Lecture 2 Chemical Bonds Atomic Orbital Theory and

Lecture 2 Chemical Bonds: Atomic Orbital Theory and Molecular Orbital Theory Dr. A. K. M. Shafiqul Islam 10. 07. 09
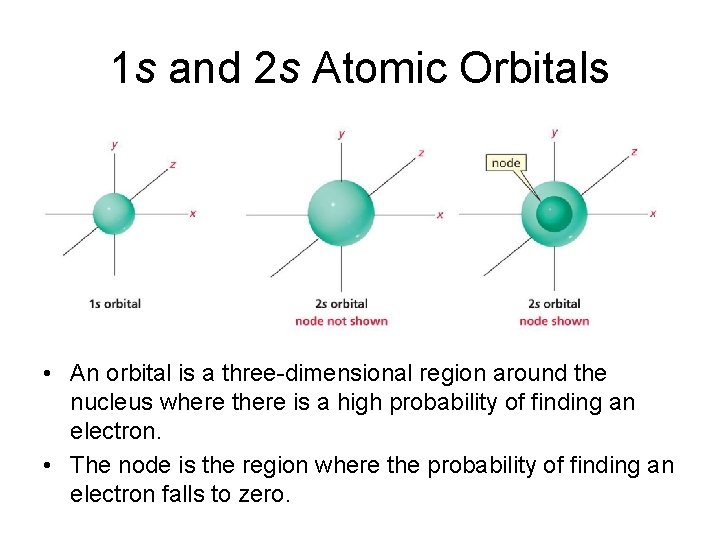
1 s and 2 s Atomic Orbitals • An orbital is a three-dimensional region around the nucleus where there is a high probability of finding an electron. • The node is the region where the probability of finding an electron falls to zero.
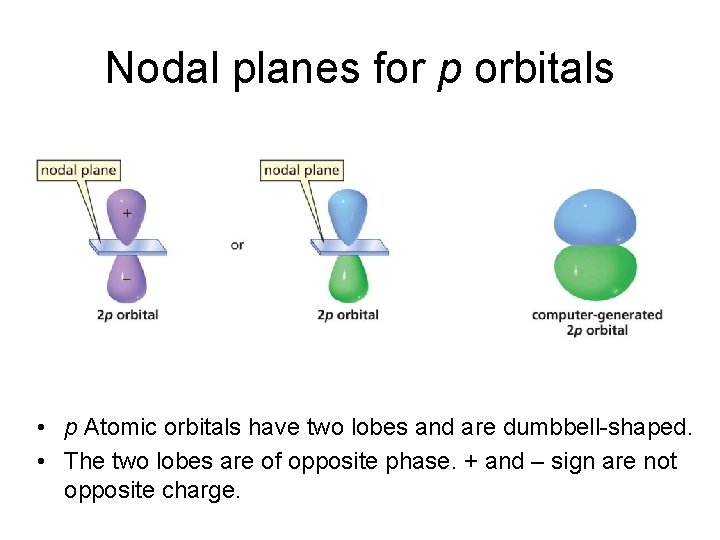
Nodal planes for p orbitals • p Atomic orbitals have two lobes and are dumbbell-shaped. • The two lobes are of opposite phase. + and – sign are not opposite charge.
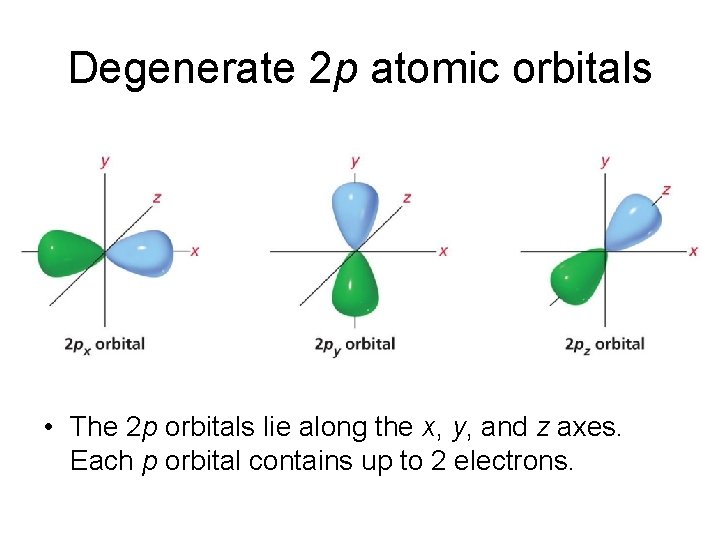
Degenerate 2 p atomic orbitals • The 2 p orbitals lie along the x, y, and z axes. Each p orbital contains up to 2 electrons.
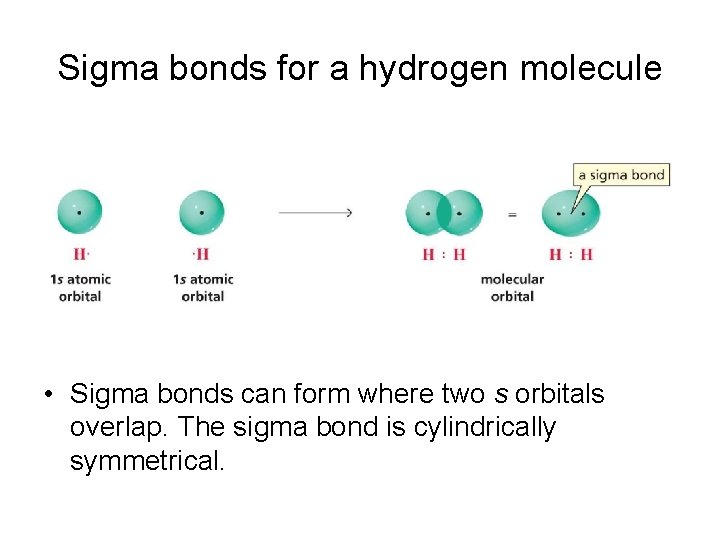
Sigma bonds for a hydrogen molecule • Sigma bonds can form where two s orbitals overlap. The sigma bond is cylindrically symmetrical.

