LECTURE 10 Wednesday 2817 Chapter 13 Alcohols Phenols




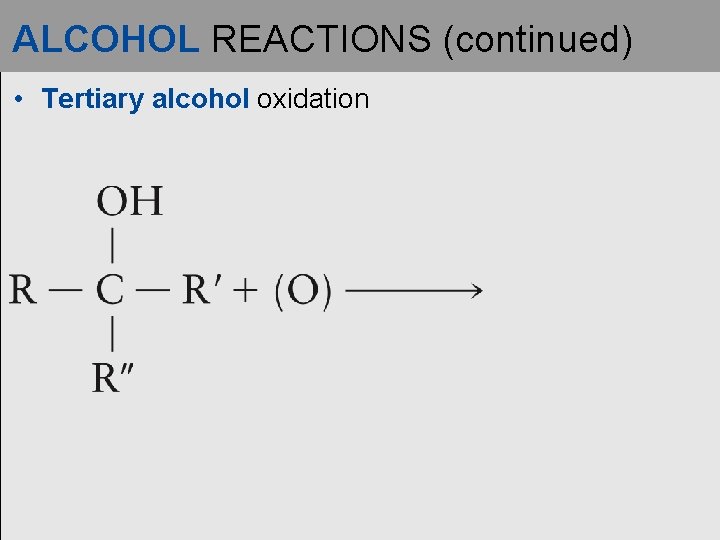












- Slides: 27

LECTURE 10 Wednesday 2/8/17

Chapter 13 Alcohols, Phenols, and Ethers

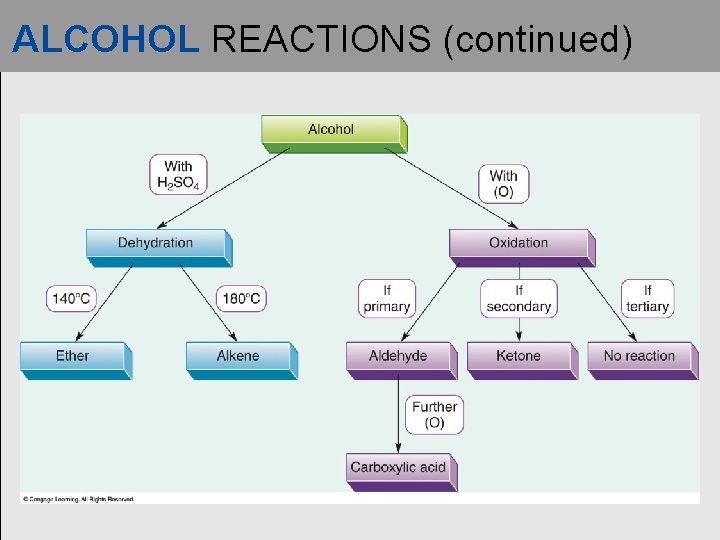
ALCOHOL REACTIONS (continued) • An oxidation reaction occurs when an alcohol loses hydrogen atoms. • Alcohol oxidations with an oxidizing (O) agent, such as K 2 Cr 2 O 7 and KMn. O 4:

ALCOHOL REACTIONS (continued) • Primary alcohol oxidation • Secondary alcohol oxidation
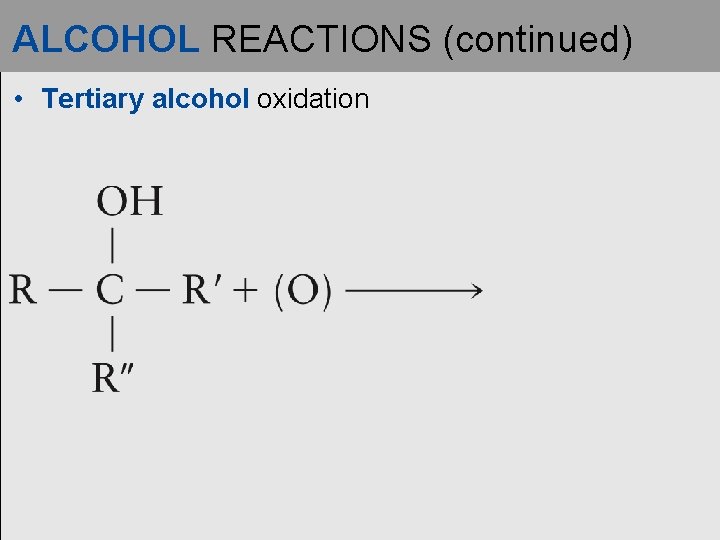
ALCOHOL REACTIONS (continued) • Tertiary alcohol oxidation

ALCOHOL OXIDATION EXAMPLES

ALCOHOL REACTIONS (continued)

