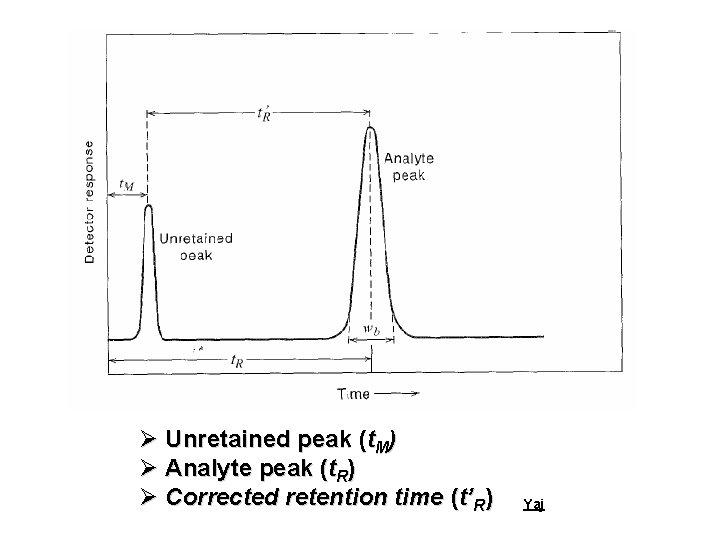
Ø Unretained peak (t. M) Ø Analyte peak (t. R) Ø Corrected retention time (t’R) Yaj

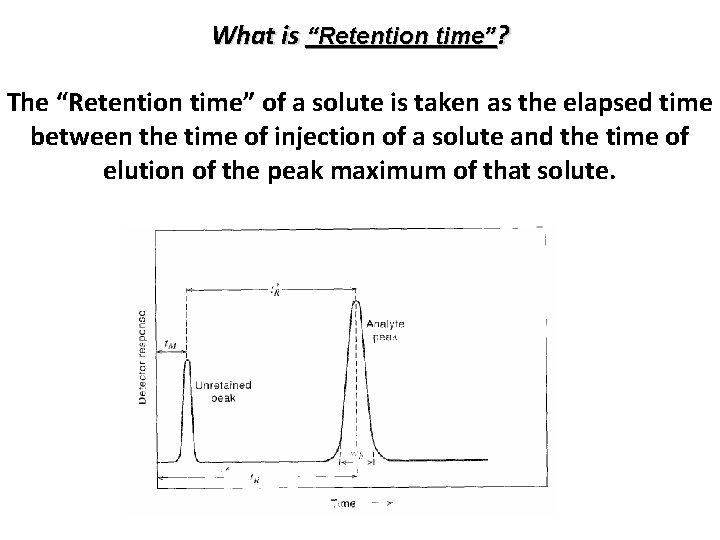
What is “Retention time”? The “Retention time” of a solute is taken as the elapsed time between the time of injection of a solute and the time of elution of the peak maximum of that solute. Yaj

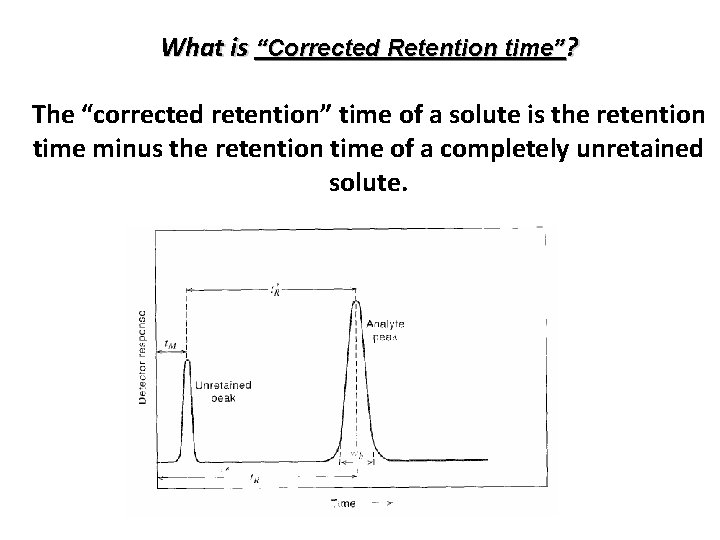
What is “Corrected Retention time”? The “corrected retention” time of a solute is the retention time minus the retention time of a completely unretained solute. Yaj

Distribution of the Analyte between two phases An analyte is in equilibrium between the two phases; Amobile <=> Astationary K, partition coefficient; defined as the molar concentration of analyte in the stationary phase divided by the molar concentration of the analyte in the mobile phase. Retention factor, k', is often used to describe the migration rate of an analyte on a column. It is also called the capacity factor. The retention factor for analyte A is defined as; k'A = t. R - t. M / t. M = t’R/t. M Ø Analytes retention factor is less than one, elution is so fast that accurate determination of the retention time is very difficult Ø High retention factors (greater than 20) mean that elution takes a very long time Ø Ideally, the retention factor for an analyte is between one Yajand five.

