Lecture 17 Spectrophotometry Emission Absorption Fluorescence sample source

Lecture 17 Spectrophotometry
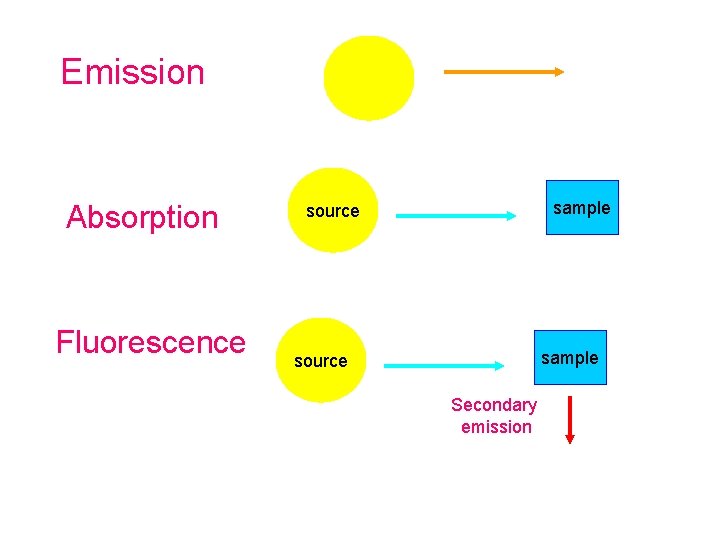
Emission Absorption Fluorescence sample source Secondary emission
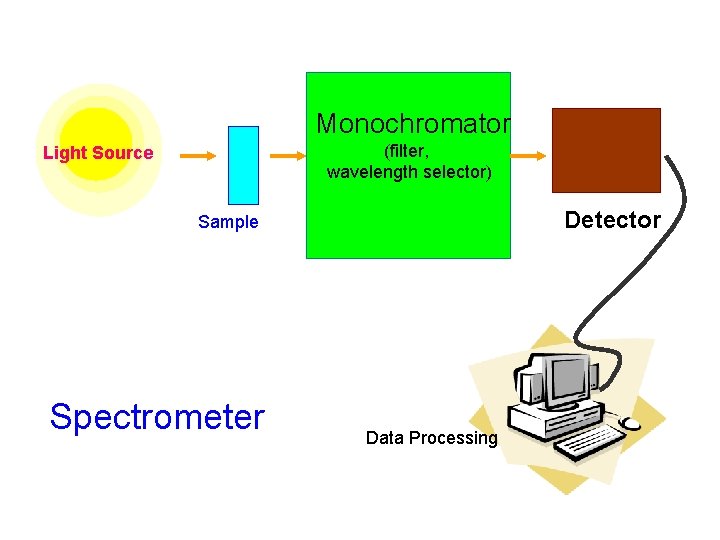
Monochromator (filter, wavelength selector) Light Source Detector Sample Spectrometer Data Processing
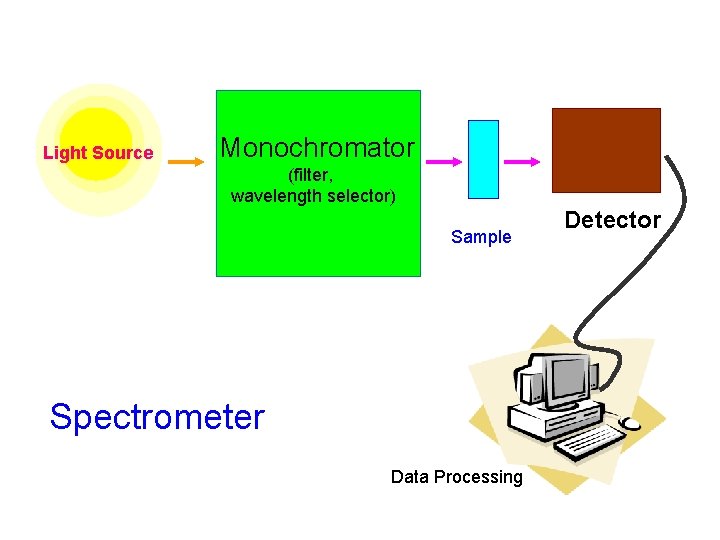
Light Source Monochromator (filter, wavelength selector) Sample Spectrometer Data Processing Detector
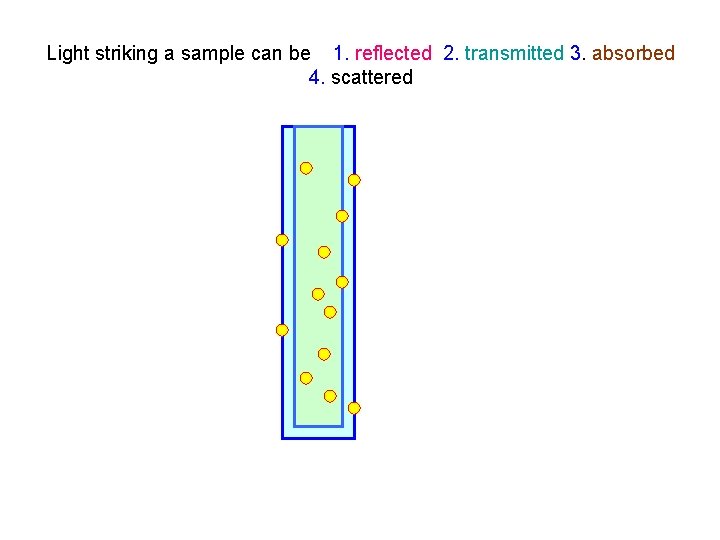
Light striking a sample can be 1. reflected 2. transmitted 3. absorbed 4. scattered
Absorbing plate
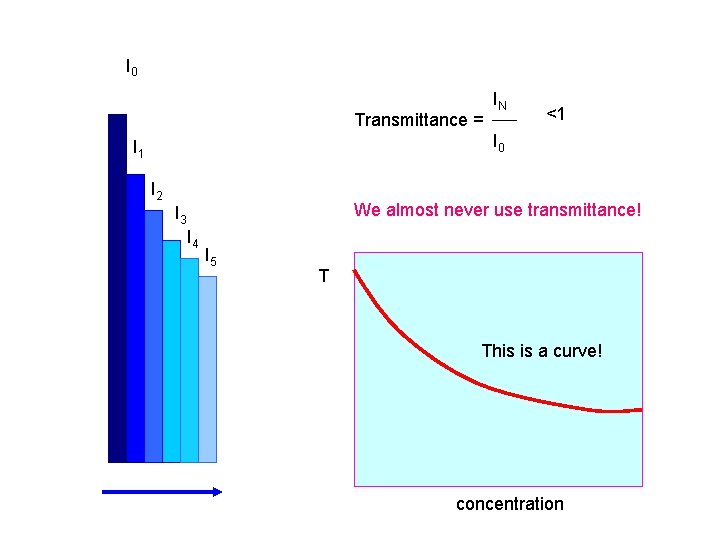
