Lecture 2 Embryonic stem cells Programming and Reprogramming

Lecture 2 Embryonic stem cells, Programming and Reprogramming • • • The discovery of ES cells Developmental origin of ES cells Signalling pathways regulating ES cells in vitro 2 i and the concept of ground state pluripotency What is reprogramming Reprogramming in normal development Experimental reprogramming and cloning Molecular basis of pluripotency Human ES cells and their application in medicine You should understand • What are ES cells and where do they come from • Methods for reprogramming somatic cells • Appplications of ES cell technology
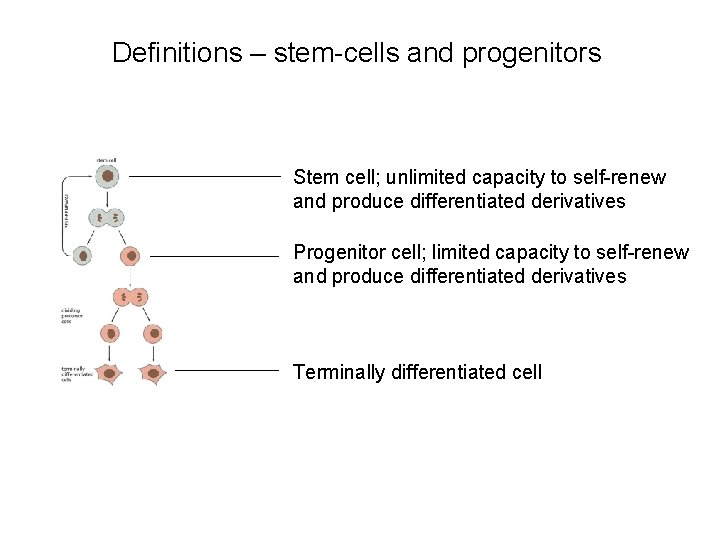
Definitions – stem-cells and progenitors Stem cell; unlimited capacity to self-renew and produce differentiated derivatives Progenitor cell; limited capacity to self-renew and produce differentiated derivatives Terminally differentiated cell

Potency of stem cells and progenitors • Totipotent; can differentiate into all cell types, including extraembryonic lineages e. g. morula cells • Pluripotent; can differentiate into all cell types of the three germ layers, ectoderm, mesoderm and endoderm e. g. ES cells • Multipotent; can differentiate into limited number of cell types, e. g. haematopoietic stem cells