Selectivity Factor (α) selectivity factor (α), , which describes the separation of two species (A and B) on the column; α = k'B / k 'A Ø species A elutes faster than species B Ø selectivity factor is always greater than one Yaj

Resolution is the measure of how well species have been separated The resolution of two species, A and B, is defined as Yaj

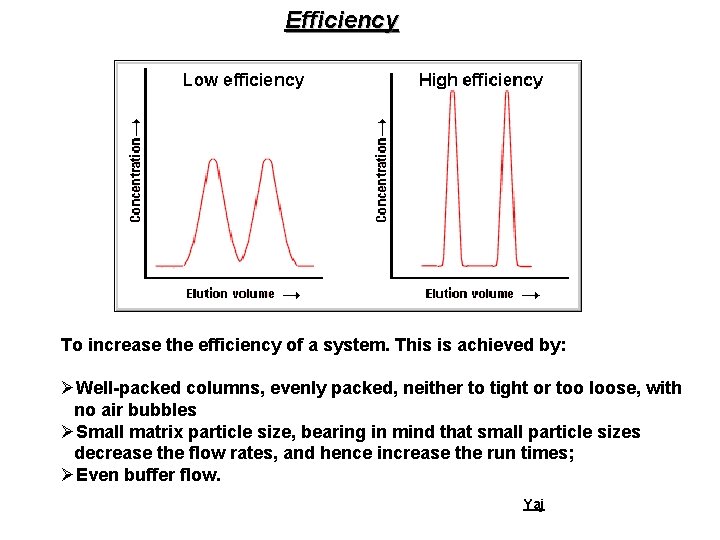
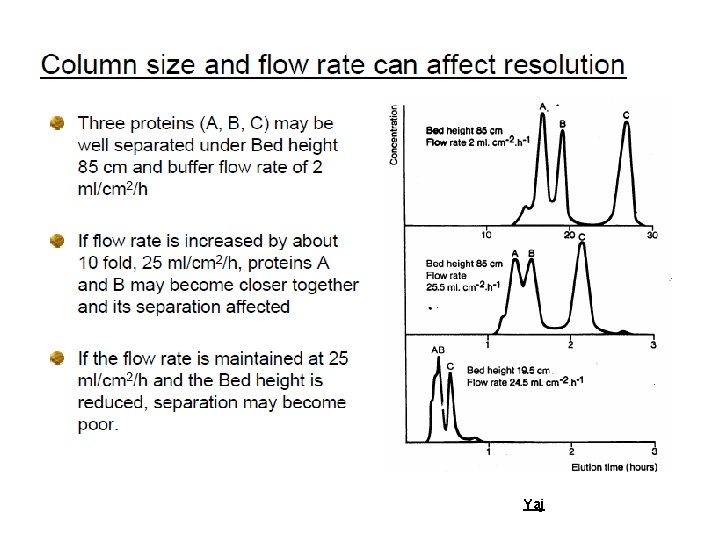
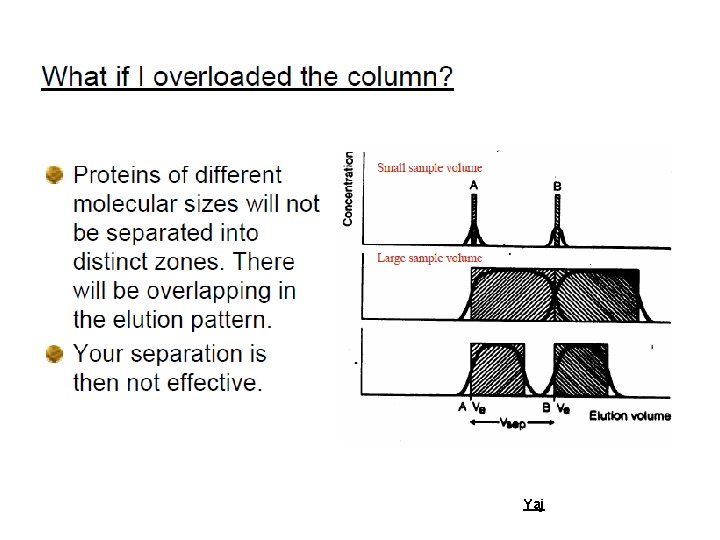
Efficiency To increase the efficiency of a system. This is achieved by: ØWell-packed columns, evenly packed, neither to tight or too loose, with no air bubbles ØSmall matrix particle size, bearing in mind that small particle sizes decrease the flow rates, and hence increase the run times; ØEven buffer flow. Yaj

