Lecture 11 Ocean Carbonate Chemistry Ocean Distributions Controls

Lecture 11: Ocean Carbonate Chemistry: Ocean Distributions Controls on Distributions What is the distribution of CO 2 added to the ocean? See Section 4. 4 Emerson and Hedges

Sarmiento and Gruber (2002) Sinks for Anthropogenic Carbon Physics Today August 2002 30 -36
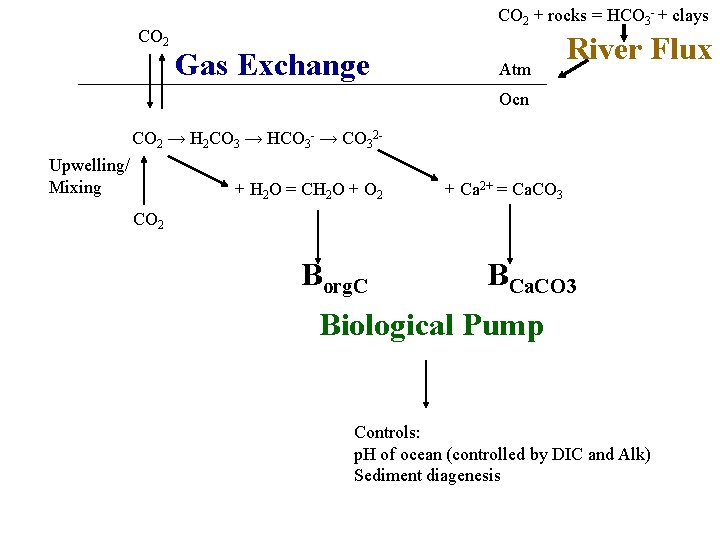
CO 2 + rocks = HCO 3 - + clays Gas Exchange Atm River Flux Ocn CO 2 → H 2 CO 3 → HCO 3 - → CO 32 Upwelling/ Mixing + H 2 O = CH 2 O + O 2 + Ca 2+ = Ca. CO 3 CO 2 Borg. C BCa. CO 3 Biological Pump Controls: p. H of ocean (controlled by DIC and Alk) Sediment diagenesis
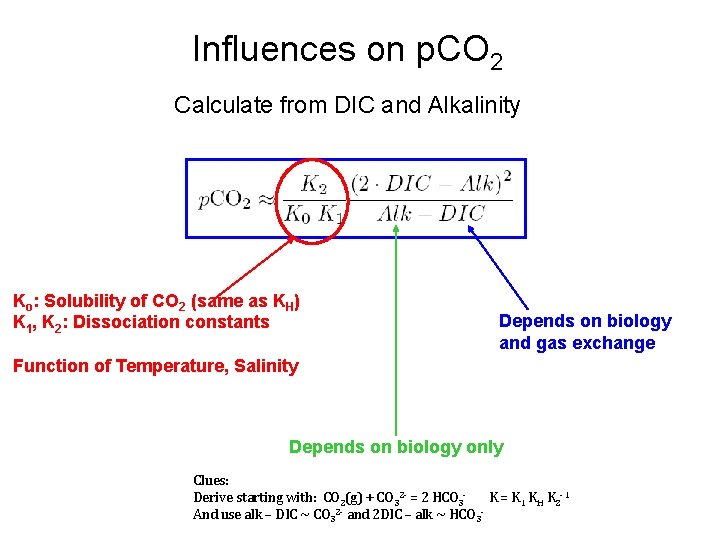
Influences on p. CO 2 Calculate from DIC and Alkalinity Ko: Solubility of CO 2 (same as KH) K 1, K 2: Dissociation constants Depends on biology and gas exchange Function of Temperature, Salinity Depends on biology only Clues: Derive starting with: CO 2(g) + CO 32 - = 2 HCO 3 K = K 1 KH K 2 -1 And use alk – DIC ~ CO 32 - and 2 DIC – alk ~ HCO 3 -
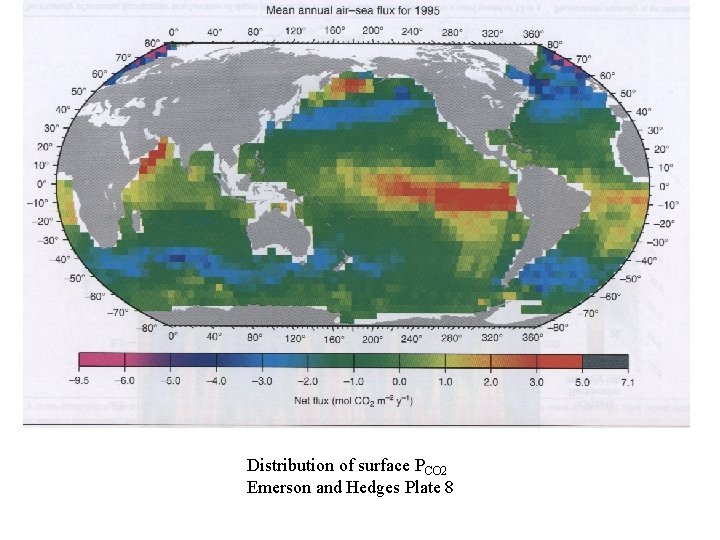
Distribution of surface PCO 2 Emerson and Hedges Plate 8
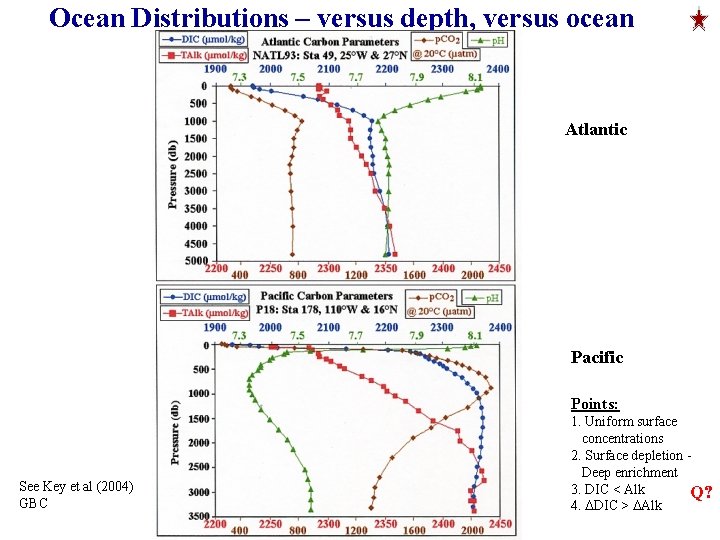