Bond length for hydrogen atoms • The change in potential energy that occurs as two 1 s atomic orbitals approach each other. • The internuclear distance at minimum energy is the length of the hydrogen-hydrogen covalent bond.
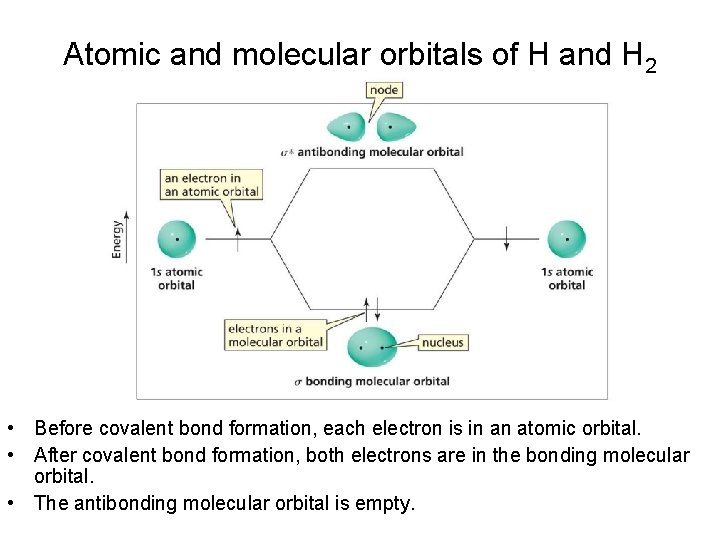
Atomic and molecular orbitals of H and H 2 • Before covalent bond formation, each electron is in an atomic orbital. • After covalent bond formation, both electrons are in the bonding molecular orbital. • The antibonding molecular orbital is empty.
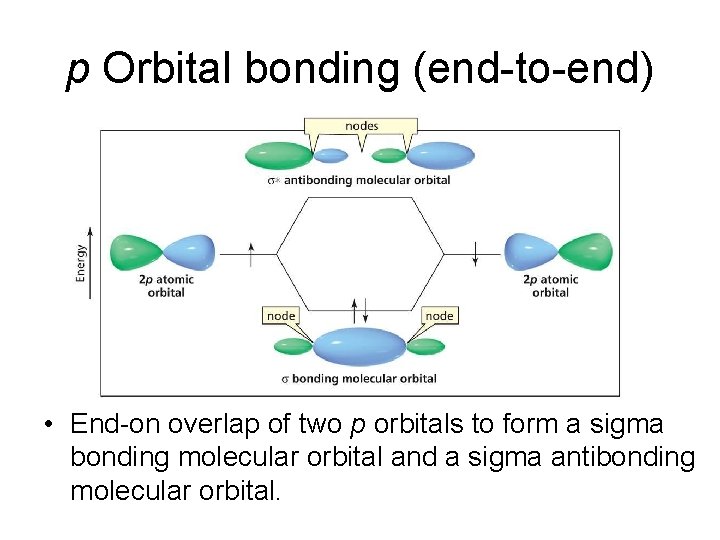
p Orbital bonding (end-to-end) • End-on overlap of two p orbitals to form a sigma bonding molecular orbital and a sigma antibonding molecular orbital.
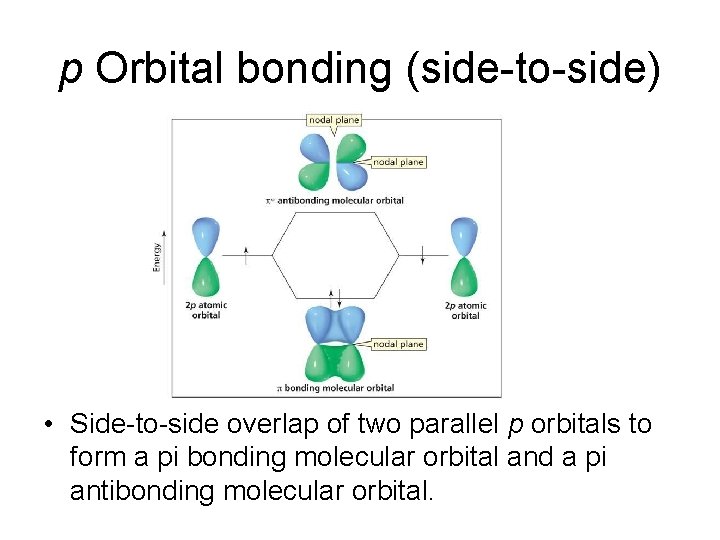
p Orbital bonding (side-to-side) • Side-to-side overlap of two parallel p orbitals to form a pi bonding molecular orbital and a pi antibonding molecular orbital.
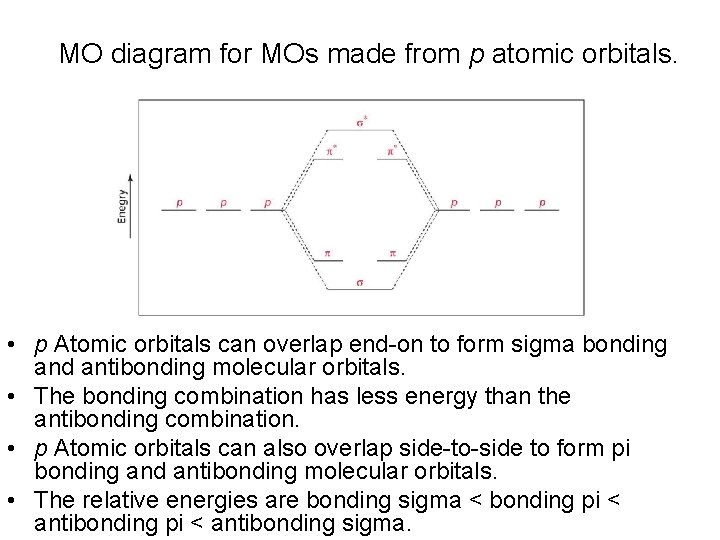
MO diagram for MOs made from p atomic orbitals. • p Atomic orbitals can overlap end-on to form sigma bonding and antibonding molecular orbitals. • The bonding combination has less energy than the antibonding combination. • p Atomic orbitals can also overlap side-to-side to form pi bonding and antibonding molecular orbitals. • The relative energies are bonding sigma < bonding pi < antibonding sigma.

