Lecture 12 Applications of Confocal Microscopy BMS 524

Lecture 12 Applications of Confocal Microscopy BMS 524 - “Introduction to Confocal Microscopy and Image Analysis” 1 Credit course offered by Purdue University Department of Basic Medical Sciences, School of Veterinary Medicine J. Paul Robinson, Ph. D. Professor of Immunopharmacology Director, Purdue University Cytometry Laboratories These slides are intended for use in a lecture series. Copies of the graphics are distributed and students encouraged to take their notes on these graphics. The intent is to have the student NOT try to reproduce the figures, but to LISTEN and UNDERSTAND the material. All material copyright J. Paul Robinson unless otherwise stated, however, the material may be freely used for lectures, tutorials and workshops. It may not be used for any commercial purpose. The text for this course is Pawley “Introduction to Confocal Microscopy”, Plenum Press, 2 nd Ed. A number of the ideas and figures in these lecture notes are taken from this text. Purdue University Cytometry Laboratories UPDATED March 2000 Slide 1 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt
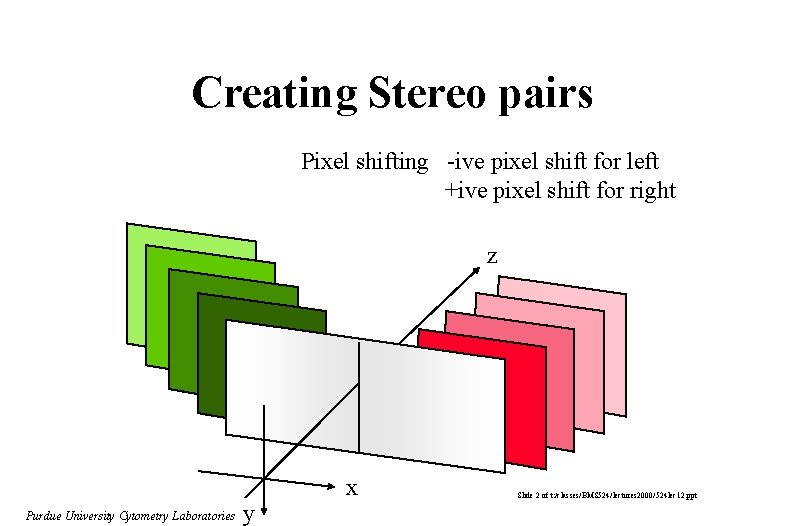
Creating Stereo pairs Pixel shifting -ive pixel shift for left +ive pixel shift for right z Purdue University Cytometry Laboratories y x Slide 2 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt
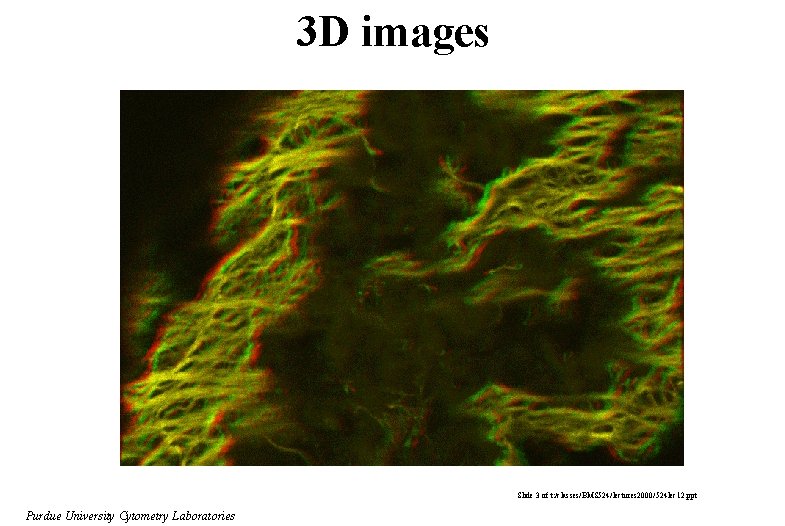