Ocean Distributions – versus depth, versus ocean Atlantic Pacific Points: See Key et al (2004) GBC 1. Uniform surface concentrations 2. Surface depletion Deep enrichment 3. DIC < Alk Q? 4. DDIC > DAlk

Controls on Ocean Distributions A) Photosynthesis/Respiration Organic matter (approximated as CH 2 O for this example) is produced and consumed as follows: CH 2 O + O 2 CO 2 + H 2 O Then: CO 2 + H 2 O H 2 CO 3* H+ + HCO 3 - H+ + CO 32 As CO 2 is produced during respiration we should observe: p. H DIC Alk PCO 2 is an acid The trends will be the opposite for photosynthesis. B) Ca. CO 3 dissolution/precipitation Ca. CO 3(s) Ca 2+ + CO 3 2 Also written as: CO 32 - is a base Ca. CO 3(s) + CO 2 + H 2 O Ca 2+ + 2 HCO 3 As Ca. CO 3(s) dissolves, CO 32 - is added to solution. We should observe: p. H DIC Alk PCO 2 Summary: DIC is from both organic matter and Ca. CO 3 Alk is only from Ca. CO 3
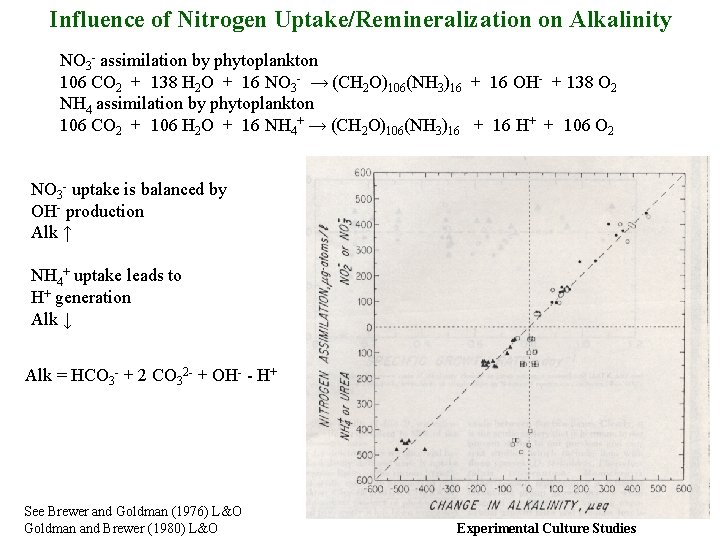
Influence of Nitrogen Uptake/Remineralization on Alkalinity NO 3 - assimilation by phytoplankton 106 CO 2 + 138 H 2 O + 16 NO 3 - → (CH 2 O)106(NH 3)16 + 16 OH- + 138 O 2 NH 4 assimilation by phytoplankton 