Carbon-oxygen pi bond formation • Side-to-side overlap of a p atomic orbital from carbon with a p atomic orbital from oxygen results in pi bonding and pi antibonding molecular orbitals

Models of methane The ball-and-stick model, the space-filling model, and the electrostatic potential map are shown for methane

Relative Energies of Atomic Orbitals

The Electronic Configurations of the Smallest Atoms
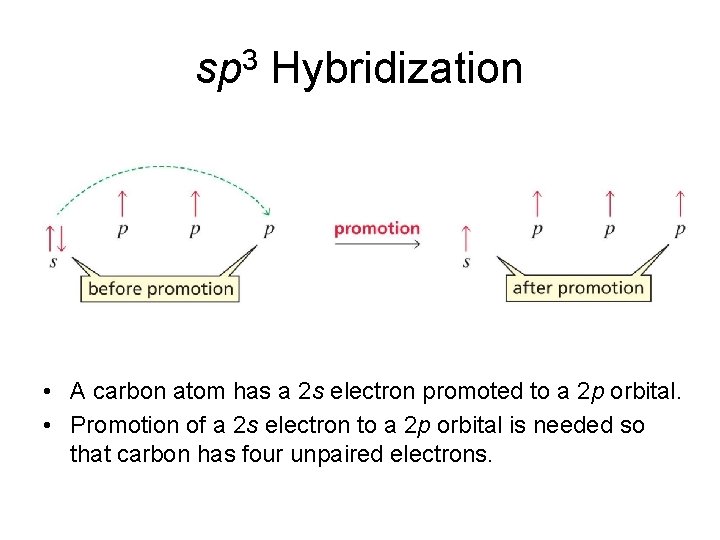
sp 3 Hybridization • A carbon atom has a 2 s electron promoted to a 2 p orbital. • Promotion of a 2 s electron to a 2 p orbital is needed so that carbon has four unpaired electrons.
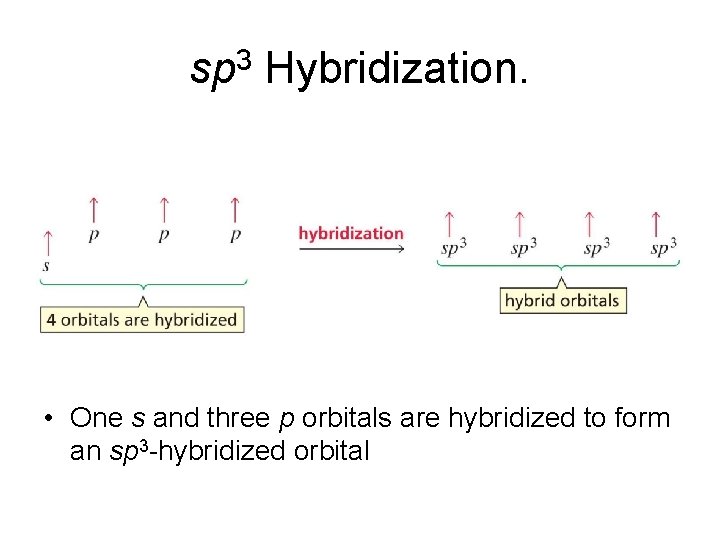
sp 3 Hybridization. • One s and three p orbitals are hybridized to form an sp 3 -hybridized orbital
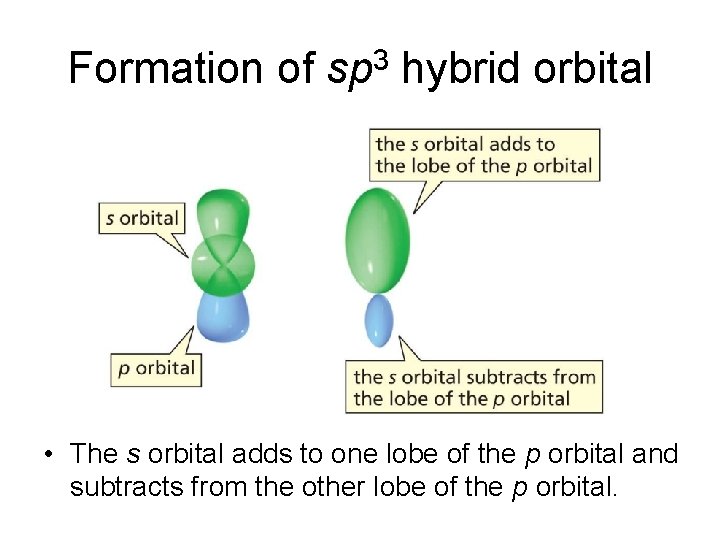
Formation of sp 3 hybrid orbital • The s orbital adds to one lobe of the p orbital and subtracts from the other lobe of the p orbital.
