Lecture 13 Carboxylic Acids Esters Amines and Amides

Lecture 13: Carboxylic Acids, Esters, Amines and Amides

�Book chapter: 14 �Pages: 472 -511

�Carbon atom of a carbonyl group is attached to a hydroxyl group forming a carboxyl group �As functional group – attached to an alkyl group or an aromatic group

IUPAC names �Replace e of the alkene with oic acid �If there are substituents – the carbon chain is numbered beginning with the carboxyl carbon

Common names of carboxylic acids �Use prefixes: form, acet, propion, butyr �Relate to the natural sources of the simple carboxylic acids

Properties of Carboxylic acids �Most polar organic compounds – functional group consists of two polar groups (-OH) and (C=O) Solubility in water: �Acids with 1 -5 C atoms are soluble in water �Length increases – the nonpolar portion reduces the solubility of acid in water �More than 5 C atoms – not very soluble in water

Acidity of Carboxylic Acids: �Importnat property: ionization in water �Hydrogen ion is transferred to a water molecule – forming of negatively charged cyrboxylate ion and hydronium ion �Generally those acids are more acidic than most other organic compounds �Weak acids; 1% of the acid molecules ionize in water
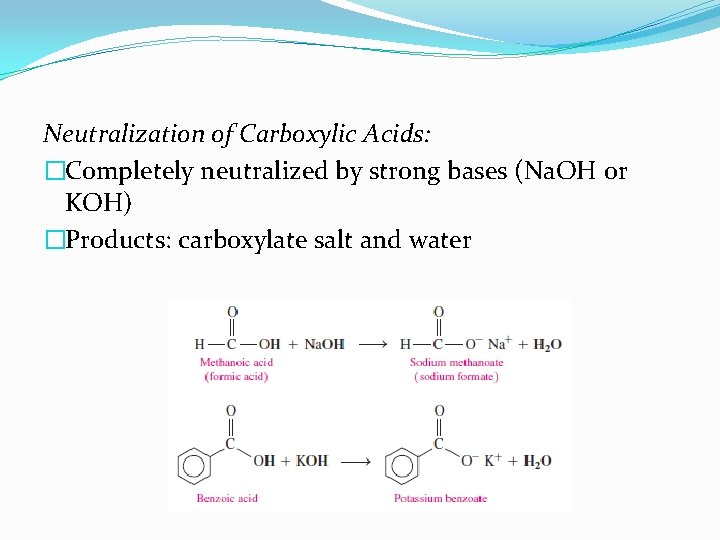
Neutralization of Carboxylic Acids: �Completely neutralized by strong bases (Na. OH or KOH) �Products: carboxylate salt and water

Chemistry Link to Health: Carboxylic Acids in Metabolism �Several are part of metabolic processes q. Example: glucose is broken down into two molecules of pyruvic acid or actually its carboxylate ion pyruvate (glycolysis) �Pyruvic acid is reduced to give lactic acid (lactate ion)

Esters �Carboxylic acid + alcohol -> ester + water �-H of cyrboxylic acid is replaced by an alkyl group �Esters contained in fats and oils (esters of glycerol and fatty acids)

Esterification �Production of esters �In the rpesence of an acid catalyst (H 2 SO 4) and heat �-OH group (carboxylic acid) and –H (alcohol) -> combined to form water

Naming esters �Two words – derived from the names of the alcohol and the acid �First word – indicates alkyl part from the alcohol �Second word – name of the carboxylate from the carboxylic acid
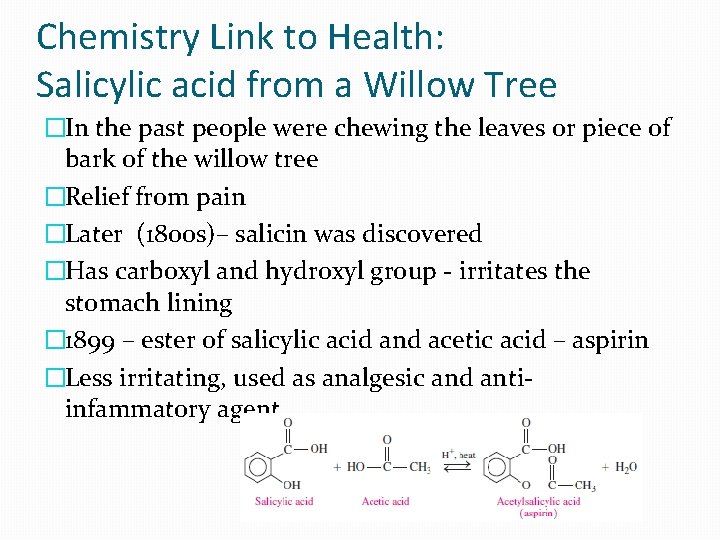
Chemistry Link to Health: Salicylic acid from a Willow Tree �In the past people were chewing the leaves or piece of bark of the willow tree �Relief from pain �Later (1800 s)– salicin was discovered �Has carboxyl and hydroxyl group - irritates the stomach lining � 1899 – ester of salicylic acid and acetic acid – aspirin �Less irritating, used as analgesic and antiinfammatory agent