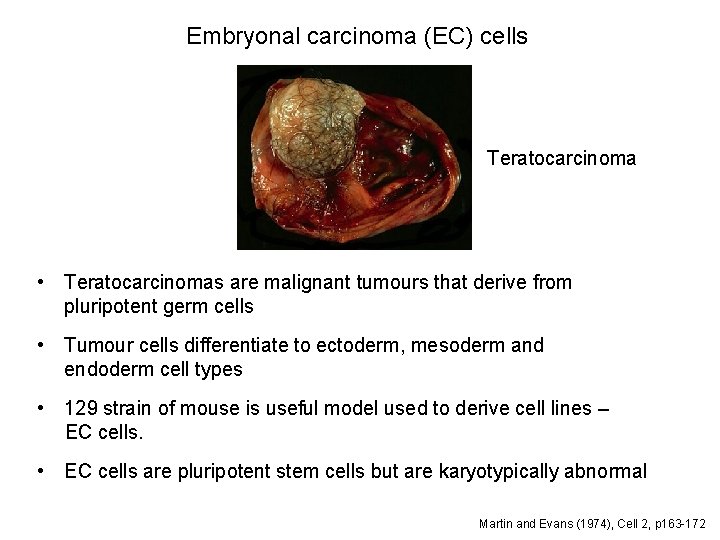
Embryonal carcinoma (EC) cells Teratocarcinoma • Teratocarcinomas are malignant tumours that derive from pluripotent germ cells • Tumour cells differentiate to ectoderm, mesoderm and endoderm cell types • 129 strain of mouse is useful model used to derive cell lines – EC cells. • EC cells are pluripotent stem cells but are karyotypically abnormal Martin and Evans (1974), Cell 2, p 163 -172
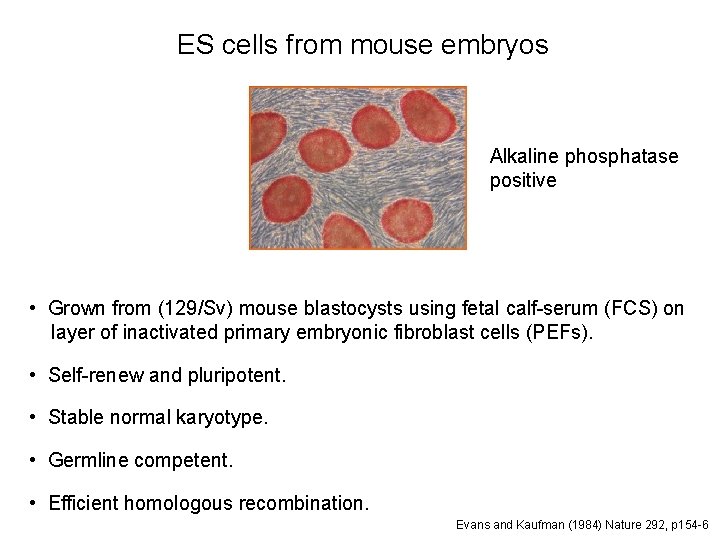
ES cells from mouse embryos Alkaline phosphatase positive • Grown from (129/Sv) mouse blastocysts using fetal calf-serum (FCS) on layer of inactivated primary embryonic fibroblast cells (PEFs). • Self-renew and pluripotent. • Stable normal karyotype. • Germline competent. • Efficient homologous recombination. Evans and Kaufman (1984) Nature 292, p 154 -6
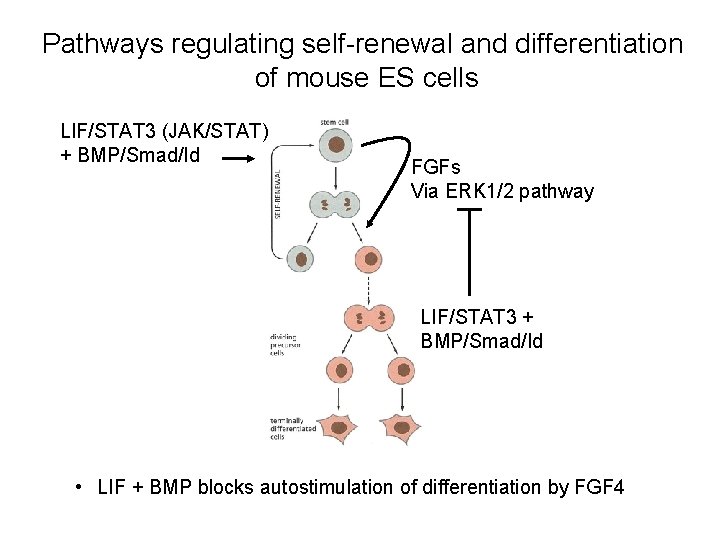
