LECTURE 10 Acids Bases and Salts ACIDS AND

LECTURE 10 Acids, Bases and Salts

ACIDS AND BASES Outlines • Properties of Acids and Bases • Acid – Base Theories • Strong and Weak Acids and Bases • Understanding Indicators • p. H Scale • Buffers and Antacids

Properties of Acids and Bases • Acids • Taste sour • Produce painful sensation on skin • React with certain metals (Mg, Zn, Fe) to produce H 2 gas • React with limestone and baking soda to produce CO 2 • Turn litmus paper red • Bases • Taste bitter • Feel slippery on skin • React with oils and greases • Turn litmus paper blue • React with acids to produce salt and water

Acids in Every Day Life Common Acids: Chemical Name Hydrochloric Acid (HCL) Common Name Muratic Acid Acetic Acid (CH 3 COOH) Vinegar Sulfuric Acid (H 2 SO 4) Auto Battery Acid Carbonic Acid (H 2 CO 3) Carbonated Water Boric Acid (H 3 BO 4) Acetylsalicylic Acid (C 16 H 12 O 6) Antiseptic Eye Drops Aspirin
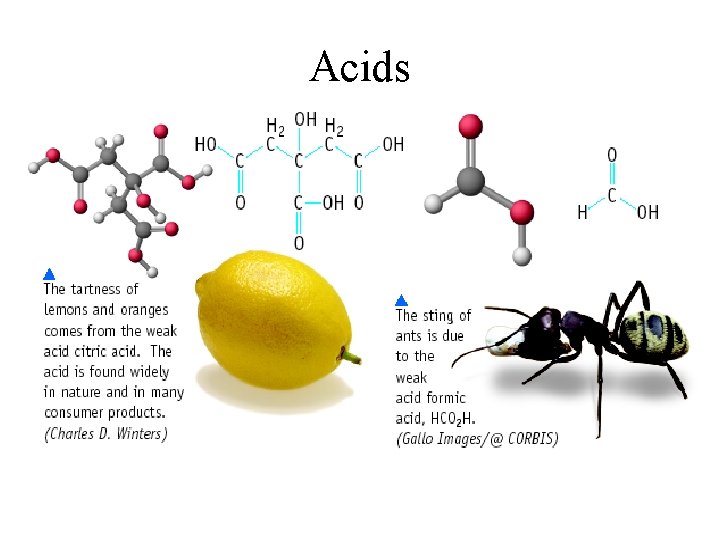
Acids
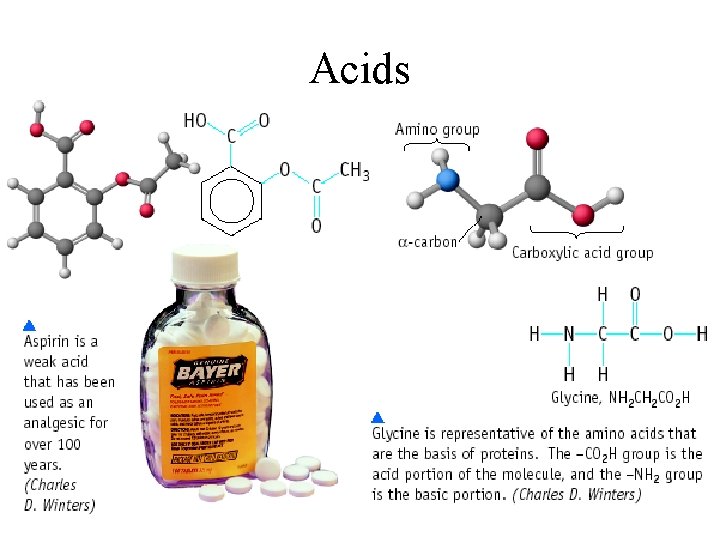
Acids

Bases in Every Day Life Common Bases : Chemical Name Common Name or Use Ammonia (NH 3) Cleaner Sodium Hydroxide (Na. OH) Lye Sodium Bicarbonate (Na. HCO 3) Baking Soda Magnesium Hydroxide (Mg(OH)2) Milk of Magnesia Calcium Carbonate (Ca. CO 3) Antacid Aluminum Hydroxide (Al(OH)3) Antacid
Bases