I 0 Transmittance = IN <1 I 0 I 1 I 2 We almost never use transmittance! I 3 I 4 I 5 T This is a curve! concentration
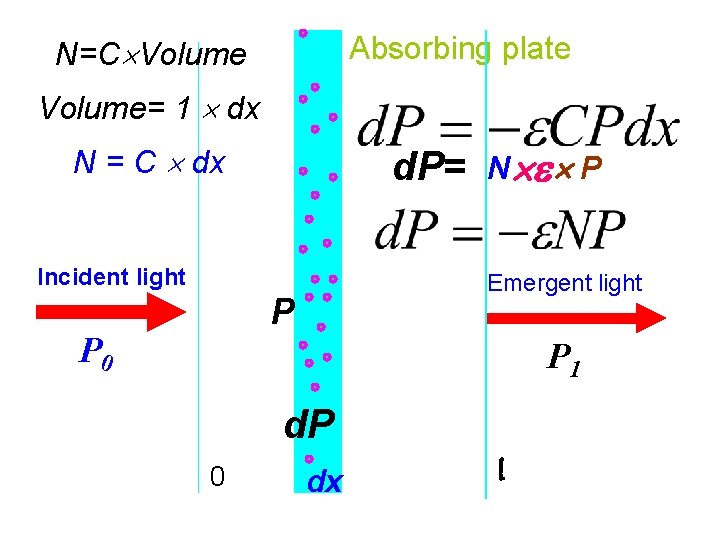
Absorbing plate N=C Volume= 1 dx N = C dx d. P= Incident light Emergent light P P 0 N P P 1 d. P 0 dx l
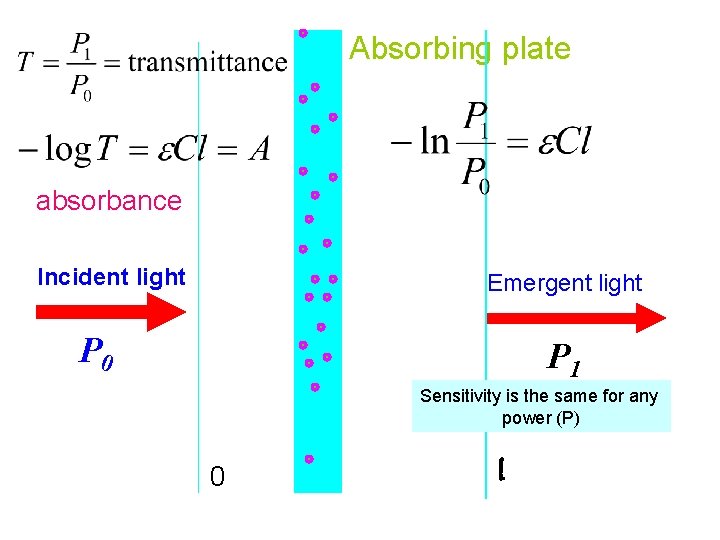
Absorbing plate absorbance Incident light Emergent light P 0 P 1 Sensitivity is the same for any power (P) 0 l
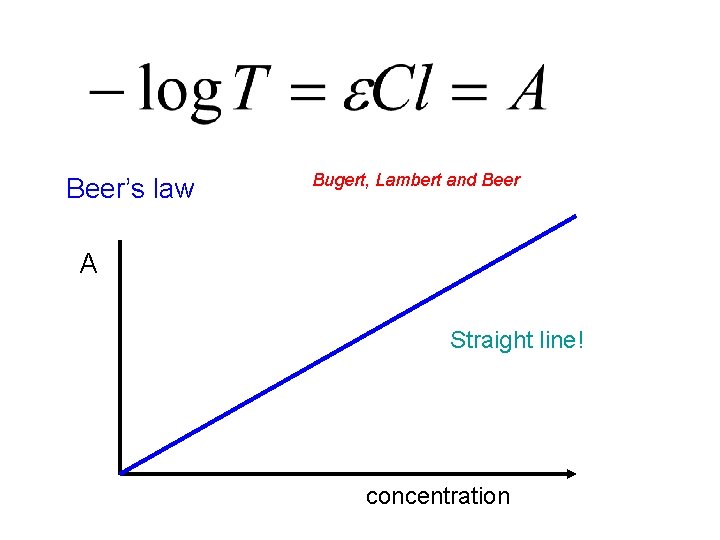
Beer’s law Bugert, Lambert and Beer A Straight line! concentration
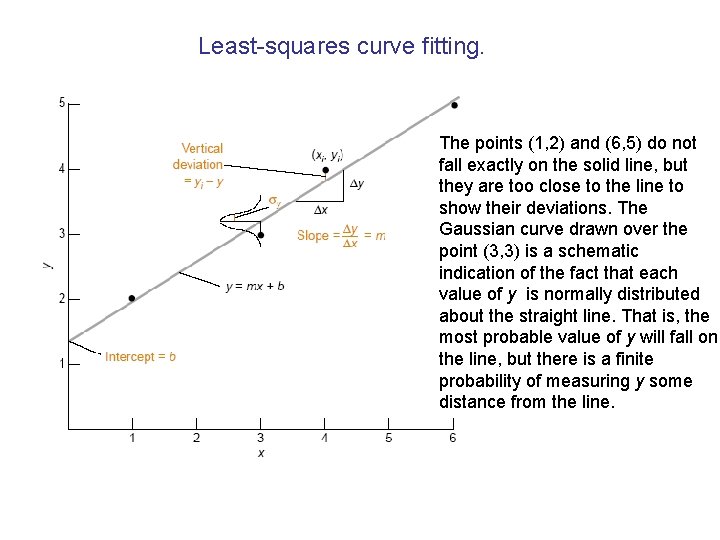
Least-squares curve fitting. The points (1, 2) and (6, 5) do not fall exactly on the solid line, but they are too close to the line to show their