Pathways regulating self-renewal and differentiation of mouse ES cells LIF/STAT 3 (JAK/STAT) + BMP/Smad/Id FGFs Via ERK 1/2 pathway LIF/STAT 3 + BMP/Smad/Id • LIF + BMP blocks autostimulation of differentiation by FGF 4
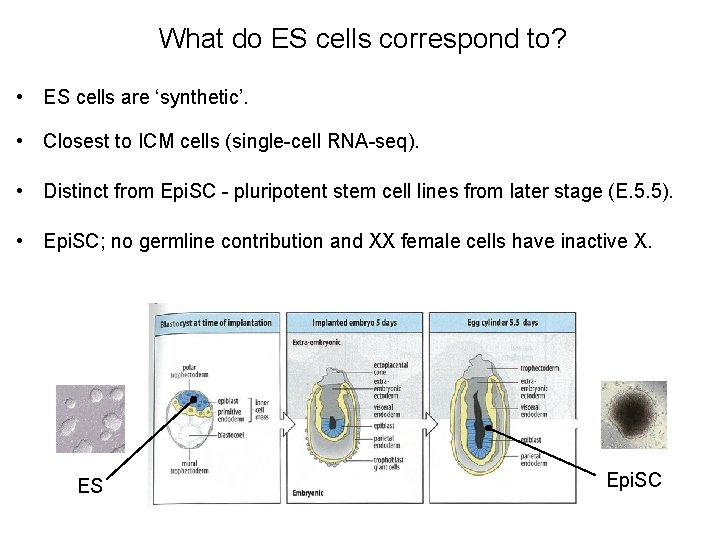
What do ES cells correspond to? • ES cells are ‘synthetic’. • Closest to ICM cells (single-cell RNA-seq). • Distinct from Epi. SC - pluripotent stem cell lines from later stage (E. 5. 5). • Epi. SC; no germline contribution and XX female cells have inactive X. ES Epi. SC

Transcription factor circuitry in ES cells Genome wide analysis; RNA-seq, Ch. IP-seq etc • Nanog, Oct 4, and Sox 2 (NOS) co-occupy many target genes. • Targets can be either activated or repressed. • NOS positively regulate one another • NOS negatively regulate master TFs for other lineages eg trophectoderm and primitive endoderm lineages. • Repressed targets ‘poised’ by epigenetic mechanisms (e. g. Polycomb). Boyer et al (2005) Cell 122, p 947 -56
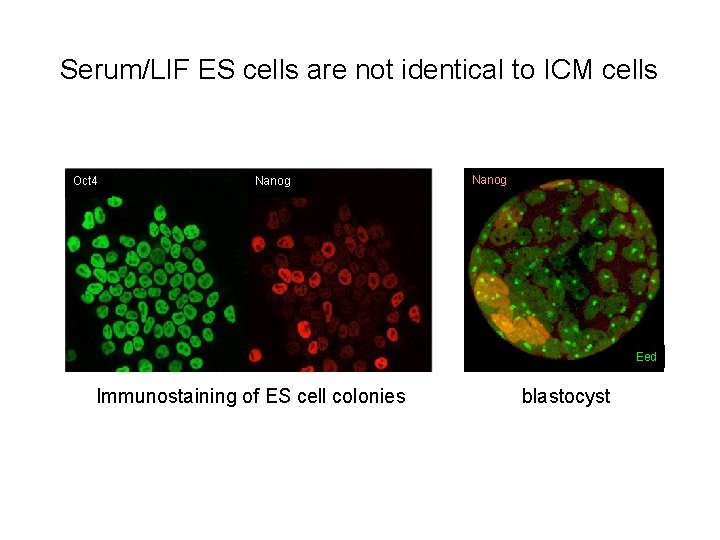
Serum/LIF ES cells are not identical to ICM cells Oct 4 Nanog Eed Immunostaining of ES cell colonies blastocyst
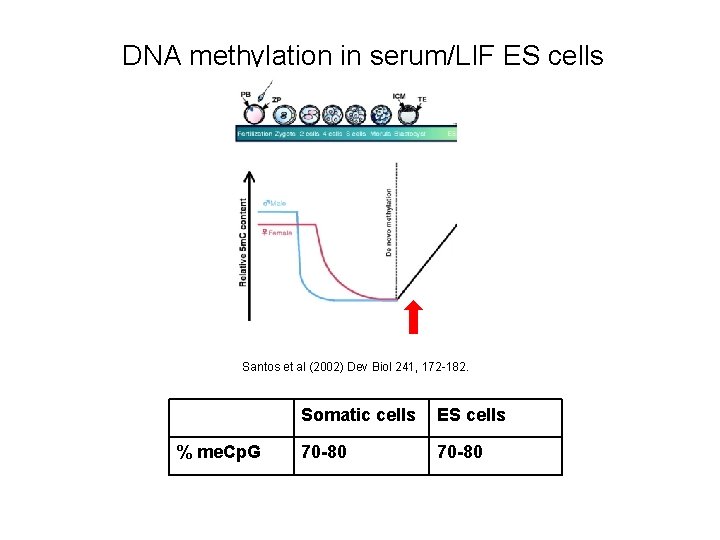