Microscopic View: • You may have noticed that all the acids contain hydrogen, while most of the bases contain the hydroxide ion (OH-). • Three main theories use these facts in their descriptions of acids and bases and their reactions: • Arrhenius Theory • Bronsted-Lowery Theory • Lewis Theory
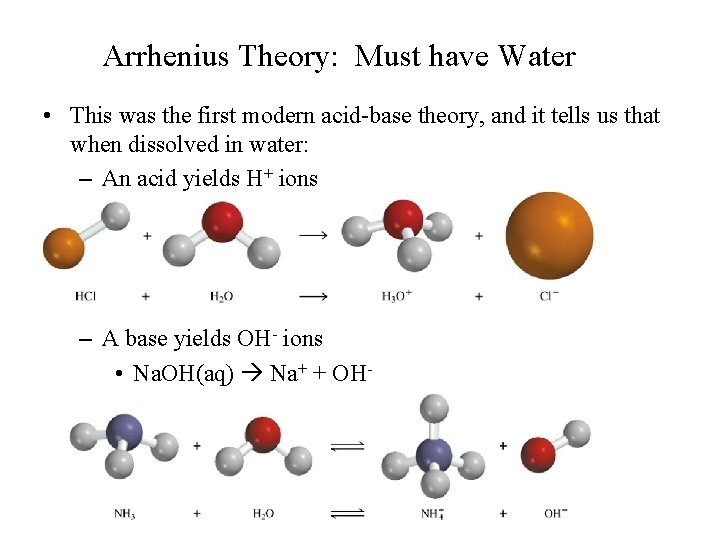
Arrhenius Theory: Must have Water • This was the first modern acid-base theory, and it tells us that when dissolved in water: – An acid yields H+ ions • HCl(aq) H+ + Cl- – A base yields OH- ions • Na. OH(aq) Na+ + OH-