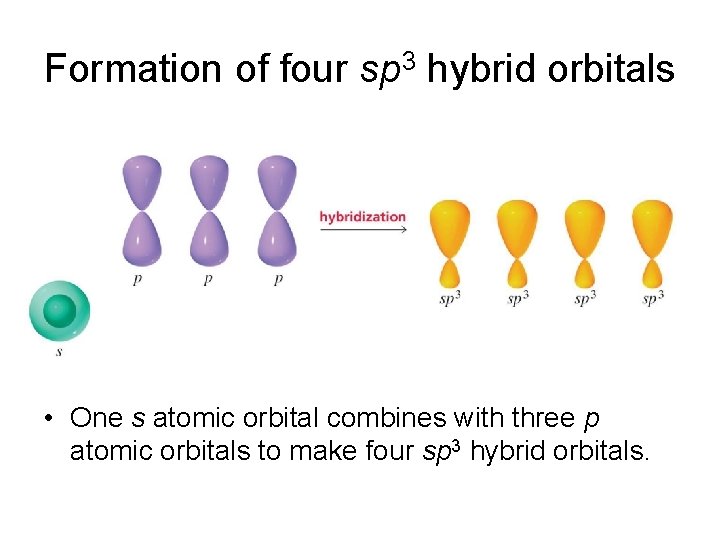
Formation of four sp 3 hybrid orbitals • One s atomic orbital combines with three p atomic orbitals to make four sp 3 hybrid orbitals.
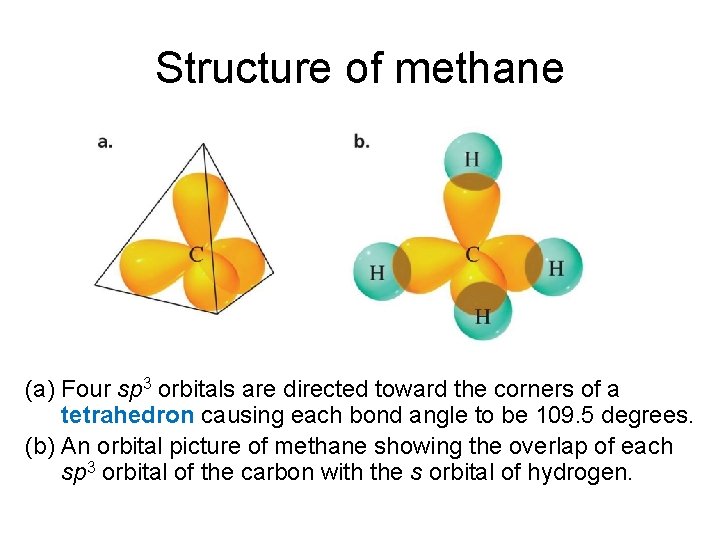
Structure of methane (a) Four sp 3 orbitals are directed toward the corners of a tetrahedron causing each bond angle to be 109. 5 degrees. (b) An orbital picture of methane showing the overlap of each sp 3 orbital of the carbon with the s orbital of hydrogen.
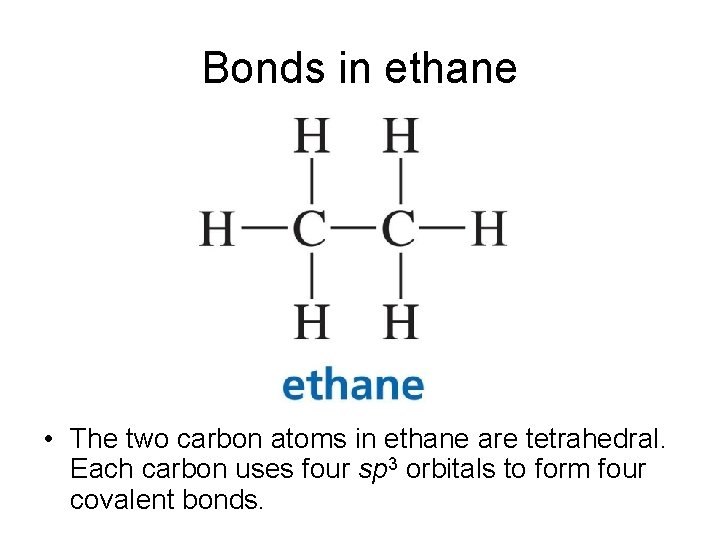
Bonds in ethane • The two carbon atoms in ethane are tetrahedral. Each carbon uses four sp 3 orbitals to form four covalent bonds.
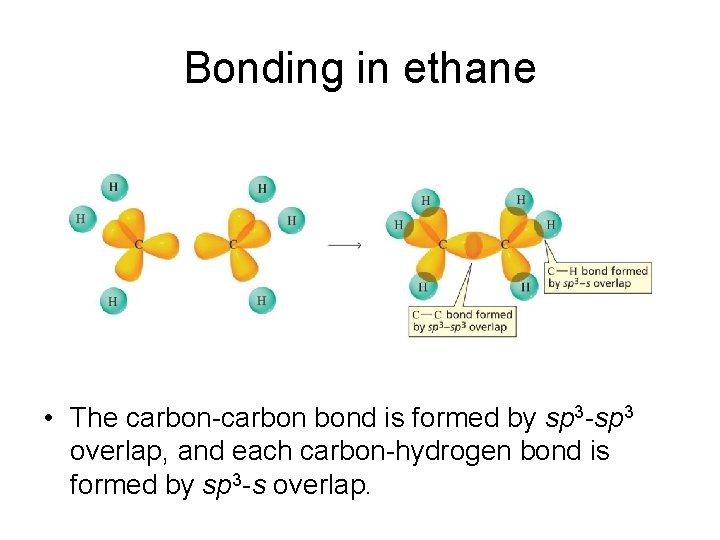
Bonding in ethane • The carbon-carbon bond is formed by sp 3 -sp 3 overlap, and each carbon-hydrogen bond is formed by sp 3 -s overlap.