3 D images Slide 3 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
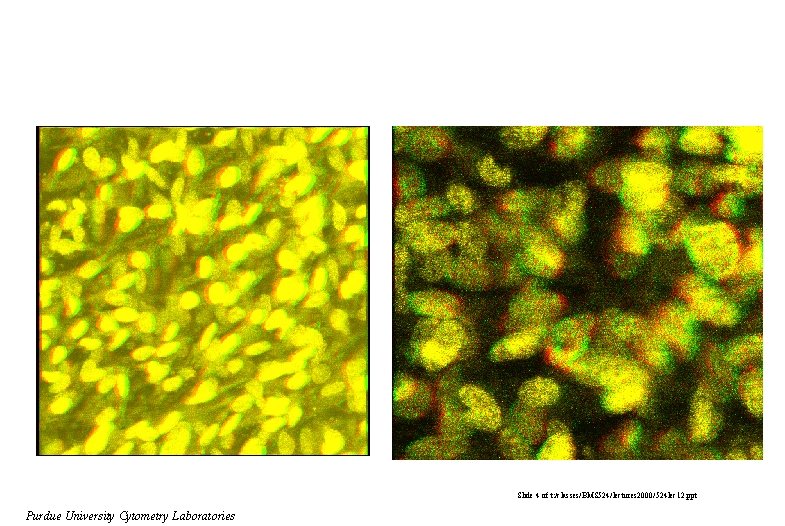
Slide 4 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
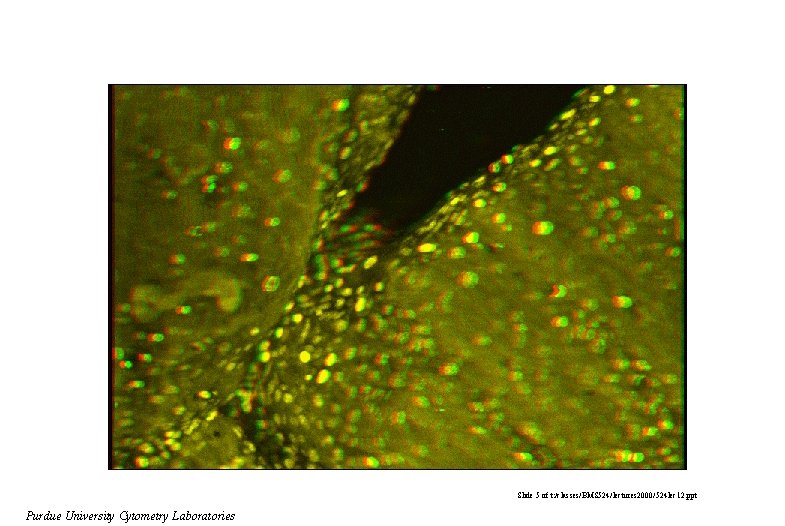
Slide 5 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
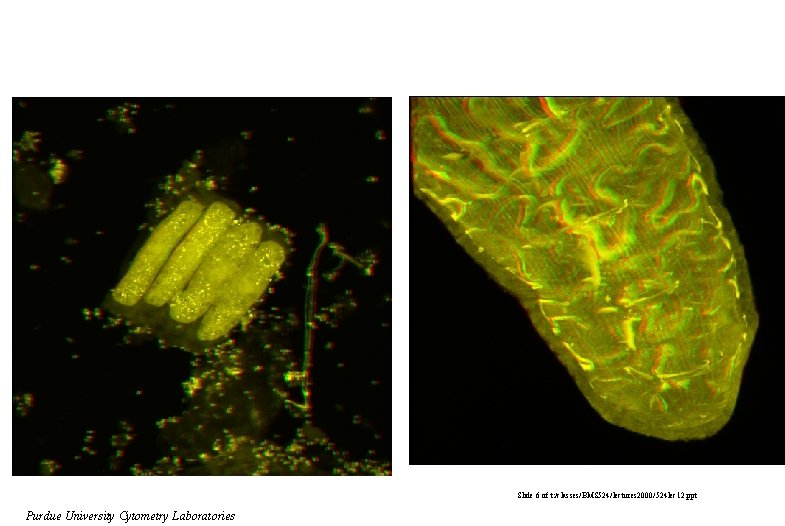
Slide 6 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
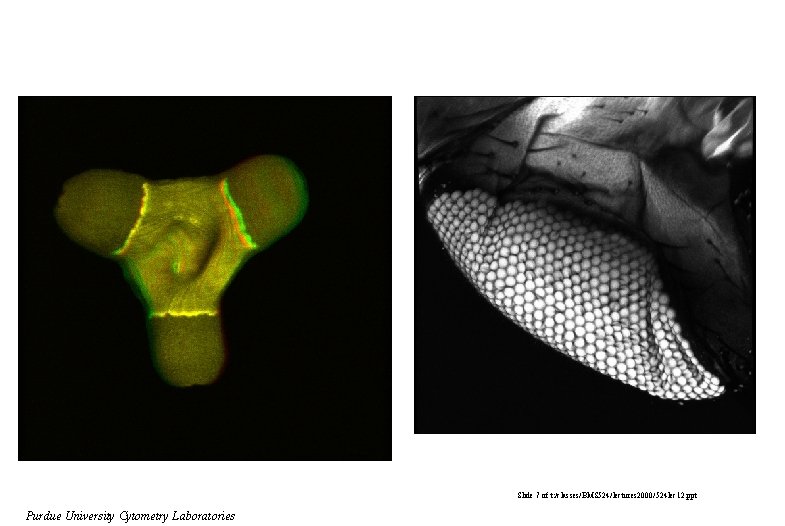
Slide 7 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Software available • SGI - Voxel. View • MAC - NIH Image • PC – – Optimus Microvoxel Lasersharp Confocal Assistant Slide 8 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Methods for visualization • Hidden object removal – Easiest methods is to reconstruct from back to front • Local Projections – Reference height above threshold – Local maximum intensity – Height at maximum intensity + Local Kalman Av. – Height at first intensity + Offset Local Ht. Intensity • Artificial lighting reflection Slide 9 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Visualization Issues Volume rendering is a computer graphics technique whereby the object or phenomenon of interest is sampled or subdivided into many cubic building blocks, called voxels (or volume elements. ) A voxel is the 3 -D counterpart of the 2 -D pixel and is a measure of unit volume. Each voxel carries one or more values for some measured or calculated property of