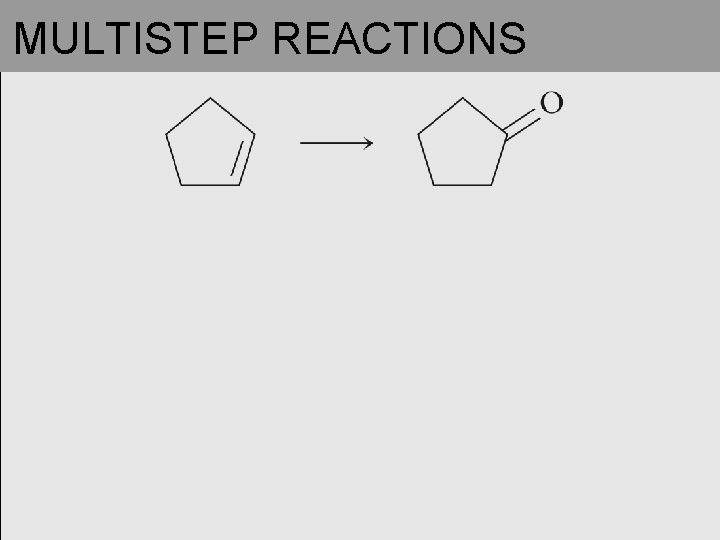
MULTISTEP REACTIONS

IMPORTANT ALCOHOLS • Methanol (wood alcohol): • Production:

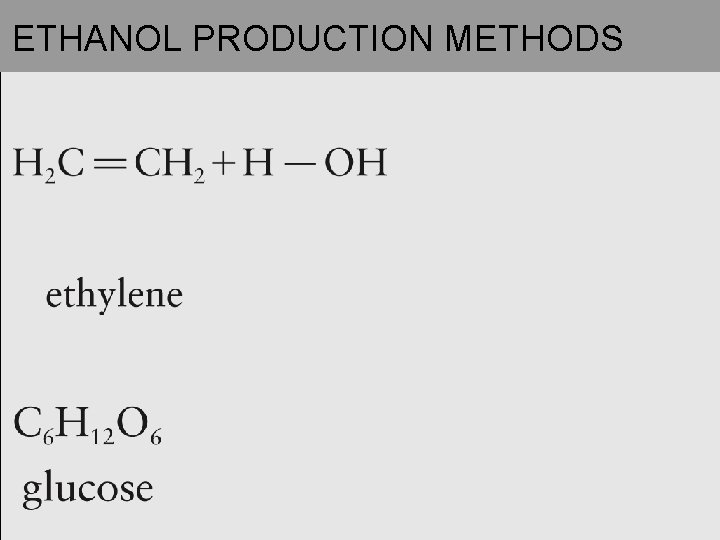
IMPORTANT ALCOHOLS (continued) • Ethanol (ethyl alcohol, grain alcohol): • Produced commercially from ethylene and through biological (yeast) ____________ of carbohydrates

ETHANOL PRODUCTION METHODS

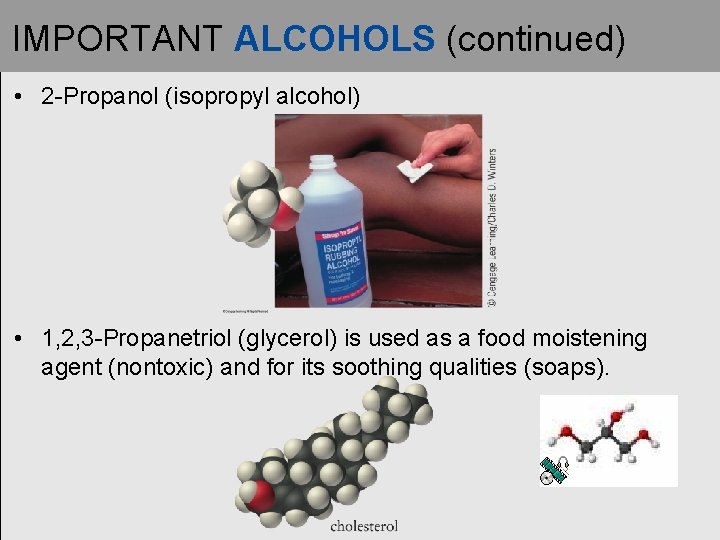
IMPORTANT ALCOHOLS (continued) • 2 -Propanol (isopropyl alcohol) • 1, 2, 3 -Propanetriol (glycerol) is used as a food moistening agent (nontoxic) and for its soothing qualities (soaps).