Yaj

Concept of Column Plates or Theoretical Plates Ø A theoretical plate in a separation processes is a hypothetical zone or stage in which two phases, establish an equilibrium with each other. Ø If the two phases are liquid and vapor for example: Ø Vapor-Liquid Equilibrium : Rate of evaporation is equal to the rate of condensation Ø A collection of theoretical plates or stages is referred to as a theoretical tray Yaj

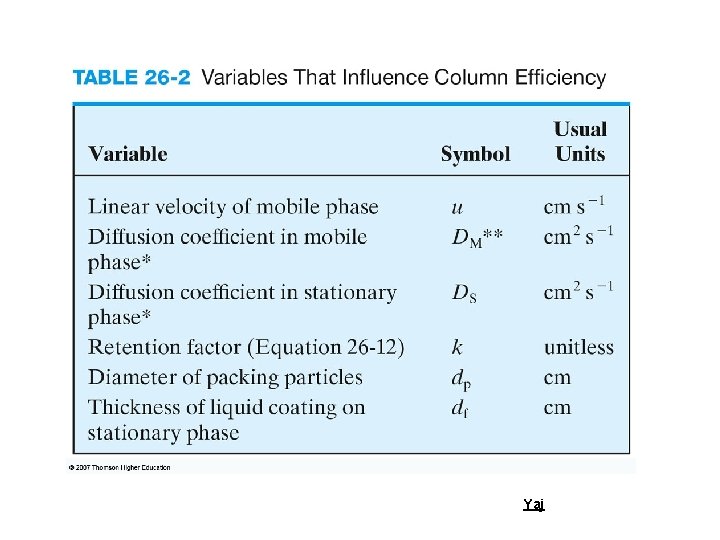
Estimating number of theoretical plates (N) from Retention time Yaj

Yaj

Yaj

Co-Relation between all that was mentioned Yaj

Points to Remember Ø Concept of Retention time Ø Partition Co-efficient and retention factor Ø Selectivity Factor Ø Resolution Ø Efficiency Ø Plate Theory of Chromatography Yaj

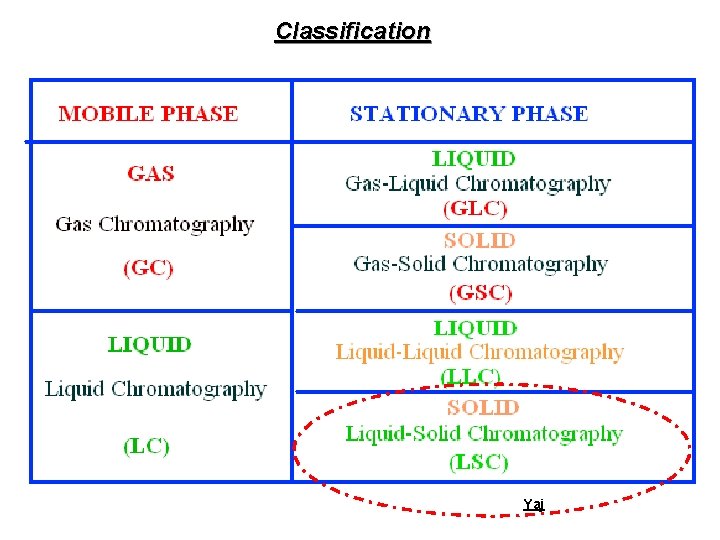
Classification Yaj

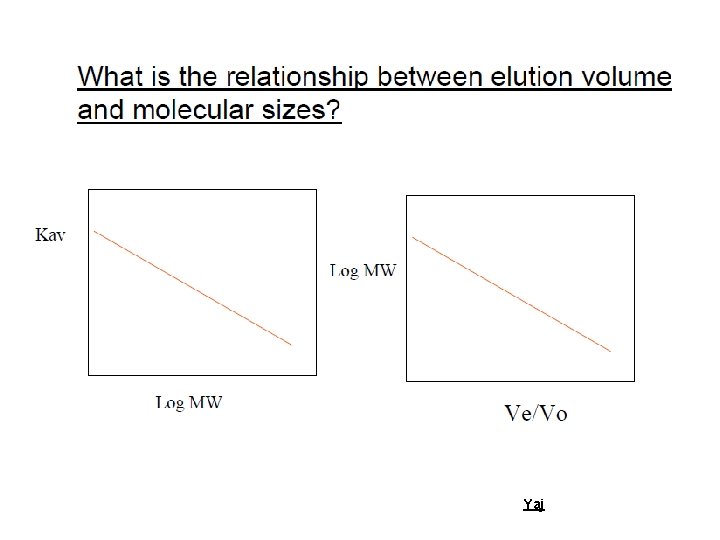
Size-Exclusion Chromatography (SEC) aka Gel-Permeation Chromatography (GPC) aka Gel Filtration Yaj

