Lecture 19 Study Design and Sample Size Estimation



























































- Slides: 61

Lecture 19: Study Design and Sample Size Estimation in TTE Studies

Study Design and Sample Size • Ideally we are involved in a study from the beginning • As statisticians (an epidemiologist) part of our role is to ensure the study is designed to address the primary hypothesis under consideration – Ensure proper study design – Ensure appropriate sample size • Power and significance level

Study Design • We’ve already talked (informally) about study design… • Our job is to ask appropriate questions – What is the study population? – Primary hypothesis? – Is the event recurrent? – Competing risks? –…

Components of Sample Size General Considerations in any hypothesis test 1. Hypothesis to be tested 2. Test statistic 3. Size of the test (i. e. a) 4. Desired power 5. Assumed effect size

Specific for Time To Event Data Additional considerations 1. Probability of an event during the study 2. Expected rate of loss (i. e. censoring) 3. Enrollment rate 4. Competing risks

Basic Considerations • Ensure precise specification of the hypothesis • Select a significance level and power appropriate for the study • What is the test statistic that will be used to test the hypothesis? – Many statistics have well known properties upon which we base our calculations – Deviations from assumptions complicate these calculations…

Null Hypothesis to be Tested • Recall for log-rank test • Where HR is assumed to be proportional for all t

Test-Statistic • Can use either – Log-rank test (or some variation there of) – HR estimated from Cox PHM • To test the null hypothesis of no difference consider the log of the hazard ratio – log(HR) ~ N when comparing two groups

Significance Level • The probability that a statistical test will reject H 0 when H 0 is actually true – Significance = a • Interpretation: For a given value under the null hypothesis, we’re going to reject the null in favor of the alternative in error (a)100% of the time.

Power • The probability that a statistical test will reject H 0 when H 0 is false – Power=1 -b • Interpretation: For a given value under the alternative hypothesis, we’re going to correctly reject the null in favor of the alternative (1 -b)100% of the time.

Choice of a and 1 -b • Generally choose a = 0. 05 • Other values can be used but should be justified – Say choose a = 0. 01, we want very string evidence of a treatment effect – Alternatively a = 0. 10 or 0. 20 might be chosen for something like a pilot study • Power generally set to 80 -99%

Effect Size • Generally assume proportional hazards • Hazard ratio: – Null: HR = 1 – HR < 1 implies longer survival in treatment B – HR > 1 implies longer survival in treatment A • Base sample size calculation on having sufficient power to detect minimum clinically important effect – For example, maybe a 30% reduction in incidence for Trt A vs. Trt B (i. e. HR = 0. 7) is clinically meaningful

Minimum Scientifically Important Difference • Definition: the smallest difference which would mandate, in the absence of serious side effects and/or excessive cost, a change in scientific practice/ understanding. • This is a scientific question, not a statistical question.

Significance, Power, and Sample Size • Sample size impacted not only by significance level, power, and research question but also practical considerations – Number of available patients – Study duration – Cost

Basic Sample Size Formula • Generic formula for total # subjects/group • Under restrictions

Design Consideration in TTE Studies • How will sample size be collected? – Enroll fixed number of patients and follow for some specified period of time – Continue study until a sufficient number of events have been observed • Other considerations – Expected event rate in each group – How much loss to follow-up expected

Study Type • Type I study – All subjects experience an event by the end of the study • Type II study – A study terminates at fixed time T resulting in administratively censored subjects. • Administrative censoring – Right censoring that occurs of subject fails to experience the event prior to the end of the study • Loss to follow-up – Occurs when a subject fails to complete the study for reasons unrelated to the event of interest

Sample Size in Time to Event Data • For many power calculations we specify significance level, power, variability, and our minimum clinically relevant difference to get our total N • In TTE studies, it is easier to specify the number of events we need to observe rather than the total number of people

Simplest Case for TTE • Trial comparing Treatment A to Treatment B • Simplest case assumes all subjects are followed to the end of the study • Also assumes all subjects in within treatment group have the same probability of experiencing the event • Finally assumes hazard rates are proportional

Required Number of Events • Za/2 and Zb are standard normal percentiles • p. A and p. B are the proportion of subjects allocated to each treatment group – i. e. equal allocation both are ½ • D is the minimum clinically relevant difference we want to detect – In this case it is the log of the hazard ratio under the alternative hypothesis

Example: Required # Events • Suppose a = 0. 05 and b = 0. 10 (90% power) – za/2 = 1. 96 and zb = 1. 282 • Equal allocation – p. Ap. B = ¼ Hazard Ratio # Events Required 0. 8 845 0. 7 331 0. 5 88 • Always round up.

How Many People? • We’ve not yet discussed event rate(s) • We’ve determined for 90% power at a significance level a = 0. 05 – To detect a 50% reduction in hazard, we need to observe 88 events • But… How many people do we actually need to enroll to get that many events?