Esters in plants �Fragnances of perfumes and flowers or fruits – esters �Small esters are volatile – smell and soluble in water; taste them

Hydrolysis of Esters Acid Hydrolysis �Heating of esters with water in presence of a strong acid �Water reacts with the ester to form a cyrboxylic acid an alcohol �Reverse of esterification reaction

Base Hydrolysis of Esters �Esters undergo hydrolysis with a strong base �Products: Carboxylate salt and corresponding alcohol �Saponification

Amines �Derivatives of ammonia (NH 3) �One or more hydrogen atoms are replaced with alkyl or aromatic groups

Naming and Classifying Amines �Common names are often used when alkyl groups bonded to the nitrogen atom are not branched �Groups are listed in alphabetical order �Prefixes di and tri indicate substituents �Classified – by counting the number of C atoms directly bonded to the nitrogen atom
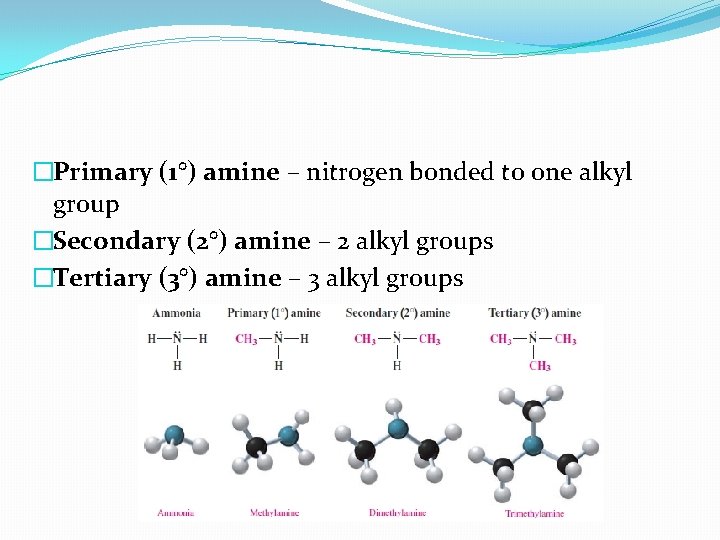
�Primary (1°) amine – nitrogen bonded to one alkyl group �Secondary (2°) amine – 2 alkyl groups �Tertiary (3°) amine – 3 alkyl groups

Aromatic amines �Use the name aniline (approved by IUPAC) �Alkyl groups attached to the nitrogen are named with the prefix N- followed by the alkyl name �Aniline – used to make dyes (indigo) �Give color to wool, cotton, silk fibers and blue jeans

Skeletal formula �Similar like for other organic compounds �We show the hydrogen atoms bonded to the N atom

Chemistry Link to Health: amines in Health and Medicine �Histamine – in response to injury or allergy �Causes blood vessels to dilate and increases the permeability of the cells �Redness and swelling in the area �Antihistamines – block the effect of histamine
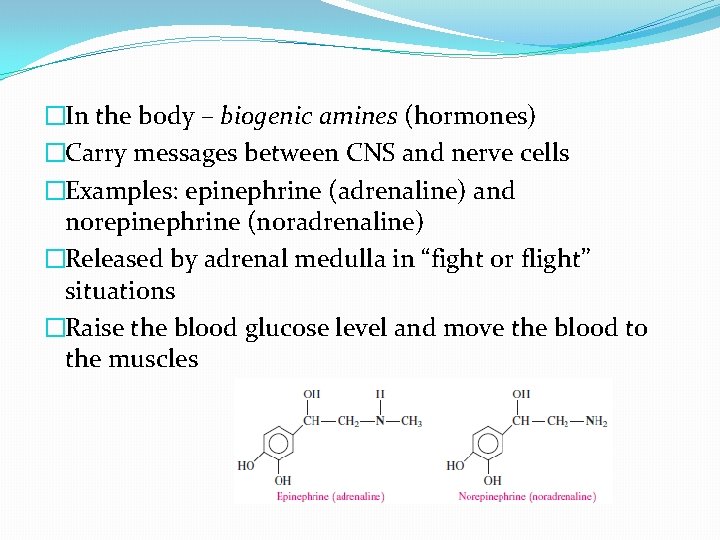
�In the body – biogenic amines (hormones) �Carry messages between CNS and nerve cells �Examples: epinephrine (adrenaline) and norepinephrine (noradrenaline) �Released by adrenal medulla in “fight or flight” situations �Raise the blood glucose level and move the blood to the muscles