Definition Type of chromatography that separates molecules according to differences in size or hydrodynamic volume as they pass through a gel filtration medium packed in a column Yaj

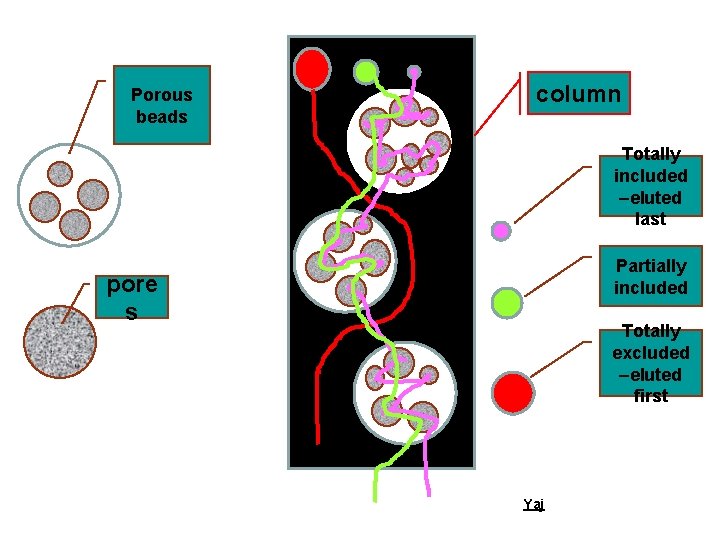
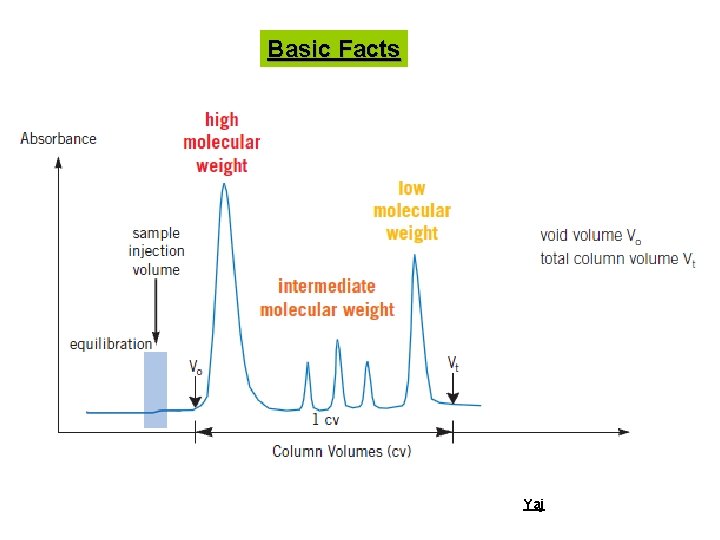
Basic Facts Ø Molecules are separated based on their size and shape using a wide array of porous materials aka gel Ø Each molecule of the gel has a porous structure Yaj