This theory also classifies the reaction between an acid and a base as a neutralization reaction, producing a neutral solution composed of a water and a salt. • HCl(aq) + Na. OH(aq) H 2 O(l) + Na. Cl(aq) • The water is formed from the combining of the H+ and OH- ions • Like all theories, this one has its limitations. • Some bases don’t have hydroxide ions. • To account for this, a new theory was developed.
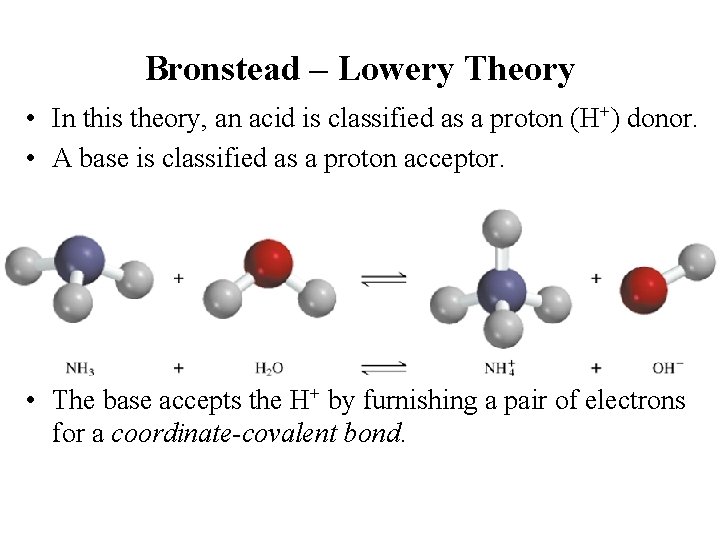
Bronstead – Lowery Theory • In this theory, an acid is classified as a proton (H+) donor. • A base is classified as a proton acceptor. • The base accepts the H+ by furnishing a pair of electrons for a coordinate-covalent bond.
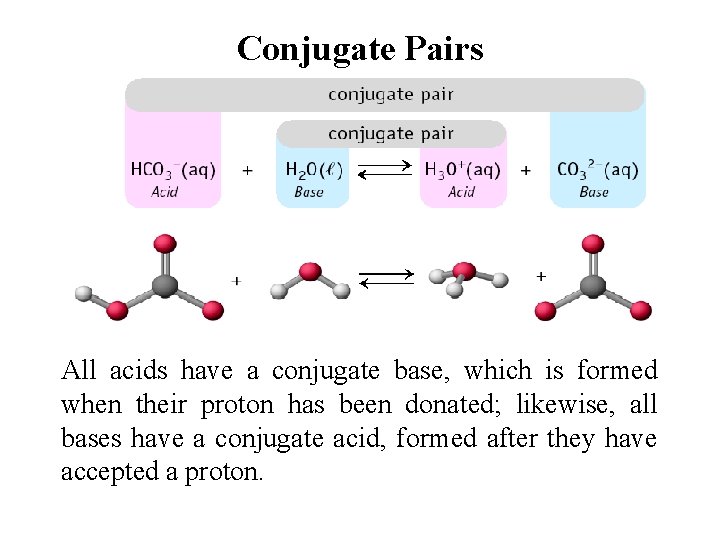
Conjugate Pairs All acids have a conjugate base, which is formed when their proton has been donated; likewise, all bases have a conjugate acid, formed after they have accepted a proton.
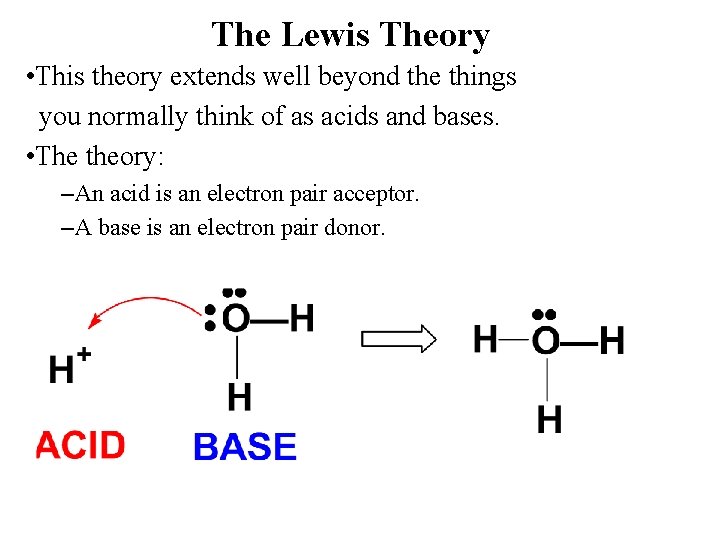
The Lewis Theory • This theory extends well beyond the things you normally think of as acids and bases. • The theory: –An acid is an electron pair acceptor. –A base is an electron pair donor.
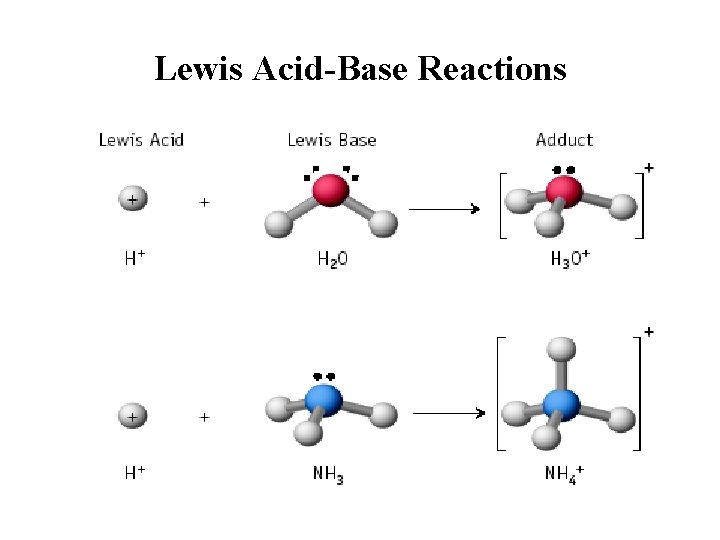
Lewis Acid-Base Reactions
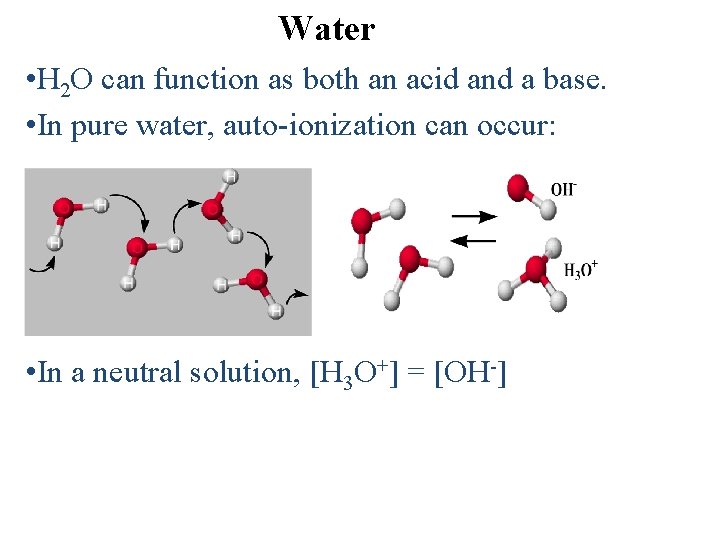
Water • H 2 O can function as both an acid and a base. • In pure water, auto-ionization can occur: • In a neutral solution, [H 3 O+] = [OH-]