the volume (such as intensity values in the case of LSCM data) and is typically represented by a unit cube. The 3 -D voxel sets are assembled from multiple 2 -D images (such as the LSCM image stack), and are displayed by projecting these images into 2 -D pixel space where they are stored in a frame buffer. Volumes rendered in this manner have been likened to a translucent suspension of particles in 3 -D space. In surface rendering, the volumetric data must first be converted into geometric primitives, by a process such as isosurfacing, isocontouring, surface extraction or border following. These primitives (such as polygon meshes or contours) are then rendered for display using conventional geometric rendering techniques. http: //www. cs. ubc. ca/spider/ladic/volviz. html Slide 10 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Additional Material • • Applications Live Cell studies Time Lapse videos exotic applications Slide 11 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Applications • Cellular Function – Esterase Activity – Oxidation Reactions – Intracellular p. H – Intracellular Calcium – Phagocytosis & Internalization – Apoptosis – Membrane Potential – Cell-cell Communication (Gap Junctions) Slide 12 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Applications • • • Conjugated Antibodies DNA/RNA Organelle Structure Cytochemical Identification Probe Ratioing Slide 13 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
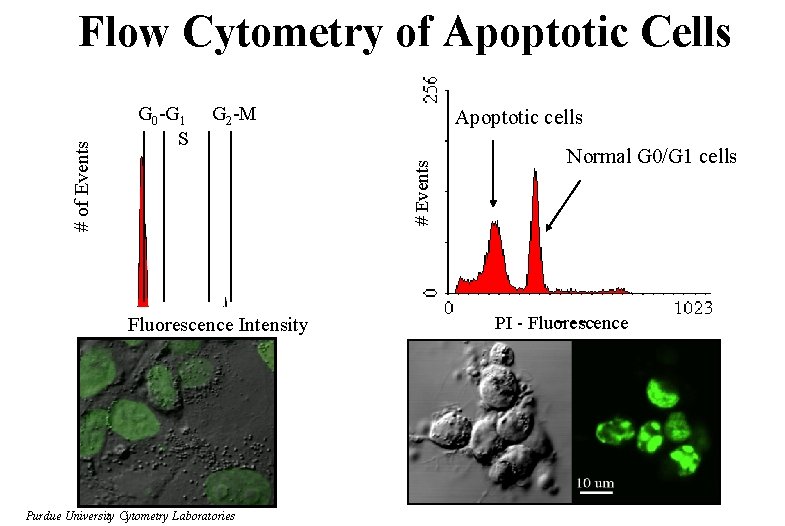
G 0 -G 1 S G 2 -M Apoptotic cells # Events # of Events Flow Cytometry of Apoptotic Cells Fluorescence Intensity Normal G 0/G 1 cells PI - Fluorescence Slide 14 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
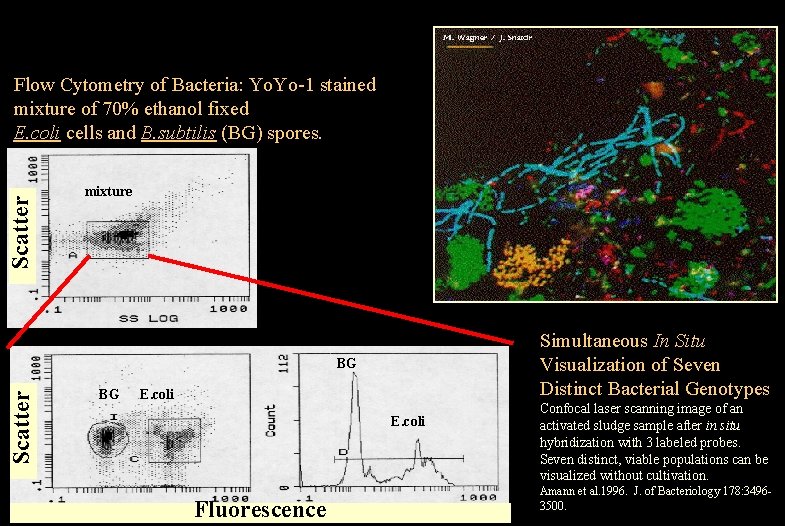
Scatter Flow Cytometry of Bacteria: Yo. Yo-1 stained mixture of 70% ethanol fixed E. coli cells and B. subtilis (BG) spores. mixture Simultaneous In Situ Visualization of Seven Distinct Bacterial Genotypes Scatter BG BG E. coli Fluorescence Purdue University Cytometry Laboratories Confocal laser scanning image of an activated sludge sample after in situ hybridization with 3 labeled probes. Seven distinct, viable populations can be visualized without cultivation. Amann et al. 1996. J. of Bacteriology Slide 15 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt 3500. 178: 3496 -
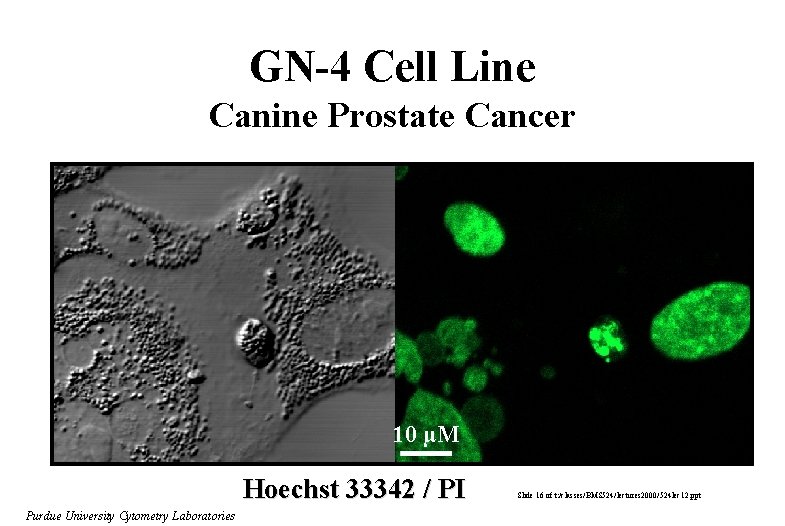
GN-4 Cell Line Canine Prostate Cancer Conjugated Linoleic Acid 200 µM 24 hours 10 µM Hoechst 33342 / PI Purdue University Cytometry Laboratories Slide 16 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt
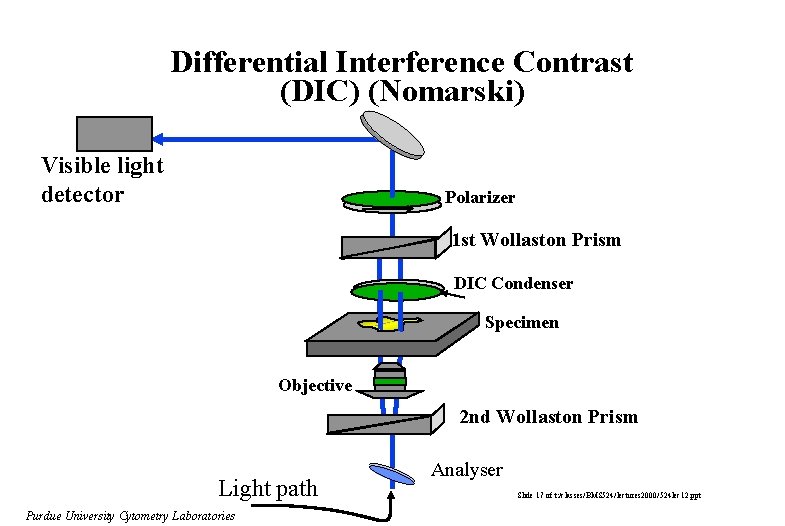
Differential Interference Contrast (DIC) (Nomarski) Visible light detector Polarizer 1 st Wollaston Prism DIC Condenser Specimen Objective 2 nd Wollaston Prism Light path Purdue University Cytometry Laboratories Analyser Slide 17 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt
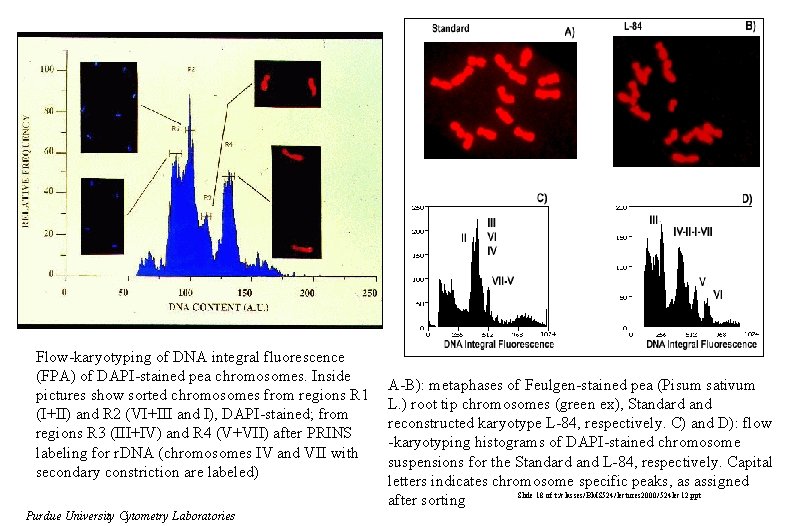
Flow-karyotyping of DNA integral fluorescence (FPA) of DAPI-stained pea chromosomes. Inside pictures show sorted chromosomes from regions R 1 (I+II) and R 2 (VI+III and I), DAPI-stained; from regions R 3 (III+IV) and R 4 (V+VII) after PRINS labeling for r. DNA (chromosomes IV and VII with secondary constriction are labeled) Purdue University Cytometry Laboratories A-B): metaphases of Feulgen-stained pea (Pisum sativum L. ) root tip chromosomes (green ex), Standard and reconstructed karyotype L-84, respectively. C) and D): flow -karyotyping histograms of DAPI-stained chromosome suspensions for the Standard and L-84, respectively. Capital letters indicates chromosome specific peaks, as assigned Slide 18 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt after sorting
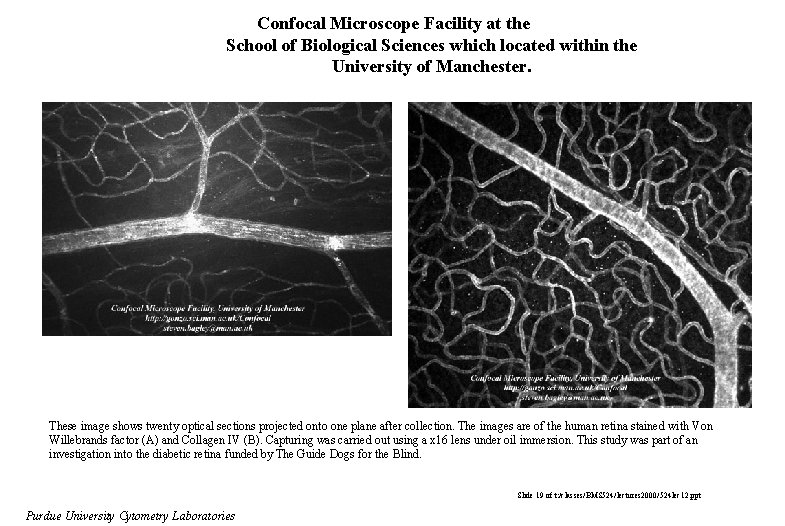
Confocal Microscope Facility at the School of Biological Sciences which located within the University of Manchester. These image shows twenty optical sections projected onto one plane after collection. The images are of the human retina stained with Von Willebrands factor (A) and Collagen IV (B). Capturing was carried out using a x 16 lens under oil immersion. This study was part of an investigation into the diabetic retina funded by The Guide Dogs for the Blind. Slide 19 