IMPORTANT ALCOHOLS (continued) • Antifreezes 1, 2 -ethanediol (ethylene glycol) • 1, 2 -propanediol (propylene glycol)

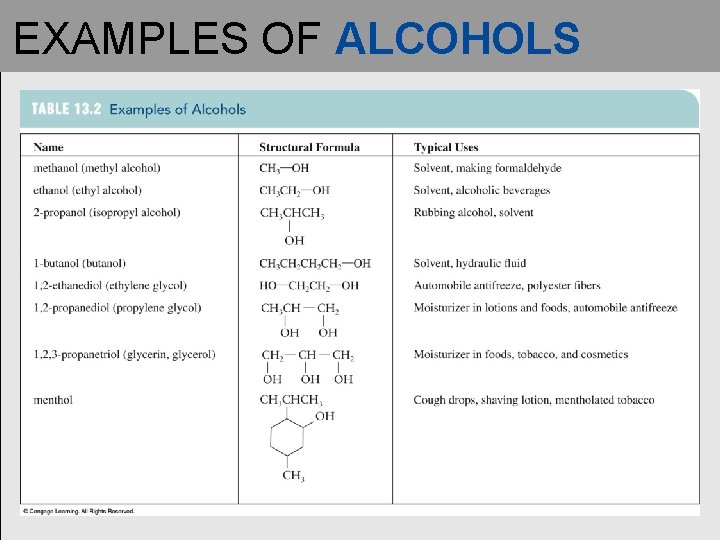
EXAMPLES OF ALCOHOLS

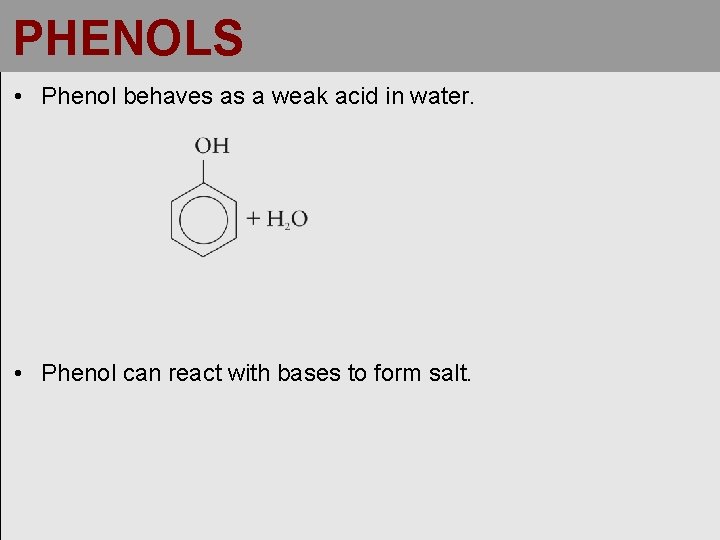
PHENOLS • Phenol behaves as a weak acid in water. • Phenol can react with bases to form salt.

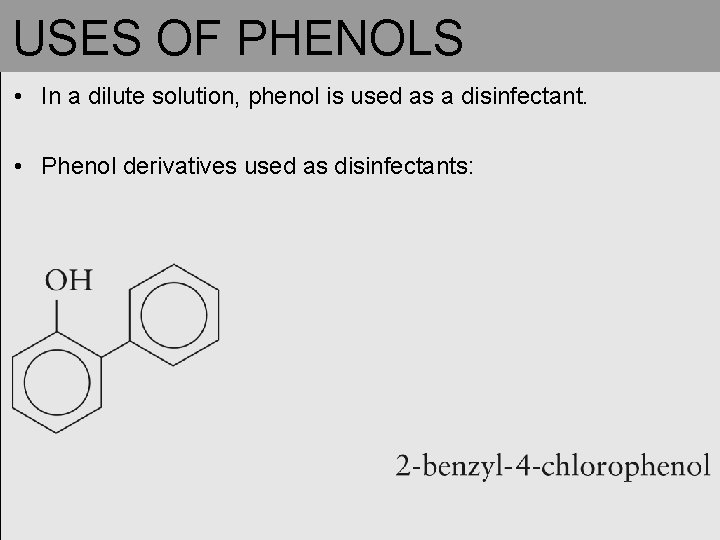
USES OF PHENOLS • In a dilute solution, phenol is used as a disinfectant. • Phenol derivatives used as disinfectants:

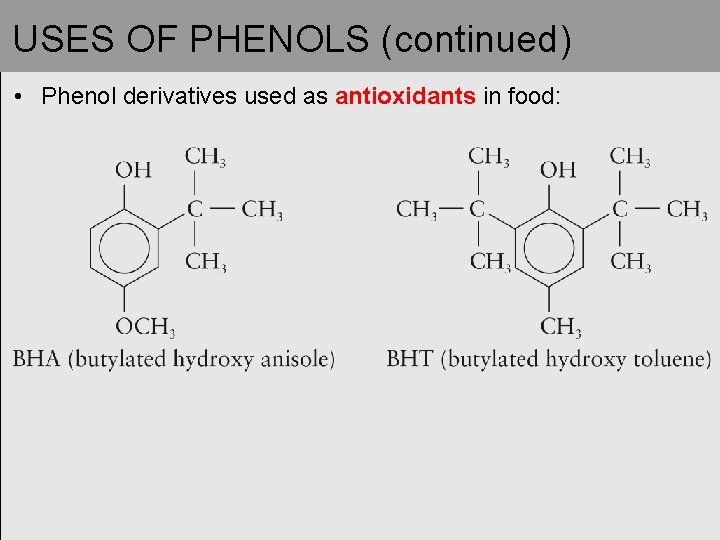
USES OF PHENOLS (continued) • Phenol derivatives used as antioxidants in food:

NAMING ETHERS • IUPAC name: Name the smaller of the two R groups as an alkoxy group attached to the parent chain by replacing the –yl ending of the R group with –oxy. • Common name: Name the groups attached to the oxygen alphabetically and add the word ether.

CYCLIC ETHERS • _________________ contain atoms other than carbon in the ring.

PROPERTIES OF ETHERS • Much less polar than • More soluble in water than _____ but less soluble than ______

PROPERTIES OF ETHERS (continued) • Hydrogen bonding of dimethyl ether: (a) with water and (b) no hydrogen bonding in the pure state.

THIOLS: THE –SH (SULFHYDRYL) GROUP • Most distinguishing characteristic • ethanethiol – • propanethiol – • 1 -propene-3 -thiol and 3, 3 -di-(1 -propenyl)disulfide – • trans-2 -butene-1 -thiol, 3 -methyl-1 -butanethiol, and methyl 1 -(trans-2 -butenyl)disulfide –

THIOL REACTIONS • Oxidation forms disulfide (-S-S-) linkages, which are important structural features of some proteins: • Specific example:

THIOL REACTIONS (continued) • Oxidation reactions can be reversed with a reducing agent (H), such as H 2, which regenerates the thiol:

THIOL REACTIONS (continued) • Thiols react with heavy metals (Pb 2+, Hg 2+) to form insoluble compounds, with adverse biological results:

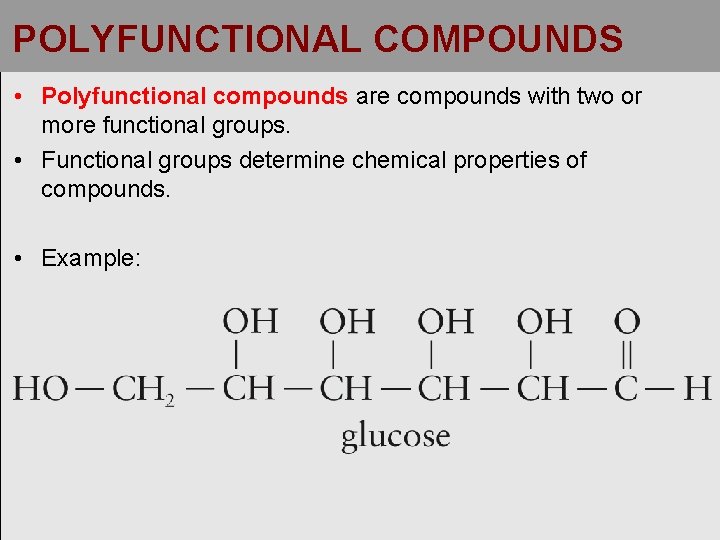
POLYFUNCTIONAL COMPOUNDS • Polyfunctional compounds are compounds with two or more functional groups. • Functional groups determine chemical properties of compounds. • Example:

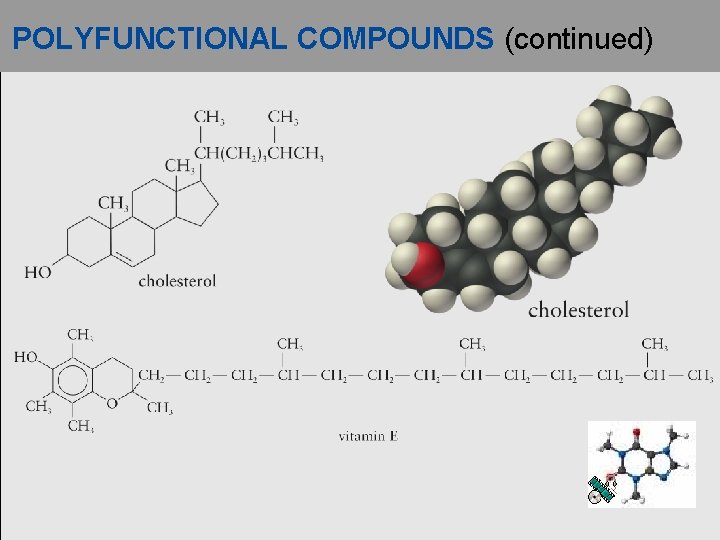
POLYFUNCTIONAL COMPOUNDS (continued)