DNA methylation in serum/LIF ES cells Santos et al (2002) Dev Biol 241, 172 -182. % me. Cp. G Somatic cells ES cells 70 -80
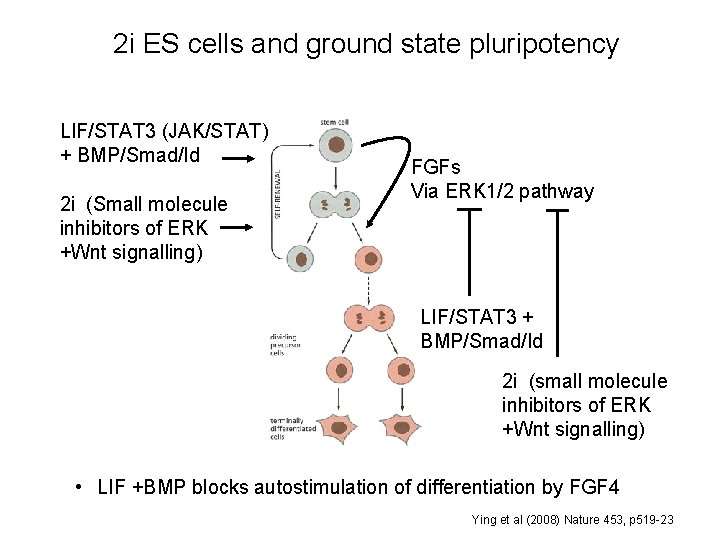
2 i ES cells and ground state pluripotency LIF/STAT 3 (JAK/STAT) + BMP/Smad/Id 2 i (Small molecule inhibitors of ERK +Wnt signalling) FGFs Via ERK 1/2 pathway LIF/STAT 3 + BMP/Smad/Id 2 i (small molecule inhibitors of ERK +Wnt signalling) • LIF +BMP blocks autostimulation of differentiation by FGF 4 Ying et al (2008) Nature 453, p 519 -23
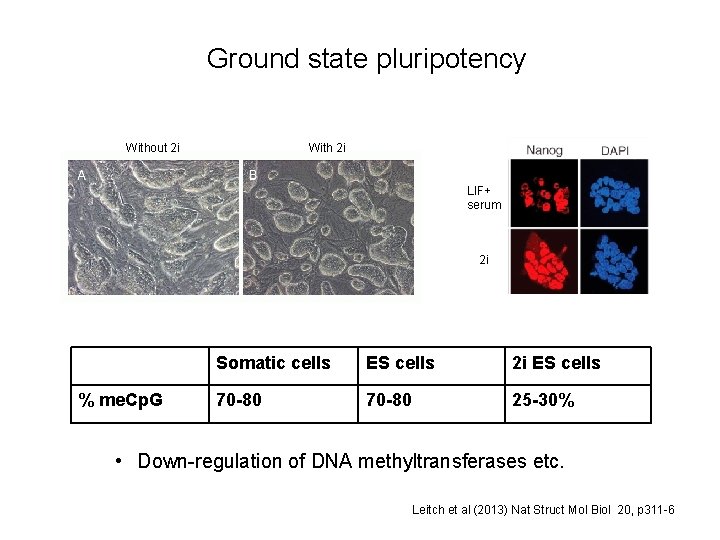
Ground state pluripotency Without 2 i With 2 i LIF+ serum 2 i % me. Cp. G Somatic cells ES cells 2 i ES cells 70 -80 25 -30% • Down-regulation of DNA methyltransferases etc. Leitch et al (2013) Nat Struct Mol Biol 20, p 311 -6

Applications of the 2 i method • ESCs from any mouse strain and from other species, eg rat (Buehr et al, 2008, Cell 135, p 1287 -98)
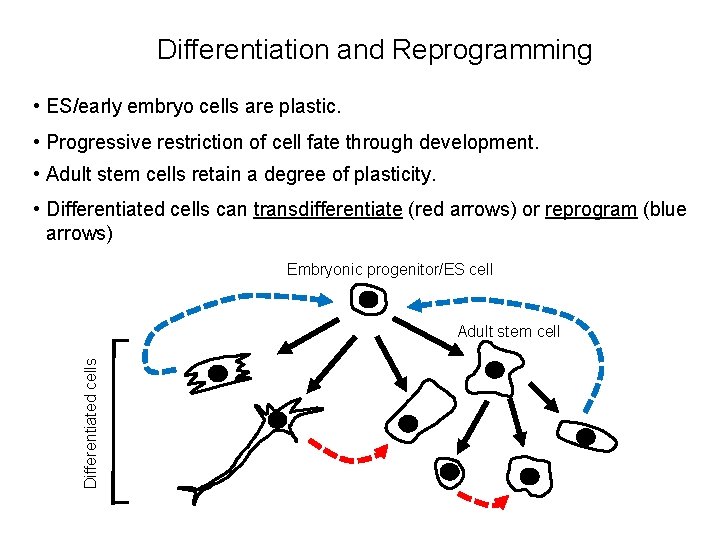
