Lecture 19 A Properties of Acids Bases Properties

Lecture 19 A Properties of Acids & Bases

Properties of Acids & Bases Acids and bases dissolve in water to form solutions that: Acids Bases • Taste sour or tart • When strong - good electrolytes (conduct electricity) • Causes indicators to change color, example: phenolphthalein (turns clear), bromothymol blue (turns yellow). • Turns litmus paper red • Release H 2 gas when they react with metals • Taste bitter and feel slippery • Electrolytes • Cause indicators to change color, ex: phenolphthalein, (turns pink) and bromothymol blue (turns gray-blue). • Turns litmus paper blue
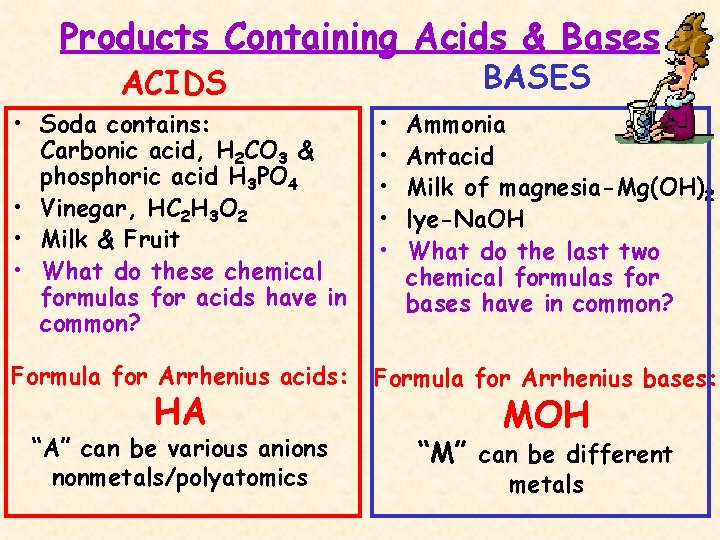
Products Containing Acids & Bases BASES ACIDS • Soda contains: Carbonic acid, H 2 CO 3 & phosphoric acid H 3 PO 4 • Vinegar, HC 2 H 3 O 2 • Milk & Fruit • What do these chemical formulas for acids have in common? • • • Formula for Arrhenius acids: Formula for Arrhenius bases: “A” can be various anions nonmetals/polyatomics “M” can be different HA Ammonia Antacid Milk of magnesia-Mg(OH)2 lye-Na. OH What do the last two chemical formulas for bases have in common? MOH metals

Name & Formulas for Acids & Bases Write the formula for the following acid and bases: Hydrobromic acid - HBr Strontium hydroxide - Sr(OH)2 Sulfurous acid - H 2 SO 3 Name the following acid and bases: HNO 3 - Nitric acid Rememorize the rules for naming acids & bases!

Lecture 19 A Arrhenius Acid -Base Theory
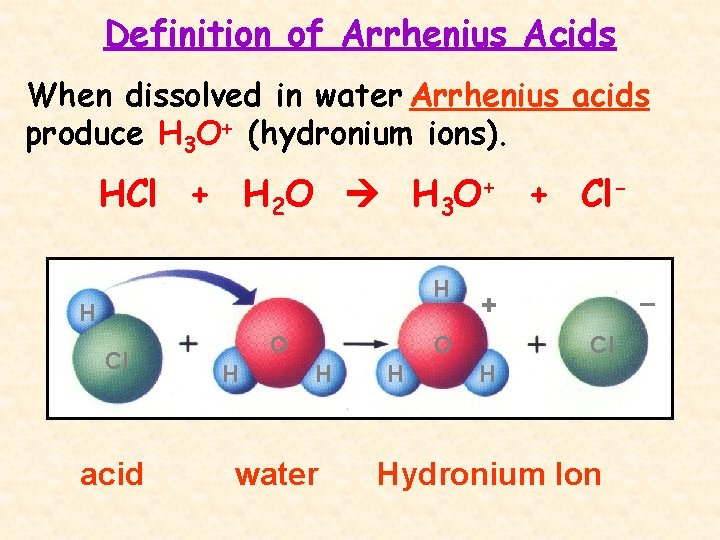
Definition of Arrhenius Acids When dissolved in water Arrhenius acids produce H 3 O+ (hydronium ions). HCl + H 2 O H 3 O+ + Cl. H H Cl acid O H water H – + Cl H Hydronium Ion