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Examples from Bio-Rad web site Paramecium labeled with an anti -tubulin-antibody showing thousands of cilia and internal microtubular structures. Image Courtesy of Ann Fleury, Michel Laurent & Andre Adoutte, Laboratoire de Biologie Cellulaire, Université, Paris-Sud, Cedex France. Whole mount of Zebra Fish larva stained with Acridine Orange, Evans Blue and Eosin. Image Courtesy of Dr. W. B. Amos, Laboratory of Molecular Biology, MRC Cambridge U. K. Slide 20 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

Examples from Bio-Rad Web site Projection of 25 optical sections of a triple-labeled rat lslet of Langerhans, acquired with a krypton/argon laser. Image courtesy of T. Clark Brelje, Martin W. Wessendorf and Robert L. Sorenseon, Dept. of Cell Biology and Neuroanatomy, University of Minnesota Medical School. This image shows a maximum brightness projection of Golgi stained neurons. Slide 21 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
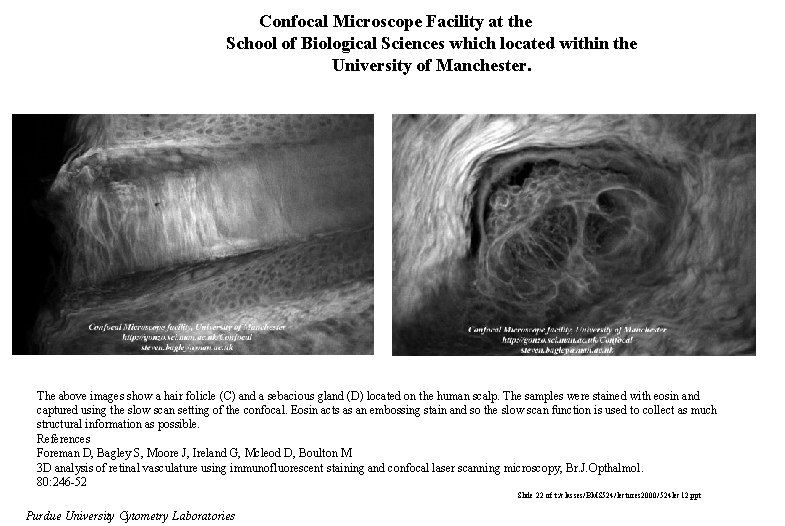
Confocal Microscope Facility at the School of Biological Sciences which located within the University of Manchester. The above images show a hair folicle (C) and a sebacious gland (D) located on the human scalp. The samples were stained with eosin and captured using the slow scan setting of the confocal. Eosin acts as an embossing stain and so the slow scan function is used to collect as much