Strong & Weak Acids • It is important to remember that acid-base strength is not the same as concentration. • Strength refers to the amount of ionization or breaking apart that a particular acid or base undergoes. • Concentration refers to the amount of acid or base that you initially have. • You can have a concentrated weak acid or a dilute strong acid…

Strong Acids • Certain acids are considered to be strong, which means they are dissociated 100% in solution: – – – HCl HNO 3 H 2 SO 4 HBr HI HCl. O 4 Hydrochloric Acid Nitric Acid Sulfuric Acid Hydrobromic Acid Hydroiodic Acid Perchloric Acid • You ought to memorize this list, because almost every other acid is weak. • The most common example is HCl.

Weak Acids • A weak acid is one which doesn't ionize fully when it is dissolved in water. • Ethanoic acid is a typical weak acid. It reacts with water to produce hydroxonium ions and ethanoate ions, but the back reaction is more successful than the forward one. The ions react very easily to reform the acid and the water. • At any one time, only about 1% of the ethanoic acid molecules have converted into ions. • The rest remain as simple ethanoic acid molecules.
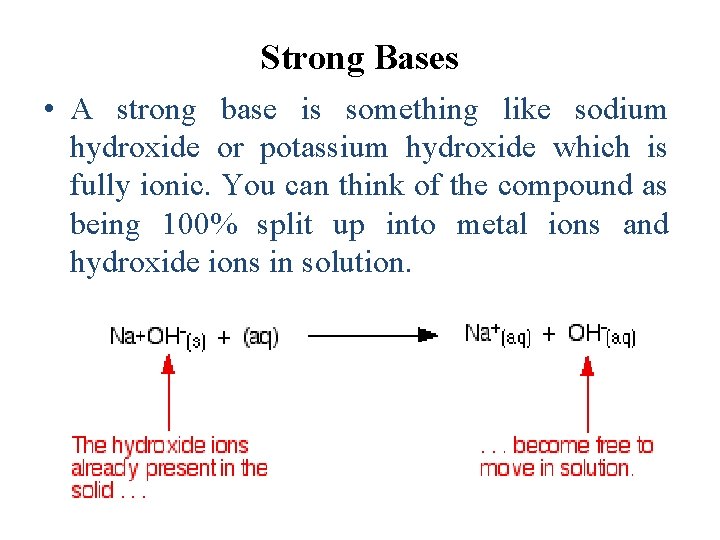
Strong Bases • A strong base is something like sodium hydroxide or potassium hydroxide which is fully ionic. You can think of the compound as being 100% split up into metal ions and hydroxide ions in solution.

Weak Bases • A weak base is one which doesn't convert fully into hydroxide ions in solution. • Ammonia is a typical weak base. Ammonia itself obviously doesn't contain hydroxide ions, but it reacts with water to produce ammonium ions and hydroxide ions. • However, the reaction is reversible, and at any one time about 99% of the ammonia is still present as ammonia molecules. Only about 1% has actually produced hydroxide ions.

Acids / Bases / Salts Recapturing…….

Objectives • • • Identify acids, bases and salts. Describe the properties of an acid and a base. Define an indicator. Draw, label and explain the p. H scale. Use indicators to determine if an unknown is an acid, a base, or a salt. • Describe a neutralization reaction. • Complete neutralization reactions.

Acids • PROTON DONOR • H+ ion

Acids • PROTON DONOR • H+ ion – A. PROPERTIES OF ACIDS: • • • Sour taste Litmus paper turns red Reacts with metal to produce H gas Contain H (most start with H) Release H+ ion in solution (forms H 30+)

Acids – B. STRONG ACIDS: • Produce many H+ ions • Good electrolytes

Acids – B. STRONG ACIDS: • Produce many H+ ions • Good electrolytes – C. WEAK ACIDS: • Produce few H+ ions • Poor electrolytes

Bases • PROTON ACCEPTOR • OH- ion

Bases • PROTON ACCEPTOR • OH- ion – A. PROPERTIES OF BASES: • • • Bitter taste Litmus paper turns blue Slippery to the touch Dissolve fats, oils, and grease Most contain OH (hydroxide) Most produce OH- ion in solution

Bases – B. STRONG BASES: • Produce many OH- ions • Good electrolytes

Bases – B. STRONG BASES: • Produce many OH- ions • Good electrolytes – C. WEAK BASES: • Produce few OH- ions • Poor electrolytes