A Simplification • Consider only the case in which each patient is followed for some specified time period, T • More general case – Patients recruited during accrual period, a – After recruitment, there is an additional follow-up period, f – First patient followed a + f – Last patient followed for f – Requires slightly more complex power calculations

Calculating Number of People • Need to consider probability of an event during the study period • Once we have this, estimating the total number of people needed is easy • NOTE: This still ignores loss to follow-up

Overall Probability of Event • Recall p. A and p. B are the proportion of subjects allocated to each treatment group • SA(T) and SB(T) are the survival distributions for the two treatment groups • How do we get values for SA(T) and SB(T)?

Estimating Survival Distributions • Crude rate – How many events are expected in each group over the course of the study? • Alternatively, we could assume S(T) ~Exp(l) • Recall S(t) = exp{-lt} • Use the assumed distribution to calculate failure probability – e. g. if l = 0. 1/unit time, then S(1) = 0. 905 – Thus, assume by t = 1, 9. 5% of subjects will have experienced the event

Example 1 • One-year study (T = 1) with p. A = p. B, l = 0. 1/yr for control group, and q. A = 0. 5

Example 1 • One-year study (T = 1) with p. A = p. B, l = 0. 1/yr for control group, and q. A = 0. 5

Example 1 • One-year study (T = 1) with p. A = p. B, l = 0. 1/yr for control group, and q. A = 0. 5

Example 1 • One-year study (T = 1) with p. A = p. B, l = 0. 1/yr for control group, and q. A = 0. 5

Example 1 • One-year study (T = 1) with p. A = p. B, l = 0. 1/yr for control group, and q. A = 0. 5

Example 2 • 5 year study (T = 5) with p. A = p. B, l = 0. 1/yr for control group, and DA = 0. 5

Example 2 • 5 year study (T = 5) with p. A = p. B, l = 0. 1/yr for control group, and DA = 0. 5

Example 2 • 5 year study (T = 5) with p. A = p. B, l = 0. 1/yr for control group, and DA = 0. 5

Example 2 • 5 year study (T = 5) with p. A = p. B, l = 0. 1/yr for control group, and DA = 0. 5

Example 2 • 5 year study (T = 5) with p. A = p. B, l = 0. 1/yr for control group, and DA = 0. 5

More General Case • Consider if subjects enroll over time – First patient followed a + f – Last patient followed for f • Proportion of patients that will survive is the average survival curve from time f to a + f • P(event) can be estimated by

Example 2 Revisited • 5 year study with p. A = p. B, l = 0. 1/yr for control group, and D = 0. 5 • But accrue patients for 2 years and follow for the remaining 3 years

Example 2 Revisited • 5 year study with p. A = p. B, l = 0. 1/yr for control group, and D = 0. 5 • But accrue patients for 2 years and follow for the remaining 3 years

Example 2 Revisited • 5 year study with p. A = p. B, l = 0. 1/yr for control group, and D = 0. 5 • But accrue patients for 2 years and follow for the remaining 3 years

Example 2 Revisited • 5 year study with p. A = p. B, l = 0. 1/yr for control group, and D = 0. 5 • But accrue patients for 2 years and follow for the remaining 3 years

Example 2 Revisited • 5 year study with p. A = p. B, l = 0. 1/yr for control group, and D = 0. 5 • But accrue patients for 2 years and follow for the remaining 3 years

Beyond Basic Considerations • Many factors may cause statistic to deviate from expected behavior – Loss to follow-up (non-administrative censoring) – Failure to comply with treatment – Non-Uniform patient entry – Non-constant hazard ratio • Failure time differ greatly from exponential – Competing risks… • Violations generally require an increase in sample size to achieve desired power

Non-Uniform Entry • Previously assumed uniform entry of subjects into the trial, which could be an error – For example patients enter trial in staggered fashion – Sample size related to total number of person years observed • Alternative “general” sample size equation proposed by Lachin and Foulkes

Lachin and Foulkes Formula • lcont and ltrt are hazard rates for each group • is overall hazard rate • f(l) is a component of the variance of s 2(l) where

Non-Uniform Entry using L-F Formulation • Assume patient entry times follow g(t) – If it is Uniform (for recruitment period a) – If it is not Uniform • If recruitment is faster than expected, power will be greater • If recruitment is slower than expected, power will be reduced

Example Alternate Distribution • Truncated exponential entry distribution

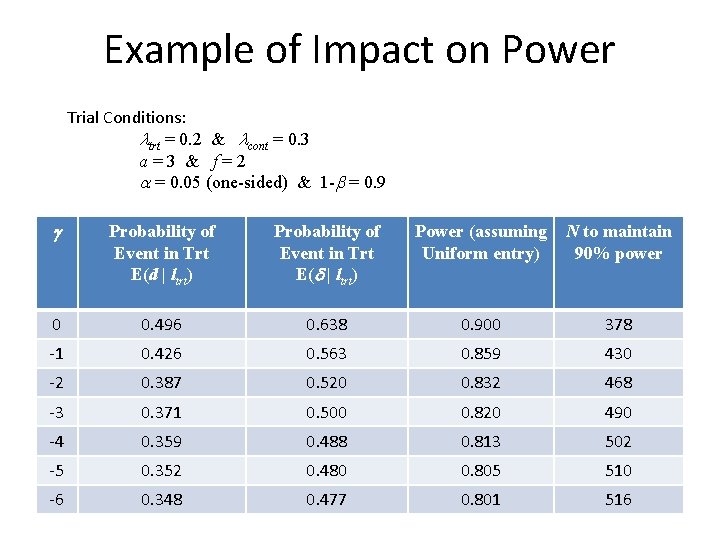
Example of Impact on Power Trial Conditions: ltrt = 0. 2 & lcont = 0. 3 a=3 & f=2 a = 0. 05 (one-sided) & 1 -b = 0. 9 g Probability of Event in Trt E(d | ltrt) Power (assuming Uniform entry) N to maintain 90% power 0 0. 496 0. 638 0. 900 378 -1 0. 426 0. 563 0. 859 430 -2 0. 387 0. 520 0. 832 468 -3 0. 371 0. 500 0. 820 490 -4 0. 359 0. 488 0. 813 502 -5 0. 352 0. 480 0. 805 510 -6 0. 348 0. 477 0. 801 516