106 CO 2 + 106 H 2 O + 16 NH 4+ → (CH 2 O)106(NH 3)16 + 16 H+ + 106 O 2 NO 3 - uptake is balanced by OH- production Alk ↑ NH 4+ uptake leads to H+ generation Alk ↓ Alk = HCO 3 - + 2 CO 32 - + OH- - H+ See Brewer and Goldman (1976) L&O Goldman and Brewer (1980) L&O Experimental Culture Studies
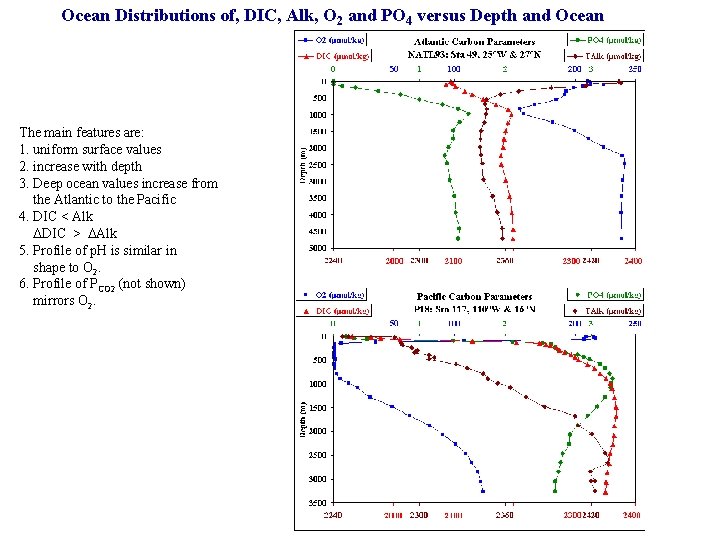
Ocean Distributions of, DIC, Alk, O 2 and PO 4 versus Depth and Ocean The main features are: 1. uniform surface values 2. increase with depth 3. Deep ocean values increase from the Atlantic to the Pacific 4. DIC < Alk DDIC > DAlk 5. Profile of p. H is similar in shape to O 2. 6. Profile of PCO 2 (not shown) mirrors O 2.
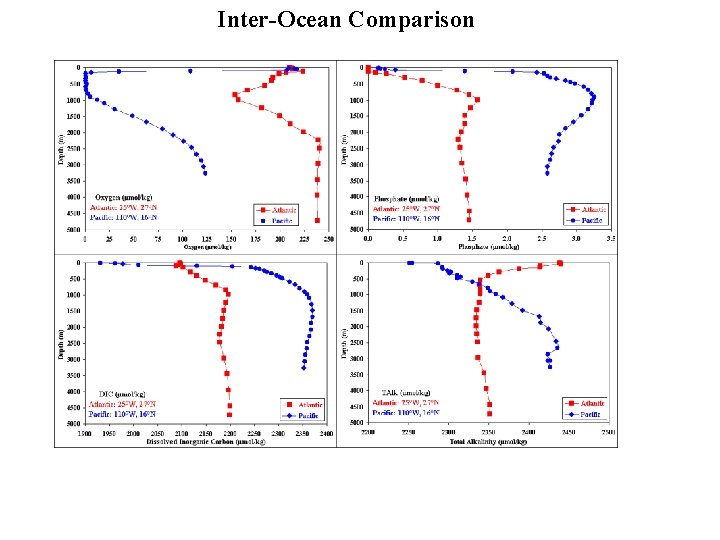
Inter-Ocean Comparison

Carbonate ion (CO 32 -) and p. H decrease from Atlantic to Pacific x 10 -3 mol kg-1 x 10 -6 mol kg-1 S = 35 T = 25°C Alk 2. 300 DIC 1. 950 CO 32246 p. H 8. 12 North Atlantic Deep Water 2. 350 2. 190 128 7. 75 Antarctic Deep Water 2. 390 2. 280 101 7. 63 Deep Atlantic to Deep Pacific DAlk = 0. 070 DDIC = 0. 180 7. 46 So DAlk/DDIC = 0. 40 Surface Water North Pacific Deep water 2. 420 Q? CO 2 Sys/CO 2 Calc 2. 370 72 CO 32 - decreases from surface to deep Atlantic to deep Pacific. These CO 32 - are from CO 2 Sys. Can Approximate as CO 32 - ≈ Alk - DIC