Indicators • Show a color change to identify an acid or base • Types: – Litmus paper • Red – acid • Blue – base – Phenolphthalein (liquid) • Clear – acid, neutral • Pink – base

Indicators • Show a color change to identify an acid or base • Types: – p. H paper • Colors represent numerical values on p. H scale – Acid / base / neutral paper • Yellow – acid • Green – neutral • Purple – base

p. H • p. H SCALE: – Range of numbers from 0 to 14 – Indicates acid, base, and neutral along with strength

What is a SALT? • A salt is a neutral substance produced from the reaction of an acid and a base. • Composed of the negative ion of an acid and the positive ion of a base. • One of the products of a Neutralization Reaction • Examples: KCl, Mg. SO 4, Na 3 PO 4

Neutralization Reaction • A neutralization reaction is the reaction of an acid with a base to produce salt and water. • Example H 2 SO 4 + Na. OH Na. HSO 4 + H 2 O

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water • HCl + Na(OH) -----> Na. Cl + H(OH)

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water • HCl + Na(OH) -----> Na. Cl + H(OH) • H(NO 3) + Na(OH) -----> Na(NO 3) + H(OH)

APPLICATIONS OF NEUTRALIZATION IN DAILY LIFE 1. TOOTHPASTE • The bacteria in the mouth can change food that contains sugar into acid. • If the acid stays in the mouth for a long time, it attacks the outside enamel of the tooth. • This can cause the tooth to decay. • Toothpaste contains a weak alkali to neutralize the acid in the mouth.

APPLICATIONS OF NEUTRALIZATION IN DAILY LIFE 2. STOMACH POWDERS • The stomach produces hydrochloric acid. Sometimes the stomach produces too much of the acid and this causes indigestion and heartburn. • Stomach powders (anti-acids) which are bases when taken, reduces the amount of the acid in the stomach. • Examples of anti-acids are bicarbonates of soda, sodium hydrogen carbonate (Na. HCO 3) and Milk of magnesia (Magnesium hydroxide, Mg(OH)2).

APPLICATIONS OF NEUTRALIZATION IN DAILY LIFE 3. LIME • Excess soil acidity is one of the important causes of crop failure. • Some crops grow well in acidity soil but many crops may grow better in neutral or alkaline soil. In order to reduce soil acidity, the soil must be limed by the addition of lime materials. • Lime materials are materials which can neutralize acids in the soil. Example of lime materials are bases such as slaked lime; (Ca(OH)2).

APPLICATIONS OF NEUTRALIZATION IN DAILY LIFE 4. Calamine lotion • The pains of bee stings are caused by the injection of acid into the skin. • It can be treated with calamine lotion, a suspension of zinc carbonate (Zn. CO 3). • You may also treat the acid by applying sodium hydrogen carbonate (Na. HCO 3).

APPLICATIONS OF NEUTRALIZATION IN DAILY LIFE 5. Weak acid and weak alkalis • The sting of a wasp injects an alkali into the skin you may treat the alkali by applying a weak acid such as vinegar (ethanoic acid). • If a concentrated acid is spilled on the floor, it should neutralize by adding a weak alkali, such as sodium hydrogen carbonate. • Only after neutralization should the spill be wiped out using plenty of water. • Likewise, a spill of a strong alkali should first be neutralized by a weak acid such as ethanoic acid (vinegar) before wiping it out.

Digestion and p. H • Digestion-process by which foods are broken down into simpler substances. • Mechanical digestion-physical process in which food is torn apart (mouth) • Chemical digestion- chemical reactions in which large molecules are broken down into smaller molecules. (stomach and small intestines)