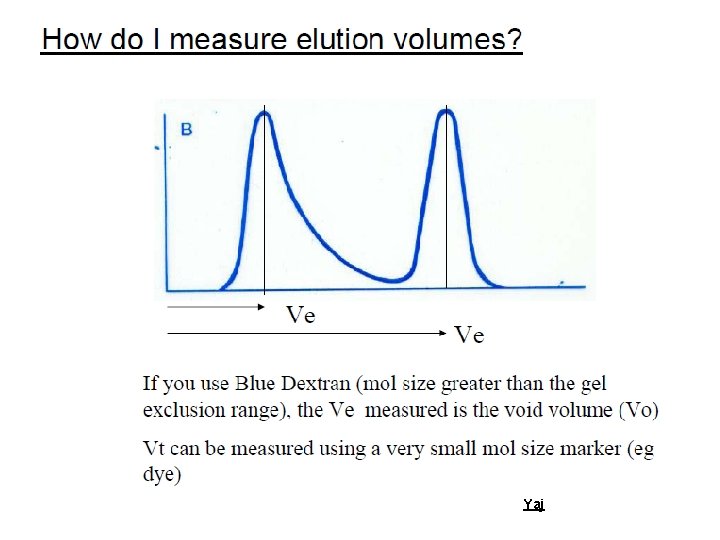
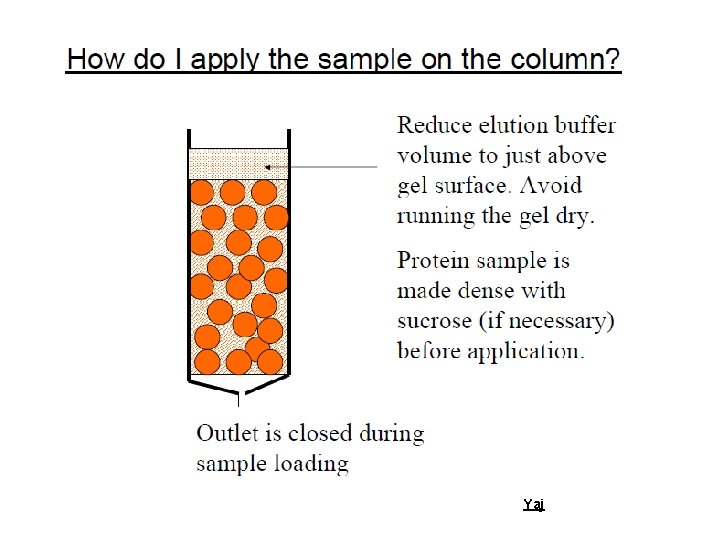
Basic Facts Ø A column of gel granules, equilibrated with appropriate buffer is set up. Molecules of different molecular sizes are eluted through this column. Large molecules will be excluded from the pores of these granules and will pass thorough the interstitial space and eluted first. Ø Large molecules will appear from the column first, followed by smaller molecules Yaj

Porous beads column Totally included –eluted last Partially included pore s Totally excluded –eluted first Yaj

Basic Facts Yaj

Types of gels used Ø Cross linked polymers Ø Uncharged and inert i. e. don’t bind or react with the materials being analyzed Ø Three types of gels are commonly used: Yaj

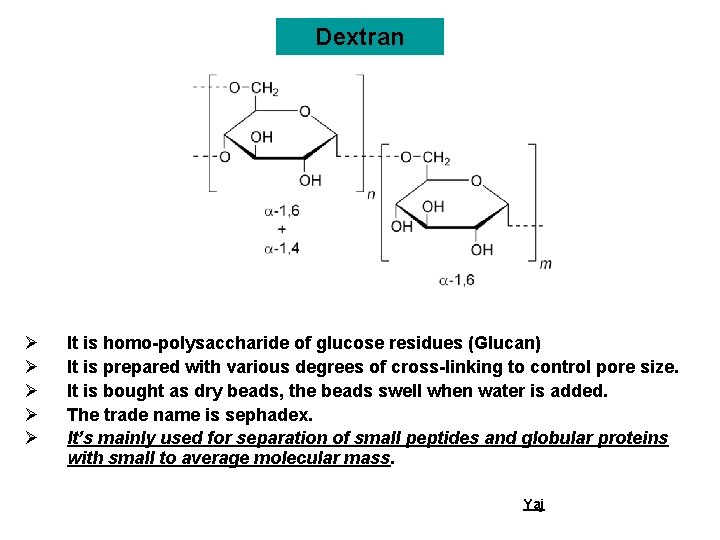
Dextran Ø Ø Ø It is homo-polysaccharide of glucose residues (Glucan) It is prepared with various degrees of cross-linking to control pore size. It is bought as dry beads, the beads swell when water is added. The trade name is sephadex. It’s mainly used for separation of small peptides and globular proteins with small to average molecular mass. Yaj

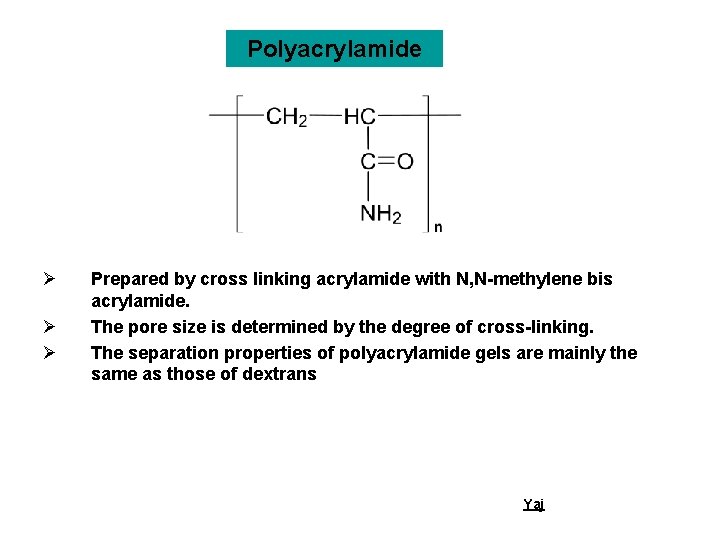
Polyacrylamide Ø Ø Ø Prepared by cross linking acrylamide with N, N-methylene bis acrylamide. The pore size is determined by the degree of cross-linking. The separation properties of polyacrylamide gels are mainly the same as those of dextrans Yaj