Composition of Sinking Particles and Predicted Changes Assume the following average elemental composition of marine particulate matter P N C Ca Si Soft Parts 1 15 105 0 0 Hard Parts 0 0 26 26 50 Composite 1 15 132 26 50 Implies Org C / Ca. CO 3 ~ 105/26 ~4/1 The impact of this material dissolving CH 2 O + O 2 CO 2 + H 2 O DDIC = 1 DAlk = 0 Ca. CO 3 Ca 2+ + CO 32 DDIC = 1 DAlk = 2 DDIC DCa Dalk 1 mol Ca. CO 3 1 1 2 4 mol org. C 4 0 0 Composite 5 1 2 Consequences: 1) DAlk/DDIC = 2/5 = 0. 40 (DIC changes more than Alk) 2) Dalk – DDIC ~ DCO 32 - = 2 – 5 = -3 (CO 3 2 - decreases)
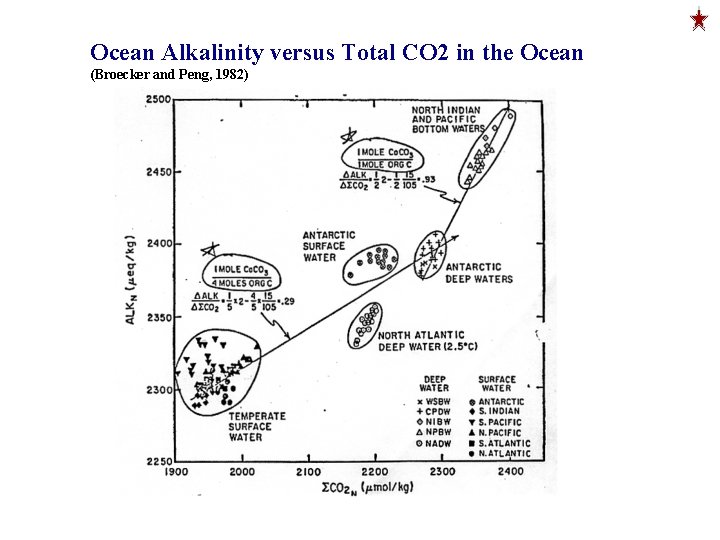
Ocean Alkalinity versus Total CO 2 in the Ocean (Broecker and Peng, 1982)
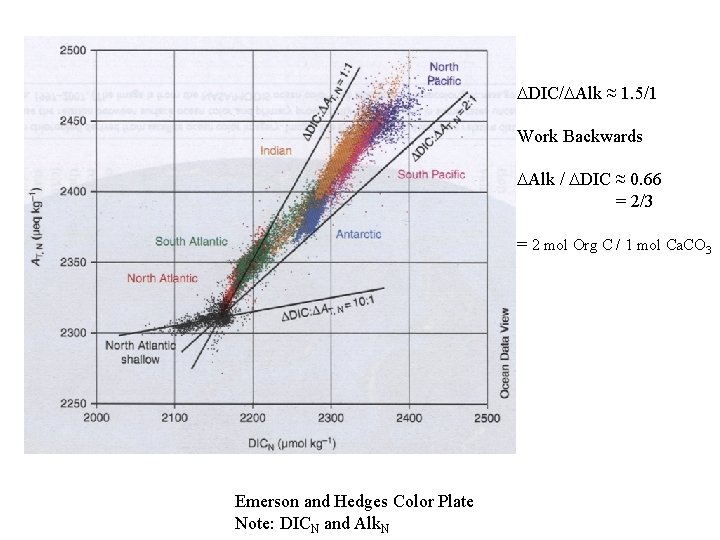
DDIC/DAlk ≈ 1. 5/1 Work Backwards DAlk / DDIC ≈ 0. 66 = 2/3 = 2 mol Org C / 1 mol Ca. CO 3 Emerson and Hedges Color Plate Note: DICN and Alk. N
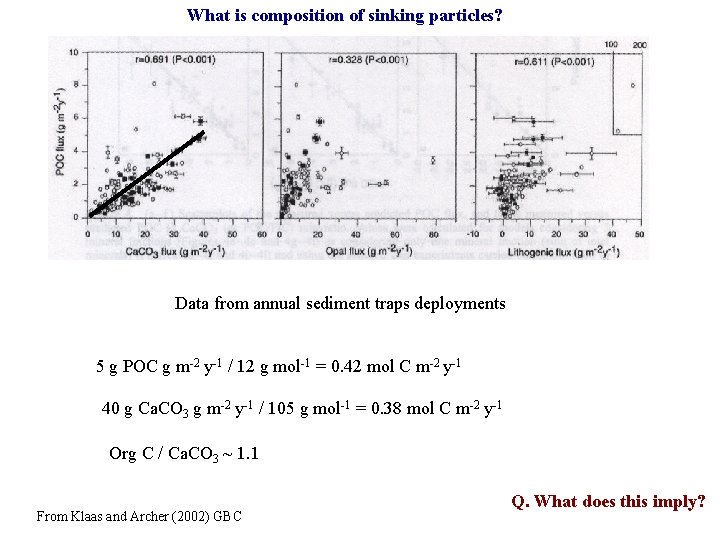