Differentiation and Reprogramming • ES/early embryo cells are plastic. • Progressive restriction of cell fate through development. • Adult stem cells retain a degree of plasticity. • Differentiated cells can transdifferentiate (red arrows) or reprogram (blue arrows) Embryonic progenitor/ES cell Differentiated cells Adult stem cell
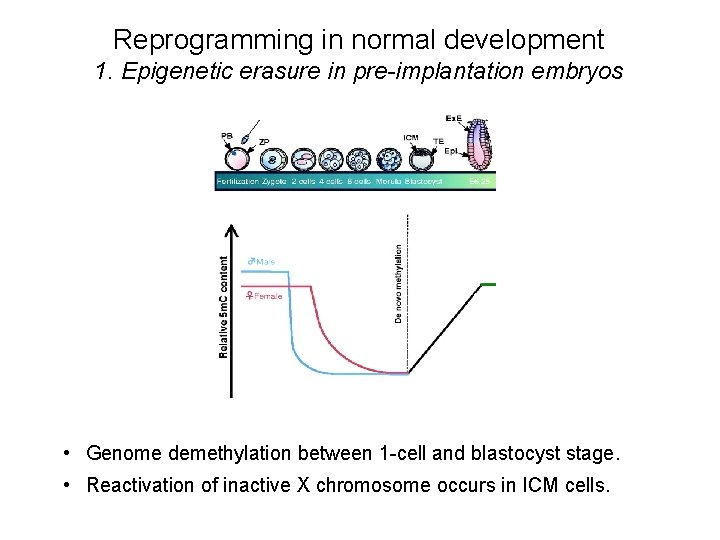
Reprogramming in normal development 1. Epigenetic erasure in pre-implantation embryos • Genome demethylation between 1 -cell and blastocyst stage. • Reactivation of inactive X chromosome occurs in ICM cells.
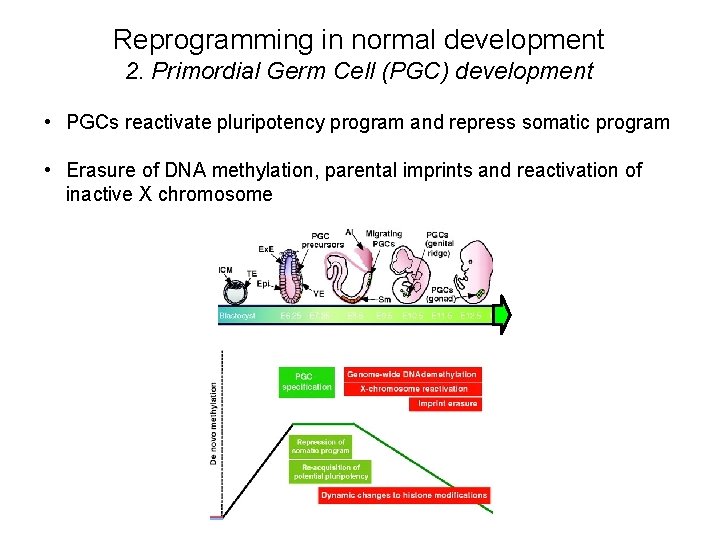
Reprogramming in normal development 2. Primordial Germ Cell (PGC) development • PGCs reactivate pluripotency program and repress somatic program • Erasure of DNA methylation, parental imprints and reactivation of inactive X chromosome
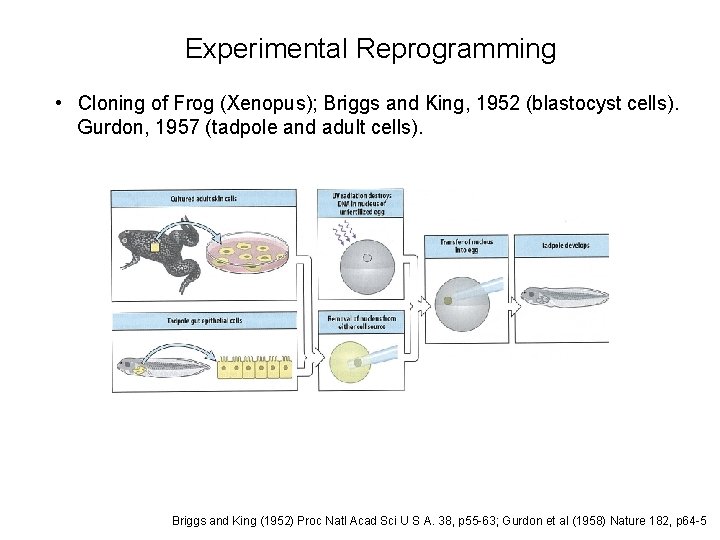
Experimental Reprogramming • Cloning of Frog (Xenopus); Briggs and King, 1952 (blastocyst cells). Gurdon, 1957 (tadpole and adult cells). Briggs and King (1952) Proc Natl Acad Sci U S A. 38, p 55 -63; Gurdon et al (1958) Nature 182, p 64 -5