Loss To Follow-Up • Again, our early expressions assumed only administrative censoring BUT clearly this is not always the case. • The Lakin-Foulkes expression can also be adapted to address random right censoring

Right Censoring Formulation • Easiest “case” – censoring times ~Exp(h) – Uniform entry into trial

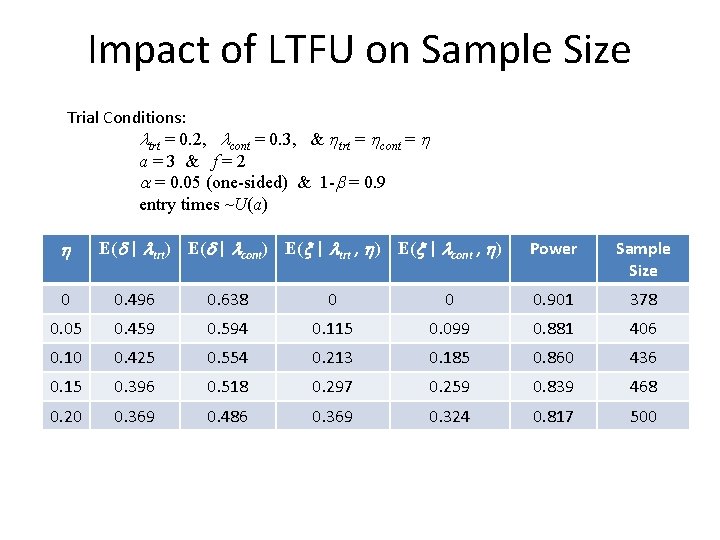
Impact of LTFU on Sample Size Trial Conditions: ltrt = 0. 2, lcont = 0. 3, & htrt = hcont = h a=3 & f=2 a = 0. 05 (one-sided) & 1 -b = 0. 9 entry times ~U(a) h E(d | ltrt) E(d | lcont) E(x | ltrt , h) E(x | lcont , h) Power Sample Size 0 0. 496 0. 638 0 0 0. 901 378 0. 05 0. 459 0. 594 0. 115 0. 099 0. 881 406 0. 10 0. 425 0. 554 0. 213 0. 185 0. 860 436 0. 15 0. 396 0. 518 0. 297 0. 259 0. 839 468 0. 20 0. 369 0. 486 0. 369 0. 324 0. 817 500

Competing Risk Setting • Same idea- we want to compare 2 treatments but now we have some competing risk(s) • Latouche et. al (2004) developed an approach for estimating sample size in data with competing risks – Extension of Schoenfeld formula – Also based on Fine and Gray model for competing risks

Recall the Fine and Gray Model • Model and Partial likelihood

Competing Risk Sample Size • Number of events • Number of subjects

Additional Considerations • There are extensions for >2 groups • If interim analyses are planned, these also need to be accounted for in the sample size analysis • Multi-center trial? • Non-proportional hazards – Methods by Halpern and Lakatos allows for nonproportional hazards – Specify event rates for groups within specific time intervals

Additional Considerations • Non-compliance – Differs from LTFU in that these people don’t comply to treatment but are still followed – There adjustments that can be made to the Lakin. Foulkes formula if non-compliance expected • Covariate adjustments? – If balanced, adjustment for covariate shouldn’t impact power – However, in cases of extreme in-balance, formulas are not valid

If Complications Exist • Consider a 3 -stage approach – Use basic formula as first estimate – Refine sample size calculation based on likely deviations from assumptions – If necessary, develop simulation to address more complicated deviations

Implementation • The Lakin and Foulkes implemented in the gs. Design package in R – Includes ability to power for stratified analysis – Can power for interim analyses • Can use proc power in SAS – Two. Sample. Survival statement • Based on log-rank but allows for different weights (Gehan, log-rank, or Tarone-Ware)

Implementation • Alternative software? – PASS • Can power for binary or non-binary covariate • Can power for scenario where an additional correlated covariate is considered – Nquery • Basic calculation based on Schoenfeld equation

All R Packages Related to Survival Analysis https: //cran. r-project. org/web/views/Survival. html

Next Time 1. Chen-Chu Wen 2. Corrine Mc. Gill