deviations. The Gaussian curve drawn over the point (3, 3) is a schematic indication of the fact that each value of y is normally distributed about the straight line. That is, the most probable value of y will fall on the line, but there is a finite probability of measuring y some distance from the line.
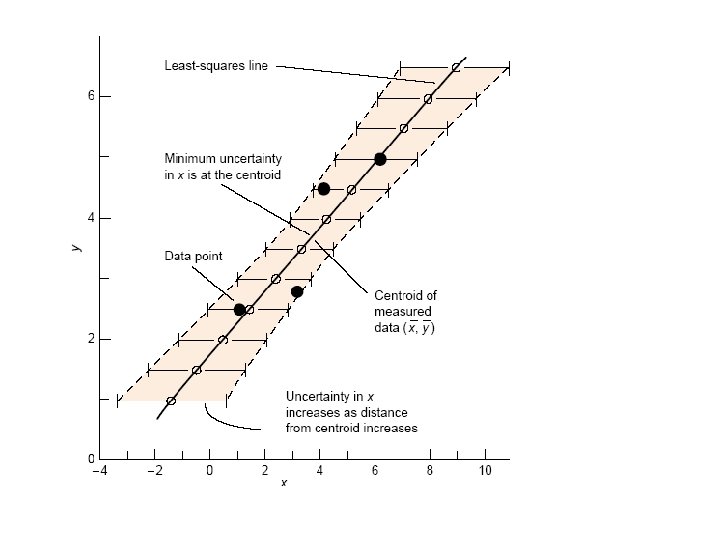

y=kx+b straight line equation k = Slope = y / x b Let us subtract blank: Y 1=kx 1 y-b = Y = kx Y 2=kx 2 - blank! One standard

Procedure: 1. Measure blank. 2. Measure standard. 3. Measure unknown. 4. Subtract blank from standard and from unknown. 5. Calculate concentration of unknown If you have several (N) standards, do it several (N) times
- Slides: 14