p. H in the Digestive System • Mouth-p. H around 7. Saliva contains amylase, an enzyme which begins to break carbohydrates into sugars. • Stomach- p. H around 2. Proteins are broken down into amino acids by the enzyme pepsin. • Small intestine-p. H around 8. Most digestion ends. Small molecules move to bloodstream toward cells that use them
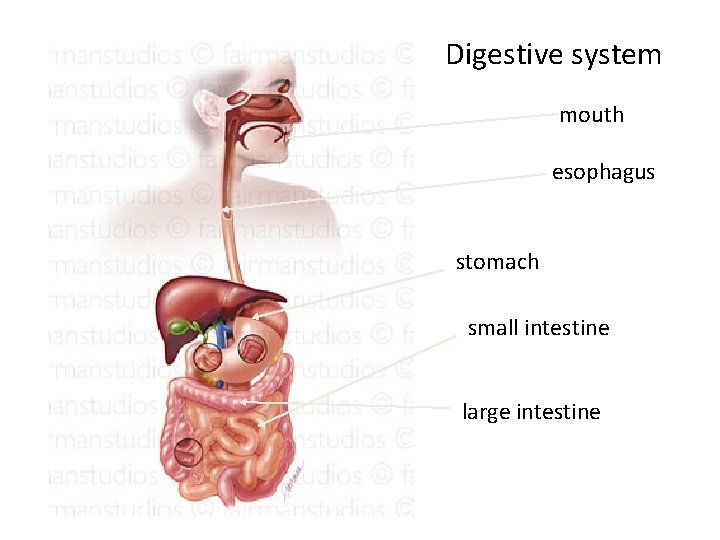
Digestive system mouth esophagus stomach small intestine large intestine

p. H in the Digestive System • Mouth-p. H around 7. Saliva contains amylase, an enzyme which begins to break carbohydrates into sugars. • Stomach- p. H around 2. Proteins are broken down into amino acids by the enzyme pepsin. • Small intestine-p. H around 8. Most digestion ends. Small molecules move to bloodstream toward cells that use them
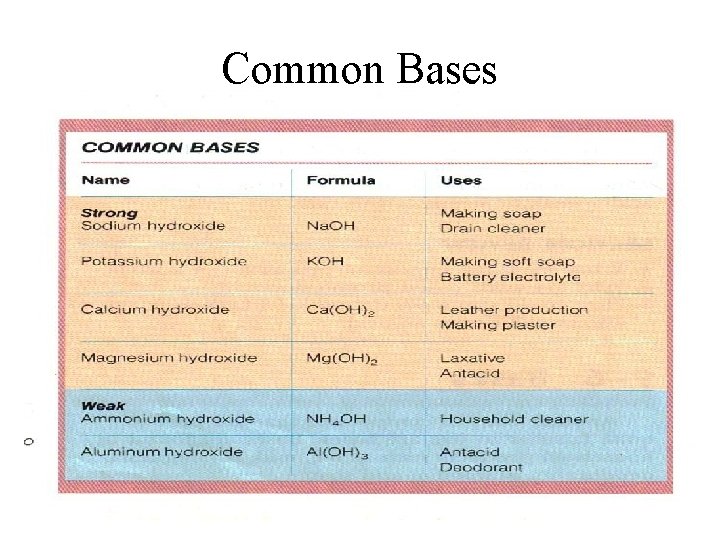
Common Bases
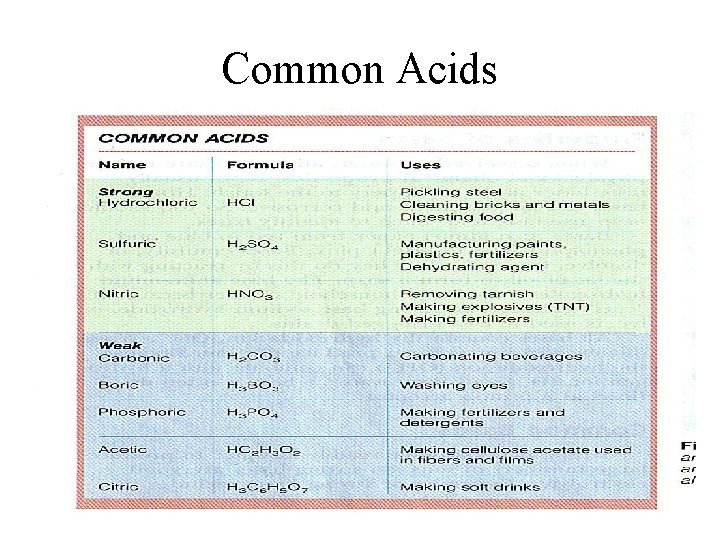
Common Acids