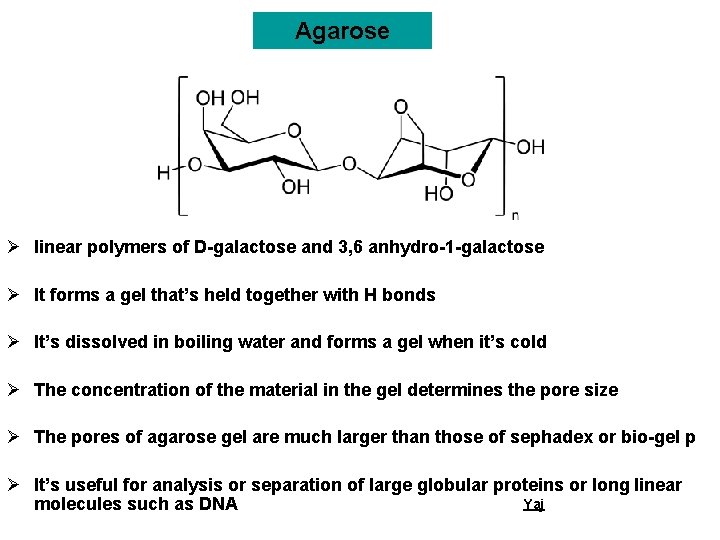
Agarose Ø linear polymers of D-galactose and 3, 6 anhydro-1 -galactose Ø It forms a gel that’s held together with H bonds Ø It’s dissolved in boiling water and forms a gel when it’s cold Ø The concentration of the material in the gel determines the pore size Ø The pores of agarose gel are much larger than those of sephadex or bio-gel p Ø It’s useful for analysis or separation of large globular proteins or long linear Yaj molecules such as DNA

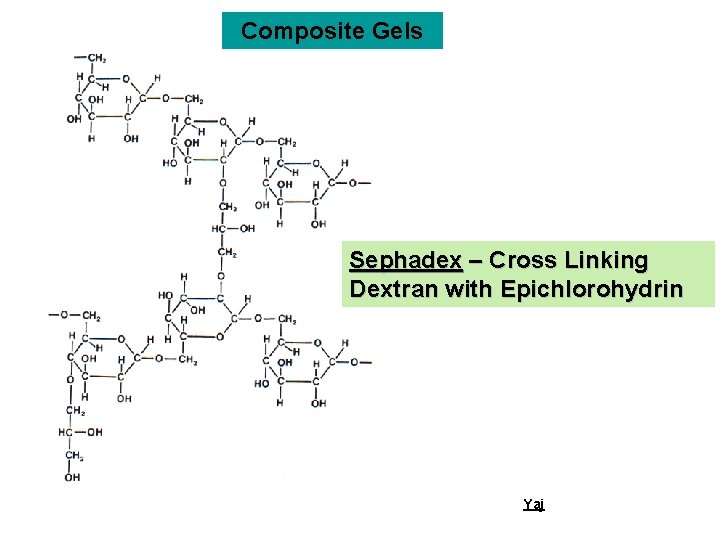
Composite Gels Sephadex – Cross Linking Dextran with Epichlorohydrin Yaj

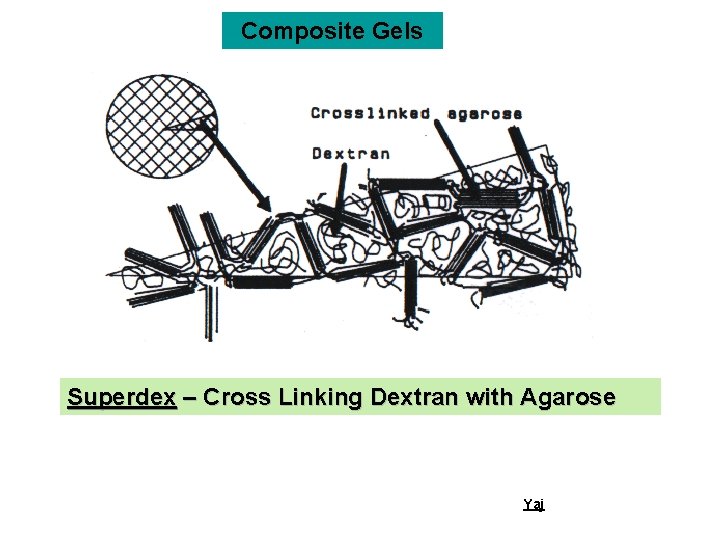
Composite Gels Superdex – Cross Linking Dextran with Agarose Yaj