Types of Acids Monoprotic Diprotic Triprotic Acids that contain one ionizable hydrogen. Acids that contain two ionizable hydrogens. Acids that contain three ionizable hydrogens. HNO 3 H 2 SO 4 H 3 PO 4
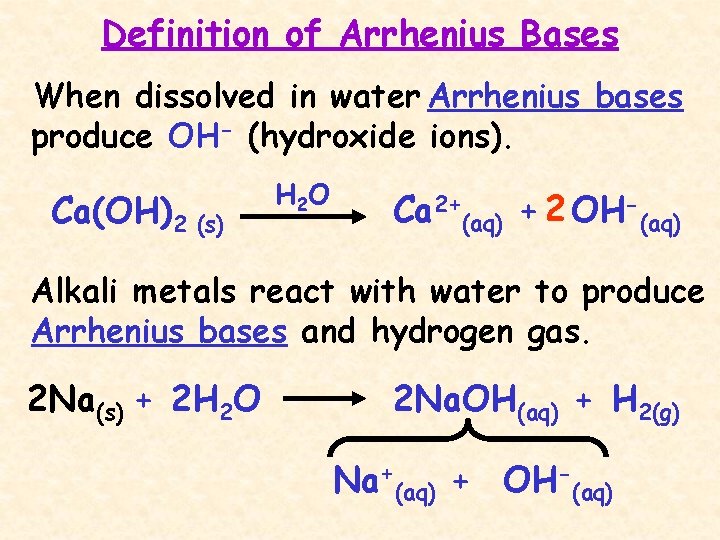
Definition of Arrhenius Bases When dissolved in water Arrhenius bases produce OH- (hydroxide ions). Ca(OH)2 (s) H 2 O Ca 2+(aq) + 2 OH-(aq) Alkali metals react with water to produce Arrhenius bases and hydrogen gas. 2 Na(s) + 2 H 2 O 2 Na. OH(aq) + H 2(g) Na+(aq) + OH-(aq)
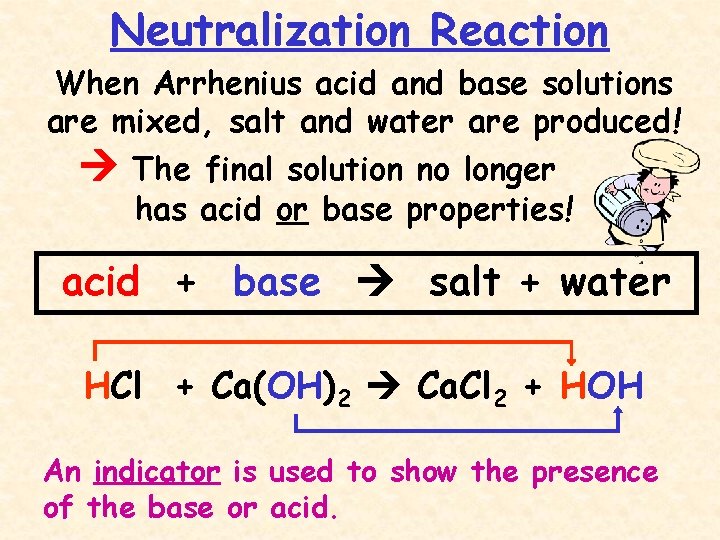
Neutralization Reaction When Arrhenius acid and base solutions are mixed, salt and water are produced! The final solution no longer has acid or base properties! acid + base salt + water HCl + Ca(OH)2 Ca. Cl 2 + HOH An indicator is used to show the presence of the base or acid.

WS 19 A Now for a little homework…
- Slides: 10