LECTURE 11 Outlines • • p. H Scale p. H of Common Substances Calculating p. H p. OH p. H Testing p. H Indicators Buffers: Controlling p. H Antacids

p. H Scale • The p. H scale is a way of expressing the strength of acids and bases. Instead of using very small numbers, we just use the NEGATIVE power of 10 on the Molarity (concentration) of the H+ or OH- ion. • Acidic: p. H < 7 • Neutral: p. H = 7 • Basic: p. H > 7

p. H of Common Substances
![Calculating p. H • p. H = -log [H+] • (Remember that the [ Calculating p. H • p. H = -log [H+] • (Remember that the [](http://slidetodoc.com/presentation_image_h/2f9b0db479a0ffdc9ce6c41e9b65c268/image-54.jpg)
Calculating p. H • p. H = -log [H+] • (Remember that the [ ] mean Molarity) Example: If [H+] = 1 X 10 -10 p. H = - log 1 X 10 -10 p. H = - (- 10) p. H = 10 Example: If [H+] = 1. 8 X 10 -5 p. H = - log 1. 8 X 10 -5 p. H = - (- 4. 74) p. H = 4. 74


p. OH and the PH scale • In terms of basicity, we can introduce a quantity p. OH. • For a basic solution, you can not directly calculate p. H. You must calculate p. OH first. p. H and p. OH are related by the following: * at 25 o. C

Understanding the p. H scale • Since the p. H scale is logarithmic, each p. H unit corresponds to an order of magnitude – [H 3 O+] = 10 -3 = 0. 001 M p. H = 3 – [H 3 O+] = 10 -2 = 0. 01 M p. H = 2 – [H 3 O+] = 10 -1 = 0. 1 M p. H = 1 – [H 3 O+] = 100 = 1 M – [H 3 O+] = 101 = 10 M p. H = 0 p. H = -1 • So, if the p. H of a solution is lowered by 3 p. H units, the H+ concentration has increased by a factor of 103, or 1000.

p. H scale and Important Notes • For this class, we will assume the temperature of all aqueous solutions to be exactly 25 o. C so that the previous expression holds true. • It is important to know the following: An acidic solution is defined as one in which [H 3 O+] > [OH-] (p. H < 7) A neutral solution is defined as one in which [H 3 O+] = [OH-] (p. H = 7) A basic solution is defined as one in which [OH-] > [H 3 O+] (p. H > 7) this will come in handy

Example • Calculate the p. H and p. OH of a solution having [H 3 O+] = 9. 14 x 10 -5 M • A solution has a p. H of 11. 4. Determine [H 3 O+] and [OH-]. Is this solution acidic or basic?

Example • What is the p. H of a 0. 040 M solution of HCl. O 4 at 25 o. C? – Perchloric acid is a strong acid, so we can assume complete dissociation. • The dissociation of the acid may also be written as: • It is a matter of preference.

Example • What is the p. H of a 0. 028 M solution of Na. OH at 25 o. C? Na. OH is a strong base (it is also a salt, and so OH- is directly produced by complete dissociation). To find p. H, we must calculate p. OH first.

p. OH • Since acids and bases are opposites, p. H and p. OH are opposites • p. OH does not really exist, but it is useful for changing bases to p. H. • p. OH looks at the perspective of a base • p. OH = - log [OH-] • Since p. H and p. OH are on opposite ends: • p. H + p. OH = 14
![p. H [H+] [OH-] p. OH p. H [H+] [OH-] p. OH](http://slidetodoc.com/presentation_image_h/2f9b0db479a0ffdc9ce6c41e9b65c268/image-63.jpg)
p. H [H+] [OH-] p. OH

p. H Testing • There are several ways to test p. H • Blue litmus paper (red = acid) • Red litmus paper (blue = basic) • p. H paper (multi-colored) • p. H meter (7 is neutral, <7 acid, >7 base) • Universal indicator (multi-colored) • Indicators like phenolphthalein • Natural indicators like red cabbage, radishes

p. H Indicators • Indicators are dyes that can be added that will change color in the presence of an acid or base. • Some indicators only work in a specific range of p. H • Once the drops are added, the sample is ruined • Some dyes are natural, like radish skin or red cabbage

Buffers: Controlling p. H • • Buffers, or buffer solutions, resist a change in p. H caused by the addition of acids or bases. There are two types of buffers: 1. Mixtures of weak acids and bases – these may be conjugate acid-base pairs, or nonconjugate acidbase pairs 2. Amphoteric species – these are substances that can act either as an acid or a base, like water

Buffers are extremely important in chemistry and biology. They maintain a nearly consistent p. H in various solutions. Our blood must maintain a p. H around 7. 35 -7. 45. If the p. H is above 7. 45 you would have a condition called alkalosis. If the p. H is below 7. 35, then one would suffer from acidosis. Acidosis leads to depression of the nervous system. Mild acidosis can result in dizziness, disorientation, or fainting; a more severe case can cause coma, or death. What would happen to the p. H of our blood if we were to eat acidic foods, such as apples, oranges, or limes? What might happen to the p. H of our blood if some of the hydrochloric acid from our stomach were to seep into our blood? The p. H would be lower in both

Despite the possibility of p. H increases or decreases, the body maintains a nearly constant p. H of 7. 4. The body uses buffers to maintain this remarkable feat. What is a buffer and how does it work?