What is composition of sinking particles? Data from annual sediment traps deployments 5 g POC g m-2 y-1 / 12 g mol-1 = 0. 42 mol C m-2 y-1 40 g Ca. CO 3 g m-2 y-1 / 105 g mol-1 = 0. 38 mol C m-2 y-1 Org C / Ca. CO 3 ~ 1. 1 From Klaas and Archer (2002) GBC Q. What does this imply?
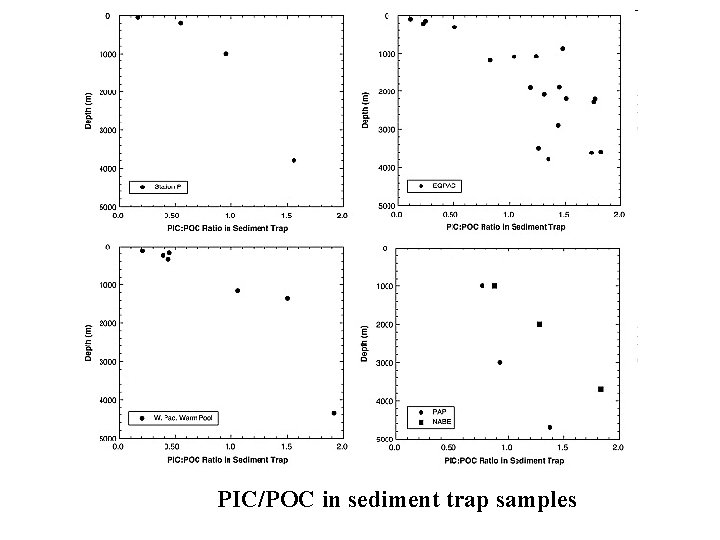
PIC/POC in sediment trap samples
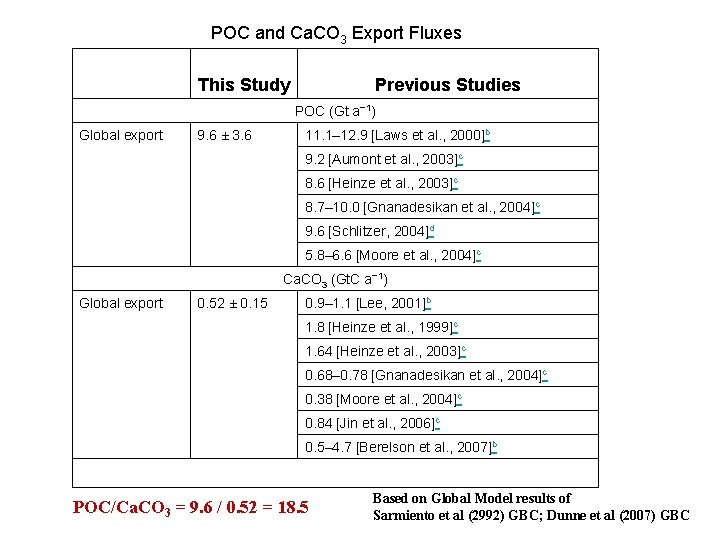
POC and Ca. CO 3 Export Fluxes This Study Previous Studies POC (Gt a− 1) Global export 9. 6 ± 3. 6 11. 1– 12. 9 [Laws et al. , 2000]b 9. 2 [Aumont et al. , 2003]c 8. 6 [Heinze et al. , 2003]c 8. 7– 10. 0 [Gnanadesikan et al. , 2004]c 9. 6 [Schlitzer, 2004]d 5. 8– 6. 6 [Moore et al. , 2004]c Ca. CO 3 (Gt. C a− 1) Global export 0. 52 ± 0. 15 0. 9– 1. 1 [Lee, 2001]b 1. 8 [Heinze et al. , 1999]c 1. 64 [Heinze et al. , 2003]c 0. 68– 0. 78 [Gnanadesikan et al. , 2004]c 0. 38 [Moore et al. , 2004]c 0. 84 [Jin et al. , 2006]c 0. 5– 4. 7 [Berelson et al. , 2007]b POC/Ca. CO 3 = 9. 6 / 0. 52 = 18. 5 Based on Global Model results of Sarmiento et al (2992) GBC; Dunne et al (2007) GBC

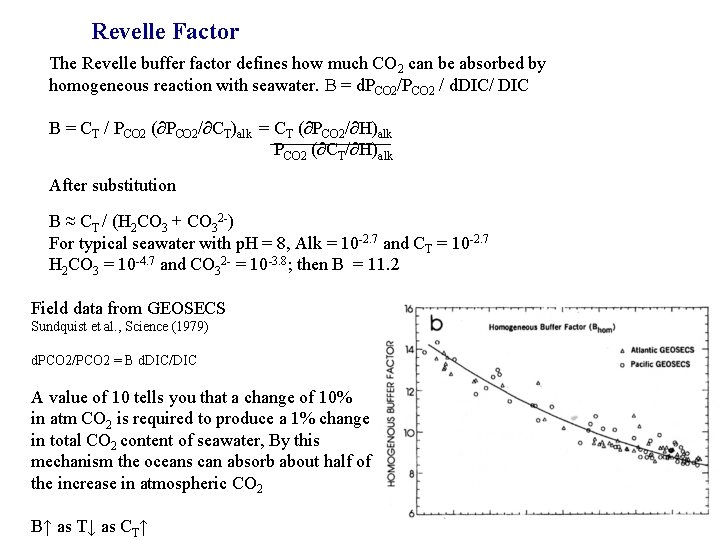