Cloning in Mammals • 1996; a sheep (Dolly) was the first cloned mammal • Extended to mouse, cat, cow and many other mammalian species. • Cloning of a mouse from a lymphocyte proves cloning of a terminally differentiated cell. • Low success rate (less than 1/100) and cloned animals have health issues. Campbell et al (1996) Nature 380, p 64 -6; Wakayama et al (1998), Nature 394, p 369 -74 ; Hochedlinger and Jaenisch (2002) Nature 415, p 1035 -8
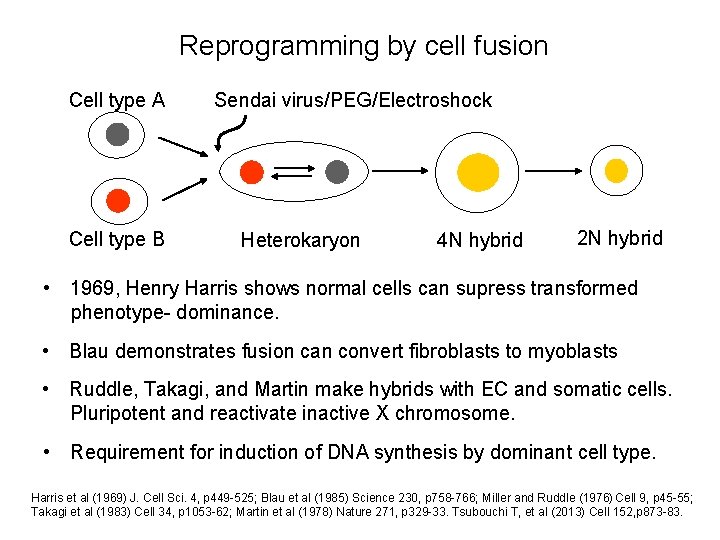
Reprogramming by cell fusion Cell type A Cell type B Sendai virus/PEG/Electroshock Heterokaryon 4 N hybrid 2 N hybrid • 1969, Henry Harris shows normal cells can supress transformed phenotype- dominance. • Blau demonstrates fusion can convert fibroblasts to myoblasts • Ruddle, Takagi, and Martin make hybrids with EC and somatic cells. Pluripotent and reactivate inactive X chromosome. • Requirement for induction of DNA synthesis by dominant cell type. Harris et al (1969) J. Cell Sci. 4, p 449 -525; Blau et al (1985) Science 230, p 758 -766; Miller and Ruddle (1976) Cell 9, p 45 -55; Takagi et al (1983) Cell 34, p 1053 -62; Martin et al (1978) Nature 271, p 329 -33. Tsubouchi T, et al (2013) Cell 152, p 873 -83.