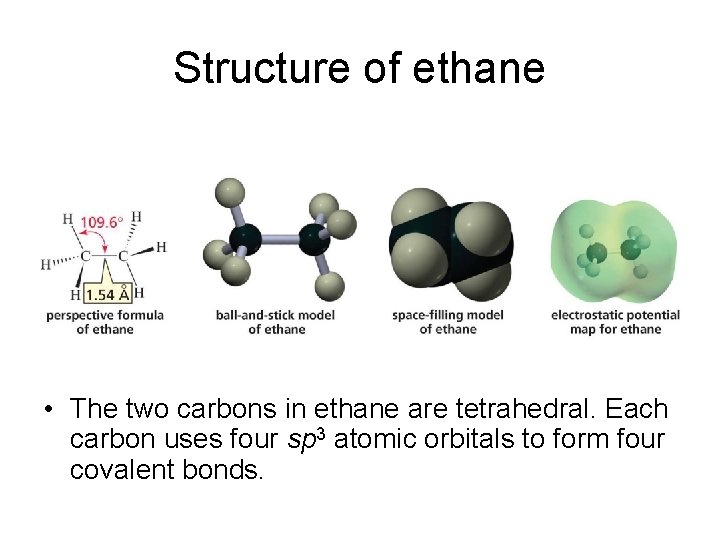
Structure of ethane • The two carbons in ethane are tetrahedral. Each carbon uses four sp 3 atomic orbitals to form four covalent bonds.
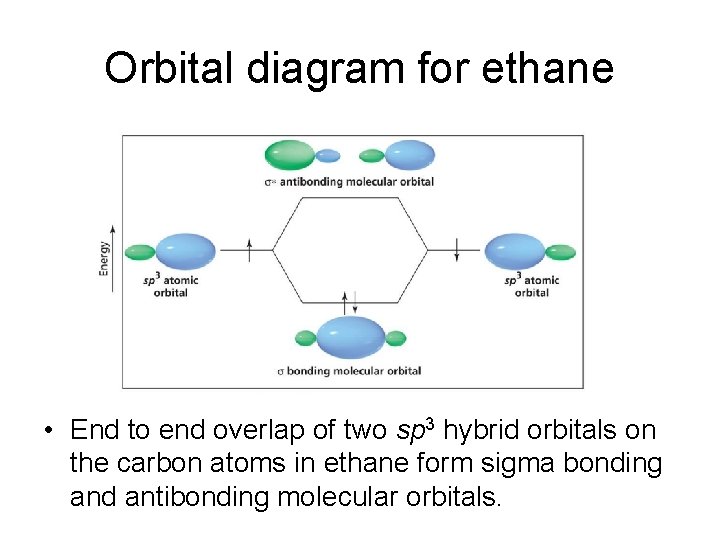
Orbital diagram for ethane • End to end overlap of two sp 3 hybrid orbitals on the carbon atoms in ethane form sigma bonding and antibonding molecular orbitals.
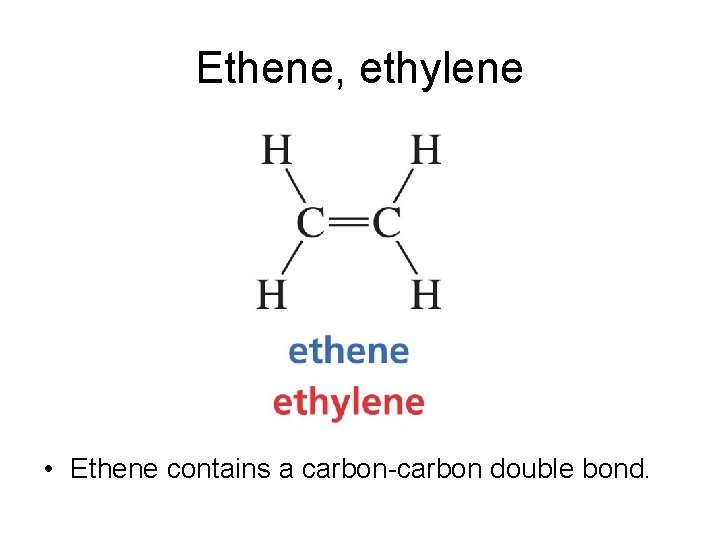
Ethene, ethylene • Ethene contains a carbon-carbon double bond.

sp 2 Hybridization • A carbon atom has a 2 s electron promoted to a 2 p orbital. • One s and two p orbitals are hybridized to form an sp 2 hybridized orbital.
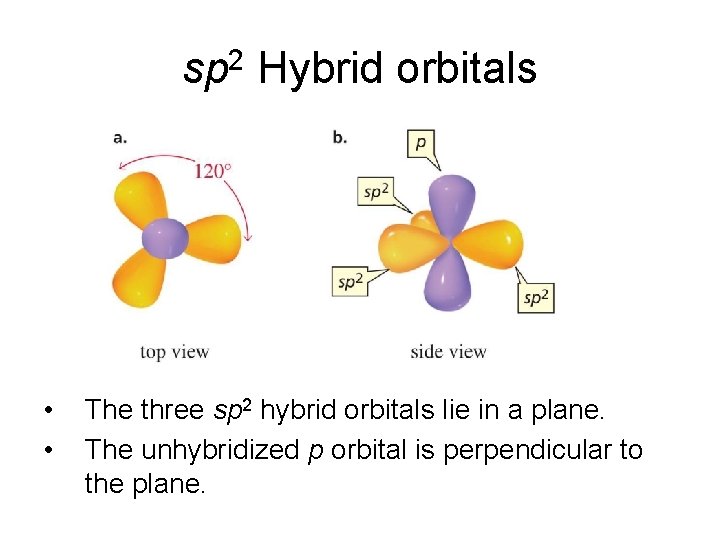
sp 2 Hybrid orbitals • • The three sp 2 hybrid orbitals lie in a plane. The unhybridized p orbital is perpendicular to the plane.