Revelle Factor The Revelle buffer factor defines how much CO 2 can be absorbed by homogeneous reaction with seawater. B = d. PCO 2/PCO 2 / d. DIC/ DIC B = CT / PCO 2 (∂PCO 2/∂CT)alk = CT (∂PCO 2/∂H)alk PCO 2 (∂CT/∂H)alk After substitution B ≈ CT / (H 2 CO 3 + CO 32 -) For typical seawater with p. H = 8, Alk = 10 -2. 7 and CT = 10 -2. 7 H 2 CO 3 = 10 -4. 7 and CO 32 - = 10 -3. 8; then B = 11. 2 Field data from GEOSECS Sundquist et al. , Science (1979) d. PCO 2/PCO 2 = B d. DIC/DIC A value of 10 tells you that a change of 10% in atm CO 2 is required to produce a 1% change in total CO 2 content of seawater, By this mechanism the oceans can absorb about half of the increase in atmospheric CO 2 B↑ as T↓ as CT↑

Revelle Factor Numerical Example (using CO 2 Sys) CO 2 + CO 32 - = HCO 3 CO 2 350 ppm + 10% = 385 ppm Atm Ocn CO 2 → H 2 CO 3 → at constant alkalinity HCO 3 - → CO 32 - DIC 11. 3 m. M 1640. 5 m. M 183. 7 1837 +1. 2 (10. 6%) +27. 7 (1. 7%) -11. 1 (-6. 0%) +17. 9 (+0. 97%) 12. 5 1668. 2 174. 2 1854. 9 The total increase in DIC of +17. 9 m. M is mostly due to a big change in HCO 3 - (+27. 7 m. M) countering a decrease in CO 32 - (-11. 1 m. M). Most of the CO 2 added to the ocean reacts with CO 32 - to make HCO 3 -. The final increase in H 2 CO 3 is a small (+1. 2 m. M) portion of the total.