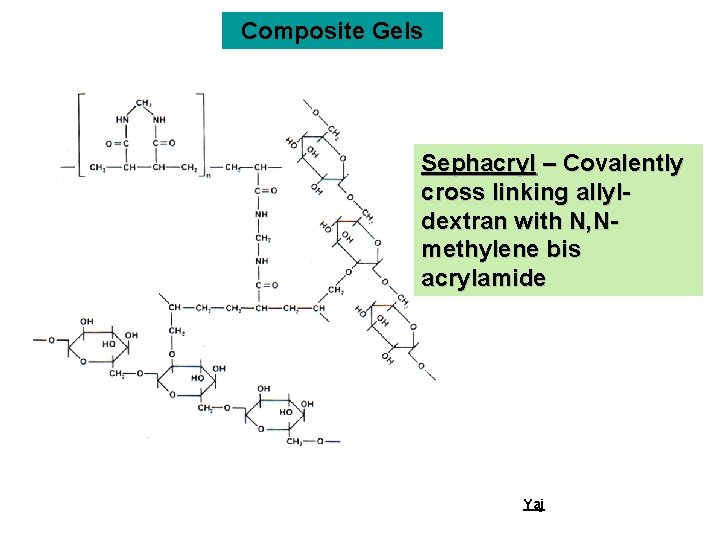
Composite Gels Sephacryl – Covalently cross linking allyldextran with N, Nmethylene bis acrylamide Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Yaj

Points to Remember Ø Size Exclusion chromatography separates on the basis of size Ø The analyte(s) do not interact chemically with the matrix – (Molecular sieve) Ø HMW analytes are eluted first, and LMW analytes later Yaj

Uses of Gel-Filtration Yaj

Yaj Separation of Proteins

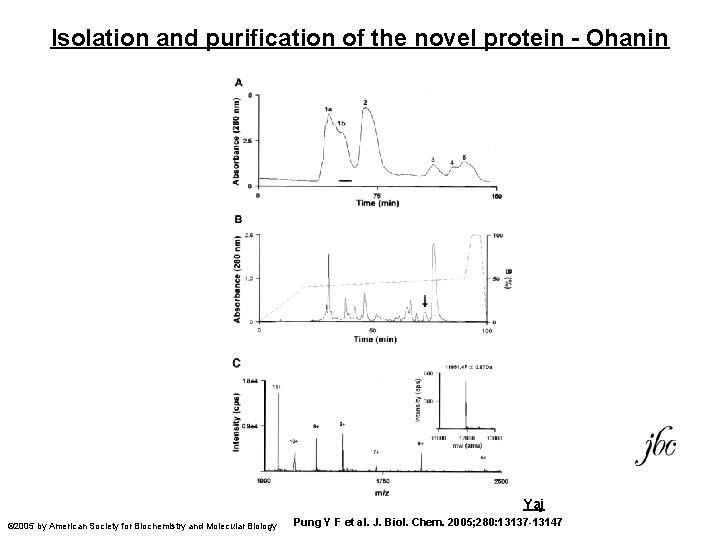
Isolation and purification of the novel protein - Ohanin Yaj © 2005 by American Society for Biochemistry and Molecular Biology Pung Y F et al. J. Biol. Chem. 2005; 280: 13137 -13147

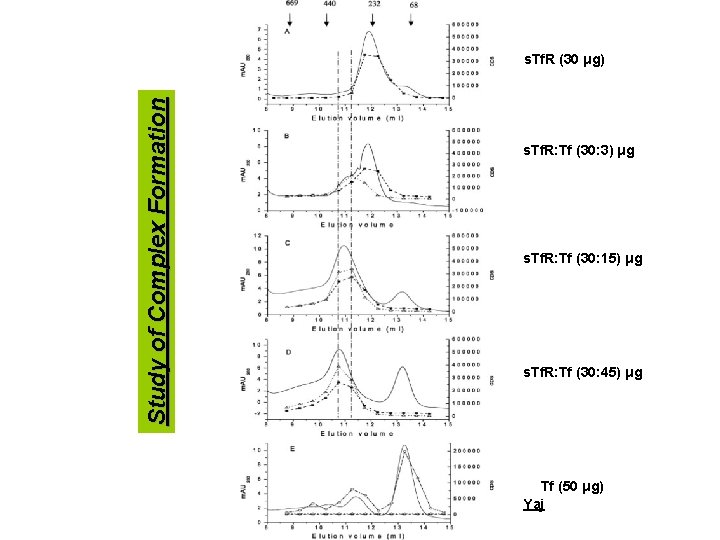
Yaj Study of Complex Formation

Study of Complex Formation s. Tf. R (30 μg) s. Tf. R: Tf (30: 3) μg s. Tf. R: Tf (30: 15) μg s. Tf. R: Tf (30: 45) μg Tf (50 μg) Yaj