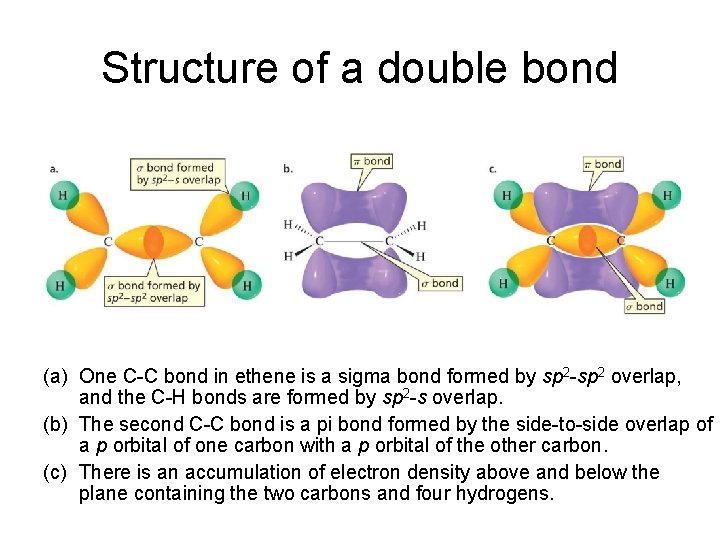
Structure of a double bond (a) One C-C bond in ethene is a sigma bond formed by sp 2 -sp 2 overlap, and the C-H bonds are formed by sp 2 -s overlap. (b) The second C-C bond is a pi bond formed by the side-to-side overlap of a p orbital of one carbon with a p orbital of the other carbon. (c) There is an accumulation of electron density above and below the plane containing the two carbons and four hydrogens.
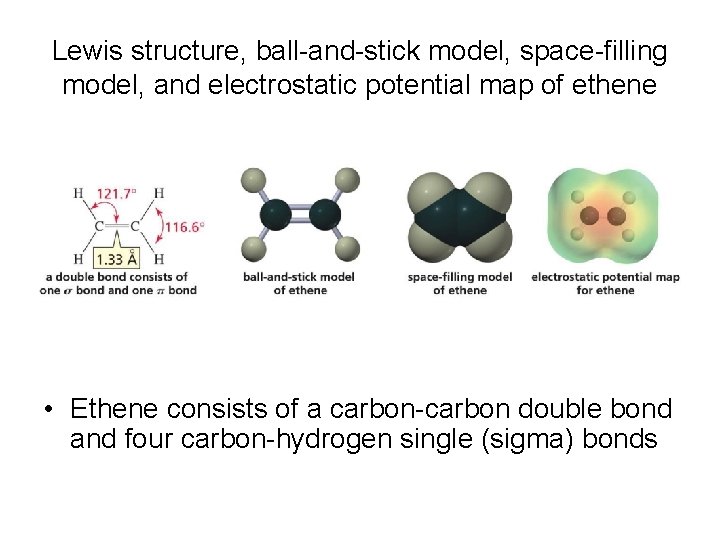
Lewis structure, ball-and-stick model, space-filling model, and electrostatic potential map of ethene • Ethene consists of a carbon-carbon double bond and four carbon-hydrogen single (sigma) bonds
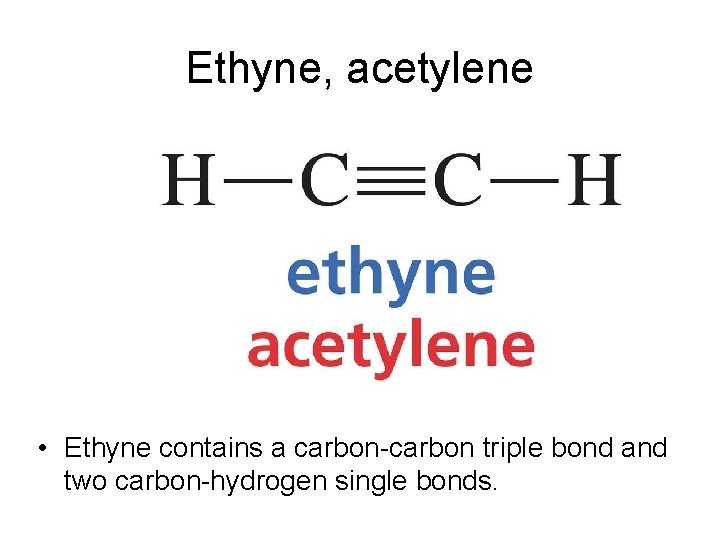
Ethyne, acetylene • Ethyne contains a carbon-carbon triple bond and two carbon-hydrogen single bonds.
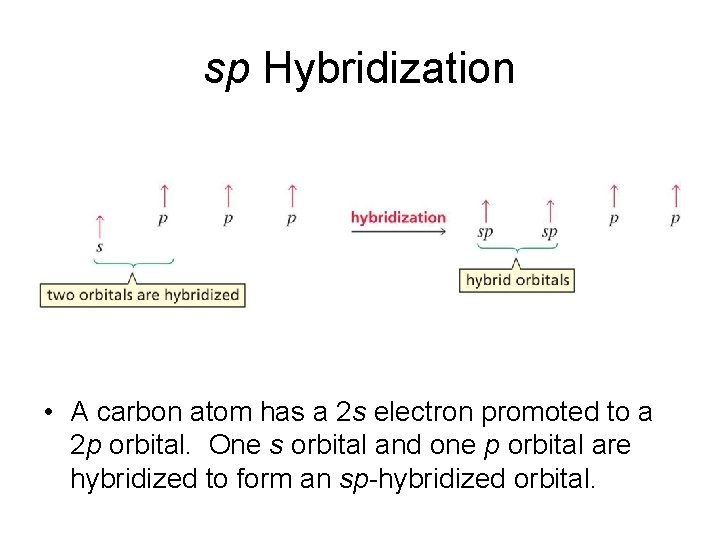
sp Hybridization • A carbon atom has a 2 s electron promoted to a 2 p orbital. One s orbital and one p orbital are hybridized to form an sp-hybridized orbital.