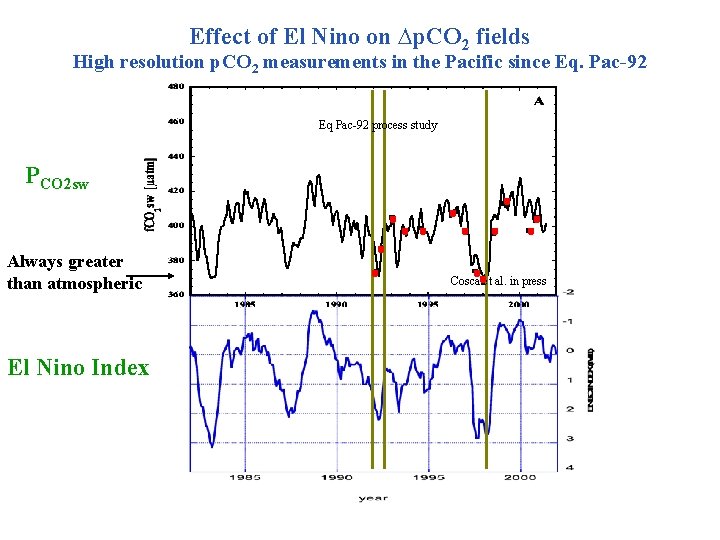
Effect of El Nino on ∆p. CO 2 fields High resolution p. CO 2 measurements in the Pacific since Eq. Pac-92 Eq Pac-92 process study PCO 2 sw Always greater than atmospheric El Nino Index Cosca et al. in press
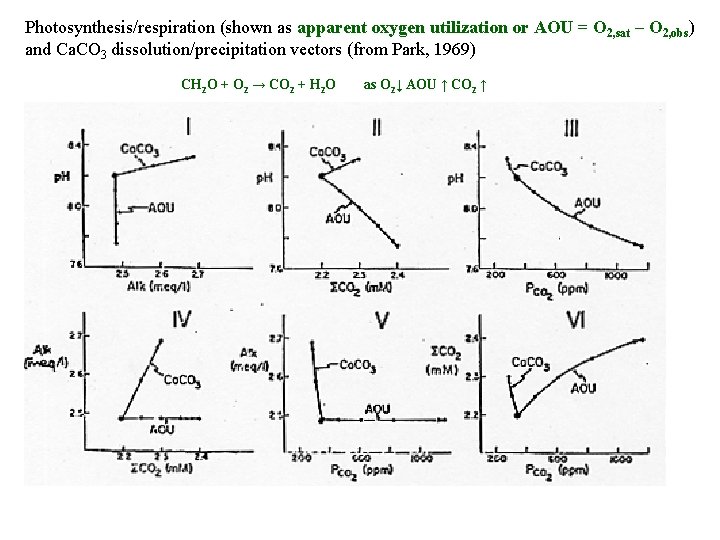
Photosynthesis/respiration (shown as apparent oxygen utilization or AOU = O 2, sat – O 2, obs) and Ca. CO 3 dissolution/precipitation vectors (from Park, 1969) CH 2 O + O 2 → CO 2 + H 2 O as O 2↓ AOU ↑ CO 2 ↑

Composition of Sinking Particles and Predicted Changes

Problem: DIC and Alk are capacity factors. p. H and PCO 2 are not. 1. 2. 3. 4. 5. Define two water parcels with T, S, DIC and Alk Calc p. H and PCO 2 for these end members Mix them 50/50 Calculate T, S, DIC, Alk, p. H and PCO 2 of the mixture. Do all properties fall along a straight line?
- Slides: 24