�Amphetamines – produced synthetically �Stimulants of the CNS �Also increase cardiovascular activity and depress the appetite �Sometimes used to bring about weight loss; can cause chemical dependency

Solubility of amines in water �Contain polar N-H bond – form hydrogen bonds with water �Primary form more bonds with water than secondary �Tertiary only with N atom in the amine

Amines react as bases in water: �Ammonia acts as base because it accepts H+ from water �Produces ammonium ion (NH 4+) and hydroxide ion (OH-)

Ammonium salts: �Example: lemon jiuce on the fish – revomes “fishy” odor by converting amines to their ammonium salts �Neutralization reaction – amines (base) react with an acid to form ammonium salt �Naming: using alkylammonium ion name followed by the name of the negative ion

�Properties of Ammonium Salts: �Ionic compounds �Solids at room temperature, odorless and soluble in water and body fluids �Amines used as drugs are usually in form of these salts �If it reacts with a strong base – converted back to the amine

Heterocyclic amines �Cyclic compound �Contains one or more nitrogen atoms in the ring �The heterocyclic amine ring consists of 5 or 6 stoms and one or more nitrogen atoms

Chemistry Link to Health: Alkaloids �Active compounds produced by plants �Contain heterocyclic amines �Alkaloid - “alkali-like” or basic characteristics �Some of them used in anesthetics, antidepressants and stimulants q. Examples: nicotine and caffeine

�Several alkaloids – used in medicine �Atropine – from nightshade; used to accelerate slow heart rates �Codeine – poppy plant; painkiller �Heroin – chemical modification of morphine; not used medically (strongly addictive)

Amides �Derivatives of cyrboxylic acids �Nitrogen replaces hydroxyl grouop
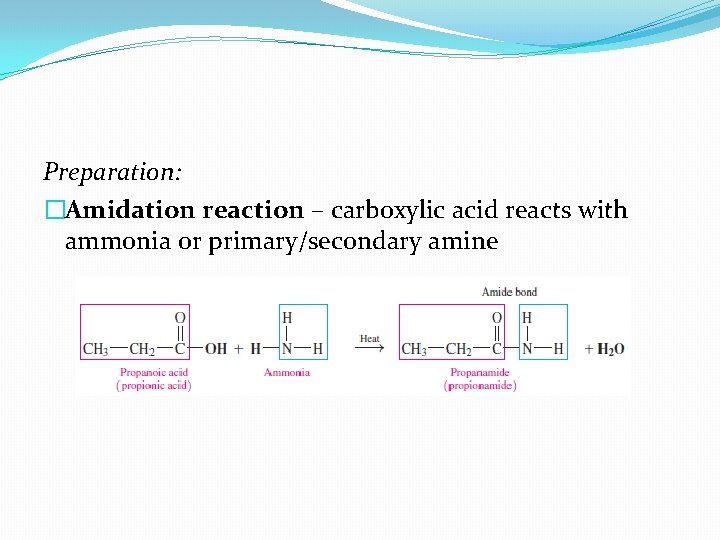
Preparation: �Amidation reaction – carboxylic acid reacts with ammonia or primary/secondary amine

Naming: �Dropping the oic acid or ic acid and adding the suffix amide �If alkyl group is attached to the nitrogen atom – prefix N- or N, N- precedes the name of the amide

�Solubility in water: �Do not have properties of bases �Amides with one to five C atoms are soluble in water (they can hydrogen bond with water molecules) �More than five C atoms – effect of hydrogen bonding is diminished; longer carbon chain decreases the solubility

Chemistry Link to Health: Amides in Health and Medicine �Simplest amide – urea (end product of protein metabolism) �Removed by kidenys �Malfunction of kidneys – urea is not removed; uremia �Used also as fertilizer to increase nitrogen in the soil

�Barbiturates – act as sedatives in small dosages or sleep inducers in larger dosages �Aspirin substituetes contain phenacetin – used in Tylenol �Reduces fever and pain, has little anti-inflammatory effect


Summary �Carboxylic acids �Properties of carboxylic acids �Esters �Acid/Base Hydrolysis of esters �Amines �Reaction of amines in water �Ammonium salts �Alkaloids �Amides
- Slides: 39