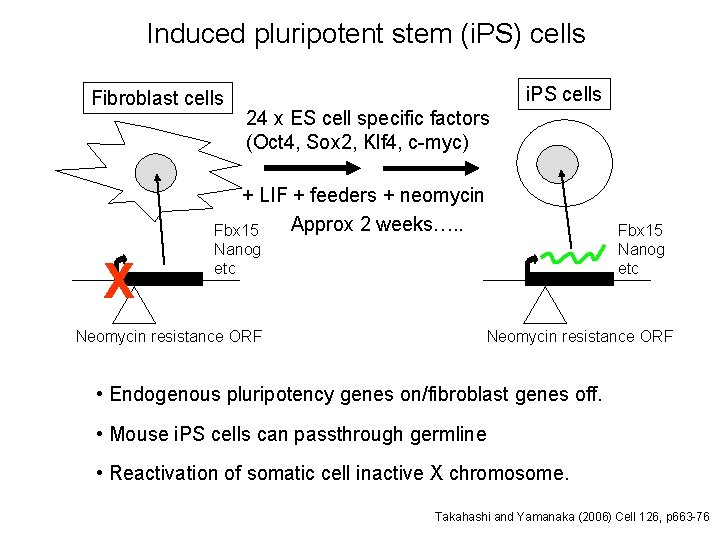
Induced pluripotent stem (i. PS) cells Fibroblast cells i. PS cells 24 x ES cell specific factors (Oct 4, Sox 2, Klf 4, c-myc) + LIF + feeders + neomycin Approx 2 weeks…. . Fbx 15 X Fbx 15 Nanog etc Neomycin resistance ORF • Endogenous pluripotency genes on/fibroblast genes off. • Mouse i. PS cells can passthrough germline • Reactivation of somatic cell inactive X chromosome. Takahashi and Yamanaka (2006) Cell 126, p 663 -76
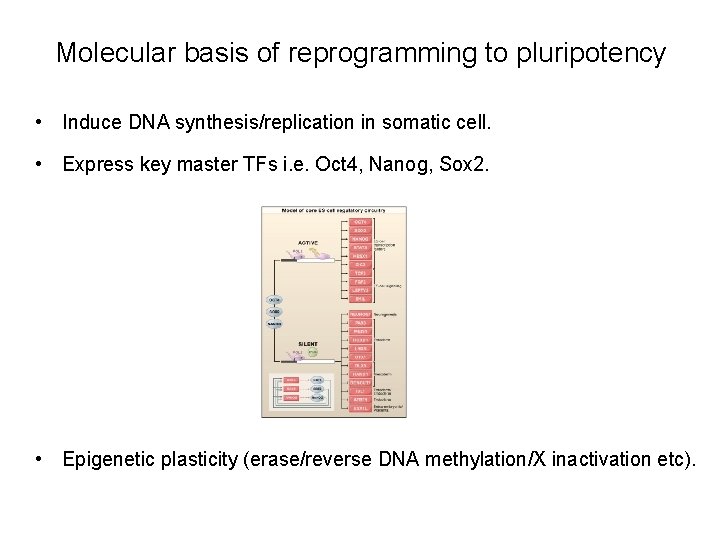
Molecular basis of reprogramming to pluripotency • Induce DNA synthesis/replication in somatic cell. • Express key master TFs i. e. Oct 4, Nanog, Sox 2. • Epigenetic plasticity (erase/reverse DNA methylation/X inactivation etc).
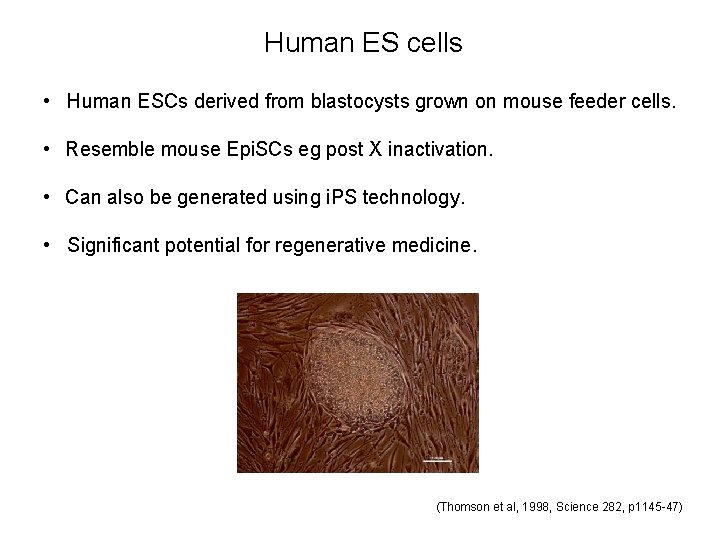