Despite the possibility of p. H increases or decreases, the body maintains a nearly constant p. H of 7. 4. The body uses buffers to maintain this remarkable feat. What is a buffer and how does it work? A buffer consists of a weak acid and the salt of its conjugate base, or a weak base and the salt of its conjugate acid. Examples: HF + Na. OH Na. F + HOH w. a. c. b.

Despite the possibility of p. H increases or decreases, the body maintains a nearly constant p. H of 7. 4. The body uses buffers to maintain this remarkable feat. What is a buffer and how does it work? A buffer consists of a weak acid and the salt of its conjugate base, or a weak base and the salt of its conjugate acid. Examples: HF + Na. OH Na. F + HOH w. a. c. b. NH 3 + HCl NH 4 Cl w. b. c. a.

Antacids • The stomach secretes hydrochloric acid to activate enzymes that break down proteins. Sometimes the stomach produces too much acid and it can work its way back up the esophagus leading to heartburn. • Antacids are compounds that neutralize the excess acid: – Bicarbonates – Na. HCO 3 and KHCO 3 – Carbonates – Ca. CO 3 and Mg. CO 3 – Hydroxides – Al(OH)3 and Mg(OH)2

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water • HCl + Na(OH) -----> Na. Cl + H(OH)

Salts • NEUTRALIZATION REACTIONS: – reaction between an acid and a base to form a salt and water • acid + base -----> salt + water • HCl + Na(OH) -----> Na. Cl + H(OH) • H(NO 3) + Na(OH) -----> Na(NO 3) + H(OH)
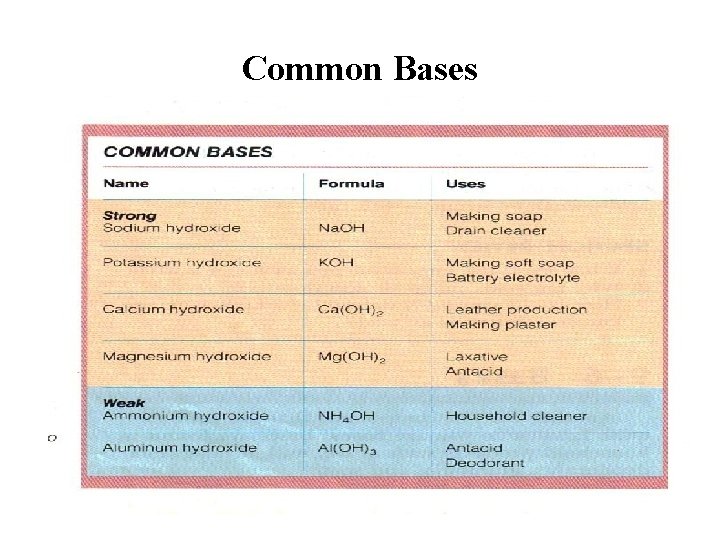
Common Bases
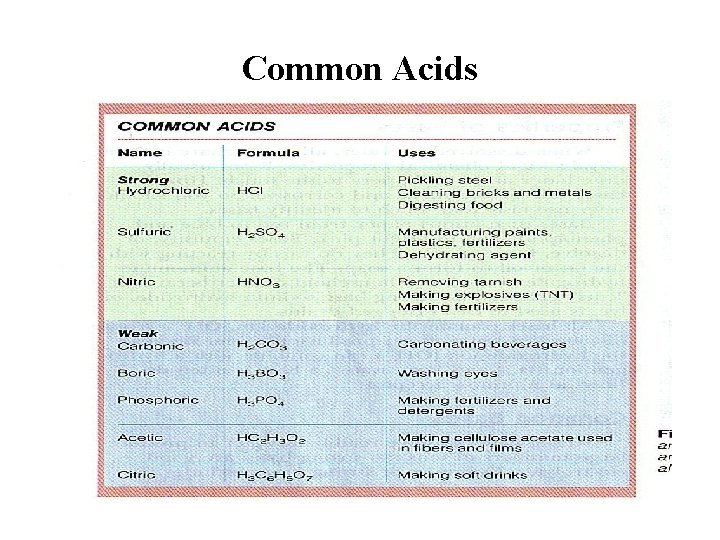
Common Acids
- Slides: 76