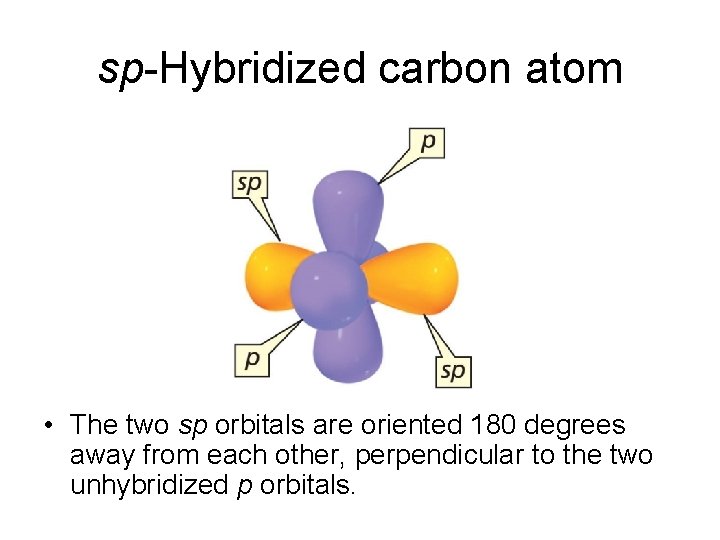
sp-Hybridized carbon atom • The two sp orbitals are oriented 180 degrees away from each other, perpendicular to the two unhybridized p orbitals.
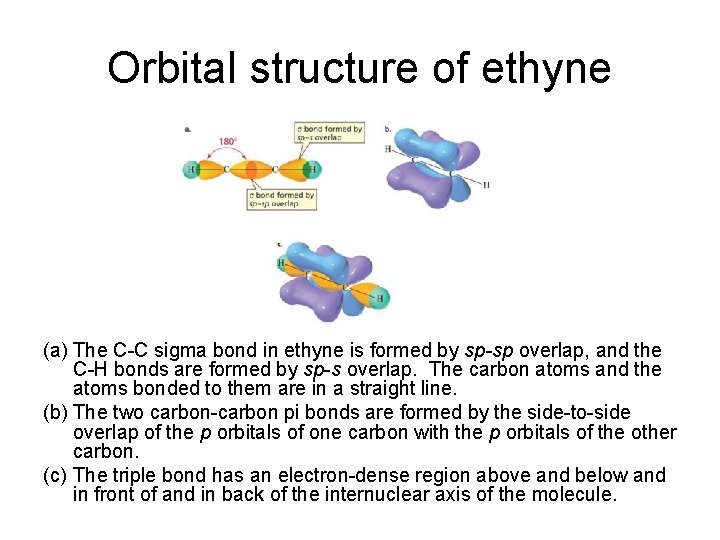
Orbital structure of ethyne (a) The C-C sigma bond in ethyne is formed by sp-sp overlap, and the C-H bonds are formed by sp-s overlap. The carbon atoms and the atoms bonded to them are in a straight line. (b) The two carbon-carbon pi bonds are formed by the side-to-side overlap of the p orbitals of one carbon with the p orbitals of the other carbon. (c) The triple bond has an electron-dense region above and below and in front of and in back of the internuclear axis of the molecule.
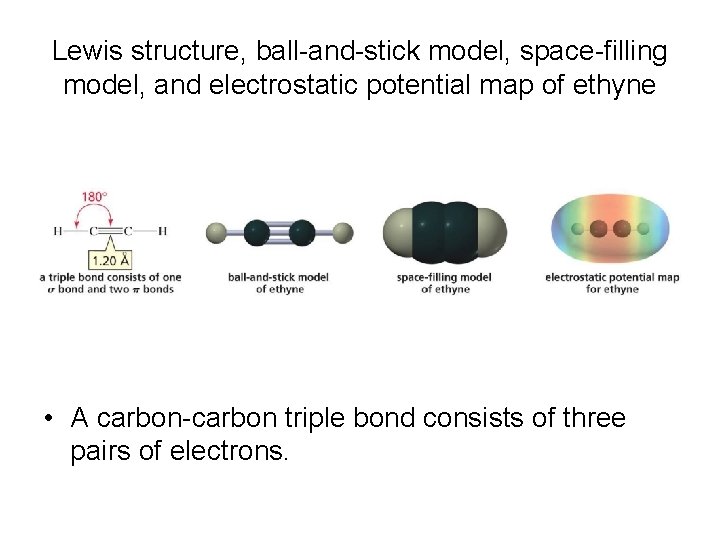
Lewis structure, ball-and-stick model, space-filling model, and electrostatic potential map of ethyne • A carbon-carbon triple bond consists of three pairs of electrons.