Human ES cells • Human ESCs derived from blastocysts grown on mouse feeder cells. • Resemble mouse Epi. SCs eg post X inactivation. • Can also be generated using i. PS technology. • Significant potential for regenerative medicine. (Thomson et al, 1998, Science 282, p 1145 -47)
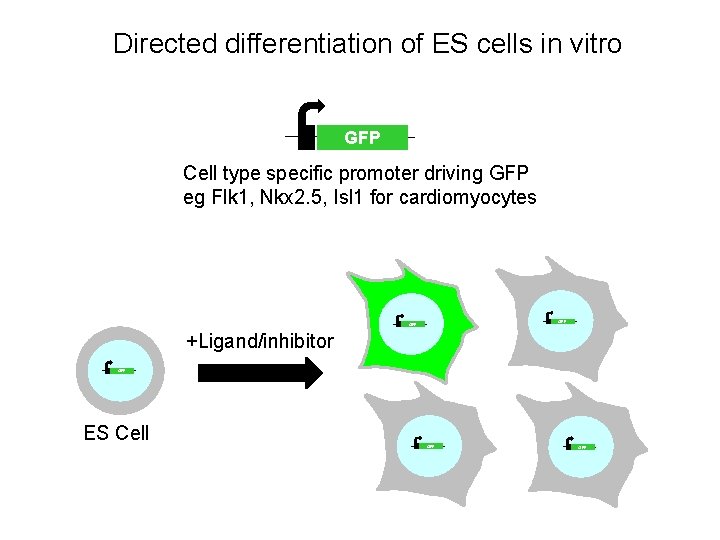
Directed differentiation of ES cells in vitro GFP Cell type specific promoter driving GFP eg Flk 1, Nkx 2. 5, Isl 1 for cardiomyocytes GFP +Ligand/inhibitor GFP ES Cell GFP
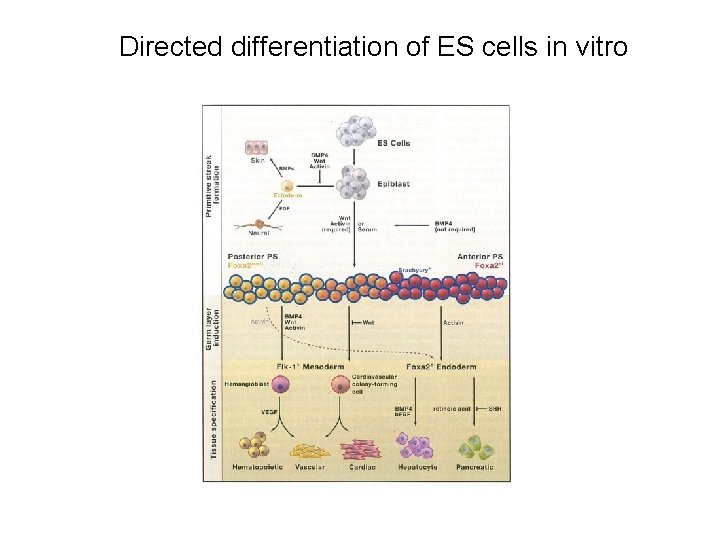
Directed differentiation of ES cells in vitro
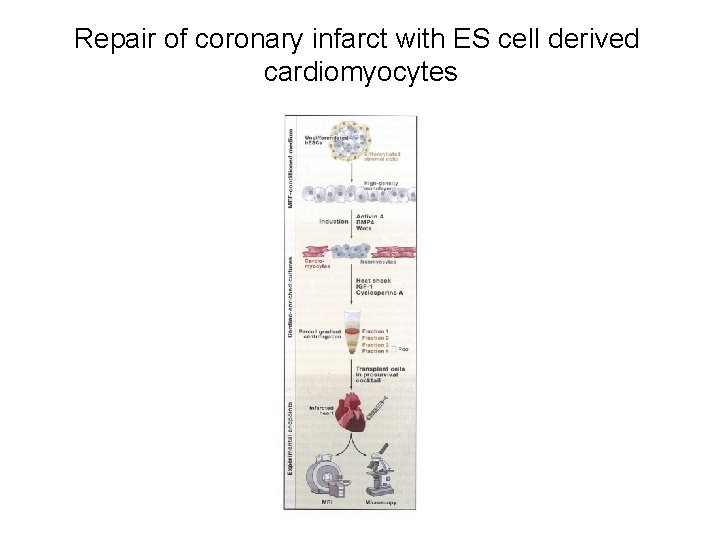
Repair of coronary infarct with ES cell derived cardiomyocytes
ES cell derived cardiomyocytes live cell imaging

Regenerative cell therapy - challenges • Cell type heterogeneity • Graft rejection • Teratocarcinoma forming potential
- Slides: 27