structural information as possible. References Foreman D, Bagley S, Moore J, Ireland G, Mcleod D, Boulton M 3 D analysis of retinal vasculature using immunofluorescent staining and confocal laser scanning microscopy, Br. J. Opthalmol. 80: 246 -52 Slide 22 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
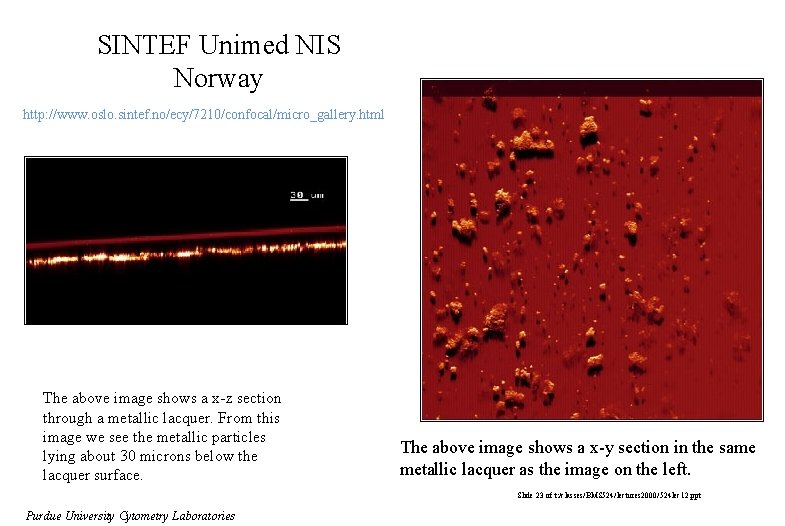
SINTEF Unimed NIS Norway http: //www. oslo. sintef. no/ecy/7210/confocal/micro_gallery. html The above image shows a x-z section through a metallic lacquer. From this image we see the metallic particles lying about 30 microns below the lacquer surface. The above image shows a x-y section in the same metallic lacquer as the image on the left. Slide 23 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

http: //www. vaytek. com/ Material from Vaytek Web site The image on the left shows an axial (top) and a lateral view of a single hamster ovary cell. The image was reconstructed from optical sections of actin-stained specimen (confocal fluorescence), using Vay. Tek's Vox. Blast software. Image courtesy of Doctors Ian S. Harper, Yuping Yuan, and Shaun Jackson of Monash University, Australia. (see Journal of Biological Chemistry 274: 36241 -36251, 1999) Slide 24 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories

http: //www. vaytek. com/vox. htm Slide 25 of t: /classes/BMS 524/lectures 2000/524 lec 12. ppt Purdue University Cytometry Laboratories
- Slides: 25