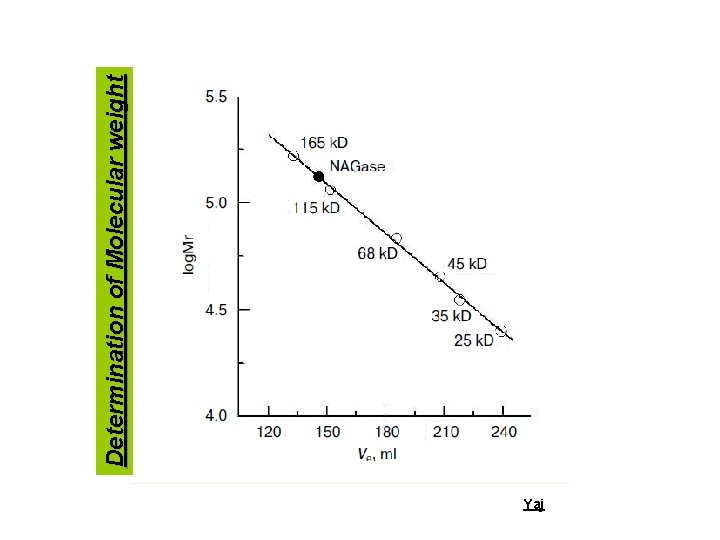
Yaj Determination of Molecular weight

Yaj Determination of Molecular weight

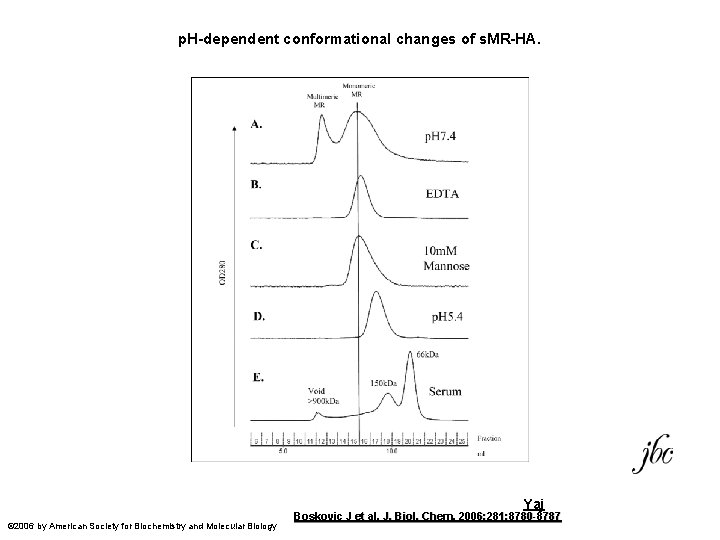
Yaj Conformational change by SEC

p. H-dependent conformational changes of s. MR-HA. Yaj © 2006 by American Society for Biochemistry and Molecular Biology Boskovic J et al. J. Biol. Chem. 2006; 281: 8780 -8787

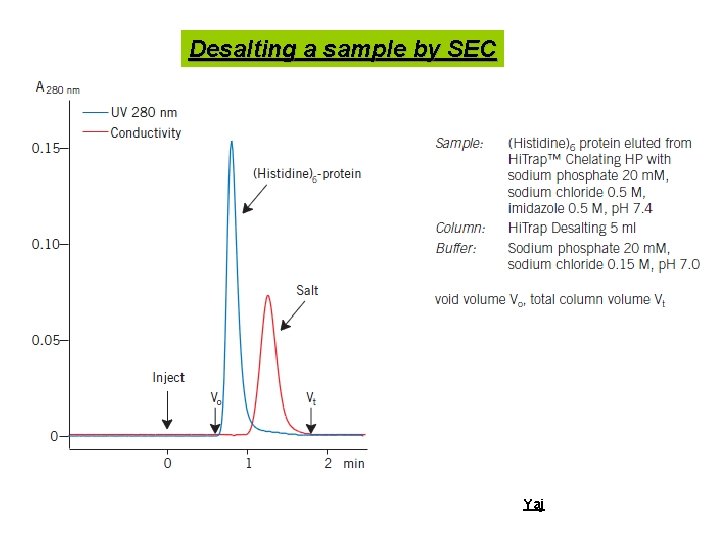
Desalting a sample by SEC Yaj

Please Go through the papers in the way that was mentioned previously Yaj