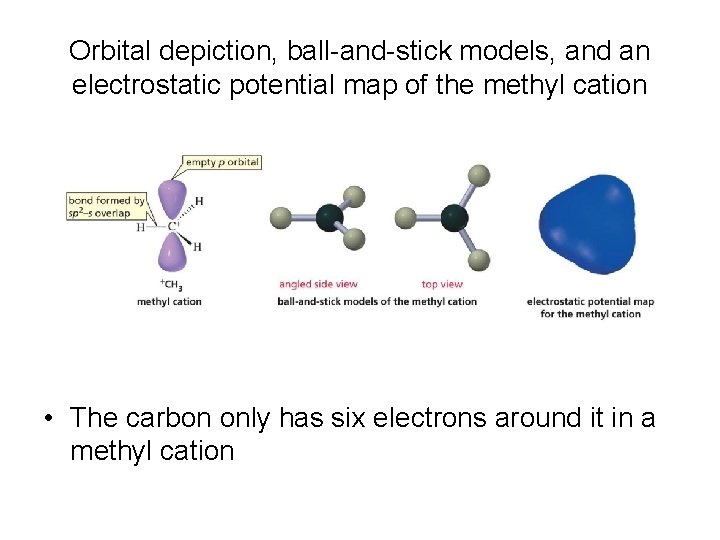
Orbital depiction, ball-and-stick models, and an electrostatic potential map of the methyl cation • The carbon only has six electrons around it in a methyl cation
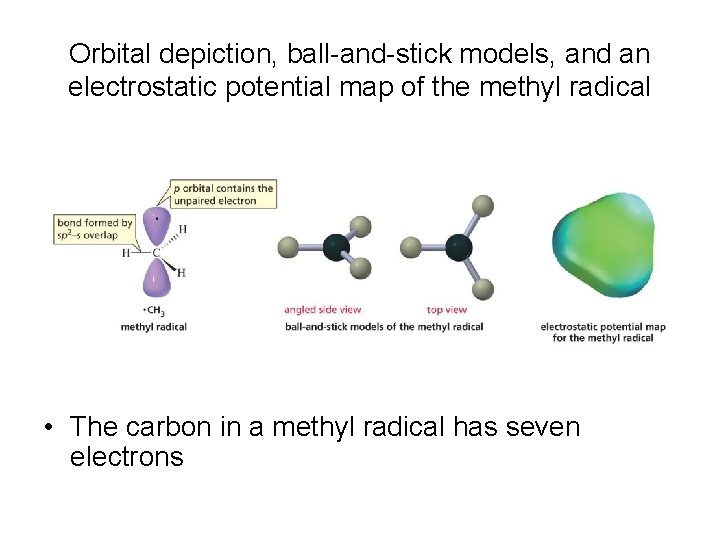
Orbital depiction, ball-and-stick models, and an electrostatic potential map of the methyl radical • The carbon in a methyl radical has seven electrons
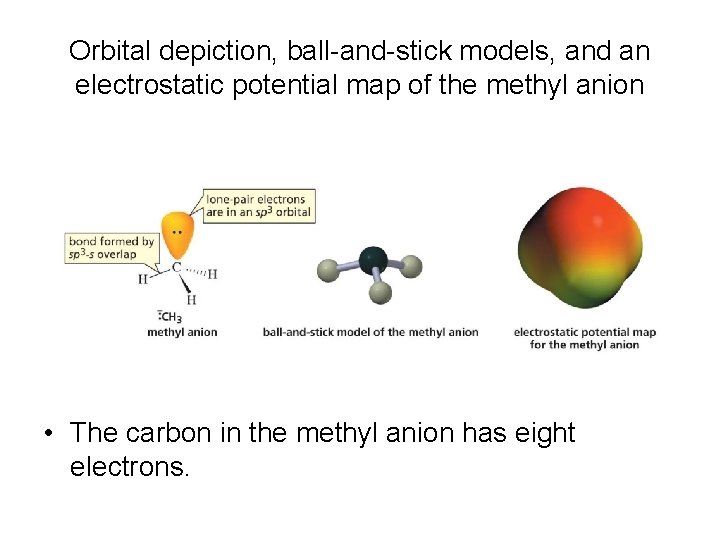
Orbital depiction, ball-and-stick models, and an electrostatic potential map of the methyl anion • The carbon in the methyl anion has eight electrons.

sp 3 Hybridization in water • One s and three p orbitals are hybridized to form an sp 3 -hybridized orbital.
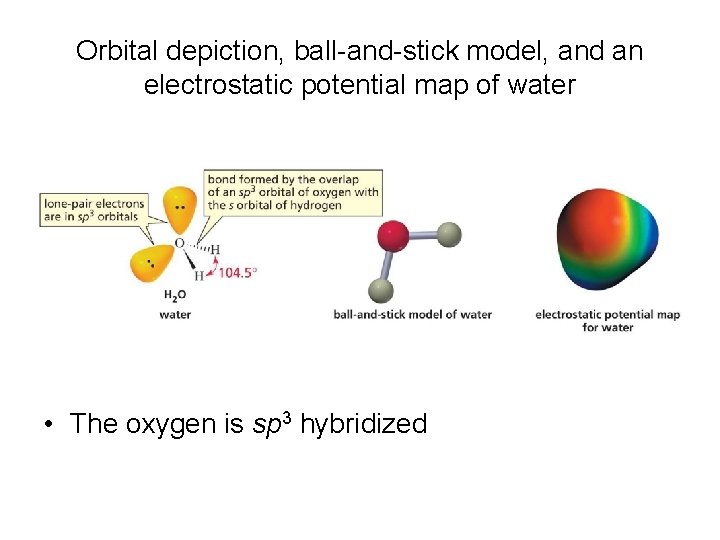
Orbital depiction, ball-and-stick model, and an electrostatic potential map of water • The oxygen is sp 3 hybridized

sp 3 Hybridization in ammonia • One s and three p orbitals are hybridized to form an sp 3 -hybridized orbital.
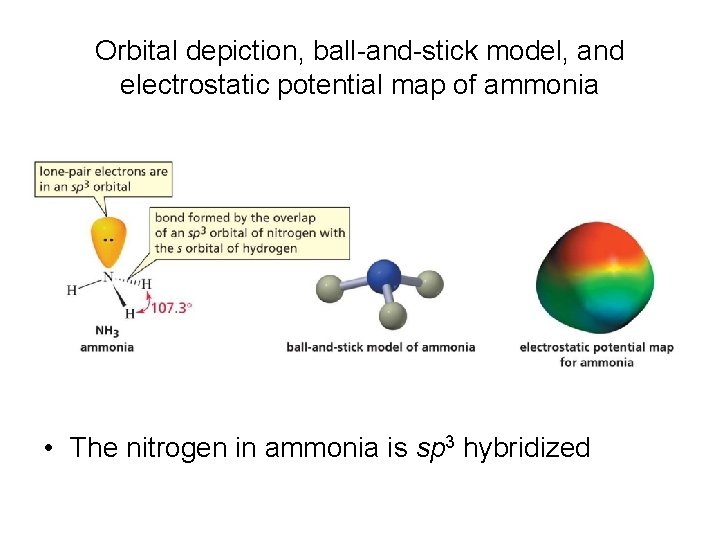
Orbital depiction, ball-and-stick model, and electrostatic potential map of ammonia • The nitrogen in ammonia is sp